
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Ferenc Sipos | -- | 2921 | 2022-11-14 14:50:16 | | | |
| 2 | Rita Xu | -1 word(s) | 2920 | 2022-11-15 02:57:31 | | | | |
| 3 | Rita Xu | -3 word(s) | 2917 | 2022-11-21 02:23:45 | | |
Video Upload Options
The basic function of the immune system is the protection of the host against infections, along with the preservation of the individual antigenic identity. The process of self-tolerance covers the discrimination between self and foreign antigens, including proteins, nucleic acids, and larger molecules. Consequently, a broken immunological self-tolerance results in the development of autoimmune or autoinflammatory disorders. Immunocompetent cells express pattern-recognition receptors on their cell membrane and cytoplasm. The majority of endogenous DNA is located intracellularly within nuclei and mitochondria. Extracellular, cell-free DNA (cfDNA) can also be detected in a variety of diseases, which has sparked interest in using cfDNA as a possible biomarker. The widespread use of liquid biopsies and the increasing demand for screening, as well as monitoring disease activity and therapy response, have enabled the revival of cfDNA research.
1. Introduction
2. Origin, Release, Amount, and Clearance of Cell-Free DNA
3. Cell-Free DNA as a Molecular Marker or a Diagnostic Tool
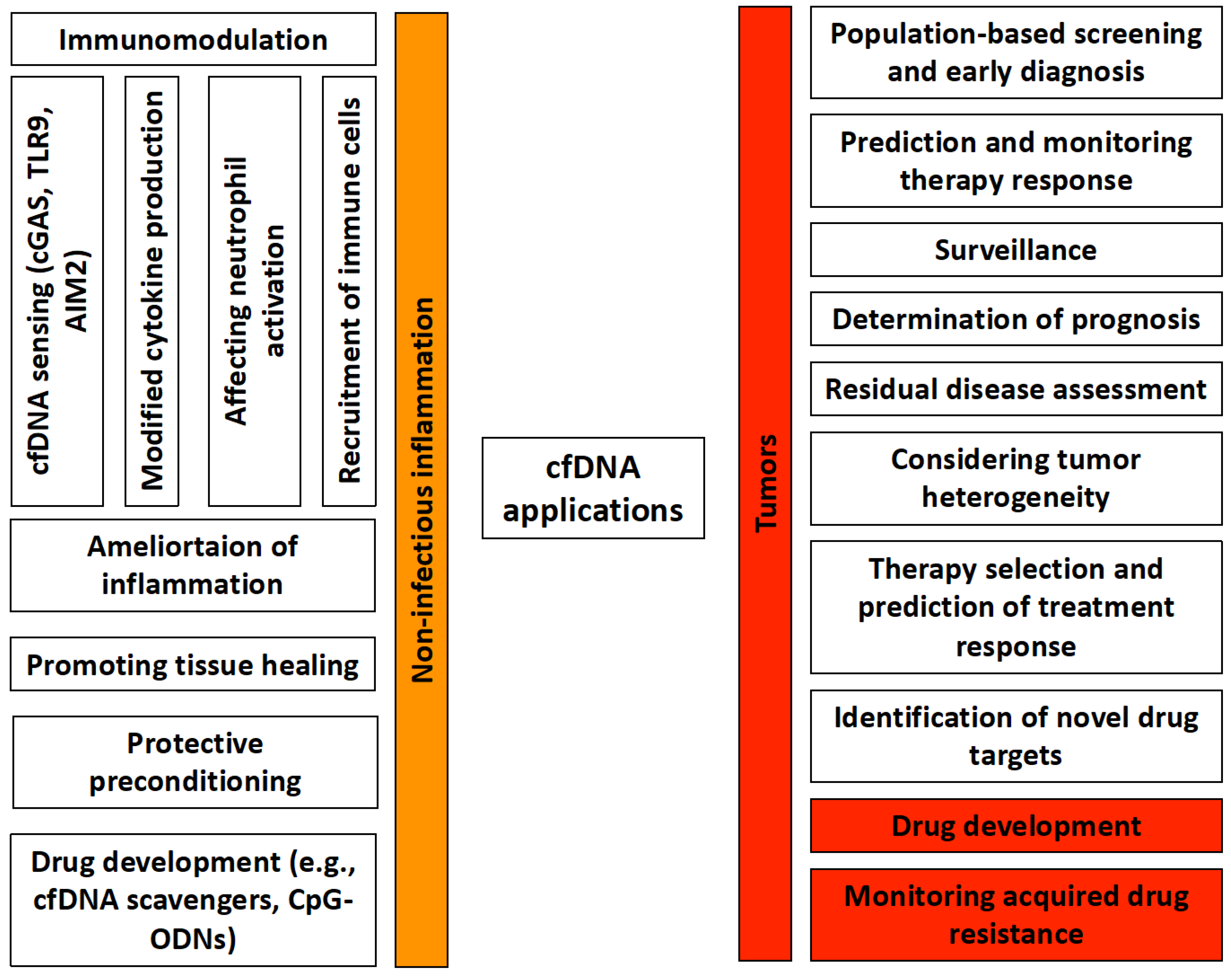
3.1. cfDNA in Prenatal Diagnosis
3.2. cfDNA in Tumors
3.3. cfDNA in Non-Tumor Disorders
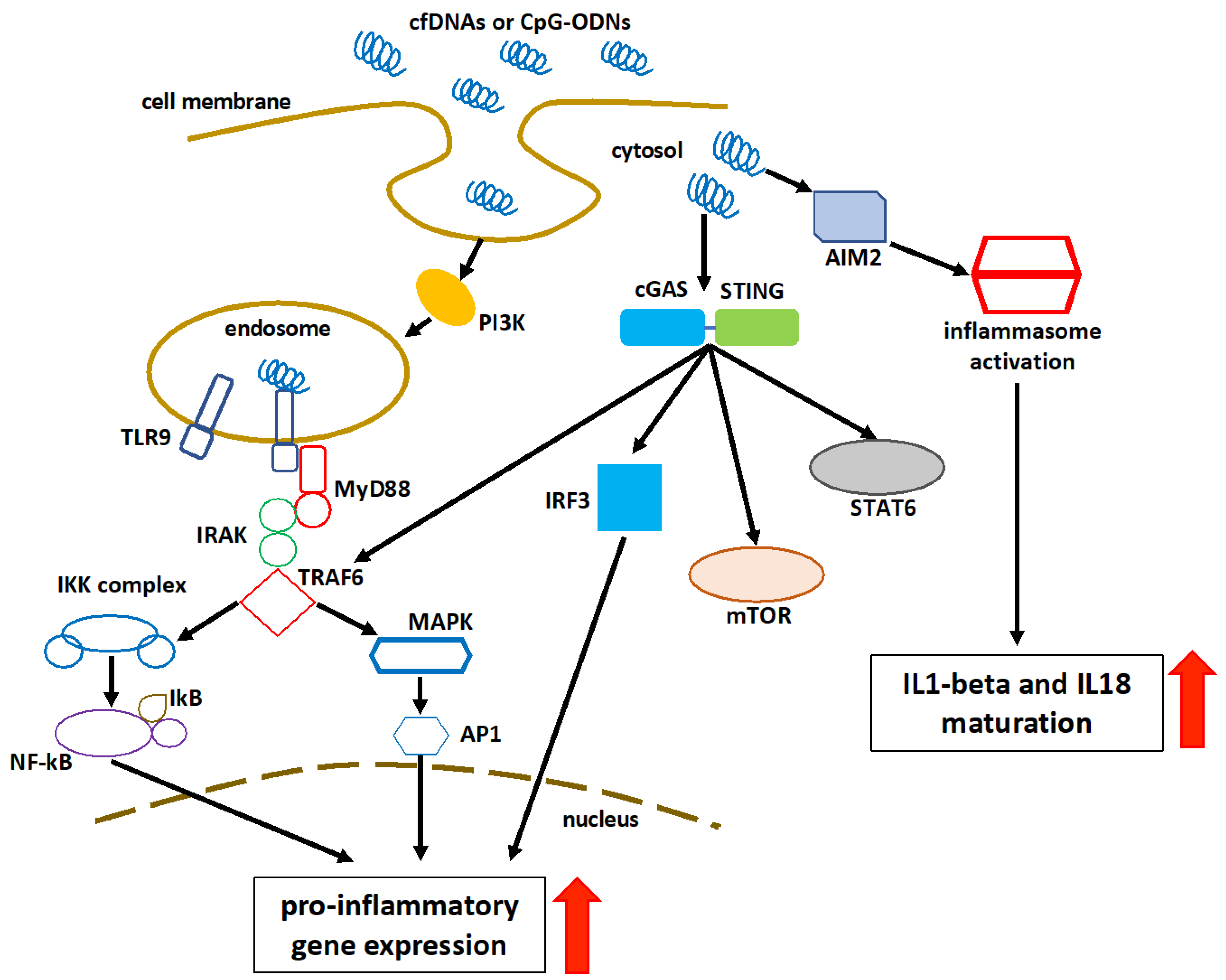
4. Recognition and Immunomodulatory Role of Cell-Free DNA
References
- Mandel, P.; Metais, P. Les acides nucléiques du plasma sanguin chez l’homme. C. R. Seances Soc. Biol. Fil. 1948, 142, 241–243.
- Tan, E.M.; Schur, P.H.; Carr, R.I.; Kunkel, H.G. Deoxybonucleic acid (DNA) and antibodies to DNA in the serum of patients with systemic lupus erythematosus. J. Clin. Investig. 1966, 45, 1732–1740.
- Leon, S.A.; Shapiro, B.; Sklaroff, D.M.; Yaros, M.J. Free DNA in the serum of cancer patients and the effect of therapy. Cancer Res. 1977, 37, 646–650.
- Lo, Y.M.; Corbetta, N.; Chamberlain, P.F.; Rai, V.; Sargent, I.L.; Redman, C.W.; Wainscoat, J.S. Presence of fetal DNA in maternal plasma and serum. Lancet 1997, 350, 485–487.
- Palomaki, G.E.; Kloza, E.M.; Lambert-Messerlian, G.M.; Haddow, J.E.; Neveux, L.M.; Ehrich, M.; van den Boom, D.; Bombard, A.T.; Deciu, C.; Grody, W.W.; et al. DNA sequencing of maternal plasma to detect Down syndrome: An international clinical validation study. Genet. Med. 2011, 13, 913–920.
- Hill, M.; Barrett, A.N.; White, H.; Chitty, L.S. Uses of cell free fetal DNA in maternal circulation. Best Pract. Res. Clin. Obs. Gynaecol. 2012, 26, 639–654.
- Banflavi, G.; Szyfter, K.; Antoni, F. Analysis of 5′-ends of short DNA fragments excreted by phytohemagglutinin stimulated lymphocytes. Biochem. Biophys. Res. Commun. 1984, 118, 140–146.
- Ranucci, R. Cell-Free DNA: Applications in Different Diseases. Methods Mol. Biol. 2019, 1909, 3–12.
- Spisák, S.; Solymosi, N.; Ittzés, P.; Bodor, A.; Kondor, D.; Vattay, G.; Barták, B.K.; Sipos, F.; Galamb, O.; Tulassay, Z.; et al. Complete genes may pass from food to human blood. PLoS ONE 2013, 8, e69805.
- Bronkhorst, A.J.; Ungerer, V.; Holdenrieder, S. The emerging role of cell-free DNA as a molecular marker for cancer management. Biomol. Detect. Quantif. 2019, 17, 100087.
- Shin, S.H.; Park, W.Y.; Park, D. Characterization of DNA lesions associated with cell-free DNA by targeted deep sequencing. BMC Med. Genom. 2021, 14, 192.
- Murao, A.; Aziz, M.; Wang, H.; Brenner, M.; Wang, P. Release mechanisms of major DAMPs. Apoptosis 2021, 26, 152–162.
- Fenech, M.; Kirsch-Volders, M.; Natarajan, A.T.; Surralles, J.; Crott, J.W.; Parry, J.; Norppa, H.; Eastmond, D.A.; Tucker, J.D.; Thomas, P. Molecular mechanisms of micronucleus, nucleoplasmic bridge and nuclear bud formation in mammalian and human cells. Mutagenesis 2011, 26, 125–132.
- Kustanovich, A.; Schwartz, R.; Peretz, T.; Grinshpun, A. Life and death of circulating cell-free DNA. Cancer Biol. Ther. 2019, 20, 1057–1067.
- Bronkhorst, A.J.; Wentzel, J.F.; Aucamp, J.; van Dyk, E.; du Plessis, L.; Pretorius, P.J. Characterization of the cell-free DNA released by cultured cancer cells. Biochim. Biophys. Acta 2016, 1863, 157–165.
- Martignano, F. Cell-Free DNA: An Overview of Sample Types and Isolation Procedures. Methods Mol. Biol. 2019, 1909, 13–27.
- Kılınç, N.; Onbaşılar, M.; Çayır, A. Evaluation of circulating cell-free nucleic acids in health workers occupationally exposed to ionizing radiation. Environ. Sci. Pollut. Res. Int. 2022, 29, 40543–40549.
- Zhong, Y.; Fan, Q.; Zhou, Z.; Wang, Y.; He, K.; Lu, J. Plasma cfDNA as a Potential Biomarker to Evaluate the Efficacy of Chemotherapy in Gastric Cancer. Cancer Manag. Res. 2020, 12, 3099–3106.
- Kadam, S.K.; Farmen, M.; Brandt, J.T. Quantitative measurement of cell-free plasma DNA and applications for detecting tumor genetic variation and promoter methylation in a clinical setting. J. Mol. Diagn. 2012, 14, 346–356.
- Hassan, S.; Shehzad, A.; Khan, S.A.; Miran, W.; Khan, S.; Lee, Y.S. Diagnostic and Therapeutic Potential of Circulating-Free DNA and Cell-Free RNA in Cancer Management. Biomedicines 2022, 10, 2047.
- MacKinnon, H.J.; Kolarova, T.R.; Katz, R.; Hedge, J.M.; Vinopal, E.; Lockwood, C.M.; Shree, R.; Delaney, S. The impact of maternal autoimmune disease on cell-free DNA test characteristics. Am. J. Obstet. Gynecol. MFM 2021, 3, 100466.
- Feng, C.S.; Lu, B.Y.; Ju, H.H.; Pan, W.J. The failure of non-invasive prenatal testing due to maternal dermatomyositis. Prenat. Diagn. 2019, 39, 958–961.
- Zhang, S.; Shu, X.; Tian, X.; Chen, F.; Lu, X.; Wang, G. Enhanced formation and impaired degradation of neutrophil extracellular traps in dermatomyositis and polymyositis: A potential contributor to interstitial lung disease complications. Clin. Exp. Immunol. 2014, 177, 134–141.
- Hashimoto, T.; Ueki, S.; Kamide, Y.; Miyabe, Y.; Fukuchi, M.; Yokoyama, Y.; Furukawa, T.; Azuma, N.; Oka, N.; Takeuchi, H.; et al. Increased Circulating Cell-Free DNA in Eosinophilic Granulomatosis with Polyangiitis: Implications for Eosinophil Extracellular Traps and Immunothrombosis. Front. Immunol. 2022, 12, 801897.
- Giaglis, S.; Daoudlarian, D.; Voll, R.E.; Kyburz, D.; Venhoff, N.; Walker, U.A. Circulating mitochondrial DNA copy numbers represent a sensitive marker for diagnosis and monitoring of disease activity in systemic lupus erythematosus. RMD Open 2021, 7, e002010.
- Camuzi Zovico, P.V.; Gasparini Neto, V.H.; Venâncio, F.A.; Soares Miguel, G.P.; Graça Pedrosa, R.; Kenji Haraguchi, F.; Barauna, V.G. Cell-free DNA as an obesity biomarker. Physiol. Res. 2020, 69, 515–520.
- Marcatti, M.; Saada, J.; Okereke, I.; Wade, C.E.; Bossmann, S.H.; Motamedi, M.; Szczesny, B. Quantification of Circulating Cell Free Mitochondrial DNA in Extracellular Vesicles with PicoGreen™ in Liquid Biopsies: Fast Assessment of Disease/Trauma Severity. Cells 2021, 10, 819.
- Otawara, M.; Roushan, M.; Wang, X.; Ellett, F.; Yu, Y.M.; Irimia, D. Microfluidic Assay Measures Increased Neutrophil Extracellular Traps Circulating in Blood after Burn Injuries. Sci. Rep. 2018, 8, 16983.
- Baumann, A.K.; Beck, J.; Kirchner, T.; Hartleben, B.; Schütz, E.; Oellerich, M.; Wedemeyer, H.; Jaeckel, E.; Taubert, R. Elevated fractional donor-derived cell-free DNA during subclinical graft injury after liver transplantation. Liver Transpl. 2022.
- Urosevic, N.; Merritt, A.J.; Inglis, T.J.J. Plasma cfDNA predictors of established bacteraemic infection. Access Microbiol. 2022, 4, acmi000373.
- Jing, Q.; Leung, C.H.C.; Wu, A.R. Cell-Free DNA as Biomarker for Sepsis by Integration of Microbial and Host Information. Clin. Chem. 2022, 68, 1184–1195.
- Sugasawa, T.; Fujita, S.I.; Kuji, T.; Ishibashi, N.; Tamai, K.; Kawakami, Y.; Takekoshi, K. Dynamics of Specific cfDNA Fragments in the Plasma of Full Marathon Participants. Genes 2021, 12, 676.
- Breitbach, S.; Tug, S.; Simon, P. Circulating cell-free DNA: An up-coming molecular marker in exercise physiology. Sports Med. 2012, 42, 565–586.
- Konuralp Atakul, B.; Koc, A.; Adiyaman, D.; Kuyucu, M.; Sahingoz Yildirim, A.G.; Saka Guvenc, M.; Erdogan, K.M.; Sengul, B.; Oztekin, D.C. Could high levels of cell-free DNA in maternal blood be associated with maternal health and perinatal outcomes? J. Obstet. Gynaecol. 2020, 40, 797–802.
- Bianchi, D.W.; Chudova, D.; Sehnert, A.J.; Bhatt, S.; Murray, K.; Prosen, T.L.; Garber, J.E.; Wilkins-Haug, L.; Vora, N.L.; Warsof, S.; et al. Noninvasive Prenatal Testing and Incidental Detection of Occult Maternal Malignancies. JAMA 2015, 314, 162–169.
- Ashoor, G.; Syngelaki, A.; Poon, L.C.; Rezende, J.C.; Nicolaides, K.H. Fetal fraction in maternal plasma cell-free DNA at 11–13 weeks’ gestation: Relation to maternal and fetal characteristics. Ultrasound Obstet. Gynecol. 2013, 41, 26–32.
- Han, D.S.C.; Ni, M.; Chan, R.W.Y.; Chan, V.W.H.; Lui, K.O.; Chiu, R.W.K.; Lo, Y.M.D. The Biology of Cell-free DNA Fragmentation and the Roles of DNASE1, DNASE1L3, and DFFB. Am. J. Hum. Genet. 2020, 106, 202–214.
- Leffler, J.; Ciacma, K.; Gullstrand, B.; Bengtsson, A.A.; Martin, M.; Blom, A.M. A subset of patients with systemic lupus erythematosus fails to degrade DNA from multiple clinically relevant sources. Arthritis Res. Ther. 2015, 17, 205.
- Barra, G.B.; Santa Rita, T.H.; de Almeida Vasques, J.; Chianca, C.F.; Nery, L.F.; Santana Soares Costa, S. EDTA-mediated inhibition of DNases protects circulating cell-free DNA from ex vivo degradation in blood samples. Clin. Biochem. 2015, 48, 976–981.
- Bodaño, A.; Amarelo, J.; González, A.; Gómez-Reino, J.J.; Conde, C. Novel DNASE I mutations related to systemic lupus erythematosus. Arthritis Rheum. 2004, 50, 4070–4071.
- Duvvuri, B.; Lood, C. Cell-Free DNA as a Biomarker in Autoimmune Rheumatic Diseases. Front. Immunol. 2019, 10, 502.
- Hartl, J.; Serpas, L.; Wang, Y.; Rashidfarrokhi, A.; Perez, O.A.; Sally, B.; Sisirak, V.; Soni, C.; Khodadadi-Jamayran, A.; Tsirigos, A.; et al. Autoantibody-mediated impairment of DNASE1L3 activity in sporadic systemic lupus erythematosus. J. Exp. Med. 2021, 218, e20201138.
- Felux, J.; Erbacher, A.; Breckler, M.; Hervé, R.; Lemeiter, D.; Mannherz, H.G.; Napirei, M.; Rammensee, H.G.; Decker, P. Deoxyribonuclease 1-Mediated Clearance of Circulating Chromatin Prevents from Immune Cell Activation and Pro-inflammatory Cytokine Production, a Phenomenon Amplified by Low Trap1 Activity: Consequences for Systemic Lupus Erythematosus. Front. Immunol. 2021, 12, 613597.
- Gaipl, U.S.; Beyer, T.D.; Heyder, P.; Kuenkele, S.; Böttcher, A.; Voll, R.E.; Kalden, J.R.; Herrmann, M. Cooperation between C1q and DNase I in the clearance of necrotic cell-derived chromatin. Arthritis Rheum. 2004, 50, 640–649.
- Fredi, M.; Bianchi, M.; Andreoli, L.; Greco, G.; Olivieri, I.; Orcesi, S.; Fazzi, E.; Cereda, C.; Tincani, A. Typing TREX1 gene in patients with systemic lupus erythematosus. Reumatismo 2015, 67, 1–7.
- Gillmore, J.D.; Hutchinson, W.L.; Herbert, J.; Bybee, A.; Mitchell, D.A.; Hasserjian, R.P.; Yamamura, K.; Suzuki, M.; Sabin, C.A.; Pepys, M.B. Autoimmunity and glomerulonephritis in mice with targeted deletion of the serum amyloid P component gene: SAP deficiency or strain combination? Immunology 2004, 112, 255–264.
- Ogden, C.A.; Kowalewski, R.; Peng, Y.; Montenegro, V.; Elkon, K.B. IGM is required for efficient complement mediated phagocytosis of apoptotic cells in vivo. Autoimmunity 2005, 38, 259–264.
- Janko, C.; Franz, S.; Munoz, L.E.; Siebig, S.; Winkler, S.; Schett, G.; Lauber, K.; Sheriff, A.; van der Vlag, J.; Herrmann, M. CRP/anti-CRP antibodies assembly on the surfaces of cell remnants switches their phagocytic clearance toward inflammation. Front. Immunol. 2011, 2, 70.
- Saevarsdottir, S.; Steinsson, K.; Ludviksson, B.R.; Grondal, G.; Valdimarsson, H. Mannan-binding lectin may facilitate the clearance of circulating immune complexes-implications from a study on C2-deficient individuals. Clin. Exp. Immunol. 2007, 148, 248–253.
- Jelin, A.C.; Sagaser, K.G.; Wilkins-Haug, L. Prenatal Genetic Testing Options. Pediatr. Clin. N. Am. 2019, 66, 281–293.
- Papageorgiou, E.A.; Karagrigoriou, A.; Tsaliki, E.; Velissariou, V.; Carter, N.P.; Patsalis, P.C. Fetal-specific DNA methylation ratio permits noninvasive prenatal diagnosis of trisomy 21. Nat. Med. 2011, 17, 510–513.
- Karakas, B.; Qubbaj, W.; Al-Hassan, S.; Coskun, S. Noninvasive Digital Detection of Fetal DNA in Plasma of 4-Week-Pregnant Women following In Vitro Fertilization and Embryo Transfer. PLoS ONE 2015, 10, e0126501.
- Yu, S.C.; Lee, S.W.; Jiang, P.; Leung, T.Y.; Chan, K.C.; Chiu, R.W.; Lo, Y.M. High-resolution profiling of fetal DNA clearance from maternal plasma by massively parallel sequencing. Clin. Chem. 2013, 59, 1228–1237.
- Rabinowitz, T.; Shomron, N. Genome-wide noninvasive prenatal diagnosis of monogenic disorders: Current and future trends. Comput. Struct. Biotechnol. J. 2020, 18, 2463–2470.
- Yaşa, B.; Şahin, O.; Öcüt, E.; Seven, M.; Sözer, S. Assessment of Fetal Rhesus D and Gender with Cell-Free DNA and Exosomes from Maternal Blood. Reprod. Sci. 2021, 28, 562–569.
- Gil, M.M.; Accurti, V.; Santacruz, B.; Plana, M.N.; Nicolaides, K.H. Analysis of cell-free DNA in maternal blood in screening for aneuploidies: Updated meta-analysis. Ultrasound Obstet. Gynecol. 2017, 50, 302–314.
- Gerson, K.D.; O’Brien, B.M. Cell-Free DNA: Screening for Single-Gene Disorders and Determination of Fetal Rhesus D Genotype. Obstet. Gynecol. Clin. N. Am. 2018, 45, 27–39.
- Chiu, E.K.L.; Hui, W.W.I.; Chiu, R.W.K. cfDNA screening and diagnosis of monogenic disorders—Where are we heading? Prenat. Diagn. 2018, 38, 52–58.
- Ulrich, B.C.; Paweletz, C.P. Cell-Free DNA in Oncology: Gearing up for Clinic. Ann. Lab. Med. 2018, 38, 1–8.
- Siravegna, G.; Mussolin, B.; Venesio, T.; Marsoni, S.; Seoane, J.; Dive, C.; Papadopoulos, N.; Kopetz, S.; Corcoran, R.B.; Siu, L.L.; et al. How liquid biopsies can change clinical practice in oncology. Ann. Oncol. 2019, 30, 1580–1590.
- Domínguez-Vigil, I.G.; Moreno-Martínez, A.K.; Wang, J.Y.; Roehrl, M.H.A.; Barrera-Saldaña, H.A. The dawn of the liquid biopsy in the fight against cancer. Oncotarget 2017, 9, 2912–2922.
- Guo, Q.; Wang, J.; Xiao, J.; Wang, L.; Hu, X.; Yu, W.; Song, G.; Lou, J.; Chen, J. Heterogeneous mutation pattern in tumor tissue and circulating tumor DNA warrants parallel NGS panel testing. Mol. Cancer 2018, 17, 131.
- Li, J.; Dittmar, R.L.; Xia, S.; Zhang, H.; Du, M.; Huang, C.C.; Druliner, B.R.; Boardman, L.; Wang, L. Cell-free DNA copy number variations in plasma from colorectal cancer patients. Mol. Oncol. 2017, 11, 1099–1111.
- Yu, D.; Liu, Z.; Su, C.; Han, Y.; Duan, X.; Zhang, R.; Liu, X.; Yang, Y.; Xu, S. Copy number variation in plasma as a tool for lung cancer prediction using Extreme Gradient Boosting (XGBoost) classifier. Thorac. Cancer 2020, 11, 95–102.
- Willis, J.; Lefterova, M.I.; Artyomenko, A.; Kasi, P.M.; Nakamura, Y.; Mody, K.; Catenacci, D.V.T.; Fakih, M.; Barbacioru, C.; Zhao, J.; et al. Validation of Microsatellite Instability Detection Using a Comprehensive Plasma-Based Genotyping Panel. Clin. Cancer Res. 2019, 25, 7035–7045.
- Müller, J.N.; Falk, M.; Talwar, J.; Neemann, N.; Mariotti, E.; Bertrand, M.; Zacherle, T.; Lakis, S.; Menon, R.; Gloeckner, C.; et al. Concordance between Comprehensive Cancer Genome Profiling in Plasma and Tumor Specimens. J. Thorac. Oncol. 2017, 12, 1503–1511.
- Park, S.; Olsen, S.; Ku, B.M.; Lee, M.S.; Jung, H.A.; Sun, J.M.; Lee, S.H.; Ahn, J.S.; Park, K.; Choi, Y.L.; et al. High concordance of actionable genomic alterations identified between circulating tumor DNA-based and tissue-based next-generation sequencing testing in advanced non-small cell lung cancer: The Korean Lung Liquid Versus Invasive Biopsy Program. Cancer 2021, 127, 3019–3028.
- Schmiegel, W.; Scott, R.J.; Dooley, S.; Lewis, W.; Meldrum, C.J.; Pockney, P.; Draganic, B.; Smith, S.; Hewitt, C.; Philimore, H.; et al. Blood-based detection of RAS mutations to guide anti-EGFR therapy in colorectal cancer patients: Concordance of results from circulating tumor DNA and tissue-based RAS testing. Mol. Oncol. 2017, 11, 208–219.
- Phallen, J.; Sausen, M.; Adleff, V.; Leal, A.; Hruban, C.; White, J.; Anagnostou, V.; Fiksel, J.; Cristiano, S.; Papp, E.; et al. Direct detection of early-stage cancers using circulating tumor DNA. Sci. Transl. Med. 2017, 9, eaan2415.
- Nesic, M.; Bødker, J.S.; Terp, S.K.; Dybkær, K. Optimization of Preanalytical Variables for cfDNA Processing and Detection of ctDNA in Archival Plasma Samples. Biomed. Res. Int. 2021, 2021, 5585148.
- Bredno, J.; Lipson, J.; Venn, O.; Aravanis, A.M.; Jamshidi, A. Clinical correlates of circulating cell-free DNA tumor fraction. PLoS ONE 2021, 16, e0256436.
- Cai, Z.; Chen, G.; Zeng, Y.; Dong, X.; Li, Z.; Huang, Y.; Xin, F.; Qiu, L.; Xu, H.; Zhang, W.; et al. Comprehensive Liquid Profiling of Circulating Tumor DNA and Protein Biomarkers in Long-Term Follow-Up Patients with Hepatocellular Carcinoma. Clin. Cancer Res. 2019, 25, 5284–5294.
- Isaksson, S.; George, A.M.; Jönsson, M.; Cirenajwis, H.; Jönsson, P.; Bendahl, P.O.; Brunnström, H.; Staaf, J.; Saal, L.H.; Planck, M. Pre-operative plasma cell-free circulating tumor DNA and serum protein tumor markers as predictors of lung adenocarcinoma recurrence. Acta Oncol. 2019, 58, 1079–1086.
- Chen, Z.; Liu, L.; Zhu, F.; Cai, X.; Zhao, Y.; Liang, P.; Ou, L.; Zhong, R.; Yu, Z.; Li, C.; et al. Dynamic monitoring serum tumor markers to predict molecular features of EGFR-mutated lung cancer during targeted therapy. Cancer Med. 2022, 11, 3115–3125.
- Tarazona, N.; Gimeno-Valiente, F.; Gambardella, V.; Zuñiga, S.; Rentero-Garrido, P.; Huerta, M.; Roselló, S.; Martinez-Ciarpaglini, C.; Carbonell-Asins, J.A.; Carrasco, F.; et al. Targeted next-generation sequencing of circulating-tumor DNA for tracking minimal residual disease in localized colon cancer. Ann. Oncol. 2019, 30, 1804–1812.
- Parsons, H.A.; Rhoades, J.; Reed, S.C.; Gydush, G.; Ram, P.; Exman, P.; Xiong, K.; Lo, C.C.; Li, T.; Fleharty, M.; et al. Sensitive Detection of Minimal Residual Disease in Patients Treated for Early-Stage Breast Cancer. Clin. Cancer Res. 2020, 26, 2556–2564.
- Powles, T.; Assaf, Z.J.; Davarpanah, N.; Banchereau, R.; Szabados, B.E.; Yuen, K.C.; Grivas, P.; Hussain, M.; Oudard, S.; Gschwend, J.E.; et al. ctDNA guiding adjuvant immunotherapy in urothelial carcinoma. Nature 2021, 595, 432–437.
- Keller, L.; Belloum, Y.; Wikman, H.; Pantel, K. Clinical relevance of blood-based ctDNA analysis: Mutation detection and beyond. Br. J. Cancer 2021, 124, 345–358.
- Kakitsuka, Y.; Sawamura, S.; Kajihara, I.; Kanemaru, H.; Honda, N.; Makino, K.; Aoi, J.; Makino, T.; Fukushima, S.; Ihn, H. Elevated circulating cell-free DNA levels in autoimmune bullous diseases. J. Dermatol. 2020, 47, e345–e346.
- Dunaeva, M.; Buddingh’, B.C.; Toes, R.E.; Luime, J.J.; Lubberts, E.; Pruijn, G.J. Decreased serum cell-free DNA levels in rheumatoid arthritis. Auto Immun. Highlights 2015, 6, 23–30.
- Truszewska, A.; Wirkowska, A.; Gala, K.; Truszewski, P.; Krzemień-Ojak, Ł.; Perkowska-Ptasińska, A.; Mucha, K.; Pączek, L.; Foroncewicz, B. Cell-free DNA profiling in patients with lupus nephritis. Lupus 2020, 29, 1759–1772.
- Xu, Y.; Song, Y.; Chang, J.; Zhou, X.; Qi, Q.; Tian, X.; Li, M.; Zeng, X.; Xu, M.; Zhang, W.; et al. High levels of circulating cell-free DNA are a biomarker of active SLE. Eur. J. Clin. Investig. 2018, 48, e13015.
- Vajpeyee, A.; Wijatmiko, T.; Vajpeyee, M.; Taywade, O.; Pandey, S.; Chauhan, P.S. Clinical Usefulness of Cell-Free DNA as a Prognostic Marker in Acute Ischemic Stroke. Neurologist 2020, 25, 11–13.
- Agiannitopoulos, K.; Samara, P.; Papadopoulou, E.; Tsamis, K.; Mertzanos, G.; Babalis, D.; Lamnissou, K. Study on the admission levels of circulating cell-free DNA in patients with acute myocardial infarction using different quantification methods. Scand. J. Clin. Lab. Investig. 2020, 80, 348–350.
- Sanchis, J.; García-Blas, S.; Ortega-Paz, L.; Dantas, A.P.; Rodríguez, E.; Abellán, L.; Brugaletta, S.; Valero, E.; Miñana, G.; Garabito, M.; et al. Cell-free DNA and Microvascular Damage in ST-segment Elevation Myocardial Infarction Treated with Primary Percutaneous Coronary Intervention. Rev. Esp. Cardiol. 2019, 72, 317–323.
- Agbor-Enoh, S.; Shah, P.; Tunc, I.; Hsu, S.; Russell, S.; Feller, E.; Shah, K.; Rodrigo, M.E.; Najjar, S.S.; Kong, H.; et al. Cell-Free DNA to Detect Heart Allograft Acute Rejection. Circulation 2021, 143, 1184–1197.
- Truszewska, A.; Foroncewicz, B.; Pączek, L. The role and diagnostic value of cell-free DNA in systemic lupus erythematosus. Clin. Exp. Rheumatol. 2017, 35, 330–336.
- Lauková, L.; Konečná, B.; Vlková, B.; Mlynáriková, V.; Celec, P.; Šteňová, E. Anti-cytokine therapy and plasma DNA in patients with rheumatoid arthritis. Rheumatol. Int. 2018, 38, 1449–1454.
- Rykova, E.; Sizikov, A.; Roggenbuck, D.; Antonenko, O.; Bryzgalov, L.; Morozkin, E.; Skvortsova, K.; Vlassov, V.; Laktionov, P.; Kozlov, V. Circulating DNA in rheumatoid arthritis: Pathological changes and association with clinically used serological markers. Arthritis Res. Ther. 2017, 19, 85.
- Dong, C.; Liu, Y.; Sun, C.; Liang, H.; Dai, L.; Shen, J.; Wei, S.; Guo, S.; Leong, K.W.; Chen, Y.; et al. Identification of Specific Joint-Inflammatogenic Cell-Free DNA Molecules from Synovial Fluids of Patients with Rheumatoid Arthritis. Front. Immunol. 2020, 11, 662.
- Glebova, K.V.; Veiko, N.N.; Nikonov, A.A.; Porokhovnik, L.N.; Kostuyk, S.V. Cell-free DNA as a biomarker in stroke: Current status, problems and perspectives. Crit. Rev. Clin. Lab. Sci. 2018, 55, 55–70.
- Grosse, G.M.; Blume, N.; Abu-Fares, O.; Götz, F.; Ernst, J.; Leotescu, A.; Gabriel, M.M.; van Gemmeren, T.; Worthmann, H.; Lichtinghagen, R.; et al. Endogenous Deoxyribonuclease Activity and Cell-Free Deoxyribonucleic Acid in Acute Ischemic Stroke: A Cohort Study. Stroke 2022, 53, 1235–1244.
- Cui, X.; Du, S.; Liu, H.; Liu, J.; Wu, Q.; Huo, Q.; Qi, Y.; Qin, X.; Yang, Y.; Li, W. The Length and Distribution of Plasma Cell-Free DNA Fragments in Stroke Patients. Biomed. Res. Int. 2020, 2020, 9054196.
- Xie, J.; Yang, J.; Hu, P. Correlations of Circulating Cell-Free DNA With Clinical Manifestations in Acute Myocardial Infarction. Am. J. Med. Sci. 2018, 356, 121–129.
- Lippi, G.; Sanchis-Gomar, F.; Cervellin, G. Cell-free DNA for diagnosing myocardial infarction: Not ready for prime time. Clin. Chem. Lab. Med. 2015, 53, 1895–1901.
- Stawski, R.; Stec-Martyna, E.; Chmielecki, A.; Nowak, D.; Perdas, E. Current Trends in Cell-Free DNA Applications. Scoping Review of Clinical Trials. Biology 2021, 10, 906.
- Schütz, E.; Fischer, A.; Beck, J.; Harden, M.; Koch, M.; Wuensch, T.; Stockmann, M.; Nashan, B.; Kollmar, O.; Matthaei, J.; et al. Graft-derived cell-free DNA, a noninvasive early rejection and graft damage marker in liver transplantation: A prospective, observational, multicenter cohort study. PLoS Med. 2017, 14, e1002286.
- Stortz, J.A.; Hawkins, R.B.; Holden, D.C.; Raymond, S.L.; Wang, Z.; Brakenridge, S.C.; Cuschieri, J.; Moore, F.A.; Maier, R.V.; Moldawer, L.L.; et al. Cell-free nuclear, but not mitochondrial, DNA concentrations correlate with the early host inflammatory response after severe trauma. Sci. Rep. 2019, 9, 13648.
- Hauser, C.J.; Otterbein, L.E. Danger signals from mitochondrial DAMPS in trauma and post-injury sepsis. Eur. J. Trauma Emerg. Surg. 2018, 44, 317–324.
- Kumar, V. The Trinity of cGAS, TLR9, and ALRs Guardians of the Cellular Galaxy Against Host-Derived Self-DNA. Front. Immunol. 2021, 11, 624597.
- Wang, H.; Zang, C.; Ren, M.; Shang, M.; Wang, Z.; Peng, X.; Zhang, Q.; Wen, X.; Xi, Z.; Zhou, C. Cellular uptake of extracellular nucleosomes induces innate immune responses by binding and activating cGMP-AMP synthase (cGAS). Sci. Rep. 2020, 10, 15385.
- Ishikawa, H.; Barber, G.N. STING is an endoplasmic reticulum adaptor that facilitates innate immune signalling. Nature 2008, 455, 674–678, Erratum in Nature 2008, 456, 274.
- Ishikawa, H.; Ma, Z.; Barber, G.N. STING regulates intracellular DNA-mediated, type I interferon-dependent innate immunity. Nature 2009, 461, 788–792.
- Hooy, R.M.; Sohn, J. The allosteric activation of cGAS underpins its dynamic signaling landscape. Elife 2018, 7, e39984.
- Luecke, S.; Holleufer, A.; Christensen, M.H.; Jønsson, K.L.; Boni, G.A.; Sørensen, L.K.; Johannsen, M.; Jakobsen, M.R.; Hartmann, R.; Paludan, S.R. cGAS is activated by DNA in a length-dependent manner. EMBO Rep. 2017, 18, 1707–1715.
- Liu, S.; Cai, X.; Wu, J.; Cong, Q.; Chen, X.; Li, T.; Du, F.; Ren, J.; Wu, Y.T.; Grishin, N.V.; et al. Phosphorylation of innate immune adaptor proteins MAVS, STING, and TRIF induces IRF3 activation. Science 2015, 347, aaa2630.
- Chen, H.; Sun, H.; You, F.; Sun, W.; Zhou, X.; Chen, L.; Yang, J.; Wang, Y.; Tang, H.; Guan, Y.; et al. Activation of STAT6 by STING is critical for antiviral innate immunity. Cell 2011, 147, 436–446.
- Abe, T.; Barber, G.N. Cytosolic-DNA-mediated, STING-dependent proinflammatory gene induction necessitates canonical NF-κB activation through TBK1. J. Virol. 2014, 88, 5328–5341.
- Saitoh, T.; Fujita, N.; Hayashi, T.; Takahara, K.; Satoh, T.; Lee, H.; Matsunaga, K.; Kageyama, S.; Omori, H.; Noda, T.; et al. Atg9a controls dsDNA-driven dynamic translocation of STING and the innate immune response. Proc. Natl. Acad. Sci. USA 2009, 106, 20842–20846.
- Liang, Q.; Seo, G.J.; Choi, Y.J.; Kwak, M.J.; Ge, J.; Rodgers, M.A.; Shi, M.; Leslie, B.J.; Hopfner, K.P.; Ha, T.; et al. Crosstalk between the cGAS DNA sensor and Beclin-1 autophagy protein shapes innate antimicrobial immune responses. Cell Host Microbe 2014, 15, 228–238.
- Latz, E.; Schoenemeyer, A.; Visintin, A.; Fitzgerald, K.A.; Monks, B.G.; Knetter, C.F.; Lien, E.; Nilsen, N.J.; Espevik, T.; Golenbock, D.T. TLR9 signals after translocating from the ER to CpG DNA in the lysosome. Nat. Immunol. 2004, 5, 190–198.
- Miyake, K.; Shibata, T.; Fukui, R.; Sato, R.; Saitoh, S.I.; Murakami, Y. Nucleic Acid Sensing by Toll-Like Receptors in the Endosomal Compartment. Front. Immunol. 2022, 13, 941931.
- Miyake, K. Nucleic acid-sensing Toll-like receptors: Beyond ligand search. Adv. Drug. Deliv. Rev. 2008, 60, 782–785.
- De Nardo, D.; Balka, K.R.; Cardona Gloria, Y.; Rao, V.R.; Latz, E.; Masters, S.L. Interleukin-1 receptor-associated kinase 4 (IRAK4) plays a dual role in myddosome formation and Toll-like receptor signaling. J. Biol. Chem. 2018, 293, 15195–15207.
- Singer, J.W.; Fleischman, A.; Al-Fayoumi, S.; Mascarenhas, J.O.; Yu, Q.; Agarwal, A. Inhibition of interleukin-1 receptor-associated kinase 1 (IRAK1) as a therapeutic strategy. Oncotarget 2018, 9, 33416–33439.
- Dong, W.; Liu, Y.; Peng, J.; Chen, L.; Zou, T.; Xiao, H.; Liu, Z.; Li, W.; Bu, Y.; Qi, Y. The IRAK-1-BCL10-MALT1-TRAF6-TAK1 cascade mediates signaling to NF-kappaB from Toll-like receptor 4. J. Biol. Chem. 2006, 281, 26029–26040.
- Wu, Z.H.; Wong, E.T.; Shi, Y.; Niu, J.; Chen, Z.; Miyamoto, S.; Tergaonkar, V. ATM- and NEMO-dependent ELKS ubiquitination coordinates TAK1-mediated IKK activation in response to genotoxic stress. Mol. Cell 2010, 40, 75–86.
- Pohar, J.; Kužnik Krajnik, A.; Jerala, R.; Benčina, M. Minimal sequence requirements for oligodeoxyribonucleotides activating human TLR9. J. Immunol. 2015, 194, 3901–3908.
- Pohar, J.; Lainšček, D.; Ivičak-Kocjan, K.; Cajnko, M.M.; Jerala, R.; Benčina, M. Short single-stranded DNA degradation products augment the activation of Toll-like receptor 9. Nat. Commun. 2017, 8, 15363.
- Pohar, J.; Yamamoto, C.; Fukui, R.; Cajnko, M.M.; Miyake, K.; Jerala, R.; Benčina, M. Selectivity of Human TLR9 for Double CpG Motifs and Implications for the Recognition of Genomic DNA. J. Immunol. 2017, 198, 2093–2104.
- Barton, G.M.; Kagan, J.C.; Medzhitov, R. Intracellular localization of Toll-like receptor 9 prevents recognition of self DNA but facilitates access to viral DNA. Nat. Immunol. 2006, 7, 49–56.
- Jansen, M.P.; Emal, D.; Teske, G.J.; Dessing, M.C.; Florquin, S.; Roelofs, J.J. Release of extracellular DNA influences renal ischemia reperfusion injury by platelet activation and formation of neutrophil extracellular traps. Kidney Int. 2017, 91, 352–364.
- Aslam, R.; Speck, E.R.; Kim, M.; Crow, A.R.; Bang, K.W.; Nestel, F.P.; Ni, H.; Lazarus, A.H.; Freedman, J.; Semple, J.W. Platelet Toll-like receptor expression modulates lipopolysaccharide-induced thrombocytopenia and tumor necrosis factor-alpha production in vivo. Blood 2006, 107, 637–641.
- Panigrahi, S.; Ma, Y.; Hong, L.; Gao, D.; West, X.Z.; Salomon, R.G.; Byzova, T.V.; Podrez, E.A. Engagement of platelet toll-like receptor 9 by novel endogenous ligands promotes platelet hyperreactivity and thrombosis. Circ. Res. 2013, 112, 103–112.
- Lugrin, J.; Martinon, F. The AIM2 inflammasome: Sensor of pathogens and cellular perturbations. Immunol. Rev. 2018, 281, 99–114.
- Wang, B.; Tian, Y.; Yin, Q. AIM2 Inflammasome Assembly and Signaling. Adv. Exp. Med. Biol. 2019, 1172, 143–155.
- Matyszewski, M.; Morrone, S.R.; Sohn, J. Digital signaling network drives the assembly of the AIM2-ASC inflammasome. Proc. Natl. Acad. Sci. USA 2018, 115, E1963–E1972.
- Morrone, S.R.; Matyszewski, M.; Yu, X.; Delannoy, M.; Egelman, E.H.; Sohn, J. Assembly-driven activation of the AIM2 foreign-dsDNA sensor provides a polymerization template for downstream ASC. Nat. Commun. 2015, 6, 7827.
- Banerjee, I.; Behl, B.; Mendonca, M.; Shrivastava, G.; Russo, A.J.; Menoret, A.; Ghosh, A.; Vella, A.T.; Vanaja, S.K.; Sarkar, S.N.; et al. Gasdermin D Restrains Type I Interferon Response to Cytosolic DNA by Disrupting Ionic Homeostasis. Immunity 2018, 49, 413–426.e5.
- Evavold, C.L.; Ruan, J.; Tan, Y.; Xia, S.; Wu, H.; Kagan, J.C. The Pore-Forming Protein Gasdermin D Regulates Interleukin-1 Secretion from Living Macrophages. Immunity 2018, 48, 35–44.e6.
- Liu, Z.; Wang, C.; Yang, J.; Zhou, B.; Yang, R.; Ramachandran, R.; Abbott, D.W.; Xiao, T.S. Crystal Structures of the Full-Length Murine and Human Gasdermin D Reveal Mechanisms of Autoinhibition, Lipid Binding, and Oligomerization. Immunity 2019, 51, 43–49.e4.
- Yan, S.; Shen, H.; Lian, Q.; Jin, W.; Zhang, R.; Lin, X.; Gu, W.; Sun, X.; Meng, G.; Tian, Z.; et al. Deficiency of the AIM2-ASC Signal Uncovers the STING-Driven Overreactive Response of Type I IFN and Reciprocal Depression of Protective IFN-γ Immunity in Mycobacterial Infection. J. Immunol. 2018, 200, 1016–1026.





