Your browser does not fully support modern features. Please upgrade for a smoother experience.

Submitted Successfully!
Thank you for your contribution! You can also upload a video entry or images related to this topic.
For video creation, please contact our Academic Video Service.
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Sukumaran Anil | -- | 3685 | 2022-11-29 20:09:21 | | | |
| 2 | Lindsay Dong | + 2 word(s) | 3687 | 2022-12-01 01:45:08 | | |
Video Upload Options
We provide professional Academic Video Service to translate complex research into visually appealing presentations. Would you like to try it?
Cite
If you have any further questions, please contact Encyclopedia Editorial Office.
Anil, S. Potential Medical Applications of Chitooligosaccharides. Encyclopedia. Available online: https://encyclopedia.pub/entry/37174 (accessed on 30 May 2026).
Anil S. Potential Medical Applications of Chitooligosaccharides. Encyclopedia. Available at: https://encyclopedia.pub/entry/37174. Accessed May 30, 2026.
Anil, Sukumaran. "Potential Medical Applications of Chitooligosaccharides" Encyclopedia, https://encyclopedia.pub/entry/37174 (accessed May 30, 2026).
Anil, S. (2022, November 29). Potential Medical Applications of Chitooligosaccharides. In Encyclopedia. https://encyclopedia.pub/entry/37174
Anil, Sukumaran. "Potential Medical Applications of Chitooligosaccharides." Encyclopedia. Web. 29 November, 2022.
Copy Citation
Chitooligosaccharides (COSs), also known as chitosan oligomers or chitooligomers, are made up of chitosan with a degree of polymerization (DP) that is less than 20 and an average molecular weight (MW) that is lower than 3.9 kDa. COS can be produced through enzymatic conversions using chitinases, physical and chemical applications, or a combination of these strategies. COS is of significant interest for pharmacological and medical applications due to its increased water solubility and non-toxicity, with a wide range of bioactivities, including antibacterial, anti-inflammatory, anti-obesity, neuroprotective, anticancer, and antioxidant effects.
chitooligosaccharides
chitin
chitosan
anti-inflammatory
antioxidant
1. Introduction
Chitin, a mucopolysaccharide, is produced by many living organisms and is usually present in a complex with other polysaccharides and proteins in insects, crustaceans, arachnids, myriapods, nematodes, algae, and fungi [1]. Chitin is a linear polysaccharide composed of (1 → 4) linked 2-acetamido-2-deoxy-β-d-glucopyranosyl units and occurs naturally in three polymorphic forms with different orientations of the microfibrils, known as α-, β-, and γ-chitin [2]. Chitin has been the focus of numerous therapeutic uses in addition to serving as a precursor for producing chitosan and chitooligosaccharides. Chitin has several uses in food, agriculture, wastewater treatment, textiles, microbiology, nanotechnology, chemistry, and material science [3]. It is biodegradable and is also considered as a promising biomaterial for tissue engineering and stem cell technologies [4].
Chitosan [poly-(β-1/4)-2-amino-2-deoxy-D-glucopyranose] is a natural nontoxic linear polysaccharide biopolymer produced through the deacetylation of chitin [5]. Commercial chitosan is made by deacetylating naturally occurring chitin and is used in dietary supplements, organic fertilizers, and cosmetics [6][7]. Chitin and chitosan can be distinguished based on the degree of acetylation of the D-glucosamine units. Chitin includes over 70% acetylated units, whereas chitosan contains less than 30% acetylation. In the presence of organic acids, including formic acid, acetic acid, and ascorbic acid, chitosan forms salt and becomes water soluble [8]. Chitosan possesses three reactive functional groups, including an amino- or N-acetamide group and two primary and secondary hydroxyl groups at the C-2, C-3, and C-6 positions. The amino- or N-acetamide groups distinguish the structure and physicochemical properties of various chitosans [9]. The fraction of N-acetylated residues (FA), degree of polymerization (DP), molecular weight (MW), MW distribution, and pattern or sequence of N-acetylation (PA) can be used to classify chitosan [10].
Chitosans with a DP of less than 20 and an average MW of less than 3.9 kDa are called chitooligosaccharides (COSs), chitosan oligomers, or chitooligomers [11][12]. Their characteristics, such as low molecular weight, low polymerization degree, and high water solubility, are superior to those of chitin and chitosan [11]. Chitooligosaccharide (COS) has a variety of biological activities and many potential uses in multiple fields, such as medicine, cosmetics, food, and agriculture [13]. In addition, chemical methods using acid, hydrogen peroxide (H2O2), or sodium nitrite (NaNO2) are also used to extract COS. There are multiple ways to obtain chitosan oligomers. These methods are categorized as enzymatic, physical, or chemical depolymerizations [14]. Chemical methods using acid [15][16], H2O2 [17], or NaNO2 [18] physical methods, such as hydrothermal [19], microwave [20], ultra-sonication [21], and gamma rays [22]. Among the chemical methods for the hydrolysis of chitosan, acid hydrolysis is probably the best known. The enzymatic depolymerization of chitosan is characterized by the enzymes’ selective cleavage of chitosan glycosidic bonds [23].
While hetero-chitooligosaccharides combine oligomers with varying DP, deacetylation degree (DD), and acetylation patterns (position of the N-acetyl residues in the chain), homo-chitooligosaccharides are oligomers made exclusively of either glucosamine (GlcN) or N-Acetylglucosamine (GlcNAc) units [3][24]. The attractive bioactive properties of COS make them suitable for various biological applications. A low molecular weight (1.5 kDa) results in aqueous solubility across a broad pH range and easy absorption through epithelial cells [10]. The physical and biological activities of chitosan and its oligomers are governed primarily by the DP, MW, and DD [3][25][26]. The most significant property of COS is its water solubility or solubility in physiological pH due to the freely accessible amino groups on the shorter chains [27]. All heterogeneous COS with a DP < 10 and DD between 50 and 100 % are completely soluble over the pH range extending from neutral to slightly alkaline, whereas commercially available chitosan with a higher DP precipitates out at such pH values [28].
COS exhibits superior solubility over a more extended pH range and at relatively higher concentrations than the corresponding chitosan, exhibiting exciting biological properties. Depending on their size, they are also soluble in other solvents, such as dimethyl sulfoxide, dimethylformamide, water, and alcohol. There has been a growing interest in modifying these oligomers to expand their applications. A low molecular weight (1.5 kDa) confers water solubility across a broad pH range and the capacity to be swiftly absorbed by epithelial cells. A wide variety of cells, including neuronal, embryonic, and bone-marrow-derived stem cells, can grow in COS with a degree of deacetylation (DD) > 85%, which is more compatible with cell growth than chitosan with lower DD levels [29]. The fabrication of compatible membrane materials, in which hydrogels are the most thoroughly investigated, is a key biomedical use of COS. Hydrogels are frequently used for therapeutic purposes, including tissue engineering and drug delivery, due to their propensity to expand in aqueous and biological fluids [30].
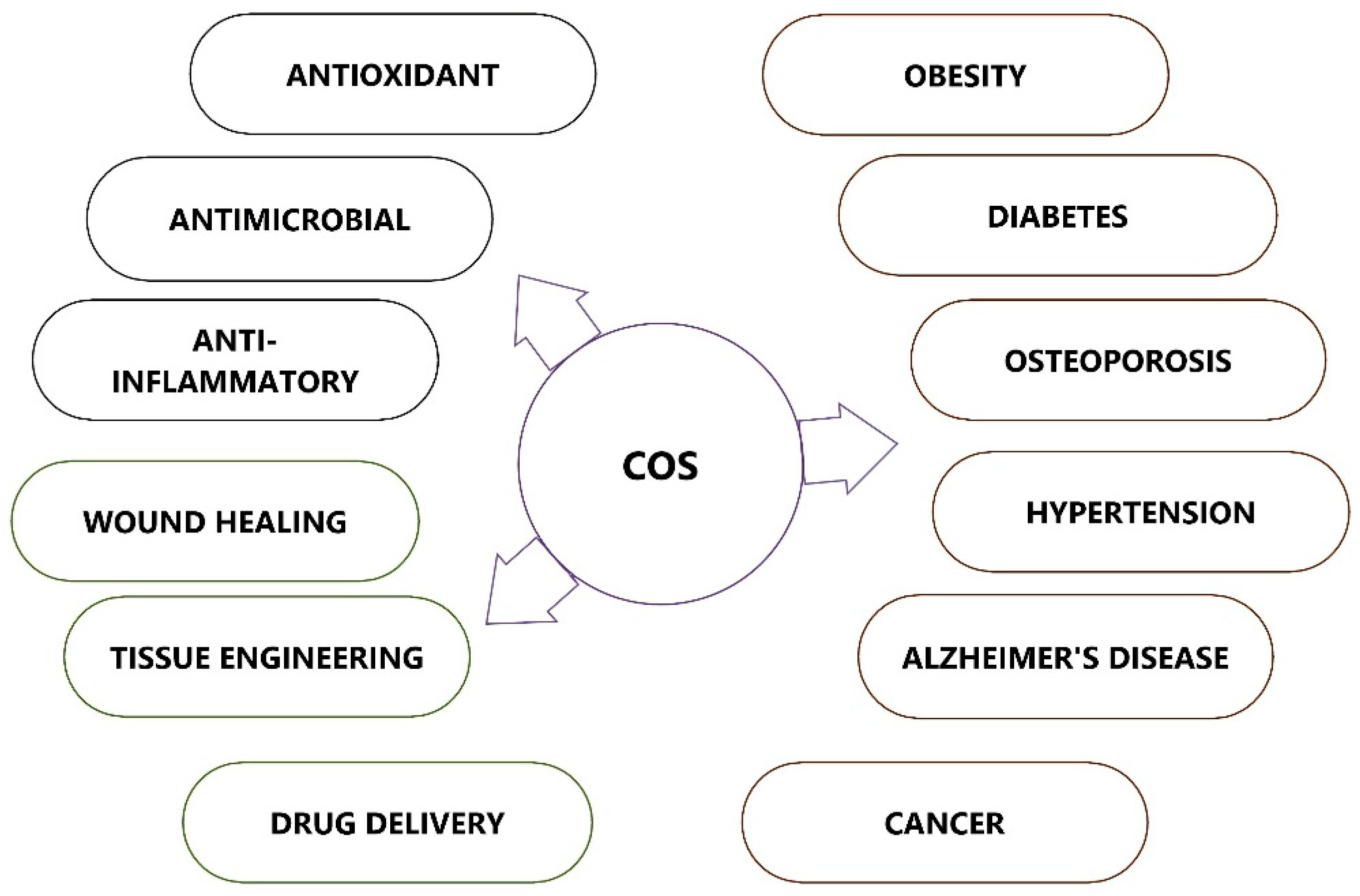
2. Therapeutic Applications of Chitooligosaccharides
COS possesses many biological activities and promising applications in multiple fields, such as medicine, cosmetics, food, and agriculture (Figure 1). COS, which is recognized as low MW and water soluble, is in much greater demand than its precursor molecule chitosan.

Figure 1. Therapeutic applications of chitooligosaccharides (COSs).
2.1. Chitooligosaccharides as Antioxidant Agents
Antioxidants can scavenge free radicals and protect the human body from highly toxic reactive oxygen species (ROS), slowing the progression of many chronic diseases. The ability of chitosan and its derivatives to scavenge free radicals and prevent oxidative damage by interrupting radial chain reactions is well established. Compared to chitosan, COS and its derivatives have better antioxidant properties [31][32]. COS antioxidant or radical-scavenging properties are primarily determined by their molecular weights and DD [31][33][34]. Several chronic diseases, such as cardiovascular diseases, atherogenesis, cancer, and Parkinson’s disease, are all linked to oxidative stress [35]. The excessive generation of ROS damages proteins, lipids, and DNA, leading to inflammation, tissue degeneration, and cellular apoptosis. ROS plays a vital role in the wound healing process at low concentrations, and an excessive level of reactive oxygen species can hinder wound healing by stimulating processes, such as inflammation and fibrosis [36].
Though the molecular mechanism underlying COS scavenging activity is unknown, the presence of hydroxyl and free amino groups in COS is thought to be responsible for its antioxidant activity. Antioxidants are noted for their positive benefits on health by protecting cells from the harmful effects of oxidation. It was demonstrated that COS and its derivatives have a high overall reducing power and can effectively remove hydroxyl radicals and superoxide anions [37]. The COS scavenging mechanism is believed to be based on the reaction of hydroxyl and superoxide anion radicals with active hydrogen atoms in COS, producing stable macromolecule radicals [32][38]. As COS can provide positrons to free radicals and transform them into more stable products, it can interrupt the chain reaction caused by free radicals [39].
2.2. Chitooligosaccharides as Antimicrobial Agents
COS is an effective antibacterial agent that inhibits the growth of various microbes, such as bacteria, fungi, and viruses [40][41]. The antimicrobial activity of COS depends on the molecular weight (MW), degree of polymerization (DP), and pattern of acetylation (PA), as well as the type of organism. Though the actual mechanism of the antimicrobial activity is not clearly understood, low-molecular-weight chitosan can penetrate bacterial cell walls, bind with DNA, and inhibit DNA transcription and mRNA synthesis [42]. Conversely, chitosan with a high molecular weight can bind to the negatively charged elements of the bacterial cell wall. As a result, it modifies the permeability of the cell, creates an impermeable layer around it, and prevents transport into the cell [43][44].
The acetylated sequences in the COS structure are necessary for antibacterial action, and COS with a greater number of acetylated sequences and fewer free amino groups possesses improved antimicrobial activity [45]. It was observed that Gram-positive and Gram-negative bacteria have varied responses to the antibacterial action of COS. Gram-negative bacteria have a negatively charged cell surface; hence, the positively charged amine group in COS can severely impede their growth. However, COS does not prevent the growth of Gram-positive bacteria very effectively. The interaction between positively charged amino groups of COS and negatively charged carboxylic acid groups of bacterial cell surfaces paves the way for the formation of polyelectrolyte complexes, resulting in the formation of an impermeable coating around the bacterial cell and the suppression of metabolic activity [46]. As a result of COS antibacterial properties, a lower pH value and more polymerization have also been deemed advantageous [47]. Chitooligosaccharides significantly block A. actinomycetemcomitan growth due to its effect on cell membrane permeability. The release of cellular components and the unregulated entry of substances from the surrounding environment results in microbial cell death [41].
COS antimicrobial activities are influenced by a variety of factors, including deoxycholic acid (DA) or dicetyl phosphate (DP), as well as other physicochemical properties and microorganism types [48]. COS can change the permeability characteristics of microbial cell membranes, preventing materials from entering or triggering the cell component’s leakage, ultimately leading to bacterial destruction. The bacterial envelope serves as the active site for COS, and membrane rupture may lead to the destruction of the microorganism. Chitosan penetration into bacterial DNA suppresses RNA transcription, an additional mechanism for killing microorganisms [49].
COS has antibacterial properties that promote tissue granulation and collagenase activity, two key factors in wound healing. Therefore, the antibacterial property of COS is responsible for its potential to promote wound healing. The antibacterial effects of COS are superior against both Gram-positive and Gram-negative microorganisms [50]. Antimicrobial action is enhanced with higher degrees of deacetylation than lower acetylation levels [51]. In a study conducted in relation to yeast, chitooligosaccharides with a degree of polymerization of 32 showed strong inhibitory efficacy [52].
2.3. Chitooligosaccharides as Anti-Inflammatory Agents
Inflammation plays a crucial role in the pathology of various diseases, such as chronic asthma, rheumatoid arthritis, multiple sclerosis, inflammatory bowel disease, psoriasis, and cancer [53][54][55][56]. It has been demonstrated that the oral administration of COS inhibits the activation of myeloperoxidase, cyclooxygenase (COX)-2, and inducible nitric oxide synthase (iNOS), as well as the levels of proinflammatory cytokines, such as interleukin (IL)-6 and tumor necrosis factor (TNF)-α [57]. COS exposure may inhibit the generation of numerous proinflammatory cytokines associated with lipopolysaccharide (LPS)-induced inflammation without impairing cell viability [58]. NF-κB (nuclear factor kappa-light-chain-enhancer of activated B cells) nucleus translocation was decreased as a result of the COS suppression of LPS-induced inflammatory gene expression, which attenuated an LPS-induced inflammatory response in vascular endothelial cells [59]. Ma et al. [60] hypothesized that COS could reduce the phosphorylation levels of mitogen-activated protein kinases (MAPKs) and activate NF-κB and activator protein 1 (AP-1) to decrease the LPS-induced interleukin 6 (IL-6) and TNF-α generation in macrophages. In a study on the effects of COS on synoviocytes in rabbit knee joints, it was found that activating AMPK reduced the expression of the enzymes iNOS and COX-2, thereby reducing synovial inflammation [61].
COS supplementation in the diets of animals has been shown to have a potent immunoenhancing effect. The results obtained from an evaluation of the COS effect on cyclophosphamide-induced immunosuppression by Mei et al. [62] showed that in mice treated orally with COS and cyclophosphamide, the delayed-type hypersensitivity reaction, macrophage phagocytosis activities, and levels of cytokines IL-2, IL-12, and interferon were significantly increased, while the production of IL-10 was decreased. COS exhibited protective benefits against ovalbumin-induced lung inflammation in induced asthmatic mouse models at a maximal dose of 16 mg/kg per day, with a significant decrease in mRNA expression and protein levels of IL-4, IL-5, IL-13, and TNF-α in lung tissue and bronchoalveolar lavage fluid [63]. The serum levels of IL-1, IL-2, IL-6, immunoglobulin (Ig) A, IgG, and IgM were all raised in early weaned pigs when COS was reintroduced into their diets. Increased cell-mediated immunity in response to weaning stress may be possible if COS can modulate the levels of specific cytokines and antibodies [64]. COS inhibits the release of nitric oxide in LPS-induced RAW 264.7 cells and BV-2 microglia, diminishes the glycerol-induced inflammatory response in rat kidneys, and decreases organ failure in LPS-induced sepsis [65][66].
2.4. Chitooligosaccharides and Their Anti-Obesity Activity
Obesity is a chronic trophic metabolic disorder primarily caused by an energy imbalance, resulting in the buildup of excess body fat. Obesity is associated with type 2 diabetes (T2D), hyperlipidemia, hypertension, cerebrovascular events, and cancer [67]. COS has excellent water solubility and lower viscosity than chitosan, and the intestine more easily absorbs it. In animal models, COS has also been shown to induce weight loss, lower triglyceride and cholesterol levels in serum, and prevent lipid buildup in hepatocytes and adipose tissues [68][69]. COS has demonstrated enhanced intestinal absorption, and studies have been performed to investigate its potential to reduce weight gain, blood triglyceride/cholesterol levels, and lipid buildup in the liver and adipose tissues [25][70][71]. COS anti-obesity activity has been the subject of numerous hypotheses, but its precise mechanism has not been fully elucidated.
When obese rats were fed COS, higher amounts of high-density lipoproteins or cardioprotective lipid-containing particles were discovered than in control rats. These lipoproteins and particles are responsible for removing excess cholesterol from tissues and transporting them to the liver [68]. Low-MW COS appears to be more effective in increasing plasma and hepatic lipoprotein lipase activity [72]. The evidence obtained using mouse models suggested that COS inhibited the expression of apolipoprotein B, which reduced the amount of cholesterol found in the serum [73]. Comparing the anti-obesity activity of COS and resistant starch (RS) to their combination, COS–RS, in rat models of induced obesity and dyslipidemia, COS–RS displayed the most significant fat and lipid-lowering benefits, followed by COS and RS [74]. Their examination of RNA sequencing revealed an increased conversion of cholesterol to bile acids. COS reduces triglycerides, total cholesterol, low-density lipoprotein cholesterol serum levels, and the expression of endoplasmic reticulum stress pathway-related factors (GRP78, GRP94, ATF4, and CHOP), and increases oxidative lipid catabolism [75]. This effect may be mediated by metabolites or directly by the molecules themselves. These findings underline the significance of developing COS as a potential drug derived from natural products for the prevention and treatment of obesity.
2.5. Chitooligosaccharides as Antidiabetic Agents
COS has demonstrated the potential to protect β cells from excessive glucose by promoting pancreatic cell proliferation, resulting in enhanced insulin production to lower glucose levels [76][77]. Reducing blood glucose levels and restoring normal insulin sensitivity were among the documented benefits of COS therapy for diabetic rats [77]. COS possesses antidiabetic properties and may reduce diabetes mellitus (DM) incidence by affecting the glucose–lipid metabolic balance and glycemic control [78]. Additionally, COS therapy can potentially improve the general health of diabetic rats, alleviate diabetic symptoms, bring blood glucose levels back to normal, and restore normal insulin sensitivity.
In addition, chitooligosaccharides can stimulate the multiplication of beta cells and restore the functional capacity of injured beta cells. COS has been shown to promote the overgrowth of beta cells and isolated pancreatic islet cells, in addition to increasing insulin release from pancreatic cells [77]. Chitooligosaccharides have the potential to significantly increase the rate of proliferation of pancreatic islet cells. In diabetic mice, it was also found that dietary COS reduced hyperglycemia by activating hepatic glucokinase and increasing peripheral tissue glucose uptake, as well as by increasing pancreatic insulin secretion and improving skeletal muscle glucose uptake [79]. In a study using streptozotocin (STZ)-induced diabetic rats, COS was able to treat hyperglycemia at a dose of 1000 mg/kg by lowering fasting serum glucose and insulin levels, thus improving O-glycosyltransferase (OGT), enhancing the index of insulin sensitivity, and reducing insulin resistance [80]. Additionally, COS significantly increases the amount of glycogen in the liver by increasing glucokinase, which facilitates the transfer of blood glucose into liver glycogen. Low-molecular-weight COS also increases the plasma adiponectin levels in prediabetic subjects [81].
2.6. Chitooligosaccharides in Osteoporosis
Supplemental chitooligosaccharides are beneficial in calcium-deficient states, such as osteoporosis. Supplementing animals’ diets with COS has been shown to improve calcium bioavailability in rat osteoporosis models induced by ovariectomy and concomitant low calcium intake [82]. A substantial reduction in the serum levels of inflammatory cytokines was observed after the oral administration of COS to older persons [83]. The mechanism behind the possible antiosteoporotic effect of COS was thought to be connected to its anti-inflammatory properties. Additionally, others have reported that COS suppressed the synthesis and expression of proinflammatory mediators in vitro [82][84].
The antiosteoporotic effect of COS may be partially explained by its anti-inflammatory effect via the downregulation of COX-2 expression levels, which provides additional evidence for the efficacy of selective COX-2 inhibition in preventing bone loss in estrogen-deficient animals and postmenopausal women [85]. The mineralization process and bone density depend on Ca2+, which provides structural support. There is evidence that COS causes an increase in bone calcium deposition [82][86].
2.7. The Antihypertensive Effect of COS
COS can effectively control hypertension by inhibiting renin or angiotensin-converting enzyme activity. COS is an angiotensin-converting enzyme (ACE) inhibitor since the active binding site of ACE is positively charged and contains hydrogen-bond acceptors and zinc as a cofactor. Hong et al. [87] evaluated the ACE inhibitory actions of COS with varying degrees of polymerization from 1 to 10 and found that the chitotriose (DP = 3) derivative was the most potent. Studies have shown that the DD is inversely proportional to the ACE inhibitory activity of COS or its derivatives [88][89]. Carboxylated and sulfated COS have been synthesized, and it has been observed that they possess a significantly greater inhibitory action against ACE than unmodified COS [88][89]. It is believed that an increase in negative charges on the molecule is responsible for the improved binding of this modified COS to the integral active site of the enzyme, thereby boosting its ACE inhibitory action. The aminoethyl-conjugated COS exhibited enhanced ACE inhibitory action due to the formation of hydrogen bonds, which promotes COS binding [90].
2.8. Chitooligosaccharides and Alzheimer’s Disease
The pathological changes in Alzheimer’s disease (AD) are caused by neuronal apoptosis, and neuronal protection is crucial in treating it [91]. A study conducted to explore the neuroprotective effect of COS against AD showed that COS significantly decreased amyloid beta-induced cell apoptosis by reducing the expression of caspase 3 and Bax/Bcl-2 ratio activation [92]. COS significantly reduced the neuronal damage caused by oxidative stress and glucose deprivation, and these findings imply that COS has the potential to serve as a neuroprotective agent against neurodegenerative diseases, such as Alzheimer’s disease [93]. The protein expression and acetylcholinesterase activity produced by amyloid β peptide in PC12 cells were inhibited by COS, which was revealed to be the first stage in the pathogenic cascade of AD [94].
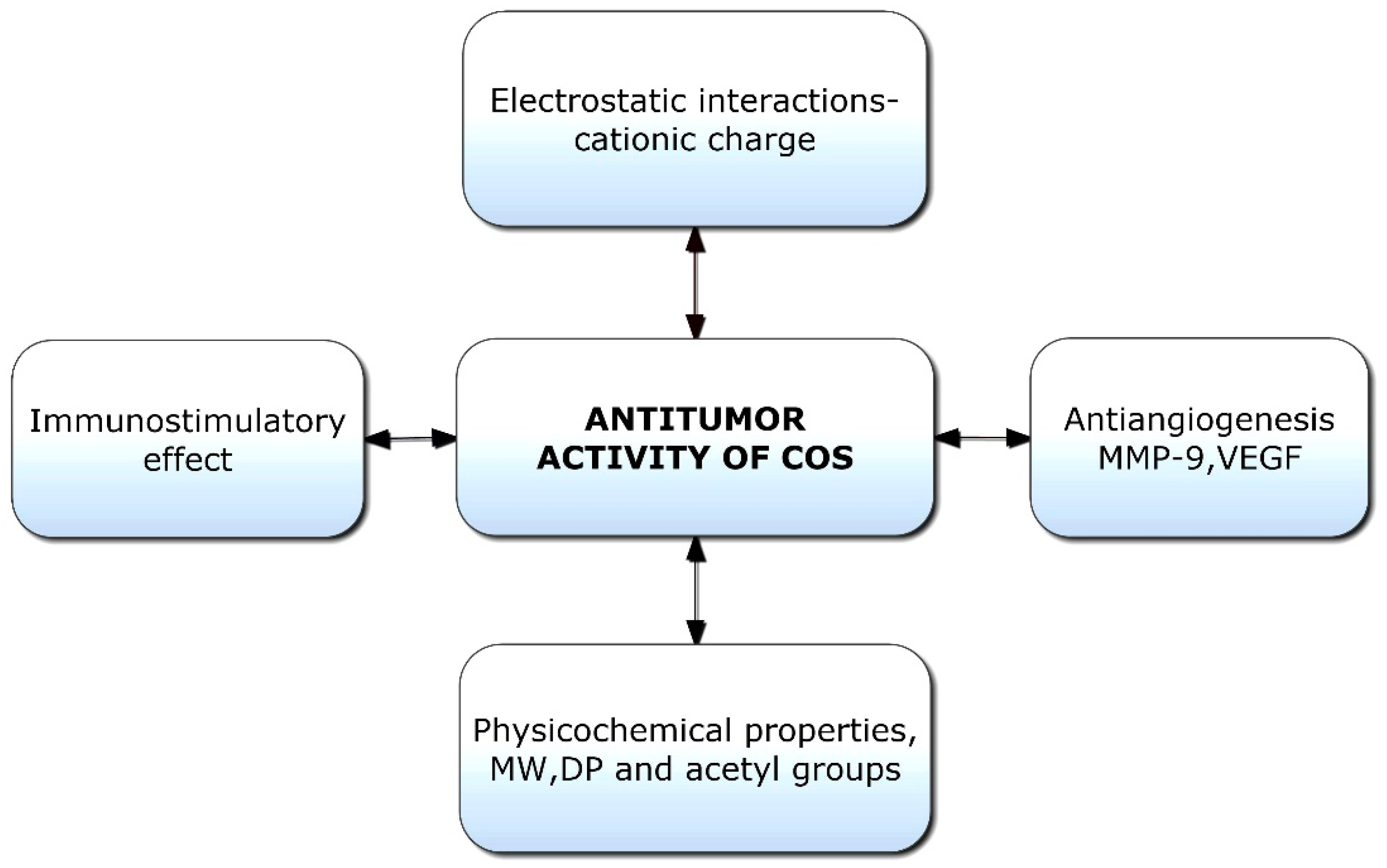
2.9. Chitooligosaccharides as Antitumor Agents
Chitooligosaccharides and their derivatives have demonstrated potent antitumor activity against human cancer cells [95]. Though limited information is available on the antitumor mechanisms of COS, some hypotheses have been proposed (Figure 2). Initially, the mechanism of anticancer activity was linked to the cationic character of COS; however, it was later hypothesized that relative molecular weight was also a crucial factor [96]. The tumor-inhibiting effect of COS is likely a result of its ability to induce lymphocyte cytokines via promoting T-cell proliferation. The antitumor mechanism of COS is essentially enhanced by acquired immunity by increasing T-cell differentiation to boost cytotoxicity and preserve T-cell activity [97]. COSs are naturally occurring polysaccharides with a cationic charge; they can promote the apoptosis of numerous cancer cells, including liver cancer, breast cancer, cervical cancer, kidney cancer, lung cancer, leukemia, and colorectal cancer cells [25].

Figure 2. Potential factors responsible for antitumor activities of chitooligosaccharides (COSs). VEGF: Vascular endothelial growth factor; MW: Molecular weight; DP: Degree of polymerization.
2.10. Chitooligosaccharides in Wound Healing
The overexpression of miR-27a and the activation of the transforming growth factor-beta (TGF-β)-1-Smad2/3 pathway are two potential mechanisms of COS in the acceleration of wound healing and tissue regeneration [98]. COS is combined with other biopolymers for wound healing due to its low molecular weight. In addition to its biological properties, COS can promote wound healing by enhancing a wound dressing’s water absorption, flexibility, and mechanical strength.
2.11. Chitooligosaccharides in Tissue Engineering
Cells, composite scaffolds, and signaling molecules are the three essential components for producing substituted tissues that repair, replace, or regenerate damaged tissues or organs. COS is comparable to glycosaminoglycans, a crucial part of the extracellular matrix of a cell (ECM). As it can create an environment that closely resembles the ECM, allowing for cell attachment and the preservation of growth factors, this makes it successful for scaffolds in tissue engineering applications [99]. In the creation of bone tissue engineering scaffolds, natural polymers, such as gelatin and COS, have been used. By regulating the genes that control osteoblast proliferation in bone tissues, COS has been shown to stimulate neuronal differentiation in PC-12 nerve cells [100]. Gelatin–COS scaffolds that were cultivated with mesenchymal stem cells (MSCs) obtained from bone marrow and capable of osteogenic differentiation demonstrated promising outcomes in the production of bone tissue [101]. Within two weeks of implantation, cell proliferation was observed inside the scaffolds; homogenous collagen distribution within the pores and calcium deposition on the scaffolds’ surfaces indicated that the cells had successfully proliferated [102].
2.12. Chitooligosaccharides in Drug Delivery
COS has excellent potential for usage in drug delivery systems (DDSs) due to its non-toxicity, biodegradability, and solubility in water [103]. Due to its solubility at physiological pH, the water solubility of COS is generally more effective for drug administration [104]. Recently, a novel alternative to red blood cells has been developed using a pectin-based COS–hydrogel microcapsule carrier designed to transport hemoglobin [105]. Oligosaccharides were used to extend the shelf life of these therapeutic drugs by several months. With COS of a 95% degree of deacetylation (DD) and an MW of 10 kDa, non-biodegradable polyethylene glycol (PEG) and cyclodextrin inclusion complexes are converted into hydrogels capable of transporting drugs.
MW and DD are influential parameters for modulating the actions of COS, offering a strategy to improve its efficacy in drug delivery systems (DDSs) [106]. COS is more significantly absorbed through negatively charged mucous membranes of tissues because it is more capable of binding under these situations [107]. DD also controls binding; at a low MW, the effect of DD on charge density is more substantial [28][108]. This may have a positive influence on the interaction of COS with oppositely charged copolymers or active medicines; nevertheless, the considerably stronger interactions can harm cell viability [109]. Therefore, when utilizing COS with a high DD in drug delivery, it is necessary to monitor and optimize the dosage rate to limit cytotoxic effects [106].
References
- Synowiecki, J.; Al-Khateeb, N.A. Production, properties, and some new applications of chitin and its derivatives. Crit. Rev. Food Sci. Nutr. 2003, 43, 145–171.
- Rudall, K.M. The chitin/protein complexes of insect cuticles. In Advances in Insect Physiology; Beament, J.W.L., Treherne, J.E., Wigglesworth, V.B., Eds.; Academic Press: Cambridge, MA, USA, 1963; Volume 1, pp. 257–313.
- Liaqat, F.; Eltem, R. Chitooligosaccharides and their biological activities: A comprehensive review. Carbohydr. Polym. 2018, 184, 243–259.
- Wan, A.C.; Tai, B.C. Chitin—A promising biomaterial for tissue engineering and stem cell technologies. Biotechnol. Adv. 2013, 31, 1776–1785.
- Lee, H.-W.; Choi, J.-W.; Han, D.P.; Lee, N.-W.; Park, S.; Yi, D.-H. Identification and production of constitutive chitosanase from bacillus sp. Hw-002. J. Microbiol. Biotechnol. 1996, 6, 12–18.
- Roy, S.; Chakraborty, T.; Begum, J.; Hasnain, M.S.; Nayak, A.K. Chapter 1-chitosan: A versatile biopolymer. In Chitosan in Biomedical Applications; Hasnain, M.S., Beg, S., Nayak, A.K., Eds.; Academic Press: Cambridge, MA, USA, 2022; pp. 1–11.
- Yeul, V.; Rayalu, S. Unprecedented chitin and chitosan: A chemical overview. J. Polym. Environ. 2012, 21, 606–614.
- Trombotto, S.; Ladaviere, C.; Delolme, F.; Domard, A. Chemical preparation and structural characterization of a homogeneous series of chitin/chitosan oligomers. Biomacromolecules 2008, 9, 1731–1738.
- Xia, W.; Liu, P.; Zhang, J.; Chen, J. Biological activities of chitosan and chitooligosaccharides. Food Hydrocoll. 2011, 25, 170–179.
- Aam, B.B.; Heggset, E.B.; Norberg, A.L.; Sorlie, M.; Varum, K.M.; Eijsink, V.G. Production of chitooligosaccharides and their potential applications in medicine. Mar. Drugs 2010, 8, 1482–1517.
- Lodhi, G.; Kim, Y.S.; Hwang, J.W.; Kim, S.K.; Jeon, Y.J.; Je, J.Y.; Ahn, C.B.; Moon, S.H.; Jeon, B.T.; Park, P.J. Chitooligosaccharide and its derivatives: Preparation and biological applications. Biomed. Res. Int. 2014, 2014, 654913.
- Mourya, V.; Inamdar, N.; Choudhari, Y.M. Chitooligosaccharides: Synthesis, characterization and applications. Polym. Sci. Ser. A 2011, 53, 583–612.
- Dong, H.; Wang, Y.; Zhao, L.; Zhou, J.; Xia, Q.; Jiang, L.; Fan, L. Purification of dp 6 to 8 chitooligosaccharides by nanofiltration from the prepared chitooligosaccharides syrup. Bioresour. Bioprocess. 2014, 1, 20.
- Jung, W.J.; Park, R.D. Bioproduction of chitooligosaccharides: Present and perspectives. Mar. Drugs 2014, 12, 5328–5356.
- Sato, K.; Saimoto, H.; Morimoto, M.; Shigemasa, Y. Depolymerization of chitin and chitosan under hydrothermal conditions. Sen’i Gakkaishi 2003, 59, 104–109.
- Xing, R.; Liu, S.; Yu, H.; Guo, Z.; Wang, P.; Li, C.; Li, Z.; Li, P. Salt-assisted acid hydrolysis of chitosan to oligomers under microwave irradiation. Carbohydr. Res. 2005, 340, 2150–2153.
- Wu, T.; Zivanovic, S.; Hayes, D.G.; Weiss, J. Efficient reduction of chitosan molecular weight by high-intensity ultrasound: Underlying mechanism and effect of process parameters. J. Agric. Food Chem. 2008, 56, 5112–5119.
- Yoksan, R.; Akashi, M.; Miyata, M.; Chirachanchai, S. Optimal gamma-ray dose and irradiation conditions for producing low-molecular-weight chitosan that retains its chemical structure. Radiat. Res. 2004, 161, 471–480.
- Domard, A.; Cartier, N. Glucosamine oligomers: 4. Solid state-crystallization and sustained dissolution. Int. J. Biol. Macromol. 1992, 14, 100–106.
- Einbu, A.; Varum, K.M. Depolymerization and de-n-acetylation of chitin oligomers in hydrochloric acid. Biomacromolecules 2007, 8, 309–314.
- Lin, F.; Jia, X.-G.; Lei, W.-X.; Li, Z.-J.; Zhang, T.-Y. Spectra analyses of chitosans degraded by hydrogen peroxide under optimal conditions. Spectrosc. Spectr. Anal. 2009, 29, 43–47.
- Morris, V.B.; Neethu, S.; Abraham, T.E.; Pillai, C.K.S.; Sharma, C.P. Studies on the condensation of depolymerized chitosans with DNA for preparing chitosan-DNA nanoparticles for gene delivery applications. J. Biomed. Mater. Res. B Appl. Biomater. 2009, 89, 282–292.
- Liang, S.; Sun, Y.; Dai, X. A review of the preparation, analysis and biological functions of chitooligosaccharide. Int. J. Mol. Sci. 2018, 19, 2197.
- Hamer, S.N.; Cord-Landwehr, S.; Biarnés, X.; Planas, A.; Waegeman, H.; Moerschbacher, B.M.; Kolkenbrock, S. Enzymatic production of defined chitosan oligomers with a specific pattern of acetylation using a combination of chitin oligosaccharide deacetylases. Sci. Rep. 2015, 5, 8716.
- Muanprasat, C.; Chatsudthipong, V. Chitosan oligosaccharide: Biological activities and potential therapeutic applications. Pharmacol. Ther. 2017, 170, 80–97.
- Naqvi, S.; Moerschbacher, B.M. The cell factory approach toward biotechnological production of high-value chitosan oligomers and their derivatives: An update. Crit. Rev. Biotechnol. 2017, 37, 11–25.
- Wang, Q.Z.; Chen, X.G.; Liu, N.; Wang, S.X.; Liu, C.S.; Meng, X.H.; Liu, C.G. Protonation constants of chitosan with different molecular weight and degree of deacetylation. Carbohydr. Polym. 2006, 65, 194–201.
- Maganti, N.; Venkat Surya, P.K.C.; Thein-Han, W.W.; Pesacreta, T.C.; Misra, R.D.K. Structure–process–property relationship of biomimetic chitosan-based nanocomposite scaffolds for tissue engineering: Biological, physico-chemical, and mechanical functions. Adv. Eng. Mater. 2011, 13, B108–B122.
- Ratanavaraporn, J.; Kanokpanont, S.; Tabata, Y.; Damrongsakkul, S. Growth and osteogenic differentiation of adipose-derived and bone marrow-derived stem cells on chitosan and chitooligosaccharide films. Carbohydr. Polym. 2009, 78, 873–878.
- Pella, M.C.G.; Lima-Tenorio, M.K.; Tenorio-Neto, E.T.; Guilherme, M.R.; Muniz, E.C.; Rubira, A.F. Chitosan-based hydrogels: From preparation to biomedical applications. Carbohydr. Polym. 2018, 196, 233–245.
- Zhao, D.; Wang, J.; Tan, L.; Sun, C.; Dong, J. Synthesis of n-furoyl chitosan and chito-oligosaccharides and evaluation of their antioxidant activity in vitro. Int. J. Biol. Macromol. 2013, 59, 391–395.
- Yang, Y.; Shu, R.; Shao, J.; Xu, G.; Gu, X. Radical scavenging activity of chitooligosaccharide with different molecular weights. Eur. Food Res. Technol. 2005, 222, 36–40.
- Laokuldilok, T.; Potivas, T.; Kanha, N.; Surawang, S.; Seesuriyachan, P.; Wangtueai, S.; Phimolsiripol, Y.; Regenstein, J.M. Physicochemical, antioxidant, and antimicrobial properties of chitooligosaccharides produced using three different enzyme treatments. Food Biosci. 2017, 18, 28–33.
- Tomida, H.; Fujii, T.; Furutani, N.; Michihara, A.; Yasufuku, T.; Akasaki, K.; Maruyama, T.; Otagiri, M.; Gebicki, J.M.; Anraku, M. Antioxidant properties of some different molecular weight chitosans. Carbohydr. Res. 2009, 344, 1690–1696.
- Garcia-Sanchez, A.; Miranda-Diaz, A.G.; Cardona-Munoz, E.G. The role of oxidative stress in physiopathology and pharmacological treatment with pro- and antioxidant properties in chronic diseases. Oxid. Med. Cell Longev. 2020, 2020, 2082145.
- Steen, E.H.; Wang, X.; Balaji, S.; Butte, M.J.; Bollyky, P.L.; Keswani, S.G. The role of the anti-inflammatory cytokine interleukin-10 in tissue fibrosis. Adv. Wound Care (New Rochelle) 2020, 9, 184–198.
- Kim, K.W.; Thomas, R.L. Antioxidative activity of chitosans with varying molecular weights. Food Chem. 2007, 101, 308–313.
- El-Sayed, S.T.; Omar, N.I.; El-Sayed, E.-S.M.; Shousha, W.G. Evaluation antioxidant and cytotoxic activities of novel chitooligosaccharides prepared from chitosan via enzymatic hydrolysis and ultrafiltration. J. Appl. Pharm. Sci. 2017, 7, 50–55.
- Feng, T.; Du, Y.; Li, J.; Wei, Y.; Yao, P. Antioxidant activity of half n-acetylated water-soluble chitosan in vitro. Eur. Food Res. Technol. 2006, 225, 133.
- Kendra, D.F.; Hadwiger, L.A. Characterization of the smallest chitosan oligomer that is maximally antifungal to Fusarium solani and elicits pisatin formation in Pisum sativum. Exp. Mycol. 1984, 8, 276–281.
- Choi, B.K.; Kim, K.Y.; Yoo, Y.J.; Oh, S.J.; Choi, J.H.; Kim, C.Y. In vitro antimicrobial activity of a chitooligosaccharide mixture against Actinobacillus actinomycetemcomitans and Streptococcus mutans. Int. J. Antimicrob. Agents 2001, 18, 553–557.
- Goy, R.C.; Britto, D.d.; Assis, O.B.G. A review of the antimicrobial activity of chitosan. Polímeros 2009, 19, 241–247.
- Goy, R.C.; Morais, S.T.B.; Assis, O.B.G. Evaluation of the antimicrobial activity of chitosan and its quaternized derivative on E. Coli and S. Aureus growth. Rev. Bras. Farmacogn. 2016, 26, 122–127.
- Zheng, L.-Y.; Zhu, J.-F. Study on antimicrobial activity of chitosan with different molecular weights. Carbohydr. Polym. 2003, 54, 527–530.
- Sanchez, A.; Mengibar, M.; Rivera-Rodriguez, G.; Moerchbacher, B.; Acosta, N.; Heras, A. The effect of preparation processes on the physicochemical characteristics and antibacterial activity of chitooligosaccharides. Carbohydr. Polym. 2017, 157, 251–257.
- Busscher, H.J.; Engels, E.; Dijkstra, R.J.; van der Mei, H.C. Influence of a chitosan on oral bacterial adhesion and growth in vitro. Eur. J. Oral Sci. 2008, 116, 493–495.
- Li, K.; Xing, R.; Liu, S.; Qin, Y.; Yu, H.; Li, P. Size and ph effects of chitooligomers on antibacterial activity against Staphylococcus aureus. Int. J. Biol. Macromol. 2014, 64, 302–305.
- Chung, Y.C.; Su, Y.P.; Chen, C.C.; Jia, G.; Wang, H.L.; Wu, J.C.; Lin, J.G. Relationship between antibacterial activity of chitosan and surface characteristics of cell wall. Acta Pharm. Sin. 2004, 25, 932–936.
- Yan, D.; Li, Y.; Liu, Y.; Li, N.; Zhang, X.; Yan, C. Antimicrobial properties of chitosan and chitosan derivatives in the treatment of enteric infections. Molecules 2021, 26, 7136.
- Benchamas, G.; Huang, G.; Huang, S.; Huang, H. Preparation and biological activities of chitosan oligosaccharides. Trends Food Sci. Technol. 2021, 107, 38–44.
- Wang, Z.; Zheng, L.; Yang, S.; Niu, R.; Chu, E.; Lin, X. N-acetylchitooligosaccharide is a potent angiogenic inhibitor both in vivo and in vitro. Biochem. Biophys. Res. Commun. 2007, 357, 26–31.
- Ganan, M.; Lorentzen, S.B.; Agger, J.W.; Heyward, C.A.; Bakke, O.; Knutsen, S.H.; Aam, B.B.; Eijsink, V.G.H.; Gaustad, P.; Sorlie, M. Antifungal activity of well-defined chito-oligosaccharide preparations against medically relevant yeasts. PLoS ONE 2019, 14, e0210208.
- Anogeianaki, A.; Angelucci, D.; Cianchetti, E.; D’Alessandro, M.; Maccauro, G.; Saggini, A.; Salini, V.; Caraffa, A.; Tete, S.; Conti, F.; et al. Atherosclerosis: A classic inflammatory disease. Int. J. Immunopathol. Pharm. 2011, 24, 817–825.
- Mantovani, A. Cancer: Inflaming metastasis. Nature 2009, 457, 36–37.
- Ngo, D.-H.; Vo, T.-S.; Ngo, D.-N.; Kang, K.-H.; Je, J.-Y.; Pham, H.N.-D.; Byun, H.-G.; Kim, S.-K. Biological effects of chitosan and its derivatives. Food Hydrocoll. 2015, 51, 200–216.
- de Andrade, R.; de Araújo, N.K.; Torres-Rêgo, M.; Furtado, A.A.; Daniele-Silva, A.; de Souza Paiva, W.; de Medeiros Dantas, J.M.; da Silva, N.S.; da Silva-Júnior, A.A.; Ururahy, M.A.G.; et al. Production and characterization of chitooligosaccharides: Evaluation of acute toxicity, healing, and anti-inflammatory actions. Int. J. Mol. Sci. 2021, 22, 10631.
- Azuma, K.; Osaki, T.; Kurozumi, S.; Kiyose, M.; Tsuka, T.; Murahata, Y.; Imagawa, T.; Itoh, N.; Minami, S.; Sato, K.; et al. Anti-inflammatory effects of orally administered glucosamine oligomer in an experimental model of inflammatory bowel disease. Carbohydr. Polym. 2015, 115, 448–456.
- Jitprasertwong, P.; Khamphio, M.; Petsrichuang, P.; Eijsink, V.G.H.; Poolsri, W.; Muanprasat, C.; Rangnoi, K.; Yamabhai, M. Anti-inflammatory activity of soluble chito-oligosaccharides (chos) on vitd3-induced human thp-1 monocytes. PLoS ONE 2021, 16, e0246381.
- Li, Y.; Liu, H.; Xu, Q.S.; Du, Y.G.; Xu, J. Chitosan oligosaccharides block lps-induced o-glcnacylation of nf-kappab and endothelial inflammatory response. Carbohydr. Polym. 2014, 99, 568–578.
- Ma, P.; Liu, H.-T.; Wei, P.; Xu, Q.-S.; Bai, X.-F.; Du, Y.-G.; Yu, C. Chitosan oligosaccharides inhibit lps-induced over-expression of il-6 and tnf-α in raw264.7 macrophage cells through blockade of mitogen-activated protein kinase (mapk) and pi3k/akt signaling pathways. Carbohydr. Polym. 2011, 84, 1391–1398.
- Kunanusornchai, W.; Witoonpanich, B.; Tawonsawatruk, T.; Pichyangkura, R.; Chatsudthipong, V.; Muanprasat, C. Chitosan oligosaccharide suppresses synovial inflammation via ampk activation: An in vitro and in vivo study. Pharm. Res. 2016, 113, 458–467.
- Mei, Y.X.; Chen, H.X.; Zhang, J.; Zhang, X.D.; Liang, Y.X. Protective effect of chitooligosaccharides against cyclophosphamide-induced immunosuppression in mice. Int. J. Biol. Macromol. 2013, 62, 330–335.
- Chung, M.J.; Park, J.K.; Park, Y.I. Anti-inflammatory effects of low-molecular weight chitosan oligosaccharides in ige–antigen complex-stimulated rbl-2h3 cells and asthma model mice. Int. Immunopharmacol. 2012, 12, 453–459.
- Yin, Y.L.; Tang, Z.R.; Sun, Z.H.; Liu, Z.Q.; Li, T.J.; Huang, R.L.; Ruan, Z.; Deng, Z.Y.; Gao, B.; Chen, L.X.; et al. Effect of galacto-mannan-oligosaccharides or chitosan supplementation on cytoimmunity and humoral immunity in early weaned piglets. Asian-Australas. J. Anim. Sci. 2008, 21, 723–731.
- Yoon, H.J.; Moon, M.E.; Park, H.S.; Im, S.Y.; Kim, Y.H. Chitosan oligosaccharide (cos) inhibits lps-induced inflammatory effects in raw 264.7 macrophage cells. Biochem. Biophys. Res. Commun. 2007, 358, 954–959.
- Vo, T.S.; Ngo, D.H.; Ta, Q.V.; Wijesekara, I.; Kong, C.S.; Kim, S.K. Protective effect of chitin oligosaccharides against lipopolysaccharide-induced inflammatory response in bv-2 microglia. Cell Immunol. 2012, 277, 14–21.
- Franks, P.W.; McCarthy, M.I. Exposing the exposures responsible for type 2 diabetes and obesity. Science 2016, 354, 69–73.
- Huang, L.; Chen, J.; Cao, P.; Pan, H.; Ding, C.; Xiao, T.; Zhang, P.; Guo, J.; Su, Z. Anti-obese effect of glucosamine and chitosan oligosaccharide in high-fat diet-induced obese rats. Mar. Drugs 2015, 13, 2732–2756.
- Choi, E.H.; Yang, H.P.; Chun, H.S. Chitooligosaccharide ameliorates diet-induced obesity in mice and affects adipose gene expression involved in adipogenesis and inflammation. Nutr. Res. 2012, 32, 218–228.
- Chen, P.; Zhao, M.; Chen, Q.; Fan, L.; Gao, F.; Zhao, L. Absorption characteristics of chitobiose and chitopentaose in the human intestinal cell line caco-2 and everted gut sacs. J. Agric. Food Chem. 2019, 67, 4513–4523.
- Pan, H.; Fu, C.; Huang, L.; Jiang, Y.; Deng, X.; Guo, J.; Su, Z. Anti-obesity effect of chitosan oligosaccharide capsules (coscs) in obese rats by ameliorating leptin resistance and adipogenesis. Mar. Drugs 2018, 16, 198.
- Zhang, J.; Zhang, W.; Mamadouba, B.; Xia, W. A comparative study on hypolipidemic activities of high and low molecular weight chitosan in rats. Int. J. Biol. Macromol. 2012, 51, 504–508.
- Kao, C.H.; Hsiang, C.Y.; Ho, T.Y. Assessment of chitosan-affected metabolic response by peroxisome proliferator-activated receptor bioluminescent imaging-guided transcriptomic analysis. PLoS ONE 2012, 7, e34969.
- Shang, W.; Si, X.; Zhou, Z.; Wang, J.; Strappe, P.; Blanchard, C. Studies on the unique properties of resistant starch and chito-oligosaccharide complexes for reducing high-fat diet-induced obesity and dyslipidemia in rats. J. Funct. Foods 2017, 38, 20–27.
- Deng, X.; Ye, Z.; Cao, H.; Bai, Y.; Che, Q.; Guo, J.; Su, Z. Chitosan oligosaccharide ameliorated obesity by reducing endoplasmic reticulum stress in diet-induced obese rats. Food Funct. 2020, 11, 6285–6296.
- Karadeniz, F.; Kim, S.K. Antidiabetic activities of chitosan and its derivatives: A mini review. Adv. Food Nutr. Res. 2014, 73, 33–44.
- Liu, B.; Liu, W.S.; Han, B.Q.; Sun, Y.Y. Antidiabetic effects of chitooligosaccharides on pancreatic islet cells in streptozotocin-induced diabetic rats. World J. Gastroenterol. 2007, 13, 725–731.
- Zhu, D.; Yan, Q.; Liu, J.; Wu, X.; Jiang, Z. Can functional oligosaccharides reduce the risk of diabetes mellitus? FASEB J. 2019, 33, 11655–11667.
- Yuan, W.P.; Liu, B.; Liu, C.H.; Wang, X.J.; Zhang, M.S.; Meng, X.M.; Xia, X.K. Antioxidant activity of chito-oligosaccharides on pancreatic islet cells in streptozotocin-induced diabetes in rats. World J. Gastroenterol. 2009, 15, 1339–1345.
- Ju, C.; Yue, W.; Yang, Z.; Zhang, Q.; Yang, X.; Liu, Z.; Zhang, F. Antidiabetic effect and mechanism of chitooligosaccharides. Biol. Pharm. Bull. 2010, 33, 1511–1516.
- Kim, H.J.; Ahn, H.Y.; Kwak, J.H.; Shin, D.Y.; Kwon, Y.I.; Oh, C.G.; Lee, J.H. The effects of chitosan oligosaccharide (go2ka1) supplementation on glucose control in subjects with prediabetes. Food Funct. 2014, 5, 2662–2669.
- Jung, W.K.; Moon, S.H.; Kim, S.K. Effect of chitooligosaccharides on calcium bioavailability and bone strength in ovariectomized rats. Life Sci. 2006, 78, 970–976.
- Kim, H.M.; Hong, S.H.; Yoo, S.J.; Baek, K.S.; Jeon, Y.J.; Choung, S.Y. Differential effects of chitooligosaccharides on serum cytokine levels in aged subjects. J. Med. Food 2006, 9, 427–430.
- Pangestuti, R.; Bak, S.S.; Kim, S.K. Attenuation of pro-inflammatory mediators in lps-stimulated bv2 microglia by chitooligosaccharides via the mapk signaling pathway. Int. J. Biol. Macromol. 2011, 49, 599–606.
- Richards, J.B.; Joseph, L.; Schwartzman, K.; Kreiger, N.; Tenenhouse, A.; Goltzman, D. The effect of cyclooxygenase-2 inhibitors on bone mineral density: Results from the canadian multicentre osteoporosis study. Osteoporos. Int. 2006, 17, 1410–1419.
- Kim, S.; Park, P.; Jung, W.; Byun, H.; Mendis, E.; Cho, Y. Inhibitory activity of phosphorylated chitooligosaccharides on the formation of calcium phosphate. Carbohydr. Polym. 2005, 60, 483–487.
- Hong, S.-P.; Kim, M.-H.; Oh, S.-W.; Han, C.-K.; Kim, Y.-H. Ace inhibitory and antihypertensive effect of chitosan oligosaccharides in shr. Korean J. Food Sci. Technol. 1998, 30, 1476–1479.
- Park, P.J.; Je, J.Y.; Kim, S.K. Angiotensin i converting enzyme (ace) inhibitory activity of hetero-chitooligosaccharides prepared from partially different deacetylated chitosans. J. Agric. Food Chem. 2003, 51, 4930–4934.
- Huang, R.; Mendis, E.; Kim, S.K. Improvement of ace inhibitory activity of chitooligosaccharides (cos) by carboxyl modification. Bioorg. Med. Chem. 2005, 13, 3649–3655.
- Ngo, D.-N.; Qian, Z.-J.; Je, J.-Y.; Kim, M.-M.; Kim, S.-K. Aminoethyl chitooligosaccharides inhibit the activity of angiotensin converting enzyme. Process. Biochem. 2008, 43, 119–123.
- Vila, M.; Przedborski, S. Targeting programmed cell death in neurodegenerative diseases. Nat. Rev. Neurosci. 2003, 4, 365–375.
- Dai, X.; Hou, W.; Sun, Y.; Gao, Z.; Zhu, S.; Jiang, Z. Chitosan oligosaccharides inhibit/disaggregate fibrils and attenuate amyloid beta-mediated neurotoxicity. Int. J. Mol. Sci. 2015, 16, 10526–10536.
- Xu, Y.; Zhang, Q.; Yu, S.; Yang, Y.; Ding, F. The protective effects of chitooligosaccharides against glucose deprivation-induced cell apoptosis in cultured cortical neurons through activation of pi3k/akt and mek/erk1/2 pathways. Brain Res. 2011, 1375, 49–58.
- Lee, S.H.; Park, J.S.; Kim, S.K.; Ahn, C.B.; Je, J.Y. Chitooligosaccharides suppress the level of protein expression and acetylcholinesterase activity induced by abeta25-35 in pc12 cells. Bioorg. Med. Chem. Lett. 2009, 19, 860–862.
- Park, J.K.; Chung, M.J.; Choi, H.N.; Park, Y.I. Effects of the molecular weight and the degree of deacetylation of chitosan oligosaccharides on antitumor activity. Int. J. Mol. Sci. 2011, 12, 266–277.
- Shen, K.T.; Chen, M.H.; Chan, H.Y.; Jeng, J.H.; Wang, Y.J. Inhibitory effects of chitooligosaccharides on tumor growth and metastasis. Food Chem. Toxicol. 2009, 47, 1864–1871.
- Zou, P.; Yang, X.; Zhang, Y.; Du, P.; Yuan, S.; Yang, D.; Wang, J. Antitumor effects of orally and intraperitoneally administered chitosan oligosaccharides (coss) on s180-bearing/residual mouse. J. Food Sci. 2016, 81, H3035–H3042.
- Okamoto, Y.; Watanabe, M.; Miyatake, K.; Morimoto, M.; Shigemasa, Y.; Minami, S. Effects of chitin/chitosan and their oligomers/monomers on migrations of fibroblasts and vascular endothelium. Biomaterials 2002, 23, 1975–1979.
- Ragetly, G.R.; Griffon, D.J.; Lee, H.-B.; Fredericks, L.P.; Gordon-Evans, W.; Chung, Y.S. Effect of chitosan scaffold microstructure on mesenchymal stem cell chondrogenesis. Acta Biomater. 2010, 6, 1430–1436.
- Yang, Y.; Liu, M.; Gu, Y.; Lin, S.; Ding, F.; Gu, X. Effect of chitooligosaccharide on neuronal differentiation of pc-12 cells. Cell Biol. Int. 2009, 33, 352–356.
- Ratanavaraporn, J.; Damrongsakkul, S.; Kanokpanont, S.; Yamamoto, M.; Tabata, Y. Osteogenic differentiation of bone-marrow-derived stem cells cultured with mixed gelatin and chitooligosaccharide scaffolds. J. Biomater. Sci. Polym. Ed. 2011, 22, 1083–1098.
- Li, J.; Chen, Y.; Yin, Y.; Yao, F.; Yao, K. Modulation of nano-hydroxyapatite size via formation on chitosan–gelatin network film in situ. Biomaterials 2007, 28, 781–790.
- Park, H.H.; Ko, S.C.; Oh, G.W.; Jang, Y.M.; Kim, Y.M.; Park, W.S.; Choi, I.W.; Jung, W.K. Characterization and biological activity of pva hydrogel containing chitooligosaccharides conjugated with gallic acid. Carbohydr. Polym. 2018, 198, 197–205.
- Sultankulov, B.; Berillo, D.; Sultankulova, K.; Tokay, T.; Saparov, A. Progress in the development of chitosan-based biomaterials for tissue engineering and regenerative medicine. Biomolecules 2019, 9, 470.
- Cherwin, A.; Namen, S.; Rapacz, J.; Kusik, G.; Anderson, A.; Wang, Y.; Kaltchev, M.; Schroeder, R.; O’Connell, K.; Stephens, S.; et al. Design of a novel oxygen therapeutic using polymeric hydrogel microcapsules mimicking red blood cells. Pharmaceutics 2019, 11, 583.
- Tabassum, N.; Ahmed, S.; Ali, M.A. Chitooligosaccharides and their structural-functional effect on hydrogels: A review. Carbohydr. Polym. 2021, 261, 117882.
- Thanou, M.; Florea, B.I.; Geldof, M.; Junginger, H.E.; Borchard, G. Quaternized chitosan oligomers as novel gene delivery vectors in epithelial cell lines. Biomaterials 2002, 23, 153–159.
- Sacco, P.; Cok, M.; Asaro, F.; Paoletti, S.; Donati, I. The role played by the molecular weight and acetylation degree in modulating the stiffness and elasticity of chitosan gels. Carbohydr. Polym. 2018, 196, 405–413.
- Chae, S.Y.; Jang, M.K.; Nah, J.W. Influence of molecular weight on oral absorption of water soluble chitosans. J. Control. Release 2005, 102, 383–394.
More
Information
Subjects:
Medicine, Research & Experimental
Contributor
MDPI registered users' name will be linked to their SciProfiles pages. To register with us, please refer to https://encyclopedia.pub/register
:
View Times:
977
Revisions:
2 times
(View History)
Update Date:
01 Dec 2022
Notice
You are not a member of the advisory board for this topic. If you want to update advisory board member profile, please contact office@encyclopedia.pub.
OK
Confirm
Only members of the Encyclopedia advisory board for this topic are allowed to note entries. Would you like to become an advisory board member of the Encyclopedia?
Yes
No
${ textCharacter }/${ maxCharacter }
Submit
Cancel
Back
Comments
${ item }
|
More
No more~
There is no comment~
${ textCharacter }/${ maxCharacter }
Submit
Cancel
${ selectedItem.replyTextCharacter }/${ selectedItem.replyMaxCharacter }
Submit
Cancel
Confirm
Are you sure to Delete?
Yes
No





