
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Sashi Debnath | -- | 1165 | 2022-12-12 16:20:46 | | | |
| 2 | Conner Chen | -23 word(s) | 1142 | 2022-12-13 10:03:39 | | |
Video Upload Options
Selenium is one of the eight necessary trace elements humans require for active health balance. It contributes in several ways to the proper functioning of selenoprotein. Selenium has received enormous interest due to its therapeutic potential against a number of ailments. Numerous chemical compounds containing selenium have been investigated for the therapy of cancer and other disorders. Unifying the selenium atom into chemical components (typically organic) greatly increased their bioactivities. Selenocysteine can substitute the effect of cysteine and shield healthy cells from the adverse effects of reactive oxygen species (ROS); in other ways, specific selenium compounds are classified as antioxidant agents that preserve the redox environment in healthy cells.
1. Introduction
2. Antioxidant Activity
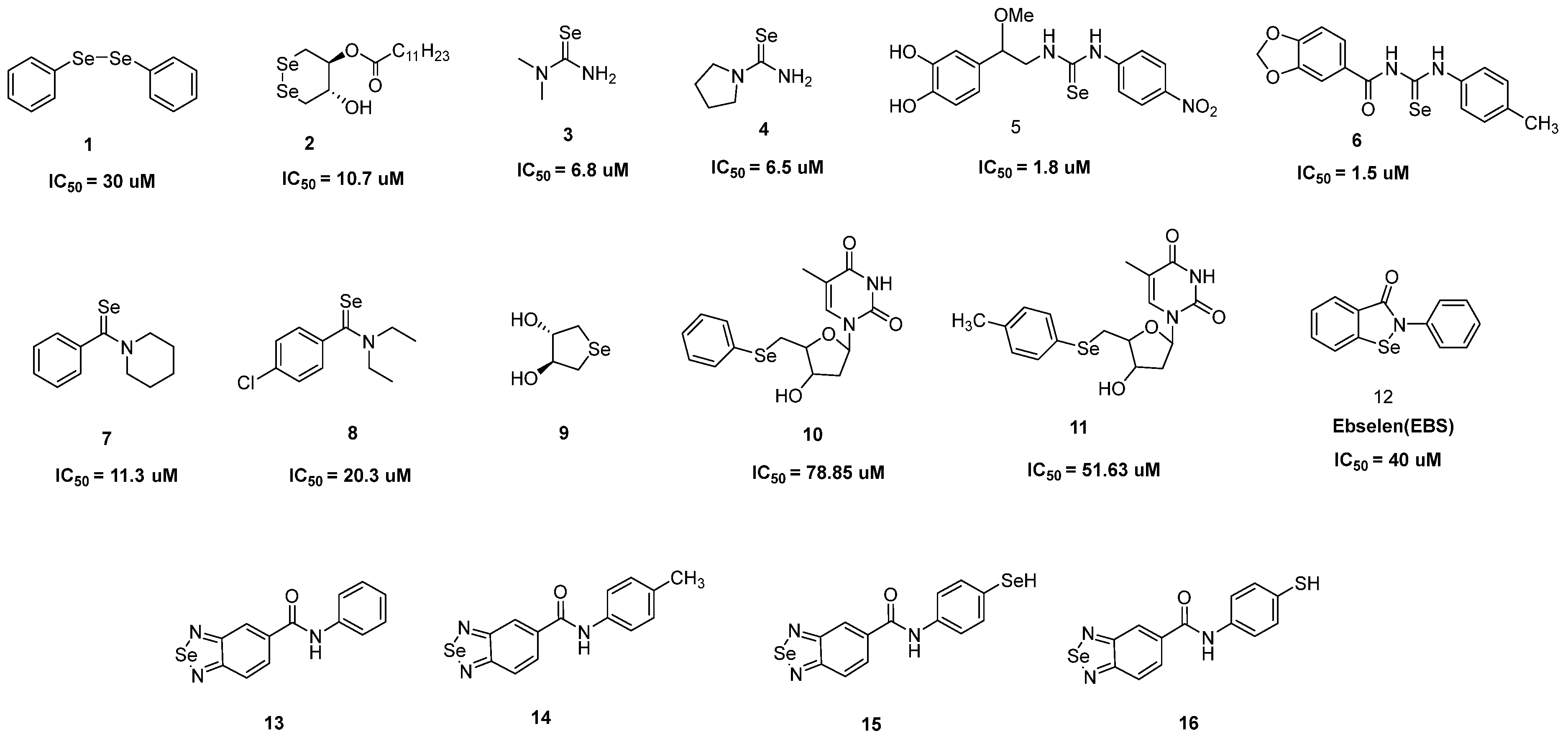
References
- Greenwood, N.N.; Earnshaw, A. Chemistry of the Elements; Elsevier: Amsterdam, The Netherlands, 2012.
- Agrawal, A.R.; Kumar, N.R.; Debnath, S.; Das, S.; Kumar, C.; Zade, S.S. Radical-cascade avenue for 3,4-fused-ring-substituted thiophenes. Org. Lett. 2018, 20, 4728–4731.
- Lenardão, E.J.; Santi, C.; Sancineto, L. Bioactive organoselenium compounds and therapeutic perspectives. In New Frontiers in Organoselenium Compounds; Springer: Berlin/Heidelberg, Germany, 2018; pp. 99–143.
- Steinbrenner, H.; Speckmann, B.; Klotz, L.-O. Selenoproteins: Antioxidant selenoenzymes and beyond. Arch. Biochem. Biophys. 2016, 595, 113–119.
- Mugesh, G.; du Mont, W.-W.; Sies, H. Chemistry of biologically important synthetic organoselenium compounds. Chem. Rev. 2001, 101, 2125–2180.
- Haug, A.; Graham, R.D.; Christophersen, O.A.; Lyons, G.H. How to use the world’s scarce selenium resources efficiently to increase the selenium concentration in food. Microb. Ecol. Health Dis. 2007, 19, 209–228.
- Krinsky, N.I.; Beecher, G.R.; Burk, R.F.; Chan, A.C.; Erdman, J.J.; Jacob, R.A.; Jialal, I.; Kolonel, L.N.; Marshall, J.R.; Taylor Mayne, P.R.; et al. Dietary reference intakes for vitamin C, vitamin E, selenium, and carotenoids. Inst. Med. 2000, 19, 95–185.
- Bedi, A.; Debnath, S.; Chandak, H.S.; Zade, S.S. Phenyl-capped cyclopenta chalcogenophenes: Synthesis, crystal structures, electrochemistry and theoretical insights. RSC Adv. 2014, 4, 35653–35658.
- Bedi, A.; Debnath, S.; Zade, S.S. Diselenolodiselenole: A selenium containing fused heterocycle for conjugated systems. Chem. Commun. 2014, 50, 13454–13456.
- Patra, A.; Agrawal, V.; Bhargav, R.; Bhardwaj, D.; Chand, S.; Sheynin, Y.; Bendikov, M. Metal free conducting PEDOS, PEDOT, and their analogues via an unusual bromine-catalyzed polymerization. Macromolecules 2015, 48, 8760–8764.
- Zhang, Z.; Li, Y.; Cai, G.; Zhang, Y.; Lu, X.; Lin, Y. Selenium heterocyclic electron acceptor with small urbach energy for as-cast high-performance organic solar cells. J. Am. Chem. Soc. 2020, 142, 18741–18745.
- Debnath, S.; Chithiravel, S.; Sharma, S.; Bedi, A.; Krishnamoorthy, K.; Zade, S.S. Selenium-Containing Fused Bicyclic Heterocycle Diselenolodiselenole: Field Effect Transistor Study and Structure–Property Relationship. ACS Appl. Mater. Interfaces 2016, 8, 18222–18230.
- Debnath, S.; Bedi, A.; Zade, S.S. Thienopentathiepine: A sulfur containing fused heterocycle for conjugated systems and their electrochemical polymerization. Polym. Chem. 2015, 6, 7658–7665.
- Mecik, P.; Pigulski, B.; Szafert, S. Serendipitous Formation of Various Selenium Heterocycles Hidden in the Classical Synthesis of Selenophene. Org. Lett. 2021, 23, 1066–1070.
- Xia, J.; Li, T.; Lu, C.; Xu, H. Selenium-containing polymers: Perspectives toward diverse applications in both adaptive and biomedical materials. Macromolecules 2018, 51, 7435–7455.
- Zhao, Z.; Yin, Z.; Chen, H.; Zheng, L.; Zhu, C.; Zhang, L.; Tan, S.; Wang, H.; Guo, Y.; Tang, Q. High-performance, air-stable field-effect transistors based on heteroatom-substituted naphthalenediimide-benzothiadiazole copolymers exhibiting ultrahigh electron mobility up to 8.5 cm V− 1 s− 1. Adv. Mater. 2017, 29, 1602410.
- Debnath, S.; Singh, S.; Bedi, A.; Krishnamoorthy, K.; Zade, S.S. Synthesis, optoelectronic, and transistor properties of BODIPY-and cyclopenta thiophene-containing π-conjugated copolymers. J. Phys. Chem. C 2015, 119, 15859–15867.
- Fan, B.; Lin, F.; Wu, X.; Zhu, Z.; Jen, A.K.-Y. Selenium-containing organic photovoltaic materials. Acc. Chem. Res. 2021, 54, 3906–3916.
- Walter, R.; Schwartz, I.; Roy, J. Can selenoamino acids act as reversible biological antioxidants. Ann. N. Y. Acad. Sci. 1972, 192, 175–180.
- Olcott, H.; Brown, W.D.; Van der Veen, J. Selenomethionine as an antioxidant. Nature 1961, 191, 1201–1202.
- Schrauzer, G.; Rhead, W. Interpretation of the methylene blue reduction test of human plasma and the possible cancer protecting effect of selenium. Experientia 1971, 27, 1069–1071.
- Shamberger, R.; Frost, D. Possible protective effect of selenium against human cancer. Can. Med. Assoc. J. 1969, 100, 682.
- Clark, L.C.; Cantor, K.P.; Allaway, W. Selenium in forage crops and cancer mortality in US counties. Arch. Environ. Health Int. J. 1991, 46, 37–42.
- May, S.W. Selenium-based drug design: Rationale and therapeutic potential. Expert Opin. Investig. Drugs 1999, 8, 1017–1030.
- Reeves, M.; Hoffmann, P. The human selenoproteome: Recent insights into functions and regulation. Cell. Mol. Life Sci. 2009, 66, 2457–2478.
- Rayman, M.P. Selenium and human health. Lancet 2012, 379, 1256–1268.
- Roman, M.; Jitaru, P.; Barbante, C. Selenium biochemistry and its role for human health. Metallomics 2014, 6, 25–54.
- Reich, H.J.; Hondal, R.J. Why nature chose selenium. ACS Chem. Biol. 2016, 11, 821–841.
- May, S.W. Selenium-based pharmacological agents: An update. Expert Opin. Investig. Drugs 2002, 11, 1261–1269.
- May, S.W.; Pollock, S.H. Selenium-based antihypertensives. Drugs 1998, 56, 959–964.
- Collery, P. Strategies for the development of selenium-based anticancer drugs. J. Trace Elem. Med. Biol. 2018, 50, 498–507.
- Wallenberg, M.; Misra, S.; Björnstedt, M. Selenium cytotoxicity in cancer. Basic Clin. Pharmacol. Toxicol. 2014, 114, 377–386.
- Straliotto, M.R.; Hort, M.A.; Fiuza, B.; Rocha, J.B.T.; Farina, M.; Chiabrando, G.; de Bem, A.F. Diphenyl diselenide modulates oxLDL-induced cytotoxicity in macrophage by improving the redox signaling. Biochimie 2013, 95, 1544–1551.
- Melo, M.; De Oliveira, I.; Grivicich, I.; Guecheva, T.; Saffi, J.; Henriques, J.; Rosa, R. Diphenyl diselenide protects cultured MCF-7 cells against tamoxifen-induced oxidative DNA damage. Biomed. Pharmacother. 2013, 67, 329–335.
- Arai, K.; Sato, Y.; Nakajima, I.; Saito, M.; Sasaki, M.; Kanamori, A.; Iwaoka, M. Glutathione peroxidase-like functions of 1,2-diselenane-4, 5-diol and its amphiphilic derivatives: Switchable catalytic cycles depending on peroxide substrates. Bioorg. Med. Chem. 2021, 29, 115866.
- Arai, K.; Ueno, H.; Asano, Y.; Chakrabarty, G.; Shimodaira, S.; Mugesh, G.; Iwaoka, M. Protein Folding in the Presence of Water-Soluble Cyclic Diselenides with Novel Oxidoreductase and Isomerase Activities. ChemBioChem 2018, 19, 207–211.
- Begines, P.; Oliete, A.; Lopez, O.; Maya, I.; Plata, G.B.; Padron, J.M.; Fernandez-Bolanos, J.G. Chalcogen-containing phenolics as antiproliferative agents. Future Med. Chem. 2018, 10, 319–334.
- Ruberte, A.C.; Ramos-Inza, S.; Aydillo, C.; Talavera, I.; Encío, I.; Plano, D.; Sanmartín, C. Novel N,N′-disubstituted acylselenoureas as potential antioxidant and cytotoxic agents. Antioxidants 2020, 9, 55.
- Tsukagoshi, H.; Koketsu, M.; Kato, M.; Kurabayashi, M.; Nishina, A.; Kimura, H. Superoxide radical-scavenging effects from polymorphonuclear leukocytes and toxicity in human cell lines of newly synthesized organic selenium compounds. FEBS J. 2007, 274, 6046–6054.
- Arai, K.; Tashiro, A.; Osaka, Y.; Iwaoka, M. Glutathione peroxidase-like activity of amino-substituted water-soluble cyclic selenides: A shift of the major catalytic cycle in methanol. Molecules 2017, 22, 354.
- Ecker, A.; Ledur, P.C.; da Silva, R.S.; Leal, D.B.R.; Rodrigues, O.E.; Ardisson-Araújo, D.; Waczuk, E.P.; da Rocha, J.B.T.; Barbosa, N.V. Chalcogenozidovudine derivatives with antitumor activity: Comparative toxicities in cultured human mononuclear cells. Toxicol. Sci. 2017, 160, 30–46.
- Nakamura, Y.; Feng, Q.; Kumagai, T.; Torikai, K.; Ohigashi, H.; Osawa, T.; Noguchi, N.; Niki, E.; Uchida, K. Ebselen, a glutathione peroxidase mimetic seleno-organic compound, as a multifunctional antioxidant: Implication for inflammation-associated carcinogenesis. J. Biol. Chem. 2002, 277, 2687–2694.
- Ruberte, A.C.; Plano, D.; Encío, I.; Aydillo, C.; Sharma, A.K.; Sanmartín, C. Novel selenadiazole derivatives as selective antitumor and radical scavenging agents. Eur. J. Med. Chem. 2018, 157, 14–27.





