
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | ANTELM PUJOL CALAFAT | -- | 1465 | 2023-01-14 17:25:03 | | | |
| 2 | Jessie Wu | Meta information modification | 1465 | 2023-01-16 02:25:53 | | | | |
| 3 | Jessie Wu | + 9 word(s) | 1474 | 2023-01-16 02:29:22 | | |
Video Upload Options
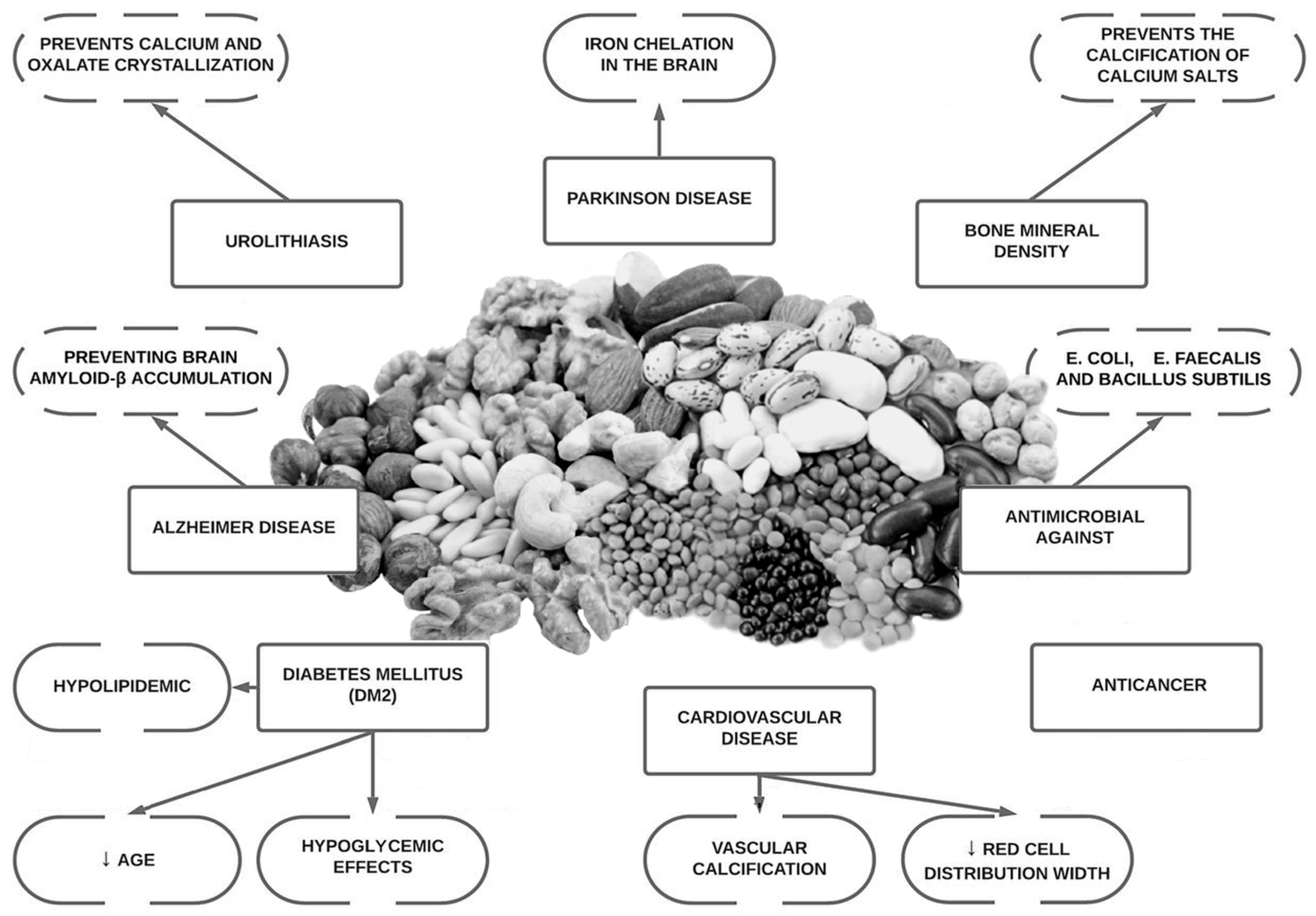
Osteoporosis is a highly prevalent bone disorder especially in post-menopausal women. Lifestyle interventions such as physical activity and nutrition play an important role in the prevention and treatment of bone mineral loss. Phytate (myo-inositol hexakisphosphate or InsP6) is the main phosphorus reservoir that is present in almost all wholegrains, legumes, and oilseeds. It is a major component of the Mediterranean and Dietary Approaches to Stop Hypertension (DASH) diets. Phytate is recognized as a nutraceutical and is classified by the Food and Drug Administration (FDA) as Generally Recognized As Safe (GRAS). Phytate has been shown to be effective in treating or preventing certain diseases. Phytate has been shown to inhibit calcium salt crystallization and, therefore, to reduce vascular calcifications, calcium renal calculi and soft tissue calcifications.
1. Introduction
2. Background and In Vitro Studies
3. Animal Studies
4. Epidemiological Studies
5. Clinical Trials in Humans
References
- Perez-Gregorio, R.; Simal-Gandara, J. A Critical Review of Bioactive Food Components, and of their Functional Mechanisms, Biological Effects and Health Outcomes. Curr. Pharm. Des. 2017, 23, 2731–2741.
- Guasch-Ferré, M.; Willett, W.C. The Mediterranean diet and health: A comprehensive overview. J. Intern. Med. 2021, 290, 549–566.
- Sanchis, P.; Molina, M.; Berga, F.; Muñoz, E.; Fortuny, R.; Costa-Bauzá, A.; Grases, F.; Buades, J.M. A Pilot Randomized Crossover Trial Assessing the Safety and Short-Term Effects of Walnut Consumption by Patients with Chronic Kidney Disease. Nutrients 2019, 12, 63.
- Buades Fuster, J.M.; Sanchís Cortés, P.; Perelló Bestard, J.; Grases Freixedas, F. Plant phosphates, phytate and pathological calcifications in chronic kidney disease. Nefrologia 2017, 37, 20–28.
- Serra-Majem, L.; Bes-Rastrollo, M.; Román-Vinas, B.; Pfrimer, K.; Sánchez-Villegas, A.; Martínez-González, M.A. Dietary patterns and nutritional adequacy in a Mediterranean country. Br. J. Nutr. 2019, 101 (Suppl. S2), S21–S28.
- Demer, L.L.; Tintut, Y. Vascular Calcification: Pathobiology of a multifaceted disease. Circulation 2008, 117, 2938–2948.
- Sanchis, P.; Buades, J.M.; Berga, F.; Gelabert, M.M.; Molina, M.; Íñigo, M.V.; García, S.; Gonzalez, J.; Bernabeu, M.R.; Costa-Bauzá, A.; et al. Protective Effect of Myo-Inositol Hexaphosphate (Phytate) on Abdominal Aortic Calcification in Patients With Chronic Kidney Disease. J. Ren. Nutr. 2016, 26, 226–236.
- Grases, F.; Costa-Bauza, A. Key Aspects of Myo-Inositol Hexaphosphate (Phytate) and Pathological Calcifications. Molecules 2019, 24, 4434.
- Sanchis, P.; López-González, Á.-A.; Costa-Bauzá, A.; Busquets-Cortés, C.; Riutord, P.; Calvo, P.; Grases, F. Understanding the Protective Effect of Phytate in Bone Decalcification Related-Diseases. Nutrients 2021, 13, 2859.
- Drake, M.T.; Clarke, B.L.; Khosla, S. Bisphosphonates: Mechanism of Action and Role in Clinical Practice. Mayo Clin. Proc. Mayo Clin. 2008, 83, 1032–1045.
- Arriero, M.D.M.; Ramis, J.M.; Perelló, J.; Monjo, M. Inositol Hexakisphosphate Inhibits Osteoclastogenesis on RAW 264.7 Cells and Human Primary Osteoclasts. PLoS ONE 2012, 7, e43187.
- Fleisch, H.; Bisaz, S. Isolation from urine of pyrophosphate, a calcification inhibitor. Am. J. Physiol. Content 1962, 203, 671–675.
- Fleish, H.; Neuman, W.F. Mechanisms of calcification: Role of collagen, polyphosphates, and phosphatase. Am. J. Physiol. Content 1961, 200, 1296–1300.
- Fleisch, H.A.; Russell, R.G.G.; Bisaz, S.; Mühlbauer, R.C.; Williams, D.A. The Inhibitory Effect of Phosphonates on the Formation of Calcium Phosphate Crystals in vitro and on Aortic and Kidney Calcification in vivo. Eur. J. Clin. Investig. 1970, 1, 12–18.
- Fleisch, H.; Graham, R.; Russell, G.; Francis, M.D. Diphosphonates Inhibit Hydroxyapatite Dissolution in vitro and Bone Resorption in Tissue Culture and in vivo. Science 1969, 165, 1262–1264.
- Grases, F.; Sanchis, P.; Prieto, R.M.; Perelló, J.; López-González, Á.A. Effect of Tetracalcium Dimagnesium Phytate on Bone Characteristics in Ovariectomized Rats. J. Med. Food 2010, 13, 1301–1306.
- Kim, O.-H.; Booth, C.J.; Choi, H.S.; Lee, J.; Kang, J.; Hur, J.; Jung, W.J.; Jung, Y.-S.; Choi, H.J.; Kim, H.; et al. High-phytate/low-calcium diet is a risk factor for crystal nephropathies, renal phosphate wasting, and bone loss. Elife 2020, 9, e52709.
- Gonzalez, A.A.L.; Grases, F.; Mari, B.; Tomas-Salva, M.; Rodriguez, A. Urinary phytate concentration and risk of fracture determined by the FRAX index in a group of postmenopausal women. Turk. J. Med. Sci. 2019, 49, 458–463.
- López-González, Á.A.; Grases, F.; Monroy, N.; Marí, B.; Vicente-Herrero, M.T.; Tur, F.; Perelló, J. Protective effect of myo-inositol hexaphosphate (phytate) on bone mass loss in postmenopausal women. Eur. J. Nutr. 2012, 52, 717–726.
- López-González, Á.A.; Grases, F.; Roca, P.; Mari, B.; Vicente-Herrero, M.; Costa-Bauzá, A.; Sanchis, P.; Prieto, R.M.; Perelló, J.; Freitas, T.P.; et al. Phytate (myo-Inositol Hexaphosphate) and Risk Factors for Osteoporosis. J. Med. Food 2008, 11, 747–752.
- López-González, Á.A.; Grases, F.; Marí, B.; Vicente-Herrero, M.T.; Costa-Bauzá, A.; Monroy, N. Influencia del consumo de fitato sobre la masa ósea en mujeres posmenopáusicas de Mallorca. Reumatol. Clínica 2011, 7, 220–223.
- Sanchis, P.; Rivera, R.; Berga, F.; Fortuny, R.; Adrover, M.; Costa-Bauza, A.; Grases, F.; Masmiquel, L. Phytate Decreases Formation of Advanced Glycation End-Products in Patients with Type II Diabetes: Randomized Crossover Trial. Sci. Rep. 2018, 8, 9619.
- Prieto, R.M.; Fiol, M.; Perello, J.; Estruch, R.; Ros, E.; Sanchis, P.; Grases, F. Effects of Mediterranean diets with low and high proportions of phytate-rich foods on the urinary phytate excretion. Eur. J. Nutr. 2010, 49, 321–326.
- Rivas, A.; Romero, A.; Mariscal-Arcas, M.; Monteagudo, C.; Feriche, B.; Lorenzo, M.L.; Olea, F. Mediterranean diet and bone mineral density in two age groups of women. Int. J. Food Sci. Nutr. 2012, 64, 155–161.
- Kontogianni, M.D.; Melistas, L.; Yannakoulia, M.; Malagaris, I.; Panagiotakos, D.B.; Yiannakouris, N. Association between dietary patterns and indices of bone mass in a sample of Mediterranean women. Nutrition 2009, 25, 165–171.
- Pérez-Rey, J.; Roncero-Martín, R.; Rico-Martín, S.; Rey-Sánchez, P.; Pedrera-Zamorano, J.D.; Pedrera-Canal, M.; López-Espuela, F.; Lavado-García, J.M. Adherence to a Mediterranean Diet and Bone Mineral Density in Spanish Premenopausal Women. Nutrients 2019, 11, 555.
- Calderon-Garcia, J.F.; Moran, J.M.; Roncero-Martin, R.; Rey-Sanchez, P.; Rodriguez-Velasco, F.J.; Pedrera-Zamorano, J.D. Dietary Habits, Nutrients and Bone Mass in Spanish Premenopausal Women: The Contribution of Fish to Better Bone Health. Nutrients 2012, 5, 10–22.
- Guimerà, J.; Martínez, A.; Bauza, J.L.; Sanchís, P.; Pieras, E.; Grases, F. Effect of phytate on hypercalciuria secondary to bone resorption in patients with urinary stones: Pilot study. Urolithiasis 2022, 50, 685–690.





