
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Naresh Kumar | -- | 5509 | 2023-01-30 15:26:15 | | | |
| 2 | Lindsay Dong | Meta information modification | 5509 | 2023-01-31 10:26:00 | | |
Video Upload Options
Endodontics is an important sub-branch of dentistry which deals with the different conditions of pulp to prevent tooth loss. Traditionally, common procedures, namely pulp capping, root canal treatment, apexification, and apexigonesis, have been considered for the treatment of different pulp conditions using selected materials. However, clinically to regenerate dental pulp, tissue engineering has been advocated as a feasible approach. New trends are emerging in terms of regenerative endodontics which have led to the replacement of diseased and non-vital teeth into the functional and healthy dentine-pulp complex. Root- canal therapy is the standard management option when dental pulp is damaged irreversibly. This treatment modality involves soft-tissue removal and then filling that gap through the obturation technique with a synthetic material. The formation of tubular dentine and pulp-like tissue formation occurs when stem cells are transplanted into the root canal with an appropriate scaffold material. To sum up tissue engineering approach includes three components: (1) scaffold, (2) differentiation, growth, and factors, and (3) the recruitment of stem cells within the pulp or from the periapical region.
1. Introduction
2. Development of Regenerative Endodontic Procedures (REP)
3. Revascularization or Revitalization
3.1. Advantages of the Revascularization Approach
-
Technically simple approach.
-
There is no need of using expensive biotechnology due to currently available instruments and medicament techniques.
-
There are almost negligible chances of immune rejection as this approach relies on the patient’s own blood.
-
Bacterial microleakage can be eliminated through the induction of stem cells into the root canal space, followed by the intra-canal barrier, inducing a blood clot.
-
The concerns of restoration retention need to be overcome.
-
When this approach is applied to immature teeth, it reinforces their root walls.
-
As the avulsed immature tooth has necrotic-pulp tissue along with an open apex, and short and intact roots; therefore, the newly formed tissue will easily reach the coronal-pulp horn because proliferation in a short distance is required. Therefore, the strategy behind the development of new tissue is to maintain the balance between the pulp-space infection and the proliferation of new tissue.
-
Additional growth of open-apex root takes place due to minimum instrumentation that will preserve viable pulp tissue.
-
The potential to regenerate more stem cells and the rapid capacity to heal the tissue in young patients needs to be recognised.
3.2. Disadvantages of the Revascularization Approach
-
The origin of where the tissue has been regenerated from is yet to be known.
-
According to researchers, effective composition and concentration of cells are mandatory for tissue engineering. However, these cells are entombed in fibrin clots; therefore, researchers do not rely on blood-clot formation for tissue engineering function.
3.3. Prerequisites for Revascularization Approach (Figure 1)
-
There should be open apices and necrotic pulp secondary to trauma.
-
In addition, open apex should be less than 1.5 mm.
-
The following agents can be incorporated to remove microorganisms from the canal.
-
The coronal seal should be effective.
-
There should be a matrix or the growth of new tissues.
-
When trying to induce bleeding, anaesthesia should be used without a vasoconstrictor [42].
-
Canals should not be instrumented.
-
Sodium hypochlorite should be used as the irrigant.
-
There should be blood-clot formation.
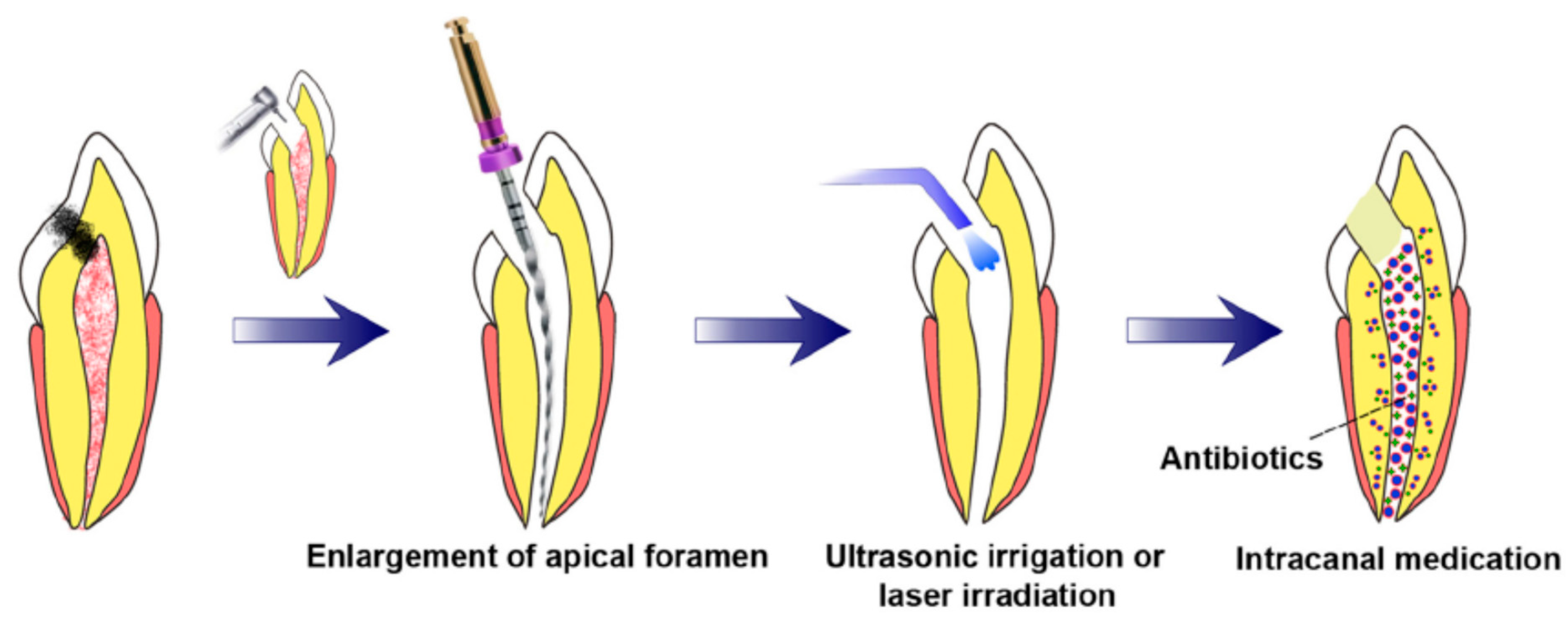
4. Postnatal Stem Cell Therapy
Bone, buccal mucosa, fat, and skin are the common sources of postnatal-stem cells. After the apex is opened, the disinfected root-canal system is injected with postnatal-stem cells. This treatment is considered the simplest technique [44]. There are numerous benefits of this type of tissue-engineering technique. Postnatal-stem cells are rationally easy to harvest, and these cells can persuade the regeneration of the pulp. Moreover, these cells are easy to deliver by syringe. In addition, application of these stem-cell therapy is used in regenerative medicine since past many years, for example, bone-marrow replacement and endodontic applications [45]. However, low survival rates are one of the major disadvantages of this technique. Moreover, these cells can migrate into different locations of the body, which presents peculiar forms of mineralization [46]. For the development of dental tissues by the differentiation of stem cells, bioactive-signalling molecules, growth factors, and scaffolds are required [47]. Consequently, with only stem cells that exclude the growth factors or scaffolds, the chance of pulpal regeneration of new tissues is very low. In this approach, the chief identification of a postnatal-stem-cell source that must be able to differentiate into the diverse cell population can be obtained [46]. However, this technique is not approved yet.4.1. Pulp Implantation
4.2. Scaffold Implantation
4.3. Three-Dimensional Cell Printing
4.4. Gene Therapy
4.5. Nitric Oxide
4.6. Platelet-Rich Plasma (PRP)
4.7. Cell Homing
In tissue regeneration, the first concept of cell homing was presented in Lancet in 2010. The concept was based on the delivery of transforming growth factor-b3 (TGFb3) without cell transplantation. This approach was first used for the regeneration of the articular cartilage [75]. However, for dental-tissue regeneration, the idea of cell homing was introduced in 2010 [76]. During cell homing, root canal of the extracted human teeth was shaped and cleaned followed by the delivery of the growth factors, scaffold, and stem cells. Residual proteins in the root canal or dentinal tubules were deactivated in the first phase. This can be done by sterilization of extracted teeth in an autoclave. This was followed by the infusion of collagen gel into a shaped and cleaned root canal that might be with or without basic fibroblast growth factors (bFGFs), vascular endothelial growth factors (VEGFs), platelet-derived growth factors (PDGFs), nerve growth factors (NGFs), or bone morphogenetic proteins (BMPs).
The prime difference between the cell homing and cell transplantation approaches is that, in the latter case, for dentine/pulp regeneration, the isolated cells (stem/progenitor) from the host are transplanted into the root canal of the host. Dental pulp-like cells have been differentiated in the cell-homing approach when growth factors are recruited into the root-canal system. Cell-homing-technique-dental-organ regeneration presents a harmonizing and/or balancing approach to cell-transplantation technique and, at the same time, this strategy has shown auspicious results in animal models [14][76][77]. Hematopoietic-stem cells were militarized and transferred to different tissues or organs using active navigation in the cell-homing approach. The ultimate outcome of this process is pulp-dentin re-cellularization and revascularization. Numerous growth factors along with cell homing will result in pulp-dentin regeneration. Tissue revascularization and regeneration-cell homing consist of two distinctive cellular processes. They are differentiation and recruitment [78][79].
5. Biomimetic Materials in Endodontics
5.1. Biointeractive Materials
5.1.1. Calcium Hydroxide
5.1.2. Calcium Sulfate
5.2. Bioactive Materials
“A bioactive material is one that elicits a specific biological response at the interface of the material which results in the formation of a bond between the tissues and the material” [98].
5.2.1. Calcium Silicate Based-Cements
Mineral Trioxide Aggregate
Biodentine
Calcium Aluminate Cement
Theracal
5.2.2. Calcium Phosphate Based Cements
Hydroxyapatite
Bioactive Glass
5.2.3. Mixture of Calcium Silicate and Phosphate Based-Cements
Bioaggregate
Endosequence Root Repair Material
5.2.4. Sealer
Endosequence BC Sealer
5.2.5. Gutta-Percha
Bioceramic Coated Gutta-Percha
5.3. Remineralizing Agents
5.3.1. Enamel Matrix Derivative (Emdogain) Remineralizing Agent
5.3.2. Dentine Matrix Derivative/Demineralized Dentin Matrix
5.4. Miscellaneous
Calcium Phosphate Cements
6. Conclusions
References
- Benyus, J.M. Biomimicry: Innovation Inspired by Nature; Morrow: New York, NY, USA, 1997.
- Vakili, V.; Shu, L.H. (Eds.) Towards biomimetic concept generation. In International Design Engineering Technical Conferences and Computers and Information in Engineering Conference; American Society of Mechanical Engineers: New York, NY, USA, 2001.
- Sonarkar, S.; Purba, R. Bioactive materials in conservative dentistry. Int. J. Contemp. Dent. Med. Rev. 2015, 2015, 340115.
- Harkness, J.M. An idea man (the life of Otto Herbert Schmitt). IEEE Eng. Med. Biol. Mag. 2004, 23, 20–41.
- Zafar, M.S.; Amin, F.; Fareed, M.A.; Ghabbani, H.; Riaz, S.; Khurshid, Z.; Kumar, N. Biomimetic Aspects of Restorative Dentistry Biomaterials. Biomimetics 2020, 5, 34.
- Kottoor, J. Biomimetic endodontics: Barriers and strategies. Health Sci. 2013, 2, 7–12.
- About, I. Biodentine: From biochemical and bioactive properties to clinical applications. G. Ital. Endod. 2016, 30, 81–88.
- Sharma, V.; Srinivasan, A.; Nikolajeff, F.; Kumar, S. Biomineralization process in hard tissues: The interaction complexity within protein and inorganic counterparts. Acta Biomater. 2021, 120, 20–37.
- Bazos, P.; Magne, P. Bio-emulation: Biomimetically emulating nature utilizing a histo-anatomic approach; structural analysis. Eur. J. Esthet. Dent. 2011, 6, 8–19.
- Andreasen, J.O.; Farik, B.; Munksgaard, E.C. Long-term calcium hydroxide as a root canal dressing may increase risk of root fracture. Dent. Traumatol. 2002, 18, 134–137.
- Hargreaves, K.M.; Diogenes, A.; Teixeira, F.B. Treatment options: Biological basis of regenerative endodontic procedures. Pediatr. Dent. 2013, 35, 129–140.
- Huang, G.T. Pulp and dentin tissue engineering and regeneration: Current progress. Regen. Med. 2009, 4, 697–707.
- Kim, S.G.; Zheng, Y.; Zhou, J.; Chen, M.; Embree, M.C.; Song, K.; Jiang, N.; Mao, J.J. Dentin and dental pulp regeneration by the patient’s endogenous cells. Restor. Dent. Endod. 2013, 28, 106–117.
- Mao, J.J.; Kim, S.G.; Zhou, J.; Ye, L.; Cho, S.; Suzuki, T.; Fu, S.Y.; Yang, R.; Zhou, X. Regenerative endodontics: Barriers and strategies for clinical translation. Dent. Clinic. 2012, 56, 639–649.
- Ostby, B.N. The role of the blood clot in endodontic therapy. An experimental histologic study. Acta Odontol. Scand. 1961, 19, 324–353.
- Murray, P.E.; Garcia-Godoy, F.; Hargreaves, K.M. Regenerative endodontics: A review of current status and a call for action. J. Endod. 2007, 33, 377–390.
- Garcia-Godoy, F.; Murray, P. Recommendations for using regenerative endodontic procedures in permanent immature traumatized teeth. Dent. Traumatol. 2012, 28, 33–41.
- Albuquerque, M.T.P.; Valera, M.C.; Nakashima, M.; Nör, J.E.; Bottino, M.C. Tissue-engineering-based Strategies for Regenerative Endodontics. J. Dent. Res. 2014, 93, 1222–1231.
- Yuan, Z.; Nie, H.; Wang, S.; Lee, C.H.; Li, A.; Fu, S.Y.; Zhou, H.; Chen, L.; Mao, J.J. Biomaterial Selection for Tooth Regeneration. Tissue Eng. Part B Rev. 2011, 17, 373–388.
- Hargreaves, K.M.; Giesler, T.; Henry, M.; Wang, Y. Regeneration potential of the young permanent tooth: What does the future hold? Pediatr. Dent. 2008, 30, 253–260.
- Huang, G.T.-J.; Sonoyama, W.; Liu, Y.; Liu, H.; Wang, S.; Shi, S. The Hidden Treasure in Apical Papilla: The Potential Role in Pulp/Dentin Regeneration and BioRoot Engineering. J. Endod. 2008, 34, 645–651.
- American Association of Endodontists. AAE Clinical Considerations for a Regenerative Procedure; American Association of Endodontists: Chicago, IL, USA, 2016.
- Sedgley, C.M.; Botero, T.M. Dental Stem Cells and Their Sources. Dent. Clin. N. Am. 2012, 56, 549–561.
- Galler, K.; D’Souza, R.; Hartgerink, J.; Schmalz, G. Scaffolds for Dental Pulp Tissue Engineering. Adv. Dent. Res. 2011, 23, 333–339.
- Widbiller, M.; Driesen, R.B.; Eidt, A.; Lambrichts, I.; Hiller, K.-A.; Buchalla, W.; Schmalz, G.; Galler, K.M. Cell Homing for Pulp Tissue Engineering with Endogenous Dentin Matrix Proteins. J. Endod. 2018, 44, 956–962.e2.
- Torabinejad, M.; Alexander, A.; Vahdati, S.A.; Grandhi, A.; Baylink, D.; Shabahang, S. Effect of Residual Dental Pulp Tissue on Regeneration of Dentin-pulp Complex: An In Vivo Investigation. J. Endod. 2018, 44, 1796–1801.
- Nygaard-Östby, B.; Hjortdal, O. Tissue formation in the root canal following pulp removal. Eur. J. Oral Sci. 1971, 79, 333–349.
- Banchs, F.; Trope, M. Revascularization of immature permanent teeth with apical periodontitis: New treatment protocol? J. Endod. 2004, 30, 196–200.
- Iwaya, S.; Ikawa, M.; Kubota, M. Revascularization of an immature permanent tooth with apical periodontitis and sinus tract. Dent. Traumat. 2001, 17, 185–187.
- Kubasad, G.C.; Ghivari, S.B. Apexification with apical plug of MTA-report of cases. Arch. Oral Sci. Res. 2011, 1, 104–107.
- Vojinović, J.; Čupić, S.; Dolić, O.; Mirjanić, Đ.; Sukara, S.; Obradović, M. Success rate of the endodontic treatment of young permanent teeth with calcium hydroxide. Growth Contemp. 2010, 4, 8.
- Nagaveni, N.; Umashankara, K.; Radhika, N.; Manjunath, S. Successful closure of the root apex in non-vital permanent incisors with wide open apices using single calcium hydroxide (caoh) dressing—Report of 2 cases. J. Clin. Exp. Dent. 2010, 1, 165–167.
- Bucal, M.O. Apexification, Apexogenesis and Pulpal Revascularization: Literature Review and Clinical Applications. St. Vincent Char. Med. Cent. J. 2018, 40, 8.
- Ford, T.P.; Shabahang, S. Management of incompletely formed roots. In Principles and Practice of Endodontics; Saunders: Philadelphia, PA, USA, 2003; pp. 388–404.
- Lovelace, T.W.; Henry, M.A.; Hargreaves, K.M.; Diogenes, A. Evaluation of the Delivery of Mesenchymal Stem Cells into the Root Canal Space of Necrotic Immature Teeth after Clinical Regenerative Endodontic Procedure. J. Endod. 2011, 37, 133–138.
- Llames, S.G.; Del Rio, M.; Larcher, F.; García, E.; García, M.; Escámez, M.J.; Jorcano, J.L.; Holguín, P.; Meana, A. Human plasma as a dermal scaffold for the generation of a completely autologous bioengineered skin. Transplantation 2004, 77, 350–355.
- Frye, C.A.; Wu, X.; Patrick, C.W. Microvascular endothelial cells sustain preadipocyte viability under hypoxic conditions. Cell. Dev. Biol. Anim. 2005, 41, 160–164.
- Hofer, S.; Mitchell, G.; Penington, A.; Morrison, W.; RomeoMeeuw, R.; Keramidaris, E.; Palmer, J.; Knight, K. The use of pimonidazole to characterise hypoxia in the internal environment of an in vivo tissue engineering chamber. Br. J. Plast. Surg. 2005, 58, 1104–1114.
- Lembert, N.; Wesche, J.; Petersen, P.; Doser, M.; Zschocke, P.; Becker, H.D.; Ammon, H.P.T. Encapsulation of Islets in Rough Surface, Hydroxymethylated Polysulfone Capillaries Stimulates VEGF Release and Promotes Vascularization after Transplantation. Cell Transplant. 2005, 14, 97–108.
- Chueh, L.-H.; Huang, G.T.-J. Immature Teeth with Periradicular Periodontitis or Abscess Undergoing Apexogenesis: A Paradigm Shift. J. Endod. 2006, 32, 1205–1213.
- Shah, N.; Logani, A.; Bhaskar, U.; Aggarwal, V. Efficacy of Revascularization to Induce Apexification/Apexogensis in Infected, Nonvital, Immature Teeth: A Pilot Clinical Study. J. Endod. 2008, 34, 919–925.
- Petrino, J.A.; Boda, K.; Shambarger, S.; Bowles, W.R.; McClanahan, S.B. Challenges in Regenerative Endodontics: A Case Series. J. Endod. 2010, 36, 536–541.
- Yang, J.; Yuan, G.; Chen, Z. Pulp Regeneration: Current Approaches and Future Challenges. Front. Physiol. 2016, 7, 58.
- Kindler, V. Postnatal stem cell survival: Does the niche, a rare harbor where to resist the ebb tide of differentiation, also provide lineage-specific instructions? J. Leukol. Biol. 2005, 78, 836–844.
- Nakashima, M.; Akamine, A. The Application of Tissue Engineering to Regeneration of Pulp and Dentin in Endodontics. J. Endod. 2005, 31, 711–718.
- Brazelton, T.R.; Blau, H.M. Optimizing Techniques for Tracking Transplanted Stem Cells In Vivo. Stem Cells 2005, 23, 1251–1265.
- Nakashima, M. Bone morphogenetic proteins in dentin regeneration for potential use in endodontic therapy. Cytokine Growth Factor Rev. 2005, 16, 369–376.
- Fukuda, J.; Khademhosseini, A.; Yeh, J.; Eng, G.; Cheng, J.; Farokhzad, O.C.; Langer, R. Micropatterned cell co-cultures using layer-by-layer deposition of extracellular matrix components. Biomaterials 2006, 27, 1479–1486.
- Huang, G.T.-J.; Sonoyama, W.; Chen, J.; Park, S.H. In vitro characterization of human dental pulp cells: Various isolation methods and culturing environments. Cell Tissue Res. 2006, 324, 225–236.
- Nakashima, M. Tissue Engineering in Endodontics. Aust. Dent. J. 2005, 31, 111–113.
- Tabata, Y. Nanomaterials of Drug Delivery Systems for Tissue Regeneration. Methods Mol. Biol. 2005, 300, 81–100.
- Boccaccini, A.R.; Blaker, J. Bioactive composite materials for tissue engineering scaffolds. Expert Rev. Med. Devices 2005, 2, 303–317.
- Kitasako, Y.; Shibata, S.; Pereira, P.N.; Tagami, J. Short-term dentin bridging of mechanically-exposed pulps capped with adhesive resin systems. Oper. Dent. 2000, 25, 155–162.
- Mjor, I.A.; Dah, E.; Cox, C.F. Healing of pulp exposures: An ultrastructural study. J. Oral Pathol. Med. 1991, 20, 496–501.
- Silva, T.; Rosa, A.; Lara, V. Dentin matrix proteins and soluble factors: Intrinsic regulatory signals for healing and resorption of dental and periodontal tissues? Oral Dis. 2004, 10, 63–74.
- Elisseeff, J.; Puleo, C.; Yang, F.; Sharma, B. Advances in skeletal tissue engineering with hydrogels. Orthod. Craniofac. Res. 2005, 8, 150–161.
- Trojani, C.; Weiss, P.; Michiels, J.-F.; Vinatier, C.; Guicheux, J.; Daculsi, G.; Gaudray, P.; Carle, G.F.; Rochet, N. Three-dimensional culture and differentiation of human osteogenic cells in an injectable hydroxypropylmethylcellulose hydrogel. Biomaterials 2005, 26, 5509–5517.
- Dusseiller, M.R.; Schlaepfer, D.; Koch, M.; Kroschewski, R.; Textor, M. An inverted microcontact printing method on topographically structured polystyrene chips for arrayed micro-3-D culturing of single cells. Biomaterials 2005, 26, 5917–5925.
- Barron, J.A.; Krizman, D.B.; Ringeisen, B.R. Laser Printing of Single Cells: Statistical Analysis, Cell Viability, and Stress. Ann. Biomed. Eng. 2005, 33, 121–130.
- Rutherford, R.B. BMP-7 gene transfer to inflamed ferret dental pulps. Eur. J. Oral Sci. 2001, 109, 422–424.
- Jüllig, M.; Zhang, W.V.; Stott, N.S. Gene therapy in orthopaedic surgery: The current status. ANZ J. Surg. 2004, 74, 46–54.
- Stolberg, S.G. Trials are halted on a gene therapy. The New York Times, 2002; p. 4.
- Kimura, H.; Esumi, H. Reciprocal regulation between nitric oxide and vascular endothelial growth factor in angiogenesis. Acta Biochim. Pol. 2003, 50, 49–59.
- Sun, B.; Slomberg, D.L.; Chudasama, S.L.; Lu, Y.; Schoenfisch, M.H. Nitric Oxide-Releasing Dendrimers as Antibacterial Agents. Biomacromolecules 2012, 13, 3343–3354.
- Backlund, C.J.; Worley, B.V.; Schoenfisch, M.H. Anti-biofilm action of nitric oxide-releasing alkyl-modified poly(amidoamine) dendrimers against Streptococcus mutans. Acta Biomater. 2016, 29, 198–205.
- Ding, R.Y.; Cheung, G.S.-P.; Chen, J.; Yin, X.; Wang, Q.Q.; Zhang, C. Pulp Revascularization of Immature Teeth with Apical Periodontitis: A Clinical Study. J. Endod. 2009, 35, 745–749.
- Anitua, E.; Sánchez, M.; Nurden, A.T.; Nurden, P.; Orive, G.; Andía, I. New insights into and novel applications for platelet-rich fibrin therapies. Trends Biotechnol. 2006, 24, 227–234.
- Ogino, Y.; Ayukawa, Y.; Kukita, T.; Koyano, K. The contribution of platelet-derived growth factor, transforming growth factor-β1, and insulin-like growth factor-I in platelet-rich plasma to the proliferation of osteoblast-like cells. Oral Surg. Oral Med. Oral Pathol. Oral Radiol. Endodontol. 2006, 101, 724–729.
- Marx, R.E. Platelet-rich plasma (PRP): What is PRP and what is not PRP? Implant Dent. 2001, 10, 225–228.
- Samadi, P.; Sheykhhasan, M.; Khoshinani, H.M. The Use of Platelet-Rich Plasma in Aesthetic and Regenerative Medicine: A Comprehensive Review. Aesthetic Plast. Surg. 2019, 43, 803–814.
- Whitman, D.H.; Berry, R.L.; Green, D.M. Platelet gel: An autologous alternative to fibrin glue with applications in oral and maxillofacial surgery. J. Oral Maxillofac. Surg. 1997, 55, 1294–1299.
- Lee, U.-L.; Jeon, S.H.; Park, J.-Y.; Choung, P.-H. Effect of platelet-rich plasma on dental stem cells derived from human impacted third molars. Regen. Med. 2011, 6, 67–79.
- Torabinejad, M.; Faras, H. A Clinical and Histological Report of a Tooth with an Open Apex Treated with Regenerative Endodontics Using Platelet-rich Plasma. J. Endod. 2012, 38, 864–868.
- DeLong, J.M.; Russell, R.P.; Mazzocca, A.D. Platelet-Rich Plasma: The PAW Classification System. Arthrosc. J. Arthrosc. Relat. Surg. 2012, 28, 998–1009.
- Lee, C.H.; Cook, J.L.; Mendelson, A.; Moioli, E.K.; Yao, H.; Mao, J.J. Regeneration of the articular surface of the rabbit synovial joint by cell homing: A proof of concept study. Lancet 2010, 376, 440–448.
- Kim, J.Y.; Xin, X.; Moioli, E.K.; Chung, J.; Lee, C.H.; Chen, M.; Fu, S.Y.; Koch, P.D.; Mao, J.J. Regeneration of Dental-Pulp-like Tissue by Chemotaxis-Induced Cell Homing. Tissue Eng. Part A 2010, 16, 3023–3031.
- Ishizaka, R.; Iohara, K.; Murakami, M.; Fukuta, O.; Nakashima, M. Regeneration of dental pulp following pulpectomy by fractionated stem/progenitor cells from bone marrow and adipose tissue. Biomaterials 2012, 33, 2109–2118.
- Rosa, V.; Botero, T.M.; Nör, J.E. Regenerative endodontics in light of the stem cell paradigm. Int. Dent. J. 2011, 61, 23–28.
- Casagrande, L.; Cordeiro, M.M.; Nör, S.A.; Nör, J.E. Dental pulp stem cells in regenerative dentistry. Odontology 2011, 99, 1–7.
- Ruparel, N.B.; Teixeira, F.B.; Ferraz, C.C.; Diogenes, A. Direct Effect of Intracanal Medicaments on Survival of Stem Cells of the Apical Papilla. J. Endod. 2012, 38, 1372–1375.
- Kahler, B.; Chugal, N.; Lin, L.M. Alkaline materials and regenerative endodontics: A review. Materials 2017, 10, 1389.
- Galler, K.M.; Buchalla, W.; Hiller, K.-A.; Federlin, M.; Eidt, A.; Schiefersteiner, M.; Schmalz, G. Influence of Root Canal Disinfectants on Growth Factor Release from Dentin. J. Endod. 2015, 41, 363–368.
- Galler, K.M.; Widbiller, M.; Buchalla, W.; Eidt, A.; Hiller, K.-A.; Hoffer, P.C.; Schmalz, G. EDTA conditioning of dentine promotes adhesion, migration and differentiation of dental pulp stem cells. Int. Endod. J. 2016, 49, 581–590.
- Althumairy, R.I.; Teixeira, F.B.; Diogenes, A. Effect of Dentin Conditioning with Intracanal Medicaments on Survival of Stem Cells of Apical Papilla. J. Endod. 2014, 40, 521–525.
- Alhadainy, H.A.; Himel, V.T. Evaluation of the sealing ability of amalgam, cavit and glass ionomer cement in the repair of furcation perforations. Oral Surg. Oral Med. Oral Pathol. Oral Radiol. 1993, 75, 362–366.
- Zaen El-Din, A.M.; Hamama, H.H.; Abo El-Elaa, M.A.; Grawish, M.E.; Mahmoud, S.H.; Neelakantan, P. The effect of four materials on direct pulp capping: An animal study. Aus. Endont. J. 2020, 46, 249–256.
- Fridland, M.; Rosado, R. Mineral trioxide aggregate solubility and porosity with different water to powder ratios. J. Endod. 2003, 29, 814–817.
- Moazami, F.; Sahebi, S.; Jamshidi, D.; Alavi, A. The long-term effect of calcium hydroxide, calcium-enriched mixture cement and mineral trioxide aggregate on dentin strength. Iran. Endod. J. 2014, 9, 185–189.
- Revathi, N.; Chandra, S.S. Merits and demerits of calcium hydroxide as a therapeutic agent: A review. Int. J. Dent. Res. 2014, 2, 1–4.
- Chen, J.; Gao, J.; Yin, H.; Liu, F.; Wang, A.; Zhu, Y.; Wu, Z.; Jiang, T.; Qin, D.; Chen, B.; et al. Size-controlled preparation of a-calcium sulphate hemihydrate starting from calcium sulphate dihydrate in the presence of modifiers and the dissolution rate in simulated body fluid. Mater. Sci. Eng. C-Mater. Biol. Appl. 2013, 33, 3256–3262.
- Peltier, L.F.; Jones, R.F. Treatment of unicameral bone cysts by curettage and packing with plaster-of-Paris pellets. J. Bone Jt. Surg. Am. 1978, 60, 820–822.
- Yoshikawa, G.; Murashima, Y.; Wadachi, R.; Sawada, N.; Suda, H. Guided bone regeneration (GBR) using membranes and calcium sulphate after apicectomy: A comparative histomorphometrical study. Int. Endod. J. 2002, 35, 255–263.
- Pecora, G.; De Leonardis, D.; Ibrahim, N.; Bovi, M.; Cornelini, R. The use of calcium sulphate in the surgical treatment of a ‘through and through’periradicular lesion. Int. Endod. J. 2001, 34, 189–197.
- Van der Stok, J.; Van Lieshout, E.M.; El-Massoudi, Y.; Van Kralingen, G.H.; Patka, P. Bone substitutes in the Netherlands–a systematic literature review. Acta Biomater. 2011, 7, 739–750.
- Yahav, A.; Kurtzman, G.M.; Katzap, M.; Dudek, D.; Baranes, D. Bone regeneration: Properties and clinical applications of biphasic calcium sulfate. Dent. Clin. 2020, 64, 453–472.
- Podaropoulos, L.; Veis, A.A.; Papadimitriou, S.; Alexandridis, C.; Kalyvas, D. Bone Regeneration Using B-Tricalcium Phosphate in a Calcium Sulfate Matrix. J. Oral Implant. 2009, 35, 28–36.
- Cai, Z.-Y.; Yang, D.-A.; Zhang, N.; Ji, C.-G.; Zhu, L.; Zhang, T. Poly (propylene fumarate)/(calcium sulphate/β-tricalcium phosphate) composites: Preparation, characterization and in vitro degradation. Acta Biomater. 2009, 5, 628–635.
- Hench, L.L.; Splinter, R.J.; Allen, W.C.; Greenlee, T.K. Bonding mechanisms at the interface of ceramic prosthetic materials. J. Biomed. Mater. Res. 1972, 5, 117–141.
- Lee, S.-J.; Monsef, M.; Torabinejad, M. Sealing ability of a mineral trioxide aggregate for repair of lateral root perforations. J. Endod. 1993, 19, 541–544.
- Asgary, S.; Parirokh, M.; Eghbal, M.J.; Brink, F. Chemical Differences between White and Gray Mineral Trioxide Aggregate. J. Endod. 2005, 31, 101–103.
- Rajasekharan, S.; Martens, L.C.; Cauwels, R.G.E.C.; Verbeeck, R.M.H. Biodentine™ material characteristics and clinical applications: A review of the literature. Eur. Arch. Paediatr. Dent. 2014, 15, 147–158.
- Camilleri, J. Hydration mechanisms of mineral trioxide aggregate. Int. Endod. J. 2007, 40, 462–470.
- Kadali, N.; Alla, R.K.; Guduri, V.; Ramaraju, A.V.; Sajjan, S.; Rudraraju, V.R. Mineral Trioxide Aggregate: An overview of composition, properties and clinical applications. Int. J. Dent. Mater. 2020, 2, 11-8.
- Aeinehchi, M.; Eslami, B.; Ghanbariha, M.; Saffar, A.S. Mineral trioxide aggregate (mta) and calcium hydroxide as pulp-capping agents in human teeth: A preliminary report. Int. Endod. J. 2003, 36, 225–235.
- White, C., Jr.; Bryant, N.N. Combined therapy of mineral trioxide aggregate and guided tissue regeneration in the treatment of external root resorption and an associated osseous defect. J. Periodontol. 2002, 73, 1517–1521.
- Tawil, P.Z.; Duggan, D.J.; Galicia, J.C. MTA: A clinical review. Compend. Contin. Educ. 2015, 36, 247.
- Cervino, G.; Laino, L.; D’Amico, C.; Russo, D.; Nucci, L.; Amoroso, G.; Gorassini, F.; Tepedino, M.; Terranova, A.; Gambino, D.; et al. Mineral Trioxide Aggregate Applications in Endodontics: A Review. Eur. J. Dent. 2020, 14, 683–691.
- Faraco, I.M., Jr.; Holland, R. Response of the pulp of dogs to capping with mineral trioxide aggregate or a calcium hydroxide cement. Dent. Traumatol. 2001, 17, 163–166.
- Dominguez, M.S.; E Witherspoon, D.; Gutmann, J.L.; Opperman, L. Histological and Scanning Electron Microscopy Assessment of Various Vital Pulp-Therapy Materials. J. Endod. 2003, 29, 324–333.
- Tziafas, D.; Pantelidou, O.; Alvanou, A.; Belibasakis, G.; Papadimitriou, S. The dentinogenic effect of mineral trioxide aggregate (MTA) in short-term capping experiments. Int. Endod. J. 2002, 35, 245–254.
- Arandi, N.Z.; Thabet, M. Minimal intervention in dentistry: A literature review on Biodentine as a bioactive pulp capping material. BioMed Res. Int. 2021.
- Nowicka, A.; Lipski, M.; Parafiniuk, M.; Sporniak-Tutak, K.; Lichota, D.; Kosierkiewicz, A.; Kaczmarek, W.; Buczkowska-Radlińska, J. Response of human dental pulp capped with biodentine and mineral trioxide aggregate. J. Endont. 2013, 39, 743–747.
- Malkondu, O.; Kazandag, M.K.; Kazazoglu, E. A review on biodentine, a contemoprary dentin replacement and repair material. BioMed. Res. Int. 2014, 2014, 160951.
- Wattanapakkavong, K.; Srisuwan, T. Release of Transforming Growth Factor Beta 1 from Human Tooth Dentin after Application of Either ProRoot MTA or Biodentine as a Coronal Barrier. J. Endod. 2019, 45, 701–705.
- Aguilar, F.G.; Garcia, L.F.R.; Pires-De-Souza, F.C.P. Biocompatibility of New Calcium Aluminate Cement (EndoBinder). J. Endod. 2012, 38, 367–371.
- Parker, K.; Sharp, J. Refractory calcium aluminate cements. Rev. Pap. 1982.
- Jacobovitz, M.; Vianna, M.E.; Pandolfelli, V.C.; Oliveira, I.R.; Rossetto, H.L.; Gomes, B.P. Root canal filling with cements based on mineral aggregates: An in vitro analysis of bacterial microleakage. Oral Surg. Oral Med. Oral Pathol. Oral Radiol. Endodontol. 2009, 108, 140–144.
- Luo, Z.; Li, D.; Kohli, M.R.; Yu, Q.; Kim, S.; He, W.-X. Effect of Biodentine™ on the proliferation, migration and adhesion of human dental pulp stem cells. J. Dent. 2014, 42, 490–497.
- Chang, S.W.; Lee, S.Y.; Ann, H.J.; Kum, K.Y.; Kim, E.C. Efects of calcium silicate endodontic cements on biocompatibility and mineralization-inducing potentials in human dental pulp cells. J. Endod. 2014, 40, 1194–1200.
- Dawood, A.; Manton, D.; Parashos, P.; Wong, R.; Palamara, J.; Stanton, D.; Reynolds, E. The physical properties and ion release of CPP-ACP-modified calcium silicate-based cements. Aust. Dent. J. 2015, 60, 434–444.
- Gandolfi, M.G.; Siboni, F.; Botero, T.; Bossù, M.; Riccitiello, F.; Prati, C. Calcium silicate and calcium hydroxide materials for pulp capping: Biointeractivity, porosity, solubility and bioactivity of current formulations. J. Appl. Biomater. Funct. Mater. 2015, 13, 43–60.
- Grech, L.; Mallia, B.; Camilleri, J. Investigation of the physical properties of tricalcium silicate cement-based root-end filling materials. Dent. Mater. 2013, 29, e20–e28.
- Camilleri, J.; Grech, L.; Galea, K.; Keir, D.; Fenech, M.; Formosa, L.; Damidot, D.; Mallia, B. Porosity and root dentine to material interface assessment of calcium silicate-based root-end flling materials. Clin. Oral Investig. 2014, 18, 1437–1446.
- Caron, G.; Azérad, J.; Faure, M.-O.; Machtou, P.; Boucher, Y. Use of a new retrograde filling material (Biodentine) for endodontic surgery: Two case reports. Int. J. Oral Sci. 2014, 6, 250–253.
- Bhavana, V.; Chaitanya, K.P.; Dola, B.; Gandi, P.; Patil, J.; Reddy, R.B. Evaluation of antibacterial and antifungal activity of new calcium-based cement (Biodentine) compared to MTA and glass ionomer cement. J. Conserv. Dent. 2015, 18, 44–46.
- Tomás-Catalá, C.J.; Collado-González, M.; García-Bernal, D.; Onate-Sánchez, R.E.; Forner, L.; Llena, C.; Lozano, A.; Moraleda, J.M.; Rodríguez-Lozano, F.J. Biocompatibility of new pulp-capping materials NeoMTA Plus, MTA Repair HP, and Biodentine on human dental pulp stem cells. J. Endod. 2018, 44, 126–132.
- Cuadros-Fernández, C.; Rodríguez, A.I.L.; Sáez-Martínez, S.; García-Binimelis, J.; About, I.; Mercade, M. Short-term treatment outcome of pulpotomies in primary molars using mineral trioxide aggregate and Biodentine: A randomized clinical trial. Clin. Oral Investig. 2016, 20, 1639–1645.
- Rajasekharan, S.; Martens, L.; Vandenbulcke, J.; Jacquet, W.; Bottenberg, P.; Cauwels, R.G.E.C. Efcacy of three diferent pulpotomy agents in primary molars—A randomised control trial. Int. Endod. J. 2016, 50, 215–228.
- Topçuoğlu, G.; Topçuoğlu, H.S. Regenerative endodontic therapy in a single visit using platelet-rich plasma and Biodentine in necrotic and asymptomatic immature molar teeth: A report of 3 cases. J. Endod. 2016, 42, 1344–1346.
- Pandolfelli, V.C.; Oliveira, I.R.; Rosseto, H.L.; Jacobovitz, M. A Composition Based on Aluminate Cement for Application in Endodontics and the Obtained Cement Product. Patent Registration. INPI 0704502-6, 2007.
- Garcia, L.F.R.; Aguilar, F.G.; Sabino, M.G.; Rossetto, H.L.; Pires-De-Souza, F. Mechanical and microstructural characterisation of new calcium aluminate cement (EndoBinder). Adv. Appl. Ceram. 2011, 110, 469–475.
- Oliveira, I.R.; Pandolfelli, V.C.; Jacobovitz, M. Chemical, physical and mechanical properties of a novel calcium aluminate endodontic cement. Int. Endod. J. 2010, 43, 1069–1076.
- Borkar, S.A.; Ataide, I. Biodentine pulpotomy several days after pulp exposure: Four case reports. J. Conserv. Dent. 2015, 18, 73–78.
- Gandolfi, M.; Siboni, F.; Prati, C. Chemical–physical properties of theracal, a novel light-curable mta-like material for pulp capping. Int. Endod. J. 2012, 45, 571–579.
- Lee, B.-N.; Chang, H.-S.; Hwang, Y.-C.; Hwang, I.-N.; Oh, W.-M. Effects of a novel light-curable material on odontoblastic differentiation of human dental pulp cells. Int. Endod. J. 2016, 50, 464–471.
- Camilleri, J. Hydration characteristics of Biodentine and Theracal used as pulp capping materials. Dent. Mater. 2014, 30, 709–715.
- Camilleri, J.; Laurent, P.; About, I. Hydration of biodentine, theracal LC, and a prototype tricalcium silicate-based dentin replacement material after pulp capping in entire tooth cultures. J. Endod. 2014, 40, 1846–1854.
- Yamamoto, S.; Han, L.; Noiri, Y.; Okiji, T. Evaluation of the Ca ion release, pH and surface apatite formation of International Journal of Dentistry 5 a prototype tricalcium silicate cement. Int. Endod. J. 2017, 50, 1–10.
- Jaffe, W.L.; Scott, D.F. Current Concepts Review—Total Hip Arthroplasty with Hydroxyapatite-Coated Prostheses*. J. Bone Jt. Surg. 1996, 78, 1918–1934.
- Moursi, A.M.; Winnard, A.V.; Winnard, P.L.; Lannutti, J.J.; Seghi, R.R. Enhanced osteoblast response to a polymethylmethacrylate–hydroxyapatite composite. Biomaterials 2002, 23, 133–144.
- Yang, J.Z.; Sultana, R.; Hu, X.Z.; Ichim, P. Novel Layered Hydroxyapatite/Tri-Calcium Phosphate–Zirconia Scaffold Composite with High Bending Strength for Load-Bearing Bone Implant Application. Int. J. Appl. Ceram. 2014, 11, 22–30.
- Sakkers, R.J.B.; Dalmeyer, R.A.J.; Brand, R.; Rozing, P.M.; van Blitterswijk, C. Assessment of bioactivity for orthopedic coatings in a gap-healing model. J. Biomed. Mater. Res. 1997, 36, 265–273.
- Roeder, R.K.; Converse, G.; Kane, R.J.; Yue, W. Hydroxyapatite-reinforced polymer biocomposites for synthetic bone substitutes. Jom 2008, 60, 38–45.
- Mendelson, B.C.; Jacobson, S.R.; Lavoipierre, A.M.; Huggins, R.J. The fate of porous hydroxyapatite granules used in facial skeletal augmentation. Aesthetic Plast. Surg. 2010, 34, 455–461.
- Dorozhkin, S.V. Calcium orthophosphates. J. Mater. Sci. 2007, 42, 1061–1095.
- Dorozhkin, S.V.; Epple, M. Biological and medical significance of calcium phosphates. Angew. Chem. Int. Ed. Engl. 2002, 41, 3130–3146.
- Martz, E.O.; Goel, V.K.; Pope, M.H.; Park, J.B. Materials and design of spinal implants—A review. J. Biomed. Mater. Res. 1997, 38, 267–288.
- Choi, J.-W.; Kong, Y.-M.; Kim, H.-E.; Lee, I.-S. Reinforcement of Hydroxyapatite Bioceramic by Addition of Ni3Al and Al2O3. J. Am. Ceram. Soc. 1998, 81, 1743–1748.
- Baino, F.; Hamzehlou, S.; Kargozar, S. Bioactive glasses: Where are we and where are we going? J. Func. Biomater. 2018, 9, 25.
- Zhang, D.; Leppäranta, O.; Munukka, E.; Ylänen, H.; Viljanen, M.K.; Eerola, E.; Hupa, M.; Hupa, L. Antibacterial effects and dissolution behavior of six bioactive glasses. J. Biomed. Mater. Res. Part A 2010, 93, 475–483.
- Gong, W.; Huang, Z.; Dong, Y.; Gan, Y.; Li, S.; Gao, X.; Chen, X. Ionic Extraction of a Novel Nano-sized Bioactive Glass Enhances Differentiation and Mineralization of Human Dental Pulp Cells. J. Endod. 2014, 40, 83–88.
- Wang, S.; Gao, X.; Gong, W.; Zhang, Z.; Chen, X.; Dong, Y. Odontogenic differentiation and dentin formation of dental pulp cells under nanobioactive glass induction. Acta Biomater. 2014, 10, 2792–2803.
- Schepers, E.; Ducheyne, P.; Barbier, L. Long term clinical evaluation of bioactive glass particles of narrow size range. Bioceramics 1996, 9, 99–102.
- Hench, L.L. Bioactive materials: The potential for tissue regeneration. J. Biomed. Mater. Res. 1998, 41, 511–518.
- Hilton, T.J. Keys to Clinical Success with Pulp Capping: A Review of the Literature. Oper. Dent. 2009, 34, 615–625.
- Macwan, C.S.; Deshpande, A. Mineral trioxide aggregate (MTA) in dentistry: A review of literature. J. Oral Res. Rev. 2014, 6, 71.
- Gholami, S.; Labbaf, S.; Houreh, A.B.; Ting, H.-K.; Jones, J.R.; Esfahani, M.-H.N. Long term effects of bioactive glass particulates on dental pulp stem cells in vitro. Biomed. Glas. 2017, 3, 96–103.
- Mukhtar-Fayyad, D. Cytocompatibility of new bioceramic-based materials on human fibroblast cells (MRC-5). Oral Surg. Oral Med. Oral Pathol. Oral Radiol. Endodontol. 2011, 112, e137–e142.
- Park, J.-W.; Hong, S.-H.; Kim, J.-H.; Lee, S.-J.; Shin, S.-J. X-ray diffraction analysis of White ProRoot MTA and Diadent BioAggregate. Oral Surg. Oral Med. Oral Pathol. Oral Radiol. Endodontol. 2010, 109, 155–158.
- Zhang, S.; Yang, X.; Fan, M. BioAggregate and iRoot BP Plus optimize the proliferation and mineralization ability of human dental pulp cells. Int. Endod. J. 2013, 46, 923–929.
- Dawood, A.E.; Parashos, P.; Wong, R.H.; Reynolds, E.C.; Manton, D.J. Calcium silicate-based cements: Composition, properties, and clinical applications. J. Investig. Clin. Dent. 2017, 8, 12195.
- Yan, P.; Yuan, Z.; Jiang, H.; Peng, B.; Bian, Z. Effect of bioaggregate on differentiation of human periodontal ligament fibroblasts. Int. Endod. J. 2010, 43, 1116–1121.
- Yuan, Z.; Peng, B.; Jiang, H.; Bian, Z.; Yan, P. Effect of Bioaggregate on Mineral-associated Gene Expression in Osteoblast Cells. J. Endod. 2010, 36, 1145–1148.
- De-Deus, G.; Canabarro, A.; Alves, G.; Linhares, A.; Senne, M.I.; Granjeiro, J.M. Optimal Cytocompatibility of a Bioceramic Nanoparticulate Cement in Primary Human Mesenchymal Cells. J. Endod. 2009, 35, 1387–1390.
- Memiş Özgül, B.; Bezgin, T.; Şahin, C.; Sarı, Ş. Resistance to leakage of various thicknesses of apical plugs of Bioaggregate using liquid filtration model. Dent. Traumatol. 2015, 31, 250–254.
- Leal, F.; De-Deus, G.; Brandão, C.; Luna, A.; Fidel, S.R.; Souza, E.M. Comparison of the root-end seal provided by bioceramic repair cements and White MTA. Int. Endod. J. 2011, 44, 662–668.
- Madfa, A.A.; Al-Sanabani, F.A.; Al-Kudami, N.H.A.-Q. Endodontic Repair Filling Materials: A Review Article. Br. J. Med. Med Res. 2014, 4, 3059–3079.
- Tuloglu, N.; Bayrak, S. Comparative evaluation of mineral trioxide aggregate and bioaggregate as apical barrier material in traumatized nonvital, immature teeth: A clinical pilot study. Niger. J. Clin. Pract. 2016, 19, 52–57.
- American Association of Endodontists. Glossary of Endodontic Terms; American Association of Endodontists: New York, NY, USA, 2003.
- Jew, R.C.; Weine, F.S.; Keene, J.J., Jr.; Smulson, M.H. A histologic evaluation of periodontal tissues adjacent to root perforations filled with Cavit. Oral Surg. Oral Med. Oral Pathol. Oral Radiol. 1982, 54, 124–135.
- Sinai, I.H. Endodontic perforations: Their prognosis and treatment. J. Am. Dent. Assoc. 1977, 95, 90–95.
- Fuss, Z.; Tsesis, I.; Lin, S. Root resorption—diagnosis, classification and treatment choices based on stimulation factors. Dent. Traumatol. 2003, 19, 175–182.
- Ingle, J.I. A standardized endodontic technique utilizing newly designed instruments and filling materials. Oral Surg. Oral Med. Oral Pathol. Oral Radiol. 1961, 14, 83–91.
- Kerekes, K.S.; Tronstad, L. Long-term results of endodontic treatment performed with a standardized technique. J. Endod. 1979, 5, 83–90.
- Farzaneh, M.; Abitbol, S.; Friedman, S. Treatment Outcome in Endodontics: The Toronto Study. Phases I and II: Orthograde Retreatment. J. Endod. 2004, 30, 627–633.
- Fagogeni, I.; Metlerska, J.; Lipski, M.; Falgowski, T.; Maciej, G.; Nowicka, A. Materials used in regenerative endodontic procedures and their impact on tooth discoloration. J. Oral Sci. 2019, 61, 379–385.
- Giacomino, C.M.; Wealleans, J.A.; Kuhn, N.; Diogenes, A. The antimicrobial effect of bioceramic sealer on an 8-week matured Enterococcus faecalis biofilm attached to root canal dentinal surface. J. Endod. 2019, 45, 51–56.
- Tomás-Catalá, C.J.; Collado-González, M.; García-Bernal, D.; Oñate-Sánchez, R.E.; Forner, L.; Llena, C.; Lozano, A.; Castelo-Baz, P.; Moraleda, J.M.; Rodríguez-Lozano, F.J. Comparative analysis of the biological effects of the endodontic bioactive cements MTA-Angelus, MTA Repair HP and NeoMTA Plus on human dental pulp stem cells. Int. Endod. J. 2017, 50, e63–e72.
- Donnermeyer, D.; Bürklein, S.; Dammaschke, T.; Schäfer, E. Endodontic sealers based on calcium silicates: A systematic review. Odontology 2019, 107, 421–436.
- Linu, S.; Lekshmi, M.; Varunkumar, V.; Joseph, V.S. Treatment Outcome Following Direct Pulp Capping Using Bioceramic Materials in Mature Permanent Teeth with Carious Exposure: A Pilot Retrospective Study. J. Endod. 2017, 43, 1635–1639.
- Liu, S.; Wang, S.; Dong, Y. Evaluation of a Bioceramic as a Pulp Capping Agent In Vitro and In Vivo. J. Endod. 2015, 41, 652–657.
- Al–Saudi, K.W.; Nabih, S.M.; Farghaly, A.M.; AboHager, E.A.-A. Pulpal repair after direct pulp capping with new bioceramic materials: A comparative histological study. Saudi Dent. J. 2019, 31, 469–475.
- Palma, P.J.; Ramos, J.C.; Martins, J.B.; Diogenes, A.; Figueiredo, M.H.; Ferreira, P.; Viegas, C.; Santos, J.M. Histologic Evaluation of Regenerative Endodontic Procedures with the Use of Chitosan Scaffolds in Immature Dog Teeth with Apical Periodontitis. J. Endod. 2017, 43, 1279–1287.
- Hess, D.; Solomon, E.; Spears, R.; He, J. Retreatability of a Bioceramic Root Canal Sealing Material. J. Endod. 2011, 37, 1547–1549.
- de Miranda Candeiro, G.T.; Correia, F.C.; Duarte, M.A.H.; Ribeiro-Siqueira, D.C.; Gavini, G. Evaluation of radiopacity, pH, release of calcium ions, and flow of a bioceramic root canal sealer. J. Endod. 2012, 38, 842–845.
- Rodríguez-Lozano, F.J.; López-García, S.; García-Bernal, D.; Tomás-Catalá, C.J.; Santos, J.M.; Llena, C.; Lozano, A.; Murcia, L.; Forner, L. Chemical composition and bioactivity potential of the new Endosequence BC Sealer formulation HiFlow. Int. Endod. J. 2020, 53, 1216–1228.
- Carvalho, C.N.; Wang, Z.; Shen, Y.; Gavini, G.; Martinelli, J.R.; Manso, A.; Haapasalo, M. Comparative analyses of ion release, pH and multispecies biofilm formation between conventional and bioactive gutta-percha. Int. Endod. J. 2016, 49, 1048–1056.
- Wolfson, E.M.; Seltzer, S. Reaction of rat connective tissue to some gutta-percha formulations. J. Endod. 1975, 1, 395–402.
- Sjögren, U.; Sundqvist, G.; Nair, P.R. Tissue reaction to gutta-percha particles of various sizes when implanted subcutaneously in guinea pigs. Eur. J. Oral Sci. 1995, 103, 313–321.
- Jitaru, S.; Hodisan, I.; Timis, L.; Lucian, A.; Bud, M. the use of bioceramics in endodontics—literature review. Med. Pharm. Rep. 2016, 89, 470–473.
- Zeichner-David, M. Is there more to enamel matrix proteins than biomineralization? Matrix Biol. 2001, 20, 307–316.
- Hoang, A.; Oates, T.; Cochran, D. In Vitro Wound Healing Responses to Enamel Matrix Derivative. J. Periodontol. 2000, 71, 1270–1277.
- Schwartz, Z.; Carnes, D.L., Jr.; Pulliam, R.; Lohmann, C.H.; Sylvia, V.L.; Liu, Y.; Dean, D.D.; Cochran, D.L.; Boyan, B.D. Porcine fetal enamel matrix derivative stimulates proliferation but not differentiation of pre-osteoblastic 2T9 cells, inhibits proliferation and stimulates differentiation of osteoblast-like MG63 cells, and increases proliferation and differentiation of normal human osteoblast NHOst cells. J. Periodontol. 2000, 71, 1287–1296.
- Sculean, A.; Kiss, A.; Miliauskaite, A.; Schwarz, F.; Arweiler, N.B.; Hannig, M. Ten-year results following treatment of intra-bony defects with enamel matrix proteins and guided tissue regeneration. J. Clin. Periodontol. 2008, 35, 817–824.
- Van der Pauw, M.T.; Van den Bos, T.; Everts, V.; Beertsen, W. Enamel matrix-derived protein stimulates attachment of periodontal ligament fibroblasts and enhances alkaline phosphatase activity and transforming growth factor β1 release of periodontal ligament and gingival fibroblasts. J. Periodontol. 2000, 71, 31–43.
- Al-Hezaimi, K.; Naghshbandi, J.; Simon, J.H.S.; Oglesby, S.; Rotstein, I. Successful treatment of a radicular groove by intentional replantation and Emdogain® therapy. Dent. Traumatol. 2004, 20, 226–228.
- Azim, A.A.; Lloyd, A.; Huang, G.T.-J. Management of Longstanding Furcation Perforation Using a Novel Approach. J. Endod. 2014, 40, 1255–1259.
- Kerezoudis, N.P.; Siskos, G.J.; Tsatsas, V. Bilateral buccal radicular groove in maxillary incisors: Case report. Int. Endod. J. 2003, 36, 898–906.
- Panzarini, S.R.; Gulinelli, J.L.; Poi, W.R.; Sonoda, C.K.; Pedrini, D.; Brandini, D.A. Treatment of root surface in delayed tooth replantation: A review of literature. Dent. Traumatol. 2008, 24, 277–282.
- Liu, G.; Xu, G.; Gao, Z.; Liu, Z.; Xu, J.; Wang, J.; Zhang, C.; Wang, S. Demineralized Dentin Matrix Induces Odontoblastic Differentiation of Dental Pulp Stem Cells. Cells Tissues Organs 2016, 201, 65–76.
- Goldberg, M.; Smith, A.J. Cells and Extracellular Matrices of Dentin and Pulp: A Biological Basis for Repair and Tissue Engineering. Crit. Rev. Oral Biol. Med. 2004, 15, 13–27.
- Murata, M.; Sato, D.; Hino, J.; Akazawa, T.; Tazaki, J.; Ito, K.; Arisue, M. Acid-insoluble human dentin as carrier material for recombinant human BMP-2. J. Biomed. Mater. Res. Part A 2012, 100, 571–577.
- Guo, W.; He, Y.; Zhang, X.; Lu, W.; Wang, C.; Yu, H.; Liu, Y.; Li, Y.; Zhou, Y.; Zhou, J.; et al. The use of dentin matrix scaffold and dental follicle cells for dentin regeneration. Biomaterials 2009, 30, 6708–6723.
- Urist, M.R.; Silverman, B.F.; Büring, K.; Dubuc, F.L.; Rosenberg, J.M. The bone induction principle. Clin. Orthop. Relat. Res. 1967, 53, 243–284.
- Gomes, M.F.; Destro, M.F.D.S.S.; Banzi, É.C.D.F.; Vieira, E.M.M.; Morosolli, A.R.C.; Goulart, M.D.G.V. Optical density of bone repair after implantation of homogenous demineralized dentin matrix in diabetic rabbits. Braz. Oral Res. 2008, 22, 2275–2280.
- Bakhshalian, N.; Hooshmand, S.; Campbell, S.C.; Kim, J.-S.; Brummel-Smith, K.; Arjmandi, B.H. Biocompatibility and Microstructural Analysis of Osteopromotive Property of Allogenic Demineralized Dentin Matrix. Int. J. Oral Maxillofac. Implant. 2013, 28, 1655–1662.
- De Oliveira, G.; Miziara, M.; da Silva, E.; Ferreira, E.; Biulchi, A.; Alves, J. Enhanced bone formation during healing process of tooth sockets filled with demineralized human dentine matrix. Aust. Dent. J. 2013, 58, 326–332.
- Epple, M.; Ganesan, K.; Heumann, R.; Klesing, J.; Kovtun, A.; Neumann, S.; Sokolova, V.J.J.C. Application of calcium phosphate nanoparticles in biomedicine. J. Mater. Chem. 2010, 20, 18–23.
- Hench, L.L. An Introduction to Bioceramics; World Scientific: Singapore, 1993.
- Legeros, R. Calcium Phosphate Materials in Restorative Dentistry: A Review. Adv. Dent. Res. 1988, 2, 164–180.
- Hench, L.L. Bioceramics: From Concept to Clinic. J. Am. Ceram. Soc. 1991, 74, 1487–1510.
- Thamaraiselvi, T.; Rajeswari, S. Biological evaluation of bioceramic materials-a review. Carbon 2004, 24, 172.
- Osborn, J.; Newesely, H. The material science of calcium phosphate ceramics. Biomaterials 1980, 1, 108–111.
- Brown, W. A new calcium phosphate, Water-setting cement. Cem. Concr. Res. 1986, 352–379.
- Costantino, P.D.; Friedman, C.D. Synthetic bone graft substitutes. Otolaryngol. Clin. N. Am. 1994, 27, 354–361.
- Sugawara, A.; Chow, L.C.; Takagi, S.; Chohayeb, H. In vitro evaluation of the sealing ability of a calcium phosphate cement when used as a root canal sealer-filler. J. Endod. 1990, 16, 162–165.
- Bilginer, S.; Esener, I.T.; Söylemezoğlu, F.; Tiftik, A.M. The investigation of biocompatibility and apical microleakage of tricalcium phosphate based root canal sealers. J. Endod. 1997, 23, 105–109.





