
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Michel Cogné | -- | 2240 | 2023-01-30 09:40:26 | | | |
| 2 | Conner Chen | + 8 word(s) | 2248 | 2023-01-30 10:33:21 | | | | |
| 3 | Conner Chen | + 1 word(s) | 2249 | 2023-01-30 10:34:24 | | | | |
| 4 | Conner Chen | + 14 word(s) | 2263 | 2023-01-31 07:50:25 | | | | |
| 5 | Conner Chen | + 2 word(s) | 2265 | 2023-01-31 09:43:19 | | |
Video Upload Options
Mature B cells notably diversify immunoglobulin (Ig) production through class switch recombination (CSR), allowing the junction of distant “switch” (S) regions. CSR is initiated by activation-induced deaminase (AID), which targets cytosines adequately exposed within single-stranded DNA of transcribed targeted S regions, with a specific affinity for WRCY motifs. In mammalian S regions, abundant G4 (G-quadruplex) DNA on the non-template strand also contributes to the formation of R-loops while the presence of G4 structures within the primary transcripts from S regions participates into recruiting AID. The ability of G4 ligands to modulate the CSR process also underlines the key role of G4 structures in the regulation of CSR.
1. Role of G4-DNA at Transcribed S Regions and in the Resolution of G4s/R-Loops Conflicts
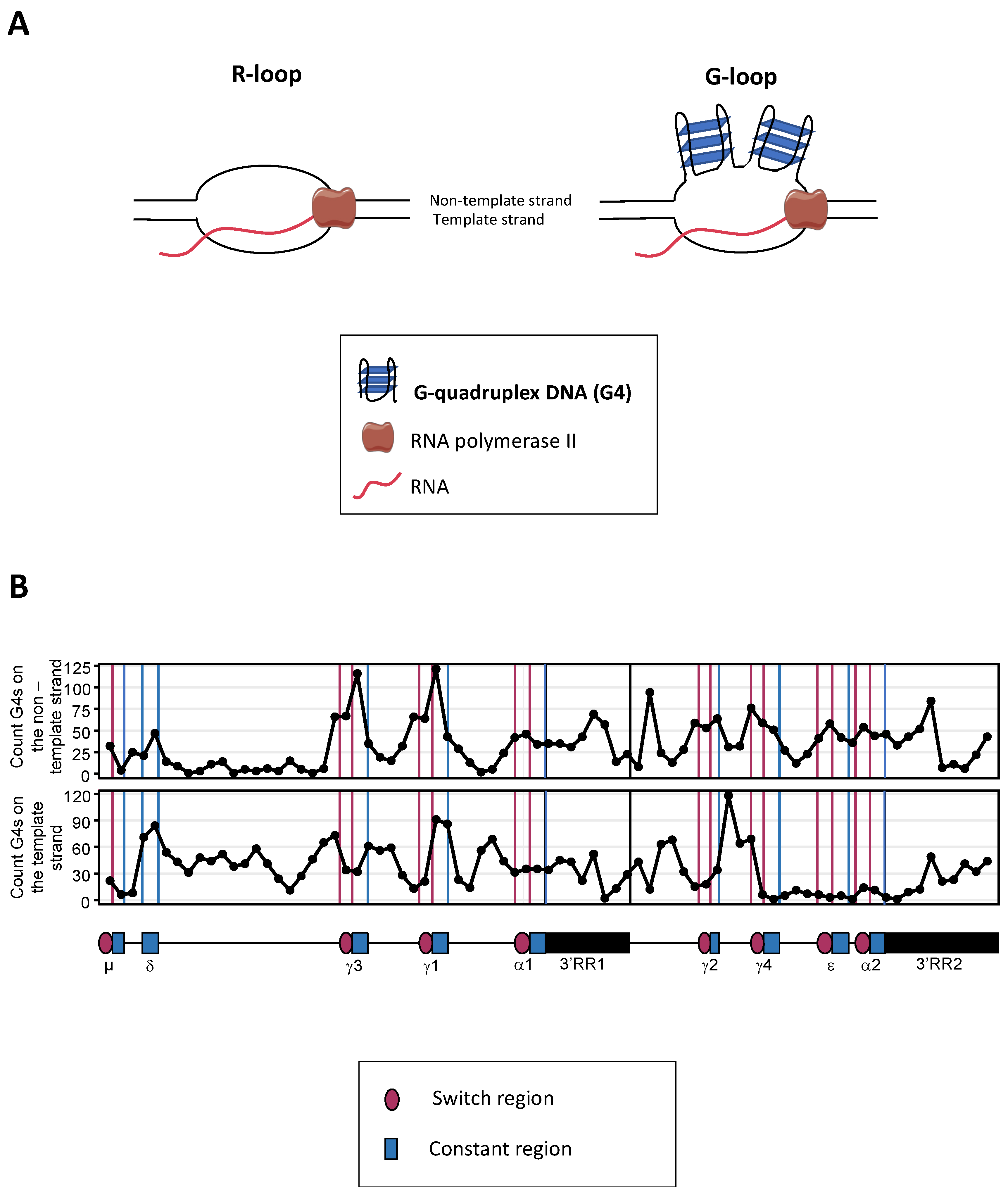
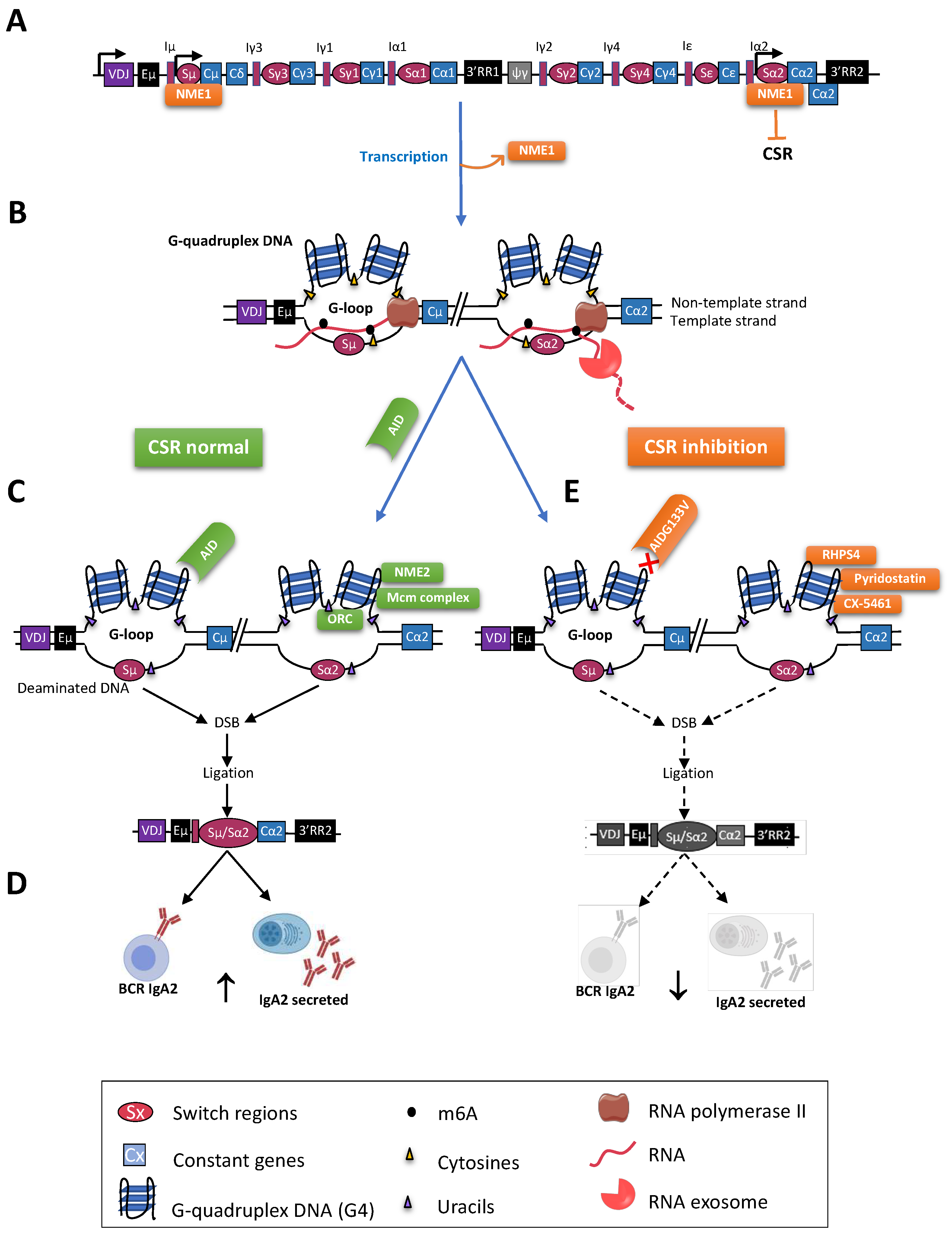
2. Role of G4-RNA Structures within S Region Transcripts
3. Connections between CSR and DNA Replication
4. A Role of G4-DNA in IgH Locus High-Dimensional Organization
References
- Williamson, J.R.; Raghuraman, M.K.; Cech, T.R. Monovalent cation-induced structure of telomeric DNA: The G-quartet model. Cell 1989, 59, 871–880.
- Dalloul, Z.; Chenuet, P.; Dalloul, I.; Boyer, F.; Aldigier, J.-C.; Laffleur, B.; El Makhour, Y.; Ryffel, B.; Quesniaux, V.F.; Togbé, D.; et al. G-quadruplex DNA targeting alters class-switch recombination in B cells and attenuates allergic inflammation. J. Allergy Clin. Immunol. 2018, 142, 1352–1355.
- Qiao, Q.; Wang, L.; Meng, F.-L.; Hwang, J.K.; Alt, F.W.; Wu, H. AID Recognizes Structured DNA for Class Switch Recombination. Mol. Cell 2017, 67, 361–373.
- Vallur, A.C.; Maizels, N. Activities of human exonuclease 1 that promote cleavage of transcribed immunoglobulin switch regions. Proc. Natl. Acad. Sci. USA 2008, 105, 16508–16512.
- Collier, D.A.; Griffin, J.A.; Wells, R.D. Non-B right-handed DNA conformations of homopurine.homopyrimidine sequences in the murine immunoglobulin C alpha switch region. J. Biol. Chem. 1988, 263, 7397–7405.
- Sen, D.; Gilbert, W. Formation of parallel four-stranded complexes by guanine-rich motifs in DNA and its implications for meiosis. Nature 1988, 334, 364–366.
- Chi, X.; Li, Y.; Qiu, X. V(D)J recombination, somatic hypermutation and class switch recombination of immunoglobulins: Mechanism and regulation. Immunology 2020, 160, 233–247.
- Abbott, R.K.; Thayer, M.; Labuda, J.; Silva, M.; Philbrook, P.; Cain, D.W.; Kojima, H.; Hatfield, S.; Sethumadhavan, S.; Ohta, A.; et al. Germinal Center Hypoxia Potentiates Immunoglobulin Class Switch Recombination. J. Immunol. 2016, 197, 4014–4020.
- Cho, S.H.; Raybuck, A.L.; Stengel, K.; Wei, M.; Beck, T.C.; Volanakis, E.; Thomas, J.W.; Hiebert, S.; Haase, V.H.; Boothby, M.R. Germinal centre hypoxia and regulation of antibody qualities by a hypoxia response system. Nature 2016, 537, 234–238.
- Matthews, A.J.; Zheng, S.; DiMenna, L.J.; Chaudhuri, J. Chapter One-Regulation of Immunoglobulin Class-Switch Recombination: Choreography of Noncoding Transcription, Targeted DNA Deamination, and Long-Range DNA Repair. Adv. Immunol. 2014, 122, 1–57.
- Nair, L.; Zhang, W.; Laffleur, B.; Jha, M.K.; Lim, J.; Lee, H.; Wu, L.; Alvarez, N.S.; Liu, Z.-P.; Munteanu, E.L.; et al. Mechanism of noncoding RNA-associated N6-methyladenosine recognition by an RNA processing complex during IgH DNA recombination. Mol. Cell 2021, 81, 3949–3964.
- Shinkura, R.; Tian, M.; Smith, M.; Chua, K.; Fujiwara, Y.; Alt, F.W. The influence of transcriptional orientation on endogenous switch region function. Nat. Immunol. 2003, 4, 435–441.
- Tashiro, J.; Kinoshita, K.; Honjo, T. Palindromic but not G-rich sequences are targets of class switch recombination. Int. Immunol. 2001, 13, 495–505.
- Zheng, S.; Kusnadi, A.; Choi, J.E.; Vuong, B.Q.; Rhodes, D.; Chaudhuri, J. NME proteins regulate class switch recombination. FEBS Lett. 2019, 593, 80–87.
- Guo, J.U.; Bartel, D.P. RNA G-quadruplexes are globally unfolded in eukaryotic cells and depleted in bacteria. Science 2016, 353, aaf5371.
- Kwok, C.K.; Marsico, G.; Sahakyan, A.B.; Chambers, V.S.; Balasubramanian, S. rG4-seq reveals widespread formation of G-quadruplex structures in the human transcriptome. Nat. Methods 2016, 13, 841–844.
- Yang, S.Y.; Monchaud, D.; Wong, J.M.Y. Global mapping of RNA G-quadruplexes (G4-RNAs) using G4RP-seq. Nat. Protoc. 2022, 17, 870–889.
- Yewdell, W.T.; Chaudhuri, J. A transcriptional serenAID: The role of noncoding RNAs in class switch recombination. Int. Immunol. 2017, 29, 183–196.
- Duquette, M.L.; Handa, P.; Vincent, J.A.; Taylor, A.F.; Maizels, N. Intracellular transcription of G-rich DNAs induces formation of G-loops, novel structures containing G4 DNA. Genes Dev. 2004, 18, 1618–1629.
- Fleming, A.M.; Nguyen, N.L.B.; Burrows, C.J. Colocalization of m6A and G-Quadruplex-Forming Sequences in Viral RNA (HIV, Zika, Hepatitis B, and SV40) Suggests Topological Control of Adenosine N6-Methylation. ACS Central Sci. 2019, 5, 218–228.
- Iwasaki, Y.; Ookuro, Y.; Iida, K.; Nagasawa, K.; Yoshida, W. Destabilization of DNA and RNA G-quadruplex structures formed by GGA repeat due to N6-methyladenine modification. Biochem. Biophys. Res. Commun. 2022, 597, 134–139.
- Czerniak, T.; Saenz, J.P. Lipid membranes modulate the activity of RNA through sequence-dependent interactions. Proc. Natl. Acad. Sci. USA 2022, 119, e2119235119.
- Zheng, S.; Vuong, B.Q.; Vaidyanathan, B.; Lin, J.-Y.; Huang, F.-T.; Chaudhuri, J. Non-coding RNA Generated following Lariat Debranching Mediates Targeting of AID to DNA. Cell 2015, 161, 762–773.
- De Almeida, C.R.; Dhir, S.; Dhir, A.; Moghaddam, A.E.; Sattentau, Q.; Meinhart, A.; Proudfoot, N.J. RNA Helicase DDX1 Converts RNA G-Quadruplex Structures into R-Loops to Promote IgH Class Switch Recombination. Mol. Cell 2018, 70, 650–662.
- Schrader, C.E.; Guikema, J.E.J.; Linehan, E.K.; Selsing, E.; Stavnezer, J. Activation-Induced Cytidine Deaminase-Dependent DNA Breaks in Class Switch Recombination Occur during G1 Phase of the Cell Cycle and Depend upon Mismatch Repair. J. Immunol. 2007, 179, 6064–6071.
- Wiedemann, E.-M.; Peycheva, M.; Pavri, R. DNA Replication Origins in Immunoglobulin Switch Regions Regulate Class Switch Recombination in an R-Loop-Dependent Manner. Cell Rep. 2016, 17, 2927–2942.
- Bochman, M.L.; Schwacha, A. The Mcm Complex: Unwinding the Mechanism of a Replicative Helicase. Microbiol. Mol. Biol. Rev. 2009, 73, 652–683.
- Zhang, X.; Zhang, Y.; Ba, Z.; Kyritsis, N.; Casellas, R.; Alt, F.W. Fundamental roles of chromatin loop extrusion in antibody class switching. Nature 2019, 575, 385–389.
- Zhang, Y.; Zhang, X.; Ba, Z.; Liang, Z.; Dring, E.W.; Hu, H.; Lou, J.; Kyritsis, N.; Zurita, J.; Shamim, M.S.; et al. The fundamental role of chromatin loop extrusion in physiological V(D)J recombination. Nature 2019, 573, 600–604.
- Haensel-Hertsch, R.; Beraldi, D.; Lensing, S.V.; Marsico, G.; Zyner, K.; Parry, A.; Di Antonio, M.; Pike, J.; Kimura, H.; Narita, M.; et al. G-quadruplex structures mark human regulatory chromatin. Nat. Genet. 2016, 48, 1267–1272.
- Li, L.; Williams, P.; Ren, W.; Wang, M.Y.; Gao, Z.; Miao, W.; Huang, M.; Song, J.; Wang, Y. YY1 interacts with guanine quadruplexes to regulate DNA looping and gene expression. Nat. Chem. Biol. 2021, 17, 161–168.
- Roach, R.J.; Garavís, M.; González, C.; Jameson, G.B.; Filichev, V.V.; Hale, T.K. Heterochromatin protein 1α interacts with parallel RNA and DNA G-quadruplexes. Nucleic Acids Res. 2020, 48, 682–693.
- Valton, A.-L.; Venev, S.V.; Mair, B.; Khokhar, E.S.; Tong, A.H.Y.; Usaj, M.; Chan, K.; Pai, A.A.; Moffat, J.; Dekker, J. A cohesin traffic pattern genetically linked to gene regulation. Nat. Struct. Mol. Biol. 2022, 29, 1239–1251.
- Laffleur, B.; Lim, J.; Zhang, W.; Chen, Y.; Pefanis, E.; Bizarro, J.; Batista, C.R.; Wu, L.; Economides, A.N.; Wang, J.; et al. Noncoding RNA processing by DIS3 regulates chromosomal architecture and somatic hypermutation in B cells. Nat. Genet. 2021, 53, 230–242.
- Ceschi, S.; Berselli, M.; Cozzaglio, M.; Giantin, M.; Toppo, S.; Spolaore, B.; Sissi, C. Vimentin binds to G-quadruplex repeats found at telomeres and gene promoters. Nucleic Acids Res. 2022, 50, 1370–1381.
- Wuerffel, R.; Wang, L.; Grigera, F.; Manis, J.; Selsing, E.; Perlot, T.; Alt, F.W.; Cogne, M.; Pinaud, E.; Kenter, A.L. S-S Synapsis during Class Switch Recombination Is Promoted by Distantly Located Transcriptional Elements and Activation-Induced Deaminase. Immunity 2007, 27, 711–722.





