Your browser does not fully support modern features. Please upgrade for a smoother experience.

Submitted Successfully!
Thank you for your contribution! You can also upload a video entry or images related to this topic.
For video creation, please contact our Academic Video Service.
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Haining Ji | -- | 1128 | 2022-11-22 10:17:23 | | | |
| 2 | Sirius Huang | Meta information modification | 1128 | 2022-11-23 02:37:39 | | |
Video Upload Options
We provide professional Academic Video Service to translate complex research into visually appealing presentations. Would you like to try it?
Cite
If you have any further questions, please contact Encyclopedia Editorial Office.
Zhao, Y.; Ji, H.; Lu, M.; Tao, J.; Ou, Y.; Wang, Y.; Chen, Y.; Huang, Y.; Wang, J.; Mao, Y. Thermochromic Smart Windows Assisted by Photothermal Nanomaterials. Encyclopedia. Available online: https://encyclopedia.pub/entry/35819 (accessed on 25 May 2026).
Zhao Y, Ji H, Lu M, Tao J, Ou Y, Wang Y, et al. Thermochromic Smart Windows Assisted by Photothermal Nanomaterials. Encyclopedia. Available at: https://encyclopedia.pub/entry/35819. Accessed May 25, 2026.
Zhao, Yong, Haining Ji, Mingying Lu, Jundong Tao, Yangyong Ou, Yi Wang, Yongxing Chen, Yan Huang, Junlong Wang, Yuliang Mao. "Thermochromic Smart Windows Assisted by Photothermal Nanomaterials" Encyclopedia, https://encyclopedia.pub/entry/35819 (accessed May 25, 2026).
Zhao, Y., Ji, H., Lu, M., Tao, J., Ou, Y., Wang, Y., Chen, Y., Huang, Y., Wang, J., & Mao, Y. (2022, November 22). Thermochromic Smart Windows Assisted by Photothermal Nanomaterials. In Encyclopedia. https://encyclopedia.pub/entry/35819
Zhao, Yong, et al. "Thermochromic Smart Windows Assisted by Photothermal Nanomaterials." Encyclopedia. Web. 22 November, 2022.
Copy Citation
Thermochromic smart windows are optical devices that can regulate their optical properties actively in response to external temperature changes. Due to their simple structures and as they do not require other additional energy supply devices, they have great potential in building energy-saving.
thermochromic
smart window
photothermal materials
phase change
1. Introduction
In recent years, with energy-saving and green development being vigorously promoted, smart windows have attracted much attention due to their outstanding energy-saving effect. The concept of “smart windows” was introduced by Granqvist in the 1980s [1]. They are energy-saving windows that regulate solar radiation by combining a dimming material [2][3][4] with a base material, such as glass. As optical devices, their core is the sensitive material attached to the glass. Under external excitation by light, electromagnetic radiation, and temperature, the sensitive materials will color or fade, thereby changing the windows’ colors and other optical properties so that they can automatically adjust the indoor temperature and light intensity according to the surrounding environment and ultimately achieve the purpose of saving energy consumption.
At present, most typical smart windows require an external power supply or heating device to dynamically tune the optical properties of materials in response to external stimuli. However, these additional devices have significant disadvantages, such as a complex structure, low lifespan, and high energy consumption, which also greatly hinder their commercial utilization. In contrast, solar-driven thermochromic smart windows can adjust the optical properties actively through the strong absorption of solar energy by photothermal materials, achieving a dual response to light and temperature. The features of low cost, simple structure, and low energy consumption are more conducive to large-scale applications in the future.
2. Thermochromic Materials for Smart Windows
There are many kinds of thermochromic materials that play an important role as substrates for smart windows. However, only those materials with phase change temperatures adjustable to room temperature (about 28 °C) can be used for building energy efficiency [5]. Therefore, the most widely used photothermal materials for smart windows are vanadium dioxide (VO2), thermochromic hydrogels, and liquid crystals, as shown in Figure 1.
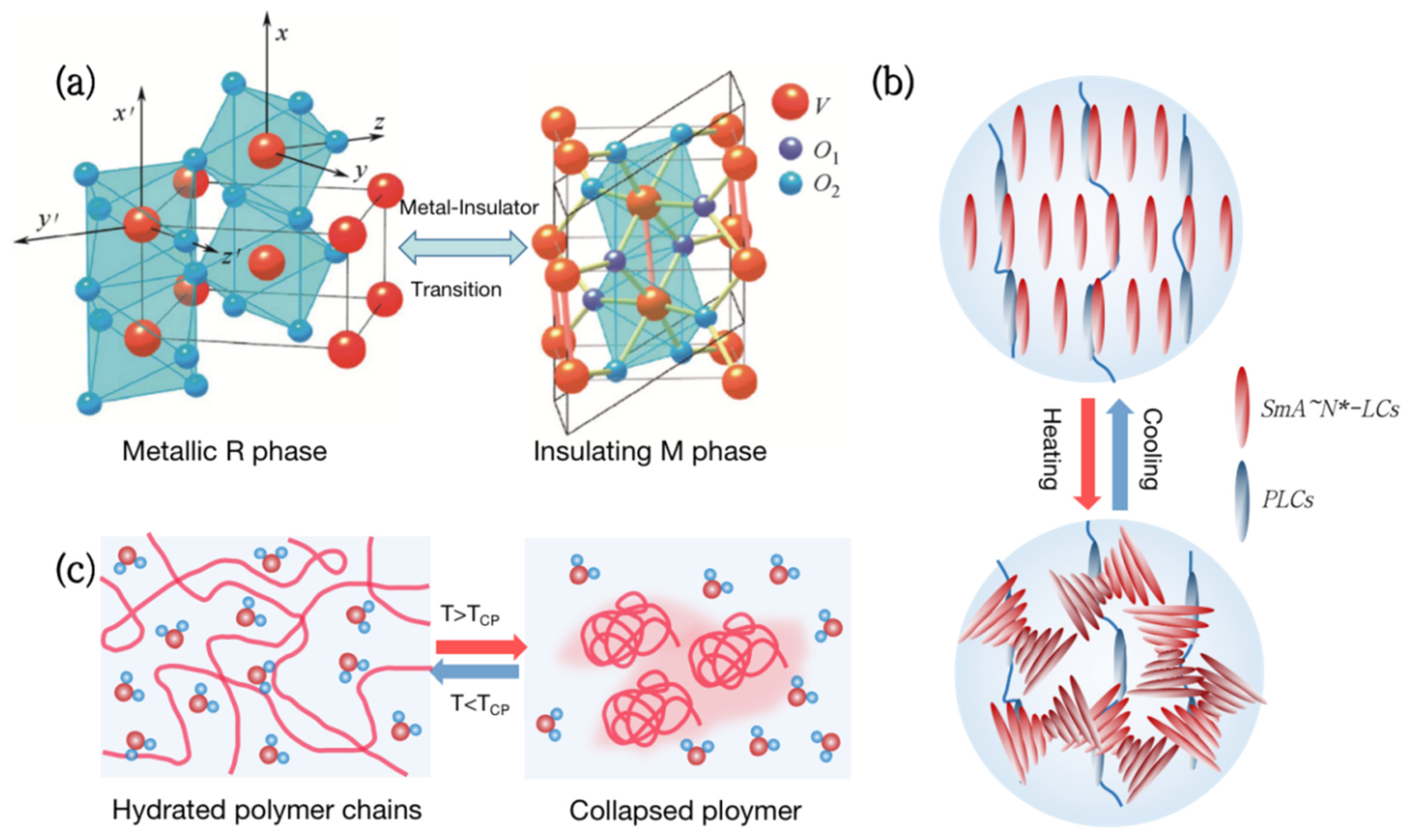
Figure 1. Types of thermochromic smart windows. (a) Metal-insulator phase transition (MIT) of vanadium dioxide (VO2). (b) Liquid crystal changes transmittance by adjusting molecular orientation in response to temperature. (c) Thermochromic hydrogels change transparency reversibly with temperature.
Vanadium dioxide (VO2) is a kind of metal-insulator phase transition (MIT) material, which is extensively utilized in thermochromic smart windows [6][7][8][9]. Its phase transition temperature is close to room temperature at 68 °C [10]. As the temperature rises, VO2 will undergo a phase transition from a semiconductor to a metal (Figure 1a), with a sudden change in infrared transmittance while keeping the visible transmittance unchanged [11]. Although VO2 films have great practical value, which are used in smart windows [12], photoelectric switches [13][14], and infrared stealth [15][16][17], the high phase transition temperature and poor optical properties are still problems to be solved.
Liquid Crystal (LC) is a special material between traditional liquids and crystalline solids. Its optical, electrical, and mechanical characteristics are anisotropic, and it can also be employed in smart windows [18][19][20][21]. From the molecular point of view, the thermochromism of Liquid Crystals can be achieved by anisotropic molecules responding to temperature and adjusting their orientation (Figure 1b). For example, the molecules inside the polymer network Liquid Crystals (PNLC) are arranged in order at low temperatures, and the refractive index of the liquid crystal and the polymer are the same, showing a highly transparent state. As the temperature rises, the liquid crystals transform into a blurred state due to the disorder of molecular orientation, and the transmittance, including near-infrared light, can change by more than 20%.
Hydrogels are both hydrophilic and hydrophobic and have been commonly employed in the field of smart windows [22][23][24][25]. Thermochromic hydrogels, such as poly(N-isopropylacrylamide) (PNIPAm) and hydroxypropyl methyl cellulose (HPMC), have a lower critical solution temperature (LCST) of around 32 °C to 40 °C. When the temperature is below the LCST, hydrophilic groups in the hydrogel form hydrogen bonds with water molecules, presenting a highly transparent state. Above the LCST, the hydrogen bonds are broken and polymers aggregate, resulting in a significant decrease in transmittance in the entire spectral range [26] (Figure 1c). Thermochromic hydrogels can reversibly change transparency with an increase in temperature, making them ideal materials for smart windows.
3. Common Photothermal Conversion Materials
Photothermal and thermochromic materials are often combined for sunlight-driven thermochromic smart windows. The strong light-absorption characteristics or localized surface plasmon resonance effects [27][28] of photothermal materials can convert solar energy into heat energy, increase the surrounding temperature, and achieve the purpose of assisting the phase transition.
3.1. Carbon-Based Nanomaterials
Carbon-based nanomaterials are a new type of photothermal material developed in recent years, such as carbon dots [29][30], graphene, and graphene oxide [31][32][33]. They have strong absorption in visible and near-infrared light and can transform solar energy into heat energy rapidly owing to their unique structures. Therefore, they show a wide range of applications in smart windows [34][35][36][37][38][39], photocatalysis [40], and tumor-targeted therapy [41].
Graphene oxide (GO) is a two-dimensional nanomaterial with a single atomic thickness obtained by the oxidation and dispersion of natural graphite. Under sunlight, most of the energy photons in visible light can be absorbed by electrons, so they are excited. When the excited-state electrons fall back to the ground state, they release heat and raise the local temperature, producing a significant photothermal effect.
3.2. Noble Metal Nanoparticles
Noble metal nanoparticles are the most researched near-infrared photothermal materials, such as Au [42][43], Ag [44], Pd [45][46], etc. Their photothermal effect is mainly derived from the strong localized surface plasmon resonance effect of nanoparticles, which is a unique phenomenon occurring in metal structures. When the frequency of the incident light matches the eigenfrequency of the free electrons in the metal, the electrons will be collectively excited and resonated. Vibrating electrons will convert kinetic energy into heat energy due to the damping effect, thus increasing the local temperature [47].
3.3. Semiconductor Nanomaterials
Due to the wide energy gap, conventional semiconductor materials need to absorb UV light with higher energy to excite the electrons and release heat in the process of falling back to the ground state. However, with the study of more kinds of semiconductor materials, it has been found that localized surface plasmon resonance (LSPR) exists not only in noble metals, but also in semiconductor materials with appreciable free carrier density, such as tin-doped indium oxide (ITO) [48][49], copper sulfide (CuxS) [50][51], titanium nitride (TiN) [52][53], etc. Compared with conventional noble metals, semiconductors can exhibit LSPR in both the ultraviolet-visible (UV-vis) and near–mid-infrared (IR) spectral regions, which significantly extends the light absorption range.
3.4. Others
Transition metal nitrides, such as titanium nitride, are versatile metal–ceramic materials with high-temperature durability, chemical stability, corrosion resistance, electrical conductivity, and also localized surface plasmon resonance properties [54][55][56]. It can replace noble metal plasmas to generate LSPR and promote material phase changes under light conditions, resulting in lower-cost thermochromic materials that are available at room temperature.
References
- Hamberg, I.; Granqvist, C. Evaporated Sn-doped In2O3 films: Basic optical properties and applications to energy-efficient windows. J. Appl. Phys. 1986, 60, 123–160.
- Jing, T. Selective Reflection of Cholesteric Liquid Crystals. Sci. Insights 2022, 40, 515–517.
- Ming, Y.; Sun, Y.; Liu, X. Optical evaluation of a Smart Transparent Insulation Material for window application. Energy Convers. Manag. X 2022, 100315.
- Huang, Z.; Chen, S.; Lv, C.; Huang, Y.; Lai, J. Infrared characteristics of VO2 thin films for smart window and laser protection applications. Appl. Phys. Lett. 2012, 101, 191905.
- Rui, S.; Lin, Y.; Junhui, H.; Jie, L. Thermochromic smart coatings. Prog. Chem. 2019, 31, 1712.
- Cui, Y.; Ke, Y.; Liu, C. Thermochromic VO2 for energy-efficient smart windows. Joule 2018, 2, 1707–1746.
- Barimah, E.; Boontan, A.; Steenson, D. Infrared optical properties modulation of VO2 thin film fabricated by ultrafast pulsed laser deposition for thermochromic smart window applications. Sci. Rep. 2022, 12, 1–10.
- Ren, H.; Hassna, O.; Li, J.; Arigong, B. A patterned phase-changing vanadium dioxide film stacking with VO2 nanoparticle matrix for high performance energy-efficient smart window applications. Appl. Phys. Lett. 2021, 118, 051901.
- Riapanitra, A.; Asakura, Y.; Yin, S. One-step hydrothermal synthesis and thermochromic properties of chlorine-doped VO2(M) for smart window application. Funct. Mater. Lett. 2020, 13, 1951008.
- Ji, H.; Liu, D.; Cheng, H.; Zhang, C.; Yang, L.; Ren, D. Infrared thermochromic properties of monoclinic VO2 nanopowders using a malic acid-assisted hydrothermal method for adaptive camouflage. RSC Adv. 2017, 7, 5189–5194.
- Ji, H.; Liu, D.; Cheng, H.; Zhang, C. Inkjet printing of vanadium dioxide nanoparticles for smart windows. J. Mater. Chem. C 2018, 6, 2424–2429.
- Ke, Y.; Zhou, C.; Zhou, Y.; Wang, S.; Chan, S.H.; Long, Y. Emerging thermal-responsive materials and integrated techniques targeting the energy-efficient smart window application. Adv. Funct. Mater. 2018, 28, 1800113.
- Markov, P.; Marvel, R.E.; Conley, H.J.; Miller, K.J.; Haglund, R.F., Jr.; Weiss, S.M. Optically monitored electrical switching in VO2. ACS Photonics 2015, 2, 1175–1182.
- Ji, H.; Liu, D.; Cheng, H.; Tao, Y. Large area infrared thermochromic VO2 nanoparticle films prepared by inkjet printing technology. Sol. Energy Mater. Sol. Cells 2019, 194, 235–243.
- Ji, H.; Liu, D.; Cheng, H. Infrared optical modulation characteristics of W-doped VO2(M) nanoparticles in the MWIR and LWIR regions. Mater. Sci. Semicond. Process. 2020, 119, 105141.
- Ji, H.; Liu, D.; Cheng, H.; Zhang, C.; Yang, L. Vanadium dioxide nanopowders with tunable emissivity for adaptive infrared camouflage in both thermal atmospheric windows. Sol. Energy Mater. Sol. Cells 2018, 175, 96–101.
- Ji, H.; Liu, D.; Zhang, C.; Cheng, H. VO2/ZnS core-shell nanoparticle for the adaptive infrared camouflage application with modified color and enhanced oxidation resistance. Sol. Energy Mater. Sol. Cells 2018, 176, 1–8.
- Meng, W.; Gao, Y.; Hu, X.; Tan, L.; Li, L.; Zhou, G.; Jiang, L. Photothermal Dual Passively Driven Liquid Crystal Smart Window. ACS Appl. Mater. Interfaces 2022, 14, 28301–28309.
- Meng, C.; Chen, E.; Wang, L.; Tang, S.; Tseng, M.; Guo, J.; Kwok, H. Color-switchable liquid crystal smart window with multi-layered light guiding structures. Opt. Express 2019, 27, 13098–13107.
- Kragt, A.; Loonen, R.; Broer, D.; Debije, M.; Schenning, A. ‘Smart’ light-reflective windows based on temperature responsive twisted nematic liquid crystal polymers. J. Polym. Sci. 2021, 59, 1278–1284.
- Oh, S.; Nam, S.; Kim, S.; Yoon, T.; Kim, W. Self-regulation of infrared using a liquid crystal mixture doped with push–pull azobenzene for energy-saving smart windows. ACS Appl. Mater. Interfaces 2021, 13, 5028–5033.
- Li, K.; Meng, S.; Xia, S.; Ren, X.; Gao, G. Durable and controllable smart windows based on thermochromic hydrogels. ACS Appl. Mater. Interfaces 2020, 12, 42193–42201.
- Xiao, X.; Shi, D.; Yang, Z.; Yu, Q.; Kaneko, D.; Chen, M. Near infrared-sensitive smart windows from Au nanorod–polymer hybrid photonic hydrogels. New J. Chem. 2021, 45, 4016–4023.
- Zhang, L.; Xia, H.; Xia, F.; Du, Y.; Wu, Y.; Gao, Y. Energy-saving smart windows with HPC/PAA hybrid hydrogels as thermochromic materials. ACS Appl. Energy Mater. 2021, 4, 9783–9791.
- Tian, J.; Peng, H.; Du, X.; Wang, H.; Cheng, X.; Du, Z. Hybrid thermochromic microgels based on UCNPs/PNIPAm hydrogel for smart window with enhanced solar modulation. J. Alloy. Compd. 2021, 858, 157725.
- Pelton, R. Poly (N-isopropylacrylamide)(PNIPAM) is never hydrophobic. J. Colloid Interface Sci. 2010, 348, 673–674.
- Sherry, L.; Jin, R.; Mirkin, C.; Schatz, G.; Van Duyne, R. Localized surface plasmon resonance spectroscopy of single silver triangular nanoprisms. Nano Lett. 2006, 6, 2060–2065.
- Agrawal, A.; Cho, S.; Zandi, O.; Ghosh, S.; Johns, R.; Milliron, D. Localized surface plasmon resonance in semiconductor nanocrystals. Chem. Rev. 2018, 118, 3121–3207.
- Kang, Z.; Lee, S. Carbon dots: Advances in nanocarbon applications. Nanoscale 2019, 11, 19214–19224.
- Mintz, K.; Bartoli, M.; Rovere, M.; Zhou, Y.; Hettiarachchi, S.; Paudyal, S.; Leblanc, R. A deep investigation into the structure of carbon dots. Carbon 2021, 173, 433–447.
- Dideikin, A.; Vul’, A. Graphene oxide and derivatives: The place in graphene family. Front. Phys. 2019, 6, 149.
- Wang, Y.; Li, S.; Yang, H.; Luo, J. Progress in the functional modification of graphene/graphene oxide: A review. RSC Adv. 2020, 10, 15328–15345.
- Farjadian, F.; Abbaspour, S.; Sadatlu, M.; Mirkiani, S.; Ghasemi, A.; Hoseini-Ghahfarokhi, M.; Hamblin, M. Recent developments in graphene and graphene oxide: Properties, synthesis, and modifications: A review. ChemistrySelect 2020, 5, 10200–10219.
- Kim, D.; Lee, E.; Lee, H.S.; Yoon, J. Energy efficient glazing for adaptive solar control fabricated with photothermotropic hydrogels containing graphene oxide. Sci. Rep. 2015, 5, 1–6.
- Zhu, C.; Lu, Y.; Peng, J.; Chen, J.; Yu, S. Photothermally sensitive poly (N-isopropylacrylamide)/graphene oxide nanocomposite hydrogels as remote light-controlled liquid microvalves. Adv. Funct. Mater. 2012, 22, 4017–4022.
- Lee, E.; Kim, D.; Yoon, J. Stepwise activation of switchable glazing by compositional gradient of copolymers. ACS Appl. Mater. Interfaces 2016, 8, 26359–26364.
- NIWAER, A.E.A.; Wang, H.; Meng, X.; He, X.; Yang, S.; Yu, H.; Zuo, F. Preparation and preliminary application of PNIPAM/laponite/graphene oxide near-infrared light responsive hydrogels. Fine Chem. 2021, 3, 6.
- Zhou, T.; Zhou, Q.; Hua, Y.; Huang, C.; Li, L.; LÜ, W. Preparation and properties of tungsten-doped vanadium dioxide/graphene composites. Fine Chem. 2020, 37, 1574–1579.
- Chang, Q.; Shen, Z.; Guo, Z.; Xue, C.; Li, N.; Yang, J.; Hu, S. Hydroxypropylmethyl cellulose modified with carbon dots exhibits light-responsive and reversible optical switching. ACS Appl. Mater. Interfaces 2021, 13, 12375–12382.
- Qin, J.; Li, R.; Lu, C.; Jiang, Y.; Tang, H.; Yang, X. Ag/ZnO/graphene oxide heterostructure for the removal of rhodamine B by the synergistic adsorption–degradation effects. Ceram. Int. 2015, 41, 4231–4237.
- Zhang, M.; Zhang, X.; Zhao, K.; Dong, Y.; Yang, W.; Liu, J.; Li, D. Assembly of gold nanorods with L-cysteine reduced graphene oxide for highly efficient NIR-triggered photothermal therapy. Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 2022, 266, 120458.
- Chen, H.; Shao, L.; Ming, T.; Sun, Z.; Zhao, C.; Yang, B.; Wang, J. Understanding the photothermal conversion efficiency of gold nanocrystals. Small 2010, 6, 2272–2280.
- Kim, H.; Lee, D. Near-infrared-responsive cancer photothermal and photodynamic therapy using gold nanoparticles. Polymers 2018, 10, 961.
- Chen, M.; He, Y.; Huang, J.; Zhu, J. Synthesis and solar photo-thermal conversion of Au, Ag, and Au-Ag blended plasmonic nanoparticles. Energy Convers. Manag. 2016, 127, 293–300.
- Xiao, J.; Fan, S.; Wang, F.; Sun, L.; Zheng, X.; Yan, C. Porous Pd nanoparticles with high photothermal conversion efficiency for efficient ablation of cancer cells. Nanoscale 2014, 6, 4345–4351.
- Tang, S.; Chen, M.; Zheng, N. Multifunctional ultrasmall Pd nanosheets for enhanced near-infrared photothermal therapy and chemotherapy of cancer. Nano Res. 2015, 8, 165–174.
- Jain, P.K.; Huang, X.; El-Sayed, I.H.; El-Sayed, M.A. Noble metals on the nanoscale: Optical and photothermal properties and some applications in imaging, sensing, biology, and medicine. Acc. Chem. Res. 2008, 41, 1578–1586.
- Blemker, M.; Gibbs, S.; Raulerson, E.; Milliron, D.; Roberts, S. Modulation of the visible absorption and reflection profiles of ITO nanocrystal thin films by plasmon excitation. ACS Photonics 2020, 7, 1188–1196.
- Wei, W.; Hong, R.; Jing, M.; Shao, W.; Tao, C.; Zhang, D. Thickness-dependent surface plasmon resonance of ITO nanoparticles for ITO/In-Sn bilayer structure. Nanotechnology 2017, 29, 015705.
- Kalanur, S.; Seo, H. Synthesis of CuxS thin films with tunable localized surface plasmon resonances. ChemistrySelect 2018, 3, 5920–5926.
- Song, G.; Han, L.; Zou, W.; Xiao, Z.; Huang, X.; Qin, Z.; Hu, J. A novel photothermal nanocrystals of Cu7S4 hollow structure for efficient ablation of cancer cells. Nano-Micro Lett. 2014, 6, 169–177.
- Zhang, J.; Chen, T.; Li, X.; Liu, Y.; Liu, Y.; Yang, H. Investigation of localized surface plasmon resonance of TiN nanoparticles in TiNxOy thin films. Opt. Mater. Express 2016, 6, 2422–2433.
- Wang, L.; Zhu, G.; Wang, M.; Yu, W.; Zeng, J.; Yu, X.; Li, Q. Dual plasmonic Au/TiN nanofluids for efficient solar photothermal conversion. Sol. Energy 2019, 184, 240–248.
- Patsalas, P.; Kalfagiannis, N.; Kassavetis, S. Optical properties and plasmonic performance of titanium nitride. Materials 2015, 8, 3128–3154.
- Gschwend, P.; Conti, S.; Kaech, A.; Maake, C.; Pratsinis, S. Silica-coated TiN particles for killing cancer cells. ACS Appl. Mater. Interfaces 2019, 11, 22550–22560.
- Jiang, W.; Fu, Q.; Wei, H.; Yao, A. TiN nanoparticles: Synthesis and application as near-infrared photothermal agents for cancer therapy. J. Mater. Sci. 2019, 54, 5743–5756.
More
Information
Subjects:
Materials Science, Coatings & Films
Contributors
MDPI registered users' name will be linked to their SciProfiles pages. To register with us, please refer to https://encyclopedia.pub/register
:
View Times:
892
Revisions:
2 times
(View History)
Update Date:
23 Nov 2022
Notice
You are not a member of the advisory board for this topic. If you want to update advisory board member profile, please contact office@encyclopedia.pub.
OK
Confirm
Only members of the Encyclopedia advisory board for this topic are allowed to note entries. Would you like to become an advisory board member of the Encyclopedia?
Yes
No
${ textCharacter }/${ maxCharacter }
Submit
Cancel
Back
Comments
${ item }
|
More
No more~
There is no comment~
${ textCharacter }/${ maxCharacter }
Submit
Cancel
${ selectedItem.replyTextCharacter }/${ selectedItem.replyMaxCharacter }
Submit
Cancel
Confirm
Are you sure to Delete?
Yes
No





