
Video Upload Options
Auricularia auricula polysaccharides (AAP) have been widely studied in the field of medicine and healthcare because of their unique structure and physiological activity. Many species of Auricularia auricula polysaccharides have been extracted, isolated, and purified by different methods, and their structures have been analyzed. Auricularia auricula polysaccharides have been proven to have beneficial effects on the human body, including slowing the aging process, controlling the intestinal system, and treating cardiovascular disorders.
1. Introduction
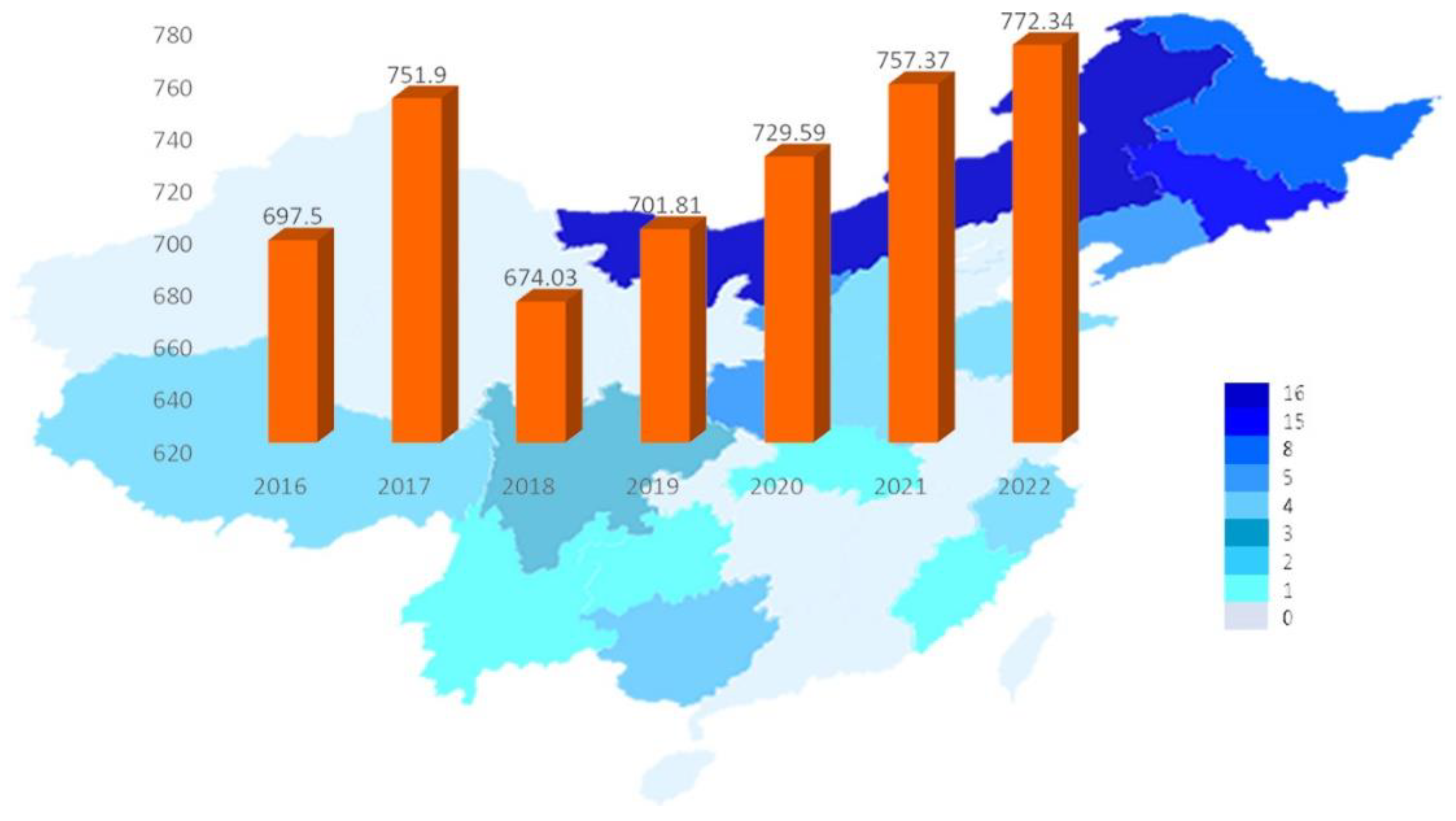
Figure 1. Production and forecast of Auricularia auricula in China from 2016–2022.
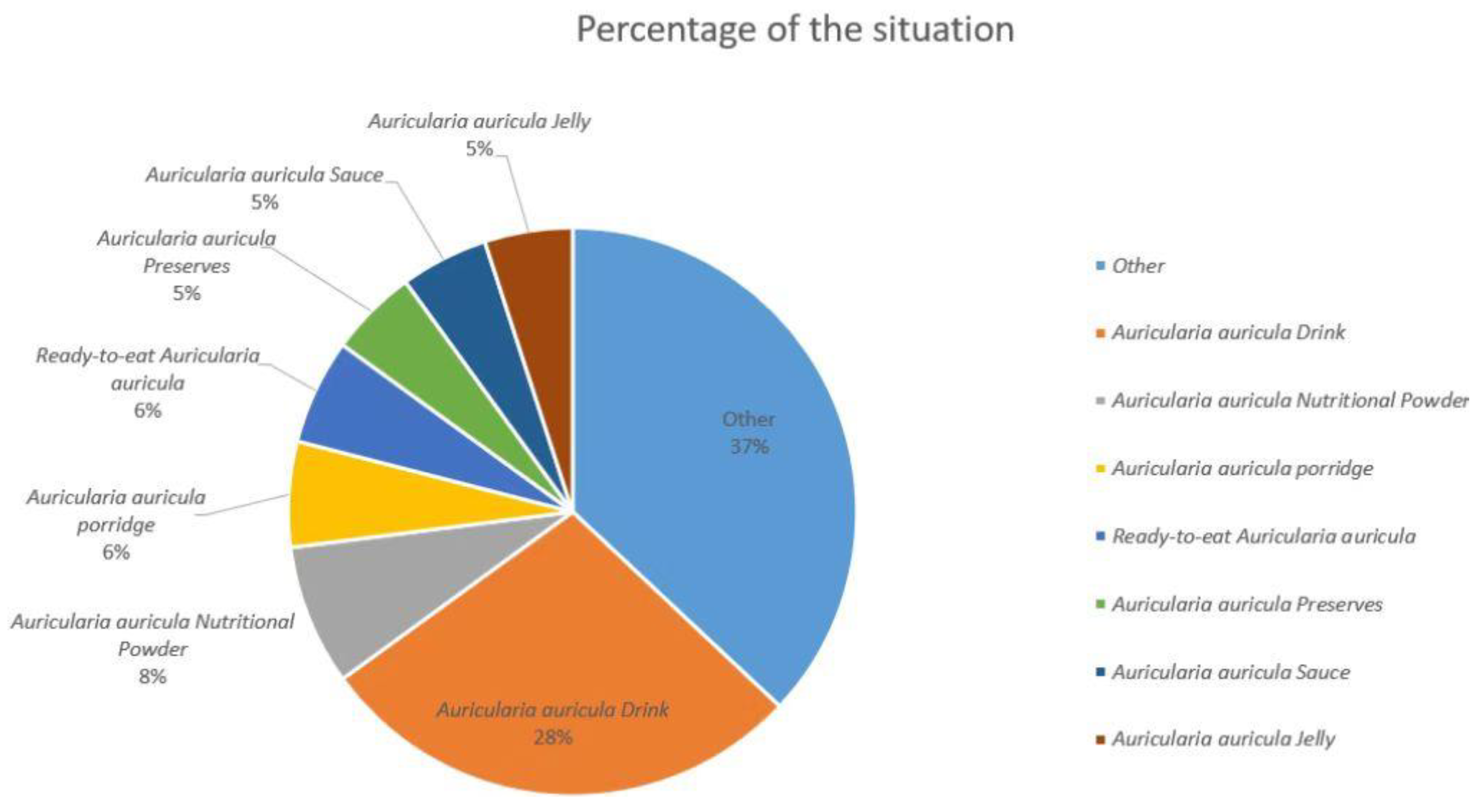
Figure 2. The proportion of commodity types in the Auricularia auricula market.
2. Pretreatment of Auricularia auricula and Extraction Technology
| Origin | Extraction Method | Time (min) | Solid-Liquid Ratio | Temperature (°C) | Other Conditions | Yield (%) | References |
|---|---|---|---|---|---|---|---|
| Jinlin County, Jilin Province | Ultrasonic Assist | 40 | 1:70 | 70 °C | Particle size of 150–200 mesh | 29.29 ± 1.41% | [28] |
| Qingchuan County, Sichuan Province | MAE | 25 | 1:25 | 95 °C | Microwave power of 860 W, pH 7.0 | 10.52% | [29] |
| Jiaohe County, Jilin Province | Pulsed Electric Field | 1.5 | 1:30 | Room temperature | HIPEF strength at 24 kV/cm, pulse number at 6, pH 8 | 14.79% | [30][31] |
| Jinan | PEG-based ultrasound-assisted | 32.44 | 1:39.27 | 91.948 °C | PEG concentration of 0.30 g/mL | 21.58% | [32] |
| Greater Khingan Mountains, Helongjiang | Neutral protease | / | 1:75 | 50 °C | E/S at 8% | 12.96% | [13] |
| Greater Khingan Mountains, Heilongjiang Province | Ultrasonic Assist Alkali method | 90 | 1:48 | 70 °C | 2.0% of NaOH concentration | 15.53% | [33] |
| Greater Khingan Mountains, Heilongjiang Province | Mannanase, β-dextranase, and cellulase | 60 | 1:80 | 83.17 °C | pH at 2.1 | 26.42% ± 0.87% | [34] |
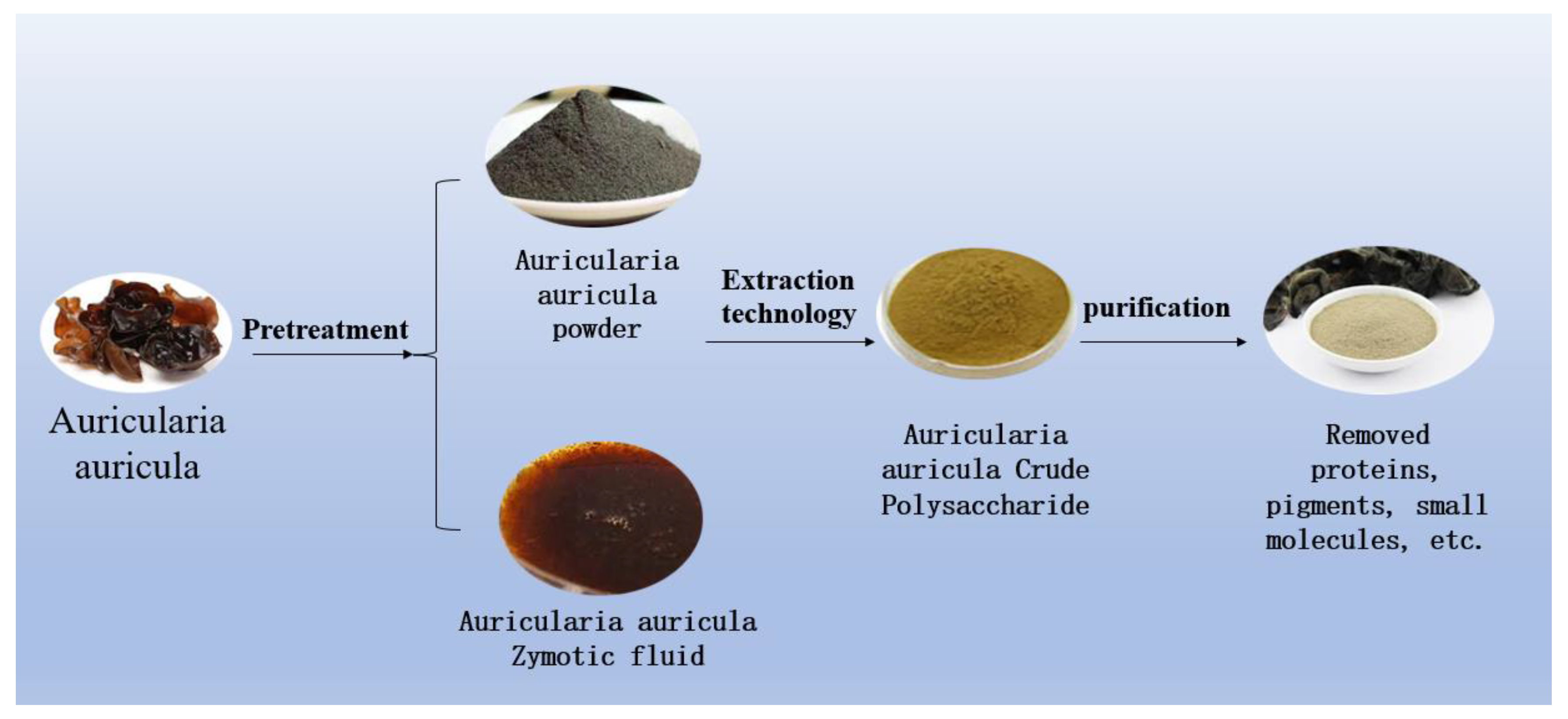
Figure 3. Auricularia auricula has two types of handling for raw materials.
3. Separation and Purification
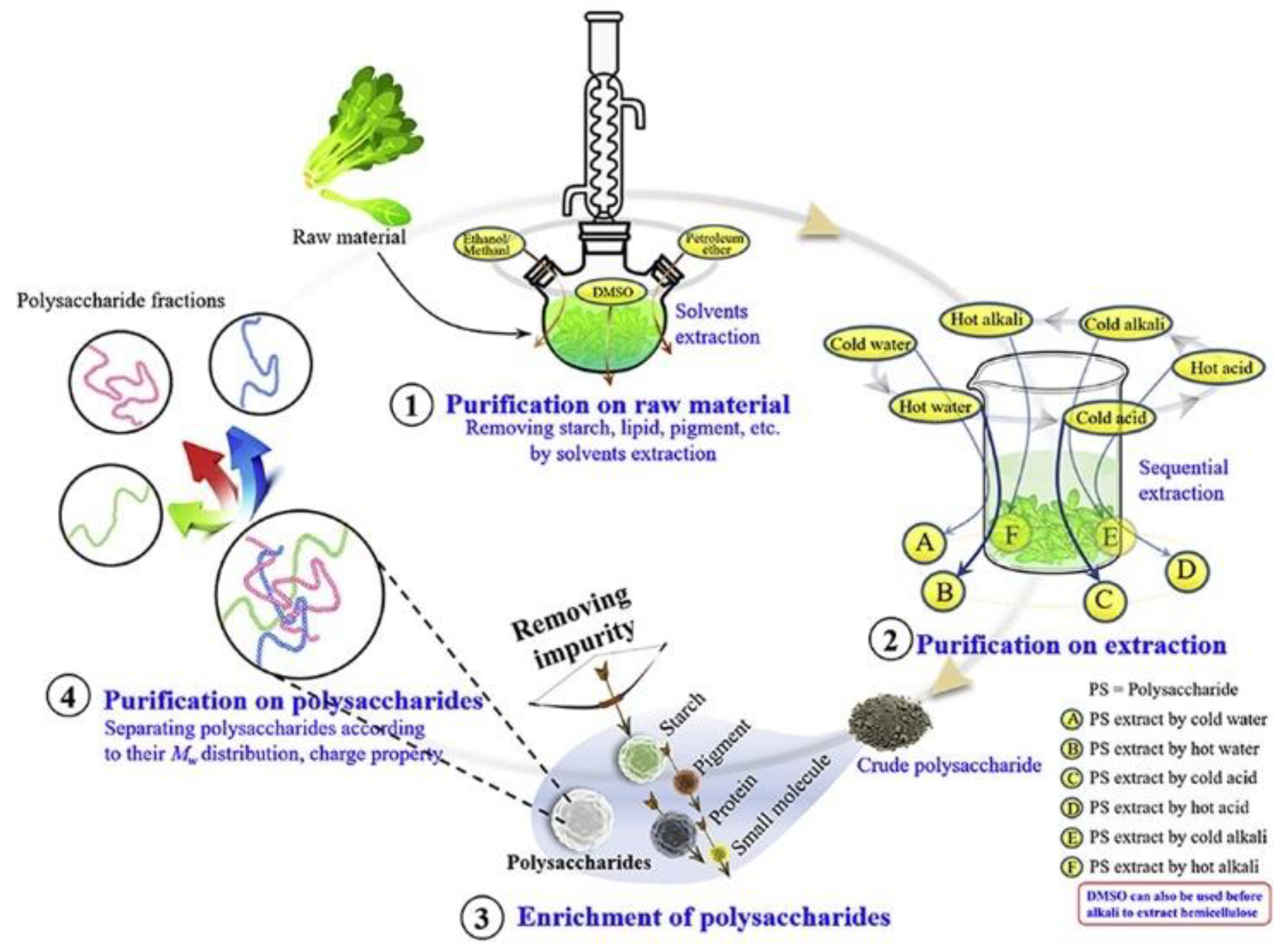
Figure 4. The purification process of polysaccharides.
3.1. Concentration Grading Method
3.2. Column Chromatography Method
3.2.1. Macroporous Resin
3.2.2. Cellulose Column Chromatography
3.2.3. Gel Column Chromatography
4. Physiological Activity and Product Development of Polysaccharides of Auricularia auricula
4.1. Physiological Activity of Auricularia auricula
4.1.1. Regulation of Intestinal Flora
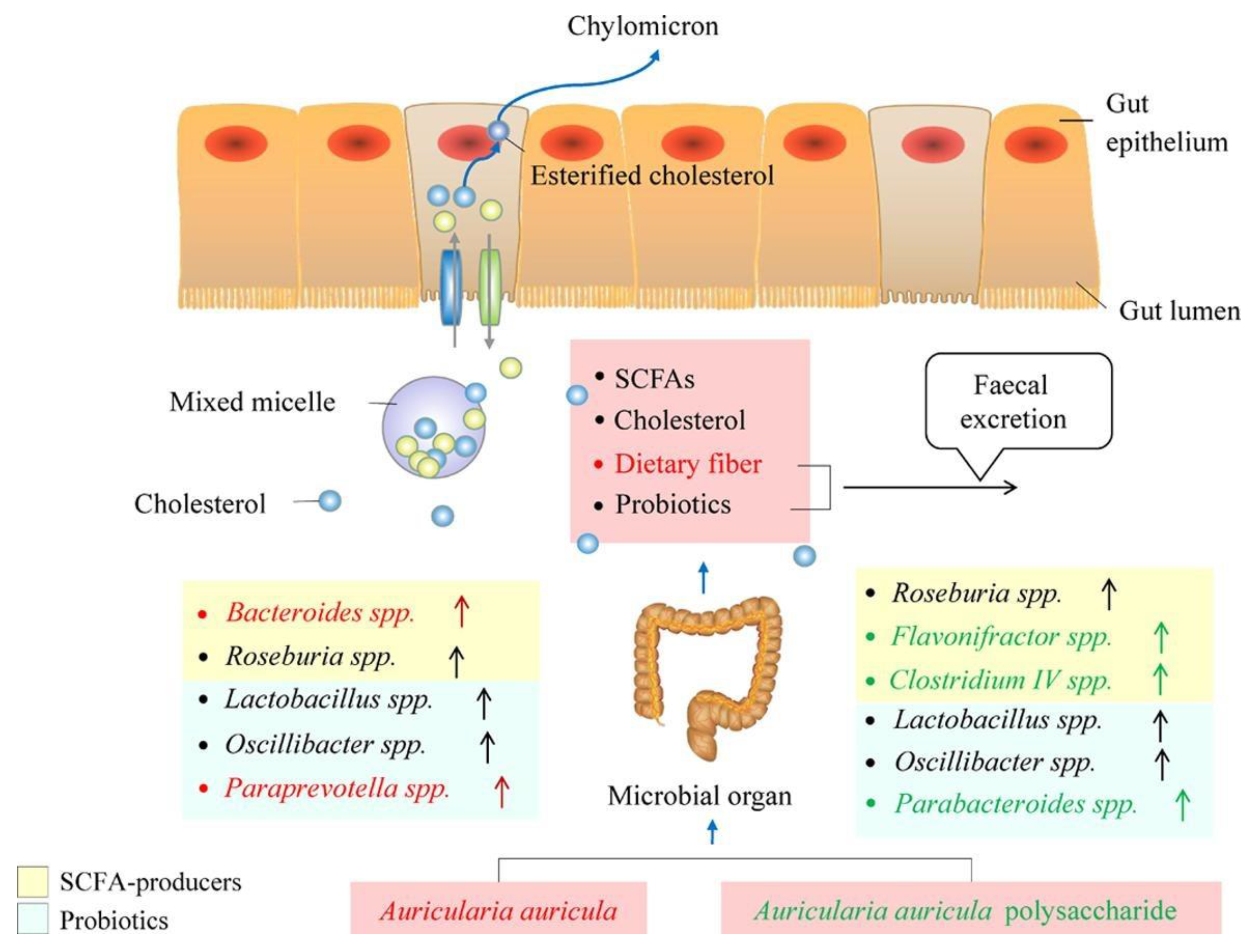
Figure 5. Mechanism of Auricularia auricula polysaccharides.
4.1.2. Anti-High Cholesterol
4.1.3. Hypoglycemic Effect
4.1.4. Anti-Cancer
4.1.5. Anti-Oxidation and Anti-Aging
4.1.6. Anti-Viral
4.2. Practical Application of Auricularia auricula Polysaccharide
4.2.1. Biological Anticorrosive Film
4.2.2. Edible Products
5. Conclusions and Future Outlook
References
- Zhao, Y.; Wang, L.; Zhang, D.; Li, R.; Cheng, T.; Zhang, Y.; Liu, X.; Wong, G.; Tang, Y.; Wang, H.J.S.R. Comparative transcriptome analysis reveals relationship of three major domesticated varieties of Auricularia auricula-judae. Sci. Rep. 2019, 9, 78.
- Cai, W.; Xie, L.; Yong, C.; Zhang, H. Purification, characterization and anticoagulant activity of the polysaccharides from green tea. Carbohydr. Polym. 2013, 92, 1086–1090.
- Bian, C.; Wang, Z.; Molecules, S.J. Extraction Optimization, Structural Characterization, and Anticoagulant Activity of Acidic Polysaccharides from Auricularia auricula-judae. Molecules 2020, 25, 710.
- Miao, J.; Regenstein, J.M.; Qiu, J.; Zhang, J.; Zhang, X.; Li, H.; Zhang, H.; Wang, Z. Isolation, structural characterization and bioactivities of polysaccharides and its derivatives from Auricularia—A review. Int. J. Biol. Macromol. 2020, 150, 102–113.
- Zhao, S.; Rong, C.; Liu, Y.; Xu, F.; Wang, S.; Duan, C.; Chen, J.; Wu, X. Extraction of a soluble polysaccharide from Auricularia polytricha and evaluation of its anti-hypercholesterolemic effect in rats. Carbohydr. Polym. 2015, 122, 39–45.
- Wu, Q.; Qin, D.; Cao, H.; Bai, Y. Enzymatic hydrolysis of polysaccharide from Auricularia auricula and characterization of the degradation product. Int. J. Biol. Macromol. 2020, 162, 127–135.
- Chen, N.; Zhang, H.; Zong, X.; Li, S.; Wang, J.; Wang, Y.; Jin, M. Polysaccharides from Auricularia auricula: Preparation, structural features and biological activities. Carbohydr. Polym. 2020, 247, 116750.
- Zhang, T.; Zhao, W.; Xie, B.; Liu, H. Effects of Auricularia auricula and its polysaccharide on diet-induced hyperlipidemia rats by modulating gut microbiota. J. Funct. Foods 2020, 72, 104038.
- Bao, Z.; Yao, L.; Zhang, X.; Lin, S. Isolation, purification, characterization, and immunomodulatory effects of polysaccharide from Auricularia auricula on RAW264.7 macrophages. J. Food Biochem. 2020, 44, e13516.
- Qian, L.; Liu, H.; Li, T.; Liu, Y.; Zhang, Z.; Zhang, Y. Purification, characterization and in vitro antioxidant activity of a polysaccharide AAP–3–1 from Auricularia auricula—ScienceDirect. Int. J. Biol. Macromol. 2020, 162, 1453–1464.
- Xu, S.; Zhang, Y.; Jiang, K. Antioxidant activity in vitro and in vivo of the polysaccharides from different varieties of Auricularia auricula. Food Funct. 2016, 7, 3868–3879.
- Chen, Z.; Zhou, X.; Zhang, J.; Zhang, H.; Geng, L.; Wang, Z.; Regenstein, J.M. Structure and radio-protective effects of sulfated Auricularia auricula polysaccharides. J. Food Biochem. 2018, 42, e12666.
- Xiao, B.; Chen, S.; Huang, Q.; Tan, J.; Zeng, J.; Yao, J.; Feng, T.; Wang, G.; Zhang, Y. The lipid lowering and antioxidative stress potential of polysaccharide from Auricularia auricula prepared by enzymatic method. Int. J. Biol. Macromol. 2021, 187, 651–663.
- Nguyen, T.L.; Chen, J.; Hu, Y.; Wang, D.; Fan, Y.; Wang, J.; Abula, S.; Zhang, J.; Qin, T.; Chen, X.; et al. In vitro antiviral activity of sulfated Auricularia auricula polysaccharides. Carbohydr. Polym. 2012, 90, 1254–1258.
- Zhang, Y.; Shi, Q.; Jiang, W.; Yao, J.; Zeng, J.; Wang, W.; Zhang, Y. Comparison of the chemical composition and antioxidant stress ability of polysaccharides from Auricularia auricula under different drying methods. Food Funct. 2022, 13, 2938–2951.
- Huang, Q.; Xiao, B.; Chen, S.; Zeng, J.; Yao, J.; Tan, J.; Wang, G.; Wang, W.; Zhang, Y. Effect of Enzyme-Assisted Extraction on the Chemical Properties and Antioxidant Activities of Polysaccharides Obtained from the Wood Ear Mushroom, Auricularia auricula (Agaricomycetes). Int. J. Med. Mushrooms 2022, 24, 49–62.
- Dobrinčić, A.; Zorić, Z.; Pedisić, S.; Repajić, M.; Roje, M.; Herceg, Z.; Čož-Rakovac, R.; Dragović-Uzelac, V. Application of Ultrasound-Assisted Extraction and Non-Thermal Plasma for Fucus virsoides and Cystoseira barbata Polysaccharides Pre-Treatment and Extraction. Processes 2022, 10, 433.
- Yang, Z.; Hu, Y.; Wu, J.; Liu, J.; Zhang, F.; Ao, H.; Zhu, Y.; He, L.; Zhang, W.; Zeng, X. High-Efficiency Production of Auricularia polytricha Polysaccharides Through Yellow Slurry Water Fermentation and Its Structure and Antioxidant Properties. Front. Microbiol. 2022, 13, 811275.
- Li, L.; Su, Y.; Feng, Y.; Hong, R. A comparison study on digestion, anti-inflammatory and functional properties of polysaccharides from four Auricularia species. Int. J. Biol. Macromol. 2020, 154, 1074–1081.
- Li, F.; Bi, H. Current Situation and Countermeasures of Auricularia auricula Industry in China. North. Hortic. 2021, 7, 142–147.
- Wang, X.; Yanfang, L.I.; Zang, F.; Miao, Y.; Wang, H.; Yin, X.; Shi, C. Disinfection Technology for Auricularia auricula in Bag Cultivation: Problems and Countermeasures. Asian J. Agric. Res. 2021, 12, 44–46.
- Wang, M.; Wang, S.; Song, W.; Zhou, D.; Wang, J.; Ding, T. The Present Situation and Prospect of Auricularia auricula Mechanized Harvesting in China. J. Chin. Agric. Mech. 2022, 43, 219–223.
- Huang, F. Analysis of Auricularia auricula industry based on spot market and e-commerce sales data. China Veg. 2022, 5, 11–15.
- Meng, X.; Guo, C.; Ma, J.; Kong, P.; Fang, B. Research Status of Auricularia auricula Products in China. Food Ind. 2021, 42, 257–259.
- Bi, H.; Li, F. Investigation and Analysis Report on the Market and Industry of Auricularia auricula in China. Farm Prod. Mart 2021, 50–53, 1009–8070.
- Sun, Z.; Cong, Y.; Li, T.; Meng, X.; Zhang, F. Enhancement of nutritional, sensory and storage stability by lactic fermentation of Auricularia auricula. J. Sci. Food Agric. 2022, 102, 5172–5180.
- Yuan, B.; Zhao, L.; Yang, W.; McClements, D.J.; Hu, Q. Enrichment of bread with nutraceutical-rich mushrooms: Impact of Auricularia auricula (Mushroom) flour upon quality attributes of wheat dough and bread. J. Food Sci. 2017, 82, 2041–2050.
- Wang, Y.; Wang, C.; Guo, M. Effects of Ultrasound Treatment on Extraction and Rheological Properties of Polysaccharides from Auricularia Cornea var. Li. Molecules 2019, 24, 939.
- Zeng, W.C.; Zhang, Z.; Gao, H.; Jia, L.R.; Chen, W.Y. Characterization of antioxidant polysaccharides from Auricularia auricular using microwave-assisted extraction. Carbohydr. Polym. 2012, 89, 694–700.
- Li, C.; Mao, X.; Xu, B. Pulsed electric field extraction enhanced anti-coagulant effect of fungal polysaccharide from Jew’s ear (Auricularia auricula). Phytochem. Anal. 2012, 24, 36–40.
- Miao, C.; Wang, Z.; Li, T.; Yin, H.; Liu, R.; Liang, W.; Yu, M.; Zhang, R. Optimization of Extraction Technology of Auricularia Auricularia Polysaccharides by Strong Electric Field. J. Anhui Agric. Sci. 2014, 42, 5.
- ZZhang, L.; Wang, M. PEG-based ultrasound-assisted extraction of polysaccharides from superfine ground Auricularia auricular. J. Food Process. Preserv. 2018, 2, e13445.
- Gu, J.; Li, Q.; Liu, J.; Ye, Z.; Feng, T.; Wang, G.; Wang, W.; Zhang, Y. Ultrasonic–assisted extraction of polysaccharides from Auricularia auricula and effects of its acid hydrolysate on the biological function of Caenorhabditis elegans. Int. J. Biol. Macromol. 2021, 167, 423–433.
- Xiao, B.; Huang, Q.; Chen, S.; Yao, J.; Zeng, J.; Shen, J.; Wang, G.; Wang, W.; Zhang, Y. Comparison on chemical features and antioxidant activity of polysaccharides from Auricularia auricula by three different enzymes. J. Food Biochem. 2022, 46, e14051.
- Sillapachaiyaporn, C.; Chuchawankul, S.; Nilkhet, S.; Moungkote, N.; Sarachana, T.; Ung, A.T.; Baek, S.J.; Tencomnao, T. Ergosterol isolated from cloud ear mushroom (Auricularia polytricha) attenuates bisphenol A-induced BV2 microglial cell inflammation. Food Res. Int. 2022, 157, 111433.
- Geng, Z.; Ye, P.; Zhou, L.; Fu, H.; Chen, X.; Wang, Y.; Wang, Y. Pasteurization of Salmonella spp. in black fungus (Auricularia auricula) powder by radio frequency heating. Int. J. Food Sci. Technol. 2022, 9, 10820132221123437.
- Yang, L.Q.; Zhang, L.M. Chemical structural and chain conformational characterization of some bioactive polysaccharides isolated from natural sources. Carbohydr. Polym. 2009, 76, 349–361.
- Tang, W.; Liu, D.; Yin, J.-Y.; Nie, S.-P. Consecutive and progressive purification of food-derived natural polysaccharide: Based on material, extraction process and crude polysaccharide. Trends Food Sci. Technol. 2020, 99, 76–87.
- Nada, S.A.; Omara, E.; Abdel-Salam, O.; Zahran, H.G. Mushroom insoluble polysaccharides prevent carbon tetrachloride-induced hepatotoxicity in rat. Food Chem. Toxicol. 2010, 48, 3184–3188.
- Johnson, W., Jr.; Heldreth, B.; Bergfeld, W.F.; Belsito, D.V.; Hill, R.A.; Klaassen, C.D.; Liebler, D.C.; Marks, J.G., Jr.; Shank, R.C.; Slaga, T.J.; et al. Safety Assessment of Galactomannans as Used in Cosmetics. Int. J. Toxicol. 2015, 34, 35S–65S.
- Han, Q.B. Critical problems stalling progress in natural bioactive polysaccharide research & development. J. Agric. Food Chem. 2018, 66, 4581–4583.
- Tziveleka, L.-A.; Ioannou, E.; Roussis, V. Ulvan, a bioactive marine sulphated polysaccharide as a key constituent of hybrid biomaterials: A review. Carbohydr. Polym. 2019, 218, 355–370.
- Liu, J.; Willför, S.; Xu, C. A review of bioactive plant polysaccharides: Biological activities, functionalization, and biomedical applications. Bioact. Carbohydr. Diet. Fibre 2015, 5, 31–61.
- Zhang, H.; Chen, J.; Li, J.; Yan, L.; Li, S.; Ye, X.; Liu, D.; Ding, T.; Linhardt, R.J.; Orfila, C.; et al. Extraction and characterization of RG-I enriched pectic polysaccharides from mandarin citrus peel. Food Hydrocoll. 2017, 79, 579–586.
- Colodel, C.; Vriesmann, L.C.; Petkowicz, C.L.D.O. Cell wall polysaccharides from Ponkan mandarin (Citrus reticulata Blanco cv. Ponkan) peel. Carbohydr. Polym. 2018, 195, 120–127.
- Shi, L. Bioactivities, isolation and purification methods of polysaccharides from natural products: A review. Int. J. Biol. Macromol. 2016, 92, 37–48.
- Lan, X.; Wang, Y.; Deng, S.; Zhao, J.; Wang, L.; Yao, K.; Jia, D. Physicochemical and rheological properties of Tremella fuciformis polysaccharide fractions by ethanol precipitation. CyTA J. Food 2021, 19, 645–655.
- Zhang, K.; Yuan, D.; Li, C.; Fu, X. Physicochemical properties and bioactivity of polysaccharides from Sargassum pallidum by fractional ethanol precipitation. Int. J. Food Sci. Technol. 2021, 56, 3536–3545.
- Chen, Y.-X.; Liu, X.-Y.; Xiao, Z.; Huang, Y.-F.; Liu, B. Antioxidant activities of polysaccharides obtained from Chlorella pyrenoidosa via different ethanol concentrations. Int. J. Biol. Macromol. 2016, 91, 505–509.
- Yang, Z.; Hu, Y.; Yue, P.; Luo, H.; Li, Q.; Li, H.; Zhang, Z.; Peng, F. Physicochemical Properties and Skin Protection Activities of Polysaccharides from Usnea longissima by Graded Ethanol Precipitation. ACS Omega 2021, 6, 25010–25018.
- Duan, S.; Huang, Q.; Shen, X.; Hu, J.; Yi, X.; Li, Z.; Ding, B. Deproteinization of four macroporous resins for rapeseed meal polysaccharides. Food Sci. Nutr. 2020, 8, 322–331.
- Xie, Y.; Guo, Q.-S.; Wang, G.-S. Preparative separation and purification of the total flavonoids in Scorzonera austriaca with macroporous resins. Molecules 2016, 21, 768.
- Chen, C.; Jin, S.; Xiang, X.; Wang, X.; Shi, Q.; Yang, M.; Ji, S.; Huang, R.; Song, C. Enrichment and Cytotoxic Activity of Curcuminoids from Turmeric Using Macroporous Resins. J. Food Sci. 2017, 82, 2024–2030.
- Wu, Y.; Han, Y.; Tao, Y.; Fan, S.; Chu, D.-T.; Ye, X.; Ye, M.; Xie, G. Ultrasound assisted adsorption and desorption of blueberry anthocyanins using macroporous resins. Ultrason. Sonochem. 2018, 48, 311–320.
- Zheng, Y.; Wang, X.-S.; Fang, J. Two acidic polysaccharides from the flowers of Chrysanthemum morifolium. J. Asian Nat. Prod. Res. 2006, 8, 217–222.
- Wang, H.; Zhong, Y.; Ma, L. Leukaemia Infection Diagnosis and Intestinal Flora Disorder. Curr. Mol. Med. 2022, 22, 2–7.
- Zhao, Q.; Shi, Q.; Zhu, Q.; Hu, Y.; Zhang, X. A mini-review of advances in intestinal flora and necrotizing enterocolitis. Lett. Appl. Microbiol. 2022, 75, 2–9.
- Hu, W.; Kong, X.; Wang, H.; Li, Y.; Luo, Y. Ischemic stroke and intestinal flora: An insight into brain-gut axis. Eur. J. Med. Res. 2022, 27, 73.
- Song, Z.; Li, S.; Li, R. An Investigation into the Correlation of Intestinal Flora with Obesity and Gestational Diabetes Mellitus. Comput. Math. Methods Med. 2022, 2022, 5677073.
- Liu, Q.; An, X.; Chen, Y.; Deng, Y.; Niu, H.; Ma, R.; Zhao, H.; Cao, W.; Wang, X.; Wang, M. Effects of Auricularia auricula Polysaccharides on Gut Microbiota and Metabolic Phenotype in Mice. Foods 2022, 11, 2700.
- Li, P.; Xiong, C.; Huang, W. Gamma-Irradiation-Induced Degradation of the Water-Soluble Polysaccharide from Auricularia polytricha and Its Anti-Hypercholesterolemic Activity. Molecules 2022, 27, 1110.
- Zhang, Y.; Li, X.; Yang, Q.; Zhang, C.; Song, X.; Wang, W.; Jia, L.; Zhang, J. Antioxidation, anti-hyperlipidaemia and hepatoprotection of polysaccharides from Auricularia auricular residue. Chem. Biol. Interact. 2021, 333, 109323.
- Liu, N.; Chen, X.; Song, J.; Chen, M.; Gong, P.; Jia, W.; Li, G. Hypoglycemic effects of Auricularia auricula polysaccharides on high fat diet and streptozotocin-induced diabetic mice using metabolomics analysis. Food Funct. 2021, 12, 9994–10007.
- Xiang, H.; Sun-Waterhouse, D.; Cui, C. Hypoglycemic polysaccharides from Auricularia auricula and Auricularia polytricha inhibit oxidative stress, NF-κB signaling and proinflammatory cytokine production in streptozotocin-induced diabetic mice. Food Sci. Hum. Wellness 2021, 10, 7.
- Xu, N.; Zhou, Y.; Lu, X.; Chang, Y. Auricularia auricula-judae (Bull.) polysaccharides improve type 2 diabetes in HFD/STZ-induced mice by regulating the AKT/AMPK signaling pathways and the gut microbiota. J. Food Sci. 2021, 86, 5479–5494.
- Ma, Y.; Wang, C.; Zhang, Q.; Peng, X.; Feng, Y.; Meng, X. The effects of polysaccharides from Auricularia auricula (Huaier) in adjuvant anti-gastrointestinal cancer therapy: A systematic review and network meta-analysis. Pharmacol. Res. 2018, 132, 80–89.
- Xu, Y.; Shen, M.; Chen, Y.; Lou, Y.; Luo, R.; Chen, J.; Zhang, Y.; Li, J.; Wang, W. Optimization of the polysaccharide hydrolysate from Auricularia auricula with antioxidant activity by response surface methodology. Int. J. Biol. Macromol. 2018, 113, 543–549.
- Asgher, M.; Qamar, S.A.; Bilal, M.; Iqbal, H.M.N. Bio-based active food packaging materials: Sustainable alternative to conventional petrochemical-based packaging materials. Food Res. Int. 2020, 137, 109625.
- Zhao, Y.; Shui, D.; Li, S.; Lin, X.; Liang, H.; Zhang, S.; Ji, C. Complexation behavior of Auricularia auricula polysaccharide and whey protein isolate: Characterization and potential beverage application. J. Food Process. Preserv. 2022, 46, e16340.





