
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Prem Gunnasegaran | -- | 2239 | 2022-12-08 12:09:53 | | | |
| 2 | Beatrix Zheng | Meta information modification | 2239 | 2022-12-09 04:43:10 | | |
Video Upload Options
Due to the combination of thermal engineering and the rapid rise of nanotechnology research over the past two decades, novel heat transfer fluids known as “nanofluids” have emerged. A “nanofluid” is a heat transfer fluid that has 1–100 nm-sized “nanoparticles”, which are suspended nanoparticles, scattered throughout the base fluid. To increase the stability of the working fluid, it is crucial to make sure the nanoparticle size is smaller than 100 nm. Water, oils, organic liquids (such as tri-ethylene-glycols, ethylene and refrigerants) and bio-fluids polymeric solutions are the most often utilized base fluids. Numerous studies throughout the years have documented diverse nanofluid preparation methods with various nanoparticle types and their heat transfer capabilities, in addition to advancing the information about nanofluids.
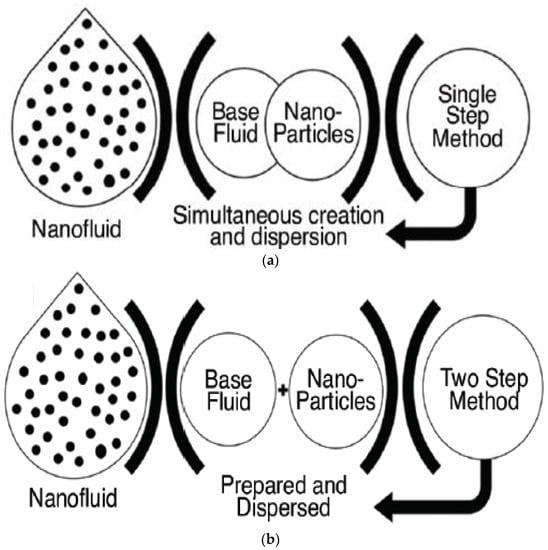
1. Nanofluid Preparation Methods
| Particular | One-Step Method | Two-Step Method |
|---|---|---|
| Synthesis process | Simultaneous production of nanoparticles and nanofluid | Production of nanoparticles either chemically or mechanically followed by dispersion of nanoparticles into the base fluid |
| Production scale | Small scale production | Large scale production |
| Cost of production | High cost | Low cost |
| Control on NPs size | Difficult and limited control over the nanoparticle size during the preparation stage | Able to control the nanoparticle size during the preparation stage |
| Particle oxidation | Oxidation of particles does not occur due to the elimination of drying, transportation and storage processes. | No such problem |
| Advantages | Reduced chances of particle agglomeration. More stable nanofluid |
Cheaper and more applicable in industry |
| Disadvantages | Residual reactants are left in the nanofluids which might cause problems during application. Can only produce in batch |
Prone to agglomeration Constant stabilizing process is needed for long term stability |
| Nanoparticles | Base Fluid | Preparation Method | Nanofluid Sonication Time | Surfactant | Reference |
|---|---|---|---|---|---|
| Metal Based | |||||
| Au | Water | Two step | 20 min | None | [9] |
| Au Ag |
DI Water | One step | - | Cationic gemini | [10] |
| Cu | Methanol | Two step | 30 min | APTMS | [11] |
| Metal-oxide Based | |||||
| TiO2 | Water | Two step | 5 h | HMDS | [12] |
| CuO | Water | Two step | 5 h | None | [13] |
| Al2O3 | DI Water | One step | - | None | [14] |
| Al2O3 | Water | Two step | 6 h | None | [15] |
| ZnO | EG | Two step | 3 h | None | [16] |
| Carbon Based | |||||
| MWCNT | Water | Two step | 20 min | None | [9] |
| COOH-CNT | DI Water | Two step | 10 min | Nonylphenol ethoxylate | [17] |
| CNT | Decane | Two step | 60 min | Oleylamine | [18] |
| MWCNT | Water | Two step | 3 h | SDBS | [19] |
| MWCNT | Kapok seed oil | One step | 6 h | none | [20] |
| Hybrid | |||||
| Ag-MWCNT (50:50) | DI Water | Two step | 30 min | SDBS | [7] |
| MgO-SWCNT (80:20) | EG | Two step | 6 h | None | [21] |
| Cu-TiO2 (36:64) |
EG/Water (50:50) | Two step | 30 min | PVP, SDBS and GA | [22] |
| ZnO-SWCNT (70:30) | EG/Water (40:60) | Two step | 7 h | None | [23] |
| ZnO-MWCNT (50:50) | EG/Water (50:50) | Two Step | 3 h | None | [24] |
| Au-TiO2 Au-Ag Au-Al Au-Ni |
DI Water | Two Step | 3 h | None | [25] |
| Ag-Fe3O4 (50:50) |
DI Water | Two Step | 3 h | None | [26] |
2. Classification of Nanofluids
2.1. Metal-Based Nanofluids
2.2. Metal Oxide-Based Nanofluids
2.3. Carbon-Based Nanofluids
2.4. Hybrid Nanofluids
3. Nanofluid Stabilization Methods
| Nanofluid | Particle Size (nm) | Concentration | Stability Period Reported | References |
|---|---|---|---|---|
| Metal Based | ||||
| Au/Water | 10–30 | 0.0001–0.004 vol% | >120 h | [9] |
| Au/DI Water Ag/DI Water |
8.6–9.4 4–33 |
- | 80 h | [10] |
| Cu/Methanol | 25–75 | 0.1–10 wt% | 4320 h | [11] |
| Metal-oxide Based | ||||
| TiO2/Water | 30–50 | 0.5–2.5 vol% | 168 h | [12] |
| CuO/Water | 30–50 | 2–4 vol% | 168 h | [13] |
| Al2O3/DI Water | 20 | 0.05–0.25 kg/m3 | - | [14] |
| Al2O3/Water | 30 | 0.5–2 vol% | 480 h | [15] |
| ZnO/EG | 10–20 | 1–5 vol% | 6 h | [16] |
| Carbon Based | ||||
| MWCNT/Water | Outer D: 50–80 Inner D: 5–15 L: 10–20 (µm) |
0.0001–0.03 vol% | <120 h | [9] |
| COOH-CNT/DI Water | D: 12–14 L: 1.5–2 (µm) |
0.1–0.3 wt% | 504 h | [17] |
| CNT/Decane | D: 15 L: 30 (µm) |
0.1 vol% | 1440 h | [18] |
| MWCNT/Water | Outer D: 50–80 Inner D: 5–15 L: 10–20 (µm) |
0.1–0.5 vol% | 1080 h | [19] |
| MWCNT/Kapok Seed Oil | D: 15.79–19.21 | 0.1 wt% | <720 h | [20] |
| Hybrid | ||||
| Ag-MWCNT/DI Water | Ag: 50 MWCNT: 20–30 |
0.01–0.05 wt% | 48 h | [7] |
| MgO-SWCNT/EG | - | 0.05–1 vol% | - | [21] |
| Cu-TiO2/EG-Water | Cu: 40–60 TiO2: <25 |
0.2–0.8 wt% | - | [22] |
| ZnO-SWCNT/EG-Water | ZnO: 10–30 SWCNT – Outer D: 1–2 Inner D: 0.8–1.6 |
0.05–1.6 vol% | - | [23] |
| ZnO-MWCNT/EG-Water | ZnO: 10–30 MWCNT - Outer D: 5–15 Inner D: 3–5 |
0.02–1 vol% | 240 h | [24] |
| Au-TiO2/DI Water Au-Ag/DI Water Au-Al/DI Water Au-Ni/DI Water |
Au: 45–85 TiO2: 15–40 Ag:30–65 Al: 50–75 Ni: 25–65 |
0.05–3 vol% | <168 h | [25] |
| Ag-Fe3O4/DI Water | 21 | 0.015 | - | [26] |
3.1. Magnetic Stirrer
3.2. Surfactants
3.3. Sonification
References
- Sridhara, V.; Satapathy, L.N. Effect of Nanoparticles on Thermal Properties Enhancement in Different Oils-A Review. Crit. Rev. Solid State Mater. Sci. 2015, 40, 399–424.
- Esfahani, M.R.; Languri, E.M.; Nunna, M.R. Effect of particle size and viscosity on thermal conductivity enhancement of graphene oxide nanofluid. Int. Commun. Heat Mass Transf. 2016, 76, 308–315.
- Asadi, A.; Pourfattah, F.; Miklós Szilágyi, I.; Afrand, M.; Żyła, G.; Seon Ahn, H.; Wongwises, S.; Minh Nguyen, H.; Arabkoohsar, A.; Mahian, O. Effect of sonication characteristics on stability, thermophysical properties, and heat transfer of nanofluids: A comprehensive review. Ultrason. Sonochem. 2019, 58, 104701.
- Babar, H.; Sajid, M.U.; Ali, H.M. Viscosity of hybrid nanofluids: A critical review. Therm. Sci. 2019, 23, 1713–1754.
- Yu, W.; France, D.; Choi, S.; Routbort, J. Review and Assessment of Nanofluid Technology for Transportation and other Applications; Argonne National Lab. (ANL): Argonne, IL, USA, 2007.
- Yu, W.; France, D.M.; Routbort, J.L.; Choi, S.U.S. Review and comparison of nanofluid thermal conductivity and heat transfer enhancements. Heat Transf. Eng. 2008, 29, 432–460.
- Sun, B.; Zhang, Y.; Yang, D.; Li, H. Experimental study on heat transfer characteristics of hybrid nanofluid impinging jets. Appl. Therm. Eng. 2019, 151, 556–566.
- Ali, H.M.; Babar, H.; Shah, T.R.; Sajid, M.U.; Qasim, M.A.; Javed, S. Preparation Techniques of TiO2 Nanofluids and Challenges: A Review. Appl. Sci. 2018, 8, 587.
- Beicker, C.L.L.; Amjad, M.; Bandarra Filho, E.P.; Wen, D. Experimental study of photothermal conversion using gold/water and MWCNT/water nanofluids. Sol. Energy Mater. Sol. Cells 2018, 188, 51–65.
- Li, D.; Fang, W.; Feng, Y.; Geng, Q.; Song, M. Stability properties of water-based gold and silver nanofluids stabilized by cationic gemini surfactants. J. Taiwan Inst. Chem. Eng. 2019, 97, 458–465.
- Graves, J.E.; Latvytė, E.; Greenwood, A.; Emekwuru, N.G. Ultrasonic preparation, stability and thermal conductivity of a capped copper-methanol nanofluid. Ultrason. Sonochem. 2019, 55, 25–31.
- Wongcharee, K.; Chuwattanakul, V.; Eiamsa-ard, S. Heat transfer of swirling impinging jets with TiO2-water nanofluids. Chem. Eng. Process. Process Intensif. 2017, 114, 16–23.
- Wongcharee, K.; Chuwattanakul, V.; Eiamsa-ard, S. Influence of CuO/water nanofluid concentration and swirling flow on jet impingement cooling. Int. Commun. Heat Mass Transf. 2017, 88, 277–283.
- Al-Zuhairy, R.C.; Kareem, Z.S.; Abdulhadi, A.A. Al2O3-water nanofluid heat transfer enhancement of a twin impingement jet. Case Stud. Therm. Eng. 2020, 19, 100626.
- Lv, J.; Chang, S.; Hu, C.; Bai, M.; Wang, P.; Zeng, K. Experimental investigation of free single jet impingement using Al2O3-water nanofluid. Int. Commun. Heat Mass Transf. 2017, 88, 126–135.
- Yu, W.; Xie, H.; Chen, L.; Li, Y. Investigation of thermal conductivity and viscosity of ethylene glycol based ZnO nanofluid. Thermochim. Acta 2009, 491, 92–96.
- Sarafraz, M.M.; Hormozi, F.; Nikkhah, V. Thermal performance of a counter-current double pipe heat exchanger working with COOH-CNT/water nanofluids. Exp. Therm. Fluid Sci. 2016, 78, 41–49.
- Xie, H.; Lee, H.; Youn, W.; Choi, M. Nanofluids containing multiwalled carbon nanotubes and their enhanced thermal conductivities. J. Appl. Phys. 2003, 94, 4967–4971.
- Muruganandam, M.; Mukesh Kumar, P.C. Experimental analysis on internal combustion engine using MWCNT/water nanofluid as a coolant. In Proceedings of the Materials Today: Proceedings; Elsevier Ltd.: Amsterdam, The Netherlands, 2020; Volume 21, pp. 248–252.
- Hameed, A.; Mukhtar, A.; Shafiq, U.; Khan, M.S.; Rashid, T.; Cornelius, B. Experimental investigation on synthesis, characterization, stability, thermo-physical properties and rheological behavior of MWCNTs-kapok seed oil based nanofluid. J. Mol. Liq. 2019, 277, 812–824.
- Hemmat Esfe, M.; Alirezaie, A.; Rejvani, M. An applicable study on the thermal conductivity of SWCNT-MgO hybrid nanofluid and price-performance analysis for energy management. Appl. Therm. Eng. 2017, 111, 1202–1210.
- Leong, K.Y.; Razali, I.; Ku Ahmad, K.Z.; Ong, H.C.; Ghazali, M.J.; Abdul Rahman, M.R. Thermal conductivity of an ethylene glycol/water-based nanofluid with copper-titanium dioxide nanoparticles: An experimental approach. Int. Commun. Heat Mass Transf. 2018, 90, 23–28.
- Hemmat Esfe, M.; Abbasian Arani, A.A.; Firouzi, M. Empirical study and model development of thermal conductivity improvement and assessment of cost and sensitivity of EG-water based SWCNT-ZnO (30%:70%) hybrid nanofluid. J. Mol. Liq. 2017, 244, 252–261.
- Hemmat Esfe, M.; Esfandeh, S.; Saedodin, S.; Rostamian, H. Experimental evaluation, sensitivity analyzation and ANN modeling of thermal conductivity of ZnO-MWCNT/EG-water hybrid nanofluid for engineering applications. Appl. Therm. Eng. 2017, 125, 673–685.
- Baniamerian, Z.; Mashayekhi, M.; Mehdipour, R. Evaporative Behavior of Gold-Based Hybrid Nanofluids. J. Thermophys. Heat Transf. 2018, 32, 284–291.
- Siricharoenpanich, A.; Wiriyasart, S.; Srichat, A.; Naphon, P. Thermal cooling system with Ag/Fe3O4nanofluids mixture as coolant for electronic devices cooling. Case Stud. Therm. Eng. 2020, 20, 100641.
- Chakraborty, S.; Panigrahi, P.K. Stability of nanofluid: A review. Appl. Therm. Eng. 2020, 174, 115259.
- Chakraborty, S.; Sarkar, I.; Behera, D.K.; Pal, S.K.; Chakraborty, S. Experimental investigation on the effect of dispersant addition on thermal and rheological characteristics of TiO2 nanofluid. Powder Technol. 2017, 307, 10–24.
- Saleh, R.; Putra, N.; Wibowo, R.E.; Septiadi, W.N.; Prakoso, S.P. Titanium dioxide nanofluids for heat transfer applications. Exp. Therm. Fluid Sci. 2014, 52, 19–29.
- Mejia, J.; Valembois, V.; Piret, J.P.; Tichelaar, F.; Van Huis, M.; Masereel, B.; Toussaint, O.; Delhalle, J.; Mekhalif, Z.; Lucas, S. Are stirring and sonication pre-dispersion methods equivalent for in vitro toxicology evaluation of SiC and TiC? J. Nanopart. Res. 2012, 14, 815.
- Xia, G.; Jiang, H.; Liu, R.; Zhai, Y. Effects of surfactant on the stability and thermal conductivity of Al2O3/de-ionized water nanofluids. Int. J. Therm. Sci. 2014, 84, 118–124.
- Yang, L.; Chen, X.; Xu, M.; Du, K. Roles of surfactants and particle shape in the enhanced thermal conductivity of TiO2 nanofluids. AIP Adv. 2016, 6, 4962659.
- Choudhary, R.; Khurana, D.; Kumar, A.; Subudhi, S. Stability analysis of Al2O3/water nanofluids. J. Exp. Nanosci. 2017, 12, 140–151.
- Afzal, A.; Nawfal, I.; Mahbubul, I.M.; Kumbar, S.S. An overview on the effect of ultrasonication duration on different properties of nanofluids. J. Therm. Anal. Calorim. 2019, 135, 393–418.
- Ali, A.R.I.; Salam, B. A review on nanofluid: Preparation, stability, thermophysical properties, heat transfer characteristics and application. SN Appl. Sci. 2020, 2, 1636.





