
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Jianhua Wang | + 2981 word(s) | 2981 | 2020-12-10 08:37:49 | | | |
| 2 | Peter Tang | -177 word(s) | 2804 | 2020-12-18 10:27:50 | | |
Video Upload Options
Antimicrobial peptides display the property of combating viruses in swine production of animal husbandry, clarify the mechanism of action of antimicrobial peptides on viruses and raise some suspending questions and prospect the future potential of antimicrobial peptides in animal husbandry.
1. Introduction
Viral infection of pigs is one of the bottlenecks restricting the development of the pig industry globally. The widely spread common porcine pathogenic viruses, including herpesvirus (such as pseudorabies virus (PRV)), coronavirus (such as porcine epidemic diarrhea virus (PEDV)), and arterivirus (such as porcine reproductive and respiratory syndrome virus (PRRSV)), have caused many serious infectious diseases and huge economic losses in the pig industry [1][2][3]. At present, only a few effective treatments are available for most viral diseases. In the past few decades, research on fighting viral infections in pigs has been mainly focused on vaccines, wherein the adaptive immunity of pigs is improved by vaccination [4][5]. However, some viruses can escape from host immunity through different strategies. It is reported that PRRSV evades the host immune response by glycosylation modification of its envelope proteins [6], and PEDV evades the host innate immune response by encoding interferon (IFN) antagonists to disrupt the innate immune pathway and hide its viral RNA to avoid exposure of viral RNA to immune sensors [7]. The emergence of new variants of viruses is one of the important causes of disease outbreaks in pigs, such as the reemergence of PRV in China since late 2011 [8], the outbreak of porcine epidemic diarrhea (PED) in China at the end of 2010 [9], and the spread of African swine fever worldwide since 2007 [10]. Besides, the development of new vaccines is usually complex, technically challenging, and time-consuming [11]. Therefore, there is an urgent need to develop novel effective agents to kill viruses and prevent their infection in swine [4].
Antimicrobial peptides (AMPs) are small proteins with potential activity against, for example, bacteria, viruses, fungi, tumors, and parasites, which are widely found in animals, plants, and microorganisms [12]. The discovery of AMPs can be traced back to 1939; Dubos isolated an antibacterial agent from a strain of soil Bacillus and found that it could protect mice from pneumococcal infection for the first time [13][14]. According to their key structural characteristics, AMPs can be usually divided into four categories: α-helix AMPs, β-folded AMPs, extended structural AMPs, and cyclic structural AMPs [15]. Since their discovery, AMPs have become important alternative drugs in the field of disease prevention and immune regulation, attracting worldwide attention [16][17]. In the field of human viral diseases, at present, AMPs have become an important direction and field in antiviral research. Previous studies have shown that the amphibian-derived AMPs, caerin 1.1 and maculatin 1.1, completely inhibited human immunodeficiency virus (HIV) [18]; arthropod-derived AMPs, cecropin A and melittin, could effectively inhibit Junin virus (JV) multiplication and impede the multiplication of herpes simplex virus (HSV) and JV, respectively [19]; plant protein kalata B1 analogs could inhibit dengue virus (DENV) [20]. The above results suggested that AMPs with antiviral activity have the potential to be antiviral drugs and could be highly expected to become clinical drugs for both human viral diseases and animal viral infections. However, there have only been a few studies on AMPs in animals compared with humans so far. It is important to pay attention to the role of AMPs in animal health because most upstream studies specialized for human usage are run with model animals.
AMPs exhibit different mechanisms on viruses, and their main antiviral mechanisms and types are summarily shown in Figure 1. (i) AMPs neutralize viruses by integrating into the viral envelope or host cell membranes, and both enveloped RNA and DNA viruses can be targeted [21][22]. Indolicidin could inactivate human immunodeficiency virus (HIV) by binding to the envelope and cracking the membrane through a membrane splitting mechanism, thereby preventing the virus from infecting the host cell [23]. (ii) AMPs bind to glycoproteins on the virus surface to inhibit viral infection. Defensin retrocyclin 2 bound to immobilized herpes simplex virus type 2 (HSV-2) glycoprotein B with high affinity so that HSV-2 could not bind to the surface of host cells [24]. (iii) AMPs can interact with specific receptors of host cell membranes, preventing virus particles from binding to host cells. HSV particles infect host cells by binding to heparan sulfate on the host cells, while the α-helix cationic polypeptide lactoferrin can prevent HSV infection by occupying heparan sulfate [25]. (iv) In addition to the above points, AMPs may also act on other stages of the viral life cycle. For example, human beta-defensin-3 (hBD-3) inhibits HIV replication by acting on entry, reverse transcription, and nuclear import of retroviral DNA [26]. Besides, some AMPs, such as cecropin D (CD), could block apoptosis induced by PRRSV at the late stage of infection, thus inhibiting the assembly, release, and transmission of the virus [27].
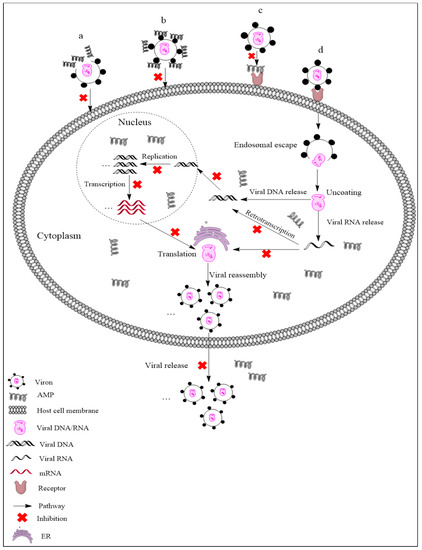
Figure 1. Antiviral mechanisms of AMPs. (a) AMPs inactivate a virus by destroying the viral envelope. (b) AMPs bind to glycoprotein on the viral envelope. (c) AMPs occupy a specific receptor on the host cell membrane to prevent viral attachment. (d) AMPs inhibit viral replication, transcription, reverse transcription, translation, and release.
As the pig industry has developed to account for the largest proportion of food animals in China, AMPs have been widely studied as antimicrobial agents and/or feed additives [28][29][30][31]. Studies have shown that the plectasin-derived peptides NZ2114 and MP1102 could effectively kill Streptococcus suis type 2 [28][29], and NZX exhibited antibacterial activity against Staphylococcus hyicus [30]. Diets supplemented with AMPs could improve the growth performance, nutrition maintenance, intestinal morphology, and immunity of weaned piglets, while also reducing the presence of harmful microorganisms in these animals [31]. In addition to antibacterial activity and immune regulation, AMPs should also be actively encouraged and utilized to combat common porcine pathogenic viruses in pig molecular breeding via genetic engineering.
2. Antimicrobial Peptides Used against Viruses in Swine
2.1. AMPs Active against PRV and PEDV
PRV is a large, enveloped, double-stranded DNA virus that is the pathogen of Aujeszky's disease and belongs to the porcine neurotropic herpesviruses [1][32]. Clinically, it is characterized by severe neurological disorders in newborn piglets and reproductive disorders in sows [32]. PEDV is also an enveloped virus; it is a positive-sense, single-stranded RNA virus, belonging to the family of Coronaviridae [2]. PEDV is the pathogen of porcine epidemic diarrhea (PED), which is an acute infectious enteropathy and is characterized by severe watery diarrhea, vomiting, and dehydration of suckling piglets, causing huge economic losses for the pig industry [33].
2.2. AMPs Active against PRRSV
Porcine reproductive and respiratory syndrome (PRRS), also known as blue-ear pig disease, is one of the most fatal infectious diseases in the pig industry around the world; it was first reported in North America and Canada in the late 1980s [34][35][36]. The pathogen of this disease is called the porcine reproductive and respiratory syndrome virus (PRRSV), which is an enveloped, single-stranded RNA virus belonging to the Arteriviridae family of Nidovirales. Its genomic RNA length is about 15.4 kb, with a 5′ cap, 3′ polyadenylation, and 10 open reading frames (ORFs) [3][37]. The main manifestations of the disease are poor reproductive performance and high miscarriage rate in pregnant sows and dyspnea in growing–finishing pigs and piglets [38]. PRRSV mainly infects porcine alveolar macrophages (PAMs) and has the characteristics of high mutation rate and high recombination rate. With the antigen variation and genetic drift of the virus, the existing vaccines are easily losing their efficacy [39][40]. Therefore, PRRSV is still the greatest challenge facing the pig industry so far, and it is urgent to develop new antiviral strategies to combat PRRSV infection [41].
2.3. Epinecidin-1 (Epi-1) Fights against FMDV
Foot and mouth disease (FMD) is a very important disease affecting livestock in the world [42] and is caused by foot and mouth disease virus (FMDV) [43]. FMDV is a nonenveloped virus belonging to the family of Picornaviridae and genus Aphthovirus [44]; it is highly infectious to pigs and other cloven-hoofed animals and imposes a significant impact on the global economy [45][46]. Common symptoms of foot and mouth disease include fever and blistering lesions in the mouth, tongue, and feet. There are seven antigenic serotypes of FMDV, including O, A, C, SAT1, SAT2, SAT3 (South African 1, 2, 3), and Asia1 (Asian 1); each serotype has multiple subtypes [47], and serotype O is the most common serotype in the world. As there is still no effective vaccine or antiviral drug, new and better drugs or candidates are being sought to combat FMDV infection.
Epinecidin-1 (Epi-1) is derived from the orange-spotted grouper, Epinephelus coioides [48], and belongs to the piscidin peptide family. The piscidin family is an evolutionarily conserved, linear, amphiphilic, antibacterial peptide family that is unique to fish and homologous to cecropins [49]. The length of complete Epi-1 cDNA is 518 base pairs, and the longest open reading frame consists of 204 base pairs and encodes a sequence of 67 amino acids [48]. Studies on the potential pharmacological activity have mainly focused on amino acid residues 22–42 of Epi-1 (Table 1) [50], which shows an α-helical structure without disulfide bonds (Figure 2g) [51]. Epi-1 has been reported to have wide activity against bacteria, fungi, and viruses and shows immune regulation [52][53][54]. Besides, Huang et al. found that the synthetic Epi-1 effectively suppresses FMDV (type O/Taw/97) by inactivating virus particles and inhibiting virus proliferation. Epi-1 not only shows a direct antiviral effect on FMDV at high concentration (10 × EC90 concentration of 125 μg/mL) but also prevents the adsorption of FMDV on BHK-21 cells at low concentration (6.2 μg/mL) [55]. Since a structured membrane is absent in FMDV, research data indicated that the application of Epi-1 in virus adsorption can effectively inhibit virus replication, and thus it is suggested that Epi-1 could interfere with the early stage of viral infection through an undisclosed mechanism [55].
2.4. Synthesized Peptides Fight against ASFV
In addition to the above several viral diseases, African swine fever (ASF) is also a viral disease that lacks effective vaccines for prevention and control in the pig breeding industry; it has spread quickly as an epidemic viral disease in China since mid-2018. As a highly infectious viral disease of pigs, ASF causes fatal hemorrhagic fever after infection, resulting in a high mortality rate of nearly 100% [56]. ASF is caused by African swine fever virus (ASFV), which is a large, enveloped, double-stranded DNA virus with icosahedral morphology and the only member of Asfarviridae family. ASFV is transmitted by arthropod soft ticks (Ornithodoros moubata), making it the only DNA virus to be transmitted via insect [57][58].
Studies have shown that ASFV utilizes dynein for internalization and intracellular transport [59], entering into host cells through dynein- and clathrin-dependent endocytosis and micropinocytosis (Figure 2) [56][60]. As a microtubular motor protein, dynein is in charge of the intracellular transport linked to microtubules. In the early stage of the virus life cycle, the virus carries out intracellular transportation along microtubules. Once the virus passes through the cytoplasm, it quickly enters into the perinuclear region or nucleus and starts to replicate [61]. P54 is the main protein of the ASFV particle membrane, which can interact with the light-chain dynein of 8 kDa (DLC8) both in vitro and in cells. This interaction allows ASFV to be transported to a viral factory located in the perinuclear area at the microtubular organizing center (MTOC), which is necessary for viral protein synthesis and replication [59]. As breaking the interaction between the virus and dynein can hinder the transportation of the virus, it should be focused on as one of the mechanisms of AMPs against ASFV.
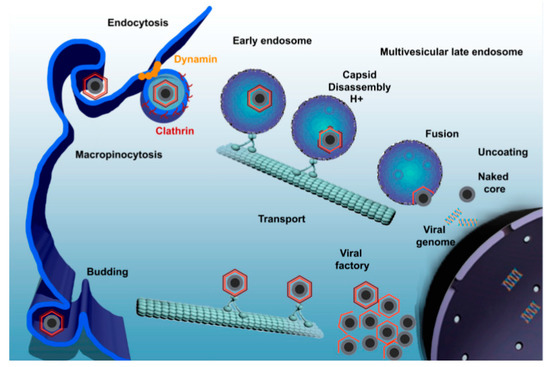
Figure 2. African swine fever virus (ASFV) enters the host cell through a complex process involving dynamin- and clathrin-mediated endocytosis and macropinocytosis. Newly synthesized virions are assembled in the viral factory and will exit the cell either by exocytosis budding at the plasma membrane or through the formation of apoptotic bodies [56].
In recent years, synthetic AMPs with specific targets have been designed to bind to receptors on the surface of host cells, rendering these binding sites unavailable to viral proteins and thus impairing viral adsorption [62][63][64]. DNBLK1 is a synthesized short peptide that consists of 28 amino acid residues and contains DLC8 binding domain; by binding to DLC8 to prevent the interaction between ASFV protein p54 and DLC8 in vitro, it may be a useful tool to retard viral replication or spread [62]. Bruno et al. indicated that DNBLK1 can reduce the infectivity, replication, and production of ASFV, and the inhibition occurs at the early stage of the ASFV infection cycle. This provides clues for the treatment of African swine fever and other diseases caused by viruses with the same transmission mechanism as ASFV [62], and more attention should be paid to this new direction.
3. Conclusions
Since there is still no effective treatment for most viral infections in animal husbandry, outbreaks of viral epidemics are generally followed by the quarantine and slaughter of infected animals, resulting in great economic losses for the breeding industry and society [33][35][42][46]. In the past few decades, research on viral diseases in pig breeding has focused on the development of vaccines [5]. Vaccination can inhibit the development of the disease [65], but with the continuous variation of viruses, traditional vaccines lose their effect on mutated virus strains, and the emergence of mutant strains leads to the outbreak of viral diseases. In the context of today's highly globalized world, viral diseases spread much faster and easier than they did centuries ago [10]. We have to face this threating challenge by utilizing more feasible options.
Therefore, in addition to the usual development of vaccines as antiviral agents, it is also of great significance to exploit new strategies to combat viruses. Currently, AMPs, due to their effective antiviral activity, are the research focus in the field of new antiviral drug development and are expected to become one of the key drivers of antiviral drug development in the future. AMPs inactivate viruses by destroying the viral envelope; binding with virus surface glycoprotein; occupying specific receptors of the host cell membrane; and inhibiting viral replication, transcription, reverse transcription, expression, and release. With the improvement of bioinformatics science, more new AMPs with antiviral activity are continuously being discovered and designed. These AMPs can resist not only porcine viruses but also Newcastle disease virus [66], duck hepatitis virus [67], bovine herpesvirus 1 [68], dengue virus [69], and other human and zoonotic viruses and thus hold great importance for research and development in theory and practice. In addition, synthetic AMPs with specific targets, such as DNBLK1 (targeting the DLC8 binding domain), have also been designed, providing a new idea and tool for the development and improvement of AMPs to resist viruses [62].
Although AMPs have great potential activity against viruses, there are still some potential problems to be solved, such as higher cost of production, shorter half-life time, and poor oral absorption of AMPs, as well as the challenge of delivery systems [70]. Some AMPs have been shown to have antiviral effects in vitro against viral diseases in animal husbandry, but their antiviral activity in vivo remains to be studied and confirmed [27][71][72]. It is known that AMPs are sensitive to trypsin and other lytic factors in vivo, especially when they are administrated orally or by blood injection. In order to improve the stability of AMPs, many studies have been carried out considering the controlled site-specific release and sustained continuous release of AMPs by nanoencapsulation [73][74] and modification of high resistance to proteolysis [75], as well including unnatural or D-amino acids substitution [76] and peptide chain cyclization [77][78]. Moreover, targeting modification is also worth considering as a powerful tool to increase killing specificity to pathogens and decrease host cell toxicity of AMPs [79][80]. In addition, many mechanisms of action between AMPs and virus molecules are still unclear and need to be further studied. However, we find it reasonable to assume that the previous fruitful findings and constructive theories from antibacterial studies with AMPs in vivo and in vitro, such as those concerning the mechanism of entry into the host cell and bactericidal details, might be shared and referenced during antiviral studies; it is confirmed from previous works that AMPs enter the blood circulation through different drug delivery routes, reach various organs, and further internalize into the cells through endocytosis and micropinocytosis [81][82][83][84]. Undoubtedly, wider and deeper new findings are highly deserving of anticipation and will attract great interest due to the unique advantages of AMPs, including their high penetration into the host cell owing to their intracellular origin, their close compatibility with the host, their hypersensitive early-warning/protection response to infection, and their low drug resistance rate owing to strong penetration and multitargeting of pathogens [30][85][86][87][88][89][90]. We strongly believe and optimistically expect that with the further elucidation of the structure, expression regulation, and mechanism of action of AMPs as a whole, the factors that limit the development of AMPs will be disclosed and overcome one by one, and more new functions of AMPs will be discovered. Eventually, AMPs will be widely utilized and commercialized in animal husbandry and even in the human health industry [16][89][91].
In summary, antimicrobial peptides, which can effectively combat viruses, are highly expected to break through their special technical bottleneck in the near future; this will enable them to support the sustainable green development of the husbandry and health industries and thus promote the general upgrading of green industry in China and the world.
References
- Ye, C.; Chen, J.; Wang, T.; Xu, J.; Zheng, H.; Wu, J.; Li, G.; Yu, Z.; Tong, W.; Cheng, X.; et al. Generation and characterization of UL41 null pseudorabies virus variant in vitro and in vivo. Virol. J. 2018, 15, 119.
- Chen, F.; Zhu, Y.; Wu, M.; Ku, X.; Ye, S.; Li, Z.; Guo, X.; He, Q. Comparative genomic analysis of classical and variant virulent parental/attenuated strains of porcine epidemic diarrhea virus. Viruses 2015, 7, 5525–5538.
- Snijder, E.J.; Meulenberg, J.J. The molecular biology of arteriviruses. J. Gen. Virol. 1998, 79, 961–979.
- Renukaradhya, G.J.; Meng, X.J.; Calvert, J.G.; Roof, M.; Lager, K.M. Inactivated and subunit vaccines against porcine reproductive and respiratory syndrome: Current status and future direction. Vaccine 2015, 33, 3065–3072.
- Paton, D.J.; Taylor, G. Developing vaccines against foot-and-mouth disease and some other exotic viral diseases of livestock. Philos. Trans. R. Soc. Lond. B Biol. Sci. 2011, 366, 2774–2781.
- Ansari, I.H.; Kwon, B.; Osorio, F.A.; Pattnaik, A.K. Influence of N-linked glycosylation of porcine reproductive and respiratory syndrome virus GP5 on virus infectivity, antigenicity, and ability to induce neutralizing antibodies. J. Virol. 2006, 80, 3994–4004.
- Li, S.; Yang, J.; Zhu, Z.; Zheng, H. Porcine epidemic diarrhea virus and the host innate immune response. Pathogens 2020, 9, 367.
- An, T.Q.; Peng, J.M.; Tian, Z.J.; Zhao, H.Y.; Li, N.; Liu, Y.M.; Chen, J.Z.; Leng, C.L.; Sun, Y.; Chang, D.; et al. Pseudorabies virus variant in Bartha-K61-vaccinated pigs, China, 2012. Emerg. Infect. Dis. 2013, 19, 1749–1755.
- Li, W.; Li, H.; Liu; Y.; Pan; Y.; Deng, F.; Song, Y.; Tang, X.; He, Q. New variants of porcine epidemic diarrhea virus, China, 2011. Emerg. Infect. Dis. 2012, 18, 1350–1353.
- Sánchez-Cordón, P.J.; Montoya, M.; Reis, A.L.; Dixon, L.K. African swine fever: A re-emerging viral disease threatening the global pig industry. Vet. J. 2018, 233, 41–48.
- Mahmoud, A. New vaccines: Challenges of discovery. Microb. Biotechnol. 2016, 9, 549–552.
- Bahar, A.A.; Ren, D. Antimicrobial peptides. Pharmaceuticals (Basel) 2013, 6, 1543–1575.
- Dubos, R.J. Studies on a bactericidal agent extracted from a soil bacillus: I. Preparation of the agent. Its activity in vitro. J. Exp. Med. 1939, 70, 1–10.
- Dubos, R.J. Studies on a bactericidal agent extracted from a soil bacillus: II. Protective effect of the bactericidal agent against experimental pneumococcus infections in mice. J. Exp. Med. 1939, 70, 11–17.
- Powers, J.P.; Hancock, R.E. The relationship between peptide structure and antibacterial activity. Peptides 2003, 24, 1681–1691.
- Zasloff, M. Antimicrobial peptides: Do they have a future as therapeutics? In Antimicrobial Peptides; Springer International Publishing: Cham, Switzerland, 2016; pp. 147–154.
- Ganz, T. Defensins: Antimicrobial peptides of innate immunity. Nat. Rev. Immunol. 2003, 3, 710–720.
- VanCompernolle, S.E.; Taylor, R.J.; Oswald-Richter, K.; Jiang, J.; Youree, B.E.; Bowie, J.H.; Tyler, M.J.; Conlon, J.M.; Wade, D.; Aiken, C.; et al. Antimicrobial peptides from amphibian skin potently inhibit human immunodeficiency virus infection and transfer of virus from dendritic cells to T cells. J. Virol. 2005, 79, 11598–11606.
- Matanic, V.C.A.; Castilla, V. Antiviral activity of antimicrobial cationic peptides against Junin virus and herpes simplex virus. Int. J. Antimicrob. Agents 2004, 23, 382–389.
- Gao, Y.; Cui, T.; Lam, Y. Synthesis and disulfide bond connectivity-activity studies of a kalata B1-inspired cyclopeptide against dengue NS2B-NS3 protease. Bioorg. Med. Chem. 2010, 18, 1331–1336.
- Bastian, A.; Schäfer, H. Human alpha-defensin 1 (HNP-1) inhibits adenoviral infection in vitro. Regul. Pept. 2001, 101, 157–161.
- Horne, W.S.; Wiethoff, C.M.; Cui, C.; Wilcoxen, K.M.; Amorin, M.; Ghadiri, M.R.; Nemerow, G.R. Antiviral cyclic D,L-alpha-peptides: Targeting a general biochemical pathway in virus infections. Bioorg. Med. Chem. 2005, 13, 5145–5153.
- Robinson, W.E., Jr.; McDougall, B.; Tran, D.; Selsted, M.E. Anti-hiv-1 activity of indolicidin, an antimicrobial peptide from neutrophils. J. Leukoc. Biol. 1998, 63, 94–100.
- Yasin, B.; Wang, W.; Pang, M.; Cheshenko, N.; Hong, T.; Waring, A.J.; Herold, B.C.; Wagar, E.A.; Lehrer, R.I. Theta defensins protect cells from infection by herpes simplex virus by inhibiting viral adhesion and entry. J. Virol. 2004, 78, 5147–5156.
- Andersen, J.H.; Jenssen, H.; Sandvik, K.; Gutteberg, T.J. Anti-hsv activity of lactoferrin and lactoferricin is dependent on the presence of heparan sulphate at the cell surface. J. Med. Virol. 2004, 74, 262–271.
- Zapata, W.; Aguilar-Jiménez, W.; Feng, Z.; Weinberg, A.; Russo, A.; Potenza, N.; Estrada, H.; Rugeles, M.T. Identification of innate immune antiretroviral factors during in vivo and in vitro exposure to HIV-1. Microbes Infect. 2016, 18, 211–219.
- Liu, X.; Guo, C.; Huang, Y.; Zhang, X.; Chen, Y. Inhibition of porcine reproductive and respiratory syndrome virus by Cecropin D in vitro. Infect. Genet. Evol. 2015, 34, 7–16.
- Jiao, J.; Mao, R.; Teng, D.; Wang, X.; Hao, Y.; Yang, N.; Wang, X.; Feng, X.; Wang, J. In vitro and in vivo antibacterial effect of NZ2114 against Streptococcus suis type 2 infection in mice peritonitis models. AMB Express 2017, 7, 44.
- Zhao, F.; Yang, N.; Wang, X.; Mao, R.; Hao, Y.; Li, Z.; Wang, X.; Teng, D.; Fan, H.; Wang, J. In vitro/vivo mechanism of action of MP1102 with low/nonresistance against Streptococcus suis type 2 strain CVCC 3928. Front. Cell. Infect. Microbiol. 2019, 9, 48.
- Liu, H.; Yang, N.; Mao, R.; Teng, D.; Hao, Y.; Wang, X.; Wang, J. A new high-yielding antimicrobial peptide NZX and its antibacterial activity against Staphylococcus hyicus in vitro/vivo. Appl. Microbiol. Biotechnol. 2020, 104, 1555–1568.
- Wu, S.; Zhang, F.; Huang, Z.; Liu, H.; Xie, C.; Zhang, J.; Thacker, P.A.; Qiao, S. Effects of the antimicrobial peptide cecropin AD on performance and intestinal health in weaned piglets challenged with Escherichia coli. Peptides 2012, 35, 225–230.
- Pomeranz, L.E.; Reynolds, A.E.; Hengartner, C.J. Molecular biology of pseudorabies virus: Impact on neurovirology and veterinary medicine. Microbiol. Mol. Biol. Rev. 2005, 69, 462–500.
- Pensaert, M.B.; de Bouck, P. A new coronavirus-like particle associated with diarrhea in swine. Arch. Virol. 1978, 58, 243–247.
- Zhao, C.; Liu, S.; Li, C.; Yang, L.; Zu, Y. In vitro evaluation of the antiviral activity of the synthetic epigallocatechin gallate analog-epigallocatechin gallate (EGCG) palmitate against porcine reproductive and respiratory syndrome virus. Viruses 2014, 6, 938–950.
- Neumann, E.J.; Kliebenstein, J.B.; Johnson, C.D.; Mabry, J.W.; Bush, E.J.; Seitzinger, A.H.; Green, A.L.; Zimmerman, J.J. Assessment of the economic impact of porcine reproductive and respiratory syndrome on swine production in the United States. J. Am. Vet. Med. Assoc. 2005, 227, 385–392.
- Wensvoort, G.; Terpstra, C.; Pol, J.M.; ter Laak, E.A.; Bloemraad, M.; de Kluyver, E.P.; Kragten, C.; van Buiten, L.; den Besten, A.; Wagenaar, F. Mystery swine disease in The Netherlands: The isolation of Lelystad virus. Vet. Q. 1991, 13, 121–130.
- Dokland, T. The structural biology of PRRSV. Virus Res. 2010, 154, 86–97.
- Kuzemtseva, L.; de la Torre, E.; Martín, G.; Soldevila, F.; Ait-Ali, T.; Mateu, E.; Darwich, L. Regulation of toll-like receptors 3, 7 and 9 in porcine alveolar macrophages by different genotype 1 strains of porcine reproductive and respiratory syndrome virus. Vet. Immunol. Immunopathol. 2014, 158, 189–198.
- Deaton, M.K.; Spear, A.; Faaberg, K.S.; Pegan, S.D. The vOTU domain of highly-pathogenic porcine reproductive and respiratory syndrome virus displays a differential substrate preference. Virology 2014, 454–455, 247–253.
- Thanawongnuwech, R.; Suradhat, S. Taming PRRSV: Revisiting the control strategies and vaccine design. Virus Res. 2010, 154, 133–140.
- Wang, L.; Xiao, S.; Gao, J.; Liu, M.; Zhang, X.; Li, M.; Zhao, G.; Mo, D.; Liu, X.; Chen, Y. Inhibition of replication of porcine reproductive and respiratory syndrome virus by hemin is highly dependent on heme oxygenase-1, but independent of iron in MARC-145 cells. Antivir. Res. 2014, 105, 39–46.
- James, A.D.; Rushton, J. The economics of foot and mouth disease. Rev. Sci. Tech. 2002, 21, 637–644.
- Wernery, U.; Kinne, J. Foot and mouth disease and similar virus infections in camelids: A review. Rev. Sci. Tech. 2012, 31, 907–918.
- Sáiz, M.; Núñez, J.I.; Jimenez-Clavero, M.A.; Baranowski, E.; Sobrino, F. Foot-and-mouth disease virus: Biology and prospects for disease control. Microbes Infect. 2002, 4, 1183–1192.
- Rweyemamu, M.; Roeder, P.; MacKay, D.; Sumption, K.; Brownlie, J.; Leforban, Y. Planning for the progressive control of foot-and-mouth disease worldwide. Transbound. Emerg. Dis. 2008, 55, 73–87.
- Knight-Jones, T.J.; Rushton, J. The economic impacts of foot and mouth disease—What are they, how big are they and where do they occur? Prev. Vet. Med. 2013, 112, 161–173.
- Cao, Y. Adjuvants for foot-and-mouth disease virus vaccines: Recent progress. Expert Rev. Vaccines 2014, 13, 1377–1385.
- Yin, Z.X.; He, W.; Chen, W.J.; Yan, J.H.; Yang, J.N.; Chan, S.M.; He, J.G. Cloning, expression and antimicrobial activity of an antimicrobial peptide, epinecidin-1, from the orange-spotted grouper, Epinephelus coioides. Aquaculture 2006, 253, 204–211.
- Masso-Silva, J.A.; Diamond, G. Antimicrobial peptides from fish. Pharmaceuticals (Basel) 2014, 7, 265–310.
- Pan, C.Y.; Chen, J.Y.; Cheng, Y.S.; Chen, C.Y.; Ni, I.H.; Sheen, J.F.; Pan, Y.L.; Kuo, C.M. Gene expression and localization of the epinecidin-1 antimicrobial peptide in the grouper (Epinephelus coioides), and its role in protecting fish against pathogenic infection. DNA Cell Biol. 2007, 26, 403–413.
- Neshani, A.; Zare, H.; Eidgahi, M.R.A.; Khaledi, A.; Ghazvini, K. Epinecidin-1, a highly potent marine antimicrobial peptide with anticancer and immunomodulatory activities. BMC Pharmacol. Toxicol. 2019, 20, 33.
- Lee, S.C.; Pan, C.Y.; Chen, J.Y. The antimicrobial peptide, epinecidin-1, mediates secretion of cytokines in the immune response to bacterial infection in mice. Peptides 2012, 36, 100–108.
- Pan, C.Y.; Chen, J.Y.; Lin, T.L.; Lin, C.H. In vitro activities of three synthetic peptides derived from epinecidin-1 and an anti-lipopolysaccharide factor against Propionibacterium acnes, Candida albicans, and Trichomonas vaginalis. Peptides 2009, 30, 1058–1068.
- Wang, Y.D.; Kung, C.W.; Chi, S.C.; Chen, J.Y. Inactivation of nervous necrosis virus infecting grouper (Epinephelus coioides) by epinecidin-1 and hepcidin 1-5 antimicrobial peptides, and downregulation of M × 2 and M × 3 gene expressions. Fish Shellfish Immunol. 2010, 28, 113–120.
- Huang, H.N.; Pan, C.Y.; Chen, J.Y. Grouper (Epinephelus coioides) antimicrobial peptide epinecidin-1 exhibits antiviral activity against foot-and-mouth disease virus in vitro. Peptides 2018, 106, 91–95.
- Galindo, I.; Alonso, C. African Swine Fever Virus: A Review. Viruses 2017, 9, 103.
- Mazur-Panasiuk, N.; Woźniakowski, G.; Niemczuk, K. The first complete genomic sequences of African swine fever virus isolated in Poland. Sci. Rep. 2019, 9, 4556.
- Anderson, E.C.; Hutchings, G.H.; Mukarati, N.; Wilkinson, P.J. African swine fever virus infection of the bushpig (Potamochoerus porcus) and its significance in the epidemiology of the disease. Vet. Microbiol. 1998, 62, 1–15.
- Alonso, C.; Miskin, J.; Hernáez, B.; Fernandez-Zapatero, P.; Soto, L.; Cantó, C.; Rodríguez-Crespo, I.; Dixon, L.; Escribano, J.M. African swine fever virus protein p54 interacts with the microtubular motor complex through direct binding to light-chain dynein. J. Virol. 2001, 75, 9819–9827.
- Hernaez, B.; Alonso, C. Dynamin- and clathrin-dependent endocytosis in African swine fever virus entry. J Virol. 2010, 84, 2100–2109.
- Döhner, K.; Gel, C.H.; Sodeik, B. Viral stop-and-go along microtubules: Taking a ride with dynein and kinesins. Trends Microbiol. 2005, 13, 320–327.
- Hernáez, B.; Tarragó, T.; Giralt, E.; Escribano, J.M.; Alonso, C. Small peptide inhibitors disrupt a high-affinity interaction between cytoplasmic dynein and a viral cargo protein. J. Virol. 2010, 84, 10792–10801.
- Raux, H.; Flamand, A.; Blondel, D. Interaction of the rabies virus P protein with the LC8 dynein light chain. J. Virol. 2000, 74, 10212–10216.
- Jacob, Y.; Badrane, H.; Ceccaldi, P.E.; Tordo, N. Cytoplasmic dynein LC8 interacts with lyssavirus phosphoprotein. J. Virol. 2000, 74, 10217–10222.
- Hu, H.; Guo, N.; Chen, S.; Guo, X.; Liu, X.; Ye, S.; Chai, Q.; Wang, Y.; Liu, B.; He, Q. Antiviral activity of Piscidin 1 against pseudorabies virus both in vitro and in vivo. Virol. J. 2019, 16, 95.
- Liu, C.G.; Jiang, L.; Liu. L.L.; Sun, L.; Zhao, W.G.; Chen, Y.Q.; Qi, T.M.; Han, Z.X.; Shao, Y.H.; Liu, S.W.; Ma, D.Y. Induction of Avian β-defensin 2 is possibly mediated by the p38 MAPK signal pathway in chicken embryo fibroblasts after newcastle disease virus infection. Front. Microbiol. 2018, 9, 751.
- Ma, D.; Lin, L.; Zhang, K.; Han, Z.X.; Shao, Y.H.; Liu, X.L.; Liu, S.W. Three novel Anas platyrhynchos avian β-defensins, upregulated by duck hepatitis virus, with antibacterial and antiviral activities. Mol. Immunol. 2011, 49, 84–96.
- Mackenzie, D.S.; Kovacs, N.J.; Snider, M.; Babiuk, L.A.; Hurk, S. Inclusion of the bovine neutrophil beta-defensin 3 with glycoprotein D of bovine herpesvirus 1 in a DNA vaccine modulates immune responses of mice and cattle. Clin. Vaccine Immunol. 2014, 21, 463–477.
- Monteiro, J.M.C.; Oliveira, M.D.; Dias, R.S.; Marcal, L.N.; Feio, R.N.; Ferreira, S.O.; Oliveira, L.L.; Silva, C.C.; Paula, S.O. The antimicrobial peptide HS-1 inhibits dengue virus infection. Virology 2018, 514, 79–87.
- Boas, L.C.P.V.; Campos, M.L.; Berlanda, R.L.A.; Neves, N.D.C.; Franco, O.L. Antiviral peptides as promising therapeutic drugs. Cell. Mol. Life Sci. CMLS 2019, 76, 3525–3542.
- Guo, C.; Cong, P.; He, Z.; Mo, D.; Zhang, W.; Chen, Y.; Liu, X. Inhibitory activity and molecular mechanism of protegrin-1 against porcine reproductive and respiratory syndrome virus in vitro. Antivir. Ther. 2015, 20, 573–582.
- Levast, B.; Hogan, D.; van Kessel, J.; Strom, S.; Walker, S.; Zhu, J.; Meurens, F.; Gerdts, V. Synthetic cationic peptide IDR-1002 and human cathelicidin LL37 modulate the cell innate response but differentially impact PRRSV replication in vitro. Front. Vet. Sci. 2019, 6, 233.
- Rai, A.; Pinto, S.; Velho, T.; Ferreira, A.; Moita, C.; Trivedi, U.; Evangelista, M.; Comune, M.; Rumbaugh, K.P.; Simoes, P.N.; et al. One-step synthesis of high-density peptide-conjugated gold nanoparticles with antimicrobial efficacy in a systemic infection model. Biomaterials 2016, 85, 99–110.
- Water, J.J.; Smart, S.; Franzyk, H.; Foged, C.; Nielsen, H.M. Nanoparticle-mediated delivery of the antimicrobial peptide plectasin against Staphylococcus aureus in infected epithelial cells. Eur. J. Pharm. Biopharm. 2015, 92, 65–73.
- Chen, H.X.; Mao, R.Y.; Teng, D.; Wang, X.M.; Hao, Y.; Feng, X.J.; Wang, J.H. Design and pharmacodynamics of recombinant NZ2114 histidine mutants with improved activity against methicillin-resistant Staphylococcus aureus. Amb Express 2017, 7, 46.
- Silva, T.; Silva, T.; Magalhães, B.; Maia, S.; Gomes, P.; Gomes, M.S. Killing of Mycobacterium avium by lactoferricin peptides: Improved activity of arginine-and D-amino-acid-containing molecules. Antimicrob. Agents Chemother. 2014, 58, 3461–3467.
- Nguyen, L.T.; Chau, J.K.; Perry, N.A.; Boer, L.; Zaat, S.A.J.; Vogel, H.J. Serum stabilities of short tryptophan-and arginine-rich antimicrobial peptide analogs. PLoS ONE 2010, 5, e12684.
- Chan, L.Y.; Zhang, V.M.; Huang, Y.H.; Waters, N.C.; Bansal, P.S.; Craik, D.J.; Daly, N.L. Cyclization of the antimicrobial peptide gomesin with native chemical ligation: Influences on stability and bioactivity. ChemBioChem 2013, 14, 617–624.
- Mao, R.Y.; Teng, D.; Wang, X.M.; Xi, D.; Zhang, Y.; Hu, X.Y.; Yang, Y.L.; Wang, J.H. Design, expression, and characterization of a novel targeted plectasin against methicillin-resistant Staphylococcus aureus. Appl. Microbiol. Biotechnol. 2013, 97, 3991–4002.
- Kuriakose, J.; Hernandez, G.V.; Nepal, M.; Brezden, A.; Pozzi, V.; Seleem, M.N.; Chmielewski, J. Targeting intracellular pathogenic bacteria with unnatural proline-rich peptides: Coupling antibacterial activity with macrophage penetration. Angew. Chem. 2013, 125, 9846–9849.
- Wang, X.; Teng, D.; Wang, X.M.; Hao, Y.; Chen, H.X.; Mao, R.Y.; Wang, J.H. Internalization, distribution, and activity of peptide H2 against the intracellular multidrug-resistant bovine mastitis-causing bacterium Staphylococcus aureus. Sci. Rep. 2019, 9, 7968.
- Li, Z.; Teng, D.; Mao, R.Y.; Wang, X.; Hao, Y.; Wang, X.M.; Wang, J.H. Improved antibacterial activity of the marine peptide N6 against intracellular Salmonella typhimurium by conjugating with the cell-penetrating peptide Tat11 via a cleavable linker. J. Med. Chem. 2018, 61, 7991–8000.
- Gomarasca, M.; Martins, T.F.C.; Greune, L.; Hardwidge, P.R.; Rüter, C. Bacterium-derived cell-penetrating peptides deliver gentamicin to kill intracellular pathogens. Antimicrob. Agents Chemother. 2014, 61, e02545-16.
- Richard, J.P.; Melikov, K.; Brooks, H.; Prevot, P.; Lebleu, B.; Chernomordik, L.V. Cellular uptake of unconjugated TAT peptide involves clathrin-dependent endocytosis and heparan sulfate receptors. J. Biol. Chem. 2005, 280, 15300–15306.
- Yang, N.; Teng, D.; Mao, R.Y.; Hao, Y.; Wang, X.; Wang, Z.L.; Wang, X.M.; Wang, J.H. A recombinant fungal defensin-like peptide-P2 combats multidrug-resistant Staphylococcus aureus and biofilms. Appl. Microbiol. Biotechnol. 2019, 103, 5193–5213.
- Wang, X.; Wang, X.M.; Teng, D.; Mao, R.Y.; Hao, Y.; Yang, N.; Li, Z.; Wang, J.H. Increased intracellular activity of MP1102 and NZ2114 against Staphylococcus aureus in vitro and in vivo. Sci. Rep. 2018, 8, 4204.
- Li, Z.; Wang, X.; Teng, D.; Mao, R.Y.; Hao, Y.; Yang, N.; Chen, H.X.; Wang, X.M.; Wang, J.H. Improved antibacterial activity of a marine peptide-N2 against intracellular Salmonella typhimurium by conjugating with cell-penetrating peptides-bLFcin6/Tat11. Eur. J. Med. Chem. 2017, 145, 263–272.
- Wang, Z.L.; Liu, X.H.; Teng, D.; Mao, R.Y.; Hao, Y.; Yang, N.; Wang, X.; Li, Z.Z.; Wang, X.M.; Wang. J.H. Development of chimeric peptides to facilitate the neutralisation of lipopolysaccharides during bactericidal targeting of multidrug-resistant Escherichia coli. Commun. Biol. 2020, 3, 41.
- Tang, H.J. The Research and Application of New Alternatives to Antibiotics (ATA) for Feed Usage; Chinese Academy of Agricultural Sciences (CAAS) Annual Report (2017); China Agricultural Science and Technology Press: Beijing, China, 2018; p. 29, ISBN 978-7-5116-3602-7.
- Zhang, Y.; Teng, D.; Mao, R.Y.; Wang, X.M.; Xi, D.; Hu, X.Y.; Wang, J.H. High expression of a plectasin-derived peptide NZ2114 in Pichia pastoris and its pharmacodynamics, postantibiotic and synergy against Staphylococcus aureus. Appl. Microbiol. Biotechnol. 2014, 98, 681–694.
- Czaplewski, L.; Bax, R.; Clokie, M.; Dawson, M.; Fairhead, H.; Fischetti, V.A.; Foster, S.; Gilmore, B.F.; Hancock, R.E.W.; Harper, D.; et al. Alternatives to antibiotics—A pipeline portfolio review. Lancet Infect. Dis. 2016, 16, 239–251.





