Your browser does not fully support modern features. Please upgrade for a smoother experience.

Submitted Successfully!
Thank you for your contribution! You can also upload a video entry or images related to this topic.
For video creation, please contact our Academic Video Service.
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Horváth László | -- | 6047 | 2022-11-02 11:10:07 | | | |
| 2 | Amina Yu | -2 word(s) | 6045 | 2022-11-03 03:10:47 | | |
Video Upload Options
We provide professional Academic Video Service to translate complex research into visually appealing presentations. Would you like to try it?
Cite
If you have any further questions, please contact Encyclopedia Editorial Office.
Horváth, L.; Kovács, �.; Csorbai, B.; Hegyi, �.; Lefler, K.; Müller, T.; Urbányi, B. Biological and Hydrobiological Background of Pond Carp Breeding. Encyclopedia. Available online: https://encyclopedia.pub/entry/32546 (accessed on 21 May 2026).
Horváth L, Kovács �, Csorbai B, Hegyi �, Lefler K, Müller T, et al. Biological and Hydrobiological Background of Pond Carp Breeding. Encyclopedia. Available at: https://encyclopedia.pub/entry/32546. Accessed May 21, 2026.
Horváth, Laszló, Éva Kovács, Balázs Csorbai, Árpád Hegyi, Kinga Lefler, Tamás Müller, Béla Urbányi. "Biological and Hydrobiological Background of Pond Carp Breeding" Encyclopedia, https://encyclopedia.pub/entry/32546 (accessed May 21, 2026).
Horváth, L., Kovács, �., Csorbai, B., Hegyi, �., Lefler, K., Müller, T., & Urbányi, B. (2022, November 02). Biological and Hydrobiological Background of Pond Carp Breeding. In Encyclopedia. https://encyclopedia.pub/entry/32546
Horváth, Laszló, et al. "Biological and Hydrobiological Background of Pond Carp Breeding." Encyclopedia. Web. 02 November, 2022.
Copy Citation
In the Central European region, there is a long tradition of breeding fish in artificially constructed ponds. As the area belongs to the temperate zone, farmed fish need to survive cold winter periods. Common carp (Cyprinus carpio L.), which is an omnivorous, bioturbating species, is well adapted to warm and cold periods and the alluvial water environment. Since the Middle Ages, a large scale, efficient carp farming methodology has been developed in the region, where production is based on natural resources (protein and fatty acid sources) of renewable water ecosystems.
common carp
carbon footprint
carp farming method
environmental protection
1. Biological and Hydrobiological Background of Pond Carp Breeding
The tiny outer surface of our planet is called the geographic envelope. All the physical, chemical and biological events happen within this envelope, which is divided into different spheres.
A characteristic part of this envelope is the gaseous atmosphere, which covers the whole surface of our planet. This surface is broken up to form continents, islands, icebergs, glaciers, water bodies (oceans, seas) and inland waters within land boundaries. A few billion years ago, organic life on Earth was born in shallow, warm marine water. Nowadays, the majority of living organisms still exist in water environments. An important part of the surface is called the hydrosphere, which includes all types of waters and is inhabited by aquatic organisms.
The lithosphere is formed by soils and rocks of lands, and it also includes the pedosphere, which is the upper 8 m layer suitable for terrestrial life, such as current human societies. Some living organisms require both terrestrial and aquatic environments, while thousands or millions of terrestrial species live in the pedosphere and in lower layers of the atmosphere.
Following the appearance of organic life, ancient living organisms inhabited all mentioned spheres, establishing the biosphere. While they occupied terrestrial and aquatic areas, they also modified their inorganic, physical and chemical environment. A good example of such modification is the activity of ancient aquatic water plants (algae and cyanobacteria), which created an oxygen-rich atmosphere for Earth over millions of years. A similar event is the current global warming aggravated by human activities through the large-scale utilization of solid and liquid carbon deposits.
In the following, a fish farming technology operating in a non-terrestrial, aqueous environment will be analysed. Experts working in aquatic environments need some basic information on the chemical and biological processes of fresh waters, mainly pond water, to be successful in fish production. The science of hydrobiology helps them to understand and regulate processes supporting fish production. Before discussing fish breeding technologies, some important facts, definitions and roles of hydrobiology are considered and discussed.
Subject to the ionic level, water bodies of the hydrosphere can be divided into two large units: (a) seas, where the ion level is high and relatively stable, and (b) fresh waters with low and variable ion levels.
Oceans, seas and inland seas resulting from marine transgressions belong to category A. Generally, a high salt content (3.5%), mainly sodium chloride, characterizes these waters. The higher the dissolved ion content of the water is, the higher its density is.
The status of waters in category B is more variable. For certain lakes that exist on special minerals/soils, the water can contain very high ion levels (soda lakes).
Other extreme cases are waters of rainy tropical areas which are nearly distilled. In general, inland waters have low ion levels.
Categorization of inland waters based on their total ion content results in three main groups:
-
Fresh waters: with a total salt content of 0–500 mg/L, with 0–700 µS/cm specific electrical conductivity.
-
Transitional waters: 500–3000 mg/L salt content.
-
Salt waters: above 3000 mg/L salt content.
The salinity of seawater is 35,000 mg L−1, approximately 3.5%, with a specific conductivity of around 70,000 µS/cm.
Waters can also be categorized according to the movement of water (lakes and rivers), depth (deep or shallow), origin (natural or artificial), length of existence (long life or astatic), etc.
2. Hydrological Cycle
Water molecules are moving constantly. During evaporation and plant transpiration, water molecules emerge from surfaces of terrestrial areas and water bodies (oceans, lakes, rivers and so on), thus creating clouds. At a certain moisture content, after cooling, rain and snow develop in clouds, fall as precipitation and the circulation starts again (Figure 1).
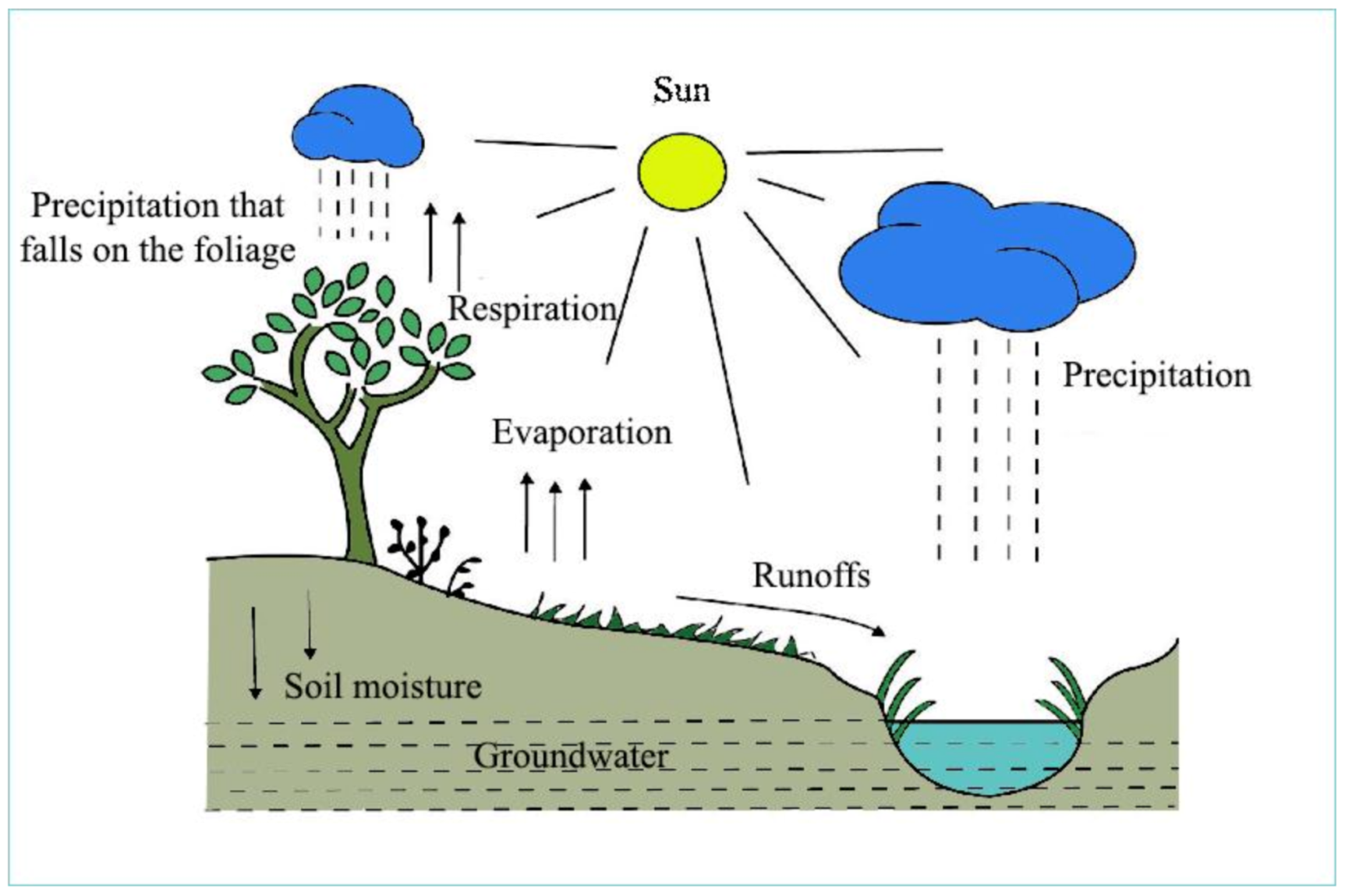
Figure 1. The hydrological cycle, water circulation of the geosphere (Padisák, 2005; modified) [1].
Water management has a significant importance for human populations, as freshwater is becoming a limited renewable resource. Water is needed both for humans and agriculture, including animal breeding, plant production and a sustainable natural life. Nowadays, the human population is gradually increasing and needs more water, while potential fresh water sources remain the same or are even decreasing. Escalated by global warming, the role of water will be more significant in the future.
3. Physical and Chemical Properties of Water Molecules
Water is essential for life. Every living creature, from the simplest microorganisms to the most advanced ones, needs water.
H2O is the most common molecule in the world and is the only compound that is present in three forms at the same time: gas, liquid and stable (microcrystalline) phases.
The structure of a water molecule explains all special properties that are characteristic for the aquatic environment. In the water molecule, two hydrogen (H) atoms with lower electro negativity are connected to a single oxygen (O) atom with high electro negativity by a strong covalent bond. The three atoms are not arranged along a linear axis, but the oxygen and the two hydrogens are positioned at an angle of 105 degrees, thus creating a dipolar molecule (see Figure 2).
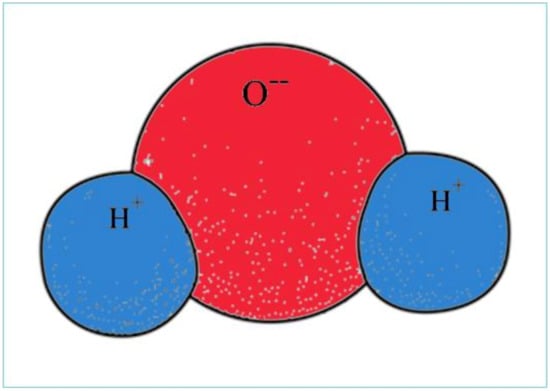
Figure 2. Bipolar water molecule.
Unbound negativity of the negatively charged oxygen also attracts the two hydrogen atoms of other water molecules. However, this bond is a weaker, secondary chemical bond, called a hydrogen bond. Accordingly, water molecules form smaller clusters, establishing a crystal-like structure.
Oxygen atoms with a slight negative charge and hydrogen atoms with a slightly positive one within a water molecule not only attract adjacent water molecules but are also able to connect to other charged atoms and compounds. Due to its bipolarity, water makes for an excellent solvent. The connection of water molecules with other atoms and ions is mutual, because atoms and compounds with different charges are formed from compounds dissolved in water, which can have a negative or positive charge. Bipolar water molecules form a shell around ions. The process is called electrolytic dissociation. On the other hand, water molecules can also form a hydrated shell around water-insoluble organic macromolecules (e.g., fats); thus, spherical droplets are developed.
The dipolar nature of water molecules also explains the specific density–temperature relationship of water (temperature anomaly).
In its solid phase, as ice, water molecules are arranged in a crystal lattice and are fixed (see Figure 3), where the distance between the atoms is constant. The volume of solid ice increases when compared to liquid water, due to the crystal lattice. Its specific gravity is less than that of water; as a result, ice floats on liquid water. During the melting of ice, the arrangement within the crystal lattice gradually disappears. The distance between molecules/atoms is largest at freezing point.
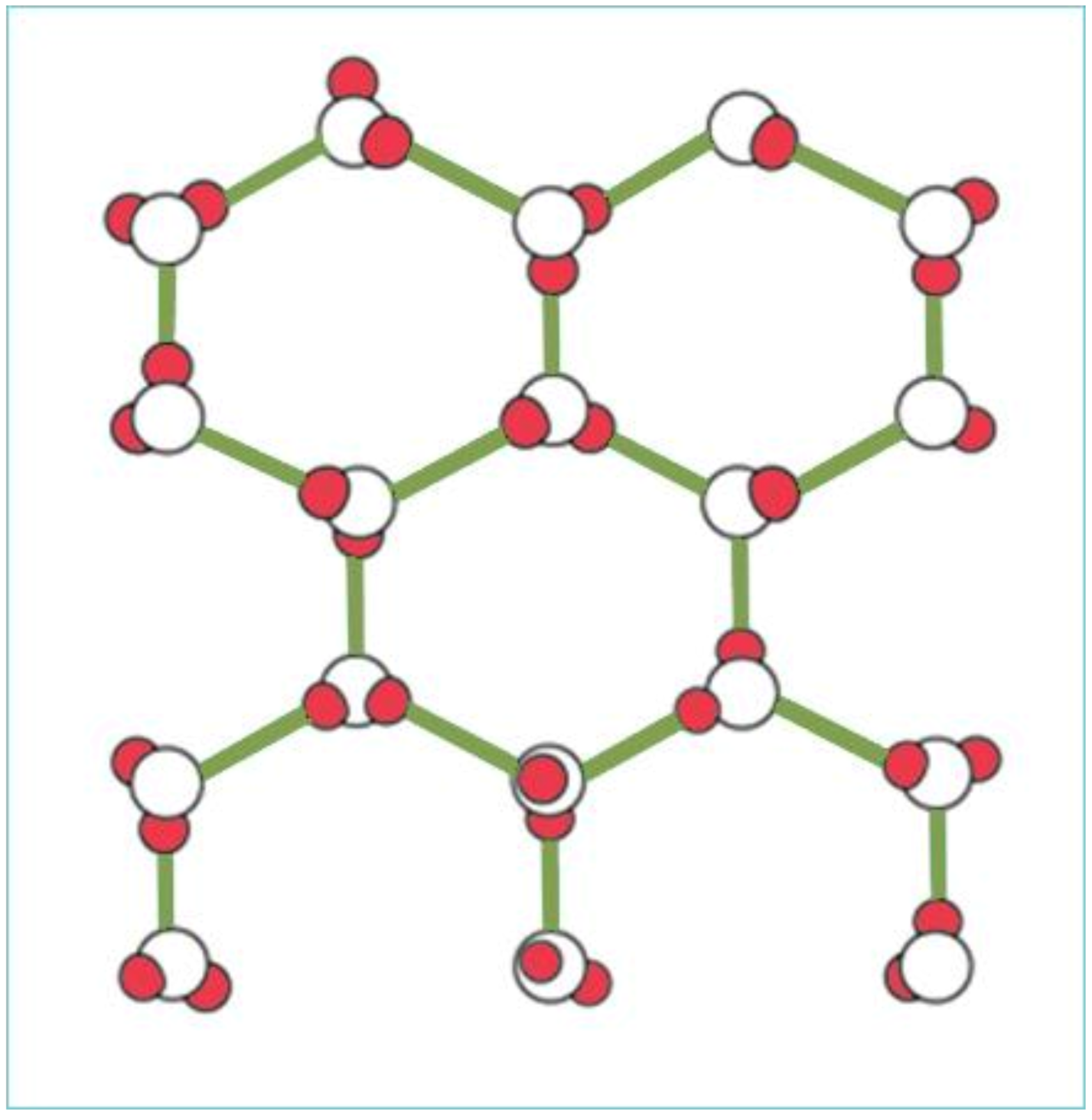
Figure 3. Crystal structure of water molecules in ice.
Additionally, there is another physical effect that affects the density of water: it increases the distance between the molecules/atoms due to heat transfer and is called Brownian motion.
The two processes influencing water density have antagonistic effects. Crystal lattice formation of gradually cooling water does not yet start at +4 °C, so there is no increase in the volume. Contrarily, thermal expansion at +4 °C is the lowest; thus, the specific gravity of water is the highest. This particular property is exceptionally important for aquatic life and is ultimately the result of the molecular structure of water.
So, the maximum density of water is above its freezing point, exactly at +4 °C. This phenomenon is called the temperature anomaly of water, which helps the deepest layers of water bodies to stay constantly at +4 °C, saving living organisms from freezing.
Water density is also affected by salinity (ion concentration), where the relationship is straight (linear). The higher the dissolved ion content of water is, the higher its density is (seawater).
4. Heat Capacity and the Route of Sunlight in Water
A further important physical property of water is its ability to absorb heat. Water warms up and cools down slowly when influenced by a changing temperature, as it has a very high heat capacity and can store a large amount of heat.
Thermal stratification of stagnant waters is related to the maximum density at 4 °C. In spring, the upper layer of waters begins to warm. When the temperature of lake water reaches 4 °C, the temperature of the whole water body becomes identical due to turbulence and vertical currents, and the temperatures of the upper epilimnion and the lower hypolimnion are equalized. In summer, the lighter warm water is located in the upper layer, forming again an epilimnion and a hypolimnion. In autumn, the process is reversed and allows for mixing to occur.
Due to the effect of wind and the movement of large fish stocks, only short-term thermal stratification occurs in shallow fishponds.
Sunlight has prominent physical and physiological impacts in water. When the biosphere is considered, this fundamental and decisive radiation is made up of mutagenic ultraviolet, visible and infrared ranges. The light absorbing ability of water is an important physical feature. In water, the light with different wavelengths is absorbed in different ways. Visual and infrared ranges are required for plant (algae) assimilation. In clean and transparent waters, the long waves of blue light from visible light penetrate the deepest [1][2].
Sunlight can only penetrate 20–30 cm deep into turbid water, while some of its heat is absorbed in the upper water layers. These layers are the best breeding mediums for unicellular algae; thus, experts of pond aquaculture should maintain the water turbidity of lakes. Bioturbation of carp and artificial agitation of the sediment can obscure pond water.
5. Cycles of Biogenic Elements within Aquatic Conditions
In addition to liquid water, inorganic carbon compounds (carbon dioxide CO2, hydrogen carbonate anion HCO3 and carbonate anion CO3) and biogenic elements, such as nitrogen and phosphorous, need to be present in pond water to enable organic production. Certain cations that bond with carbon compounds, especially magnesium and calcium cations in the aqueous medium also play a key role in pond production (further discussed in the next chapter).
From an environmental point of view, it is particularly important to analyse chemical bonds of elements at an atomic level in order to prove that carbon dioxide that has entered or was generated in pond water cannot escape into the atmosphere; it does not increase the level of carbon dioxide in the air but circulates in the system until being fixed by algae.
For a better understanding and management of the biological production and energy transfer of pond ecosystems, basic structures and reactions of elements/compounds at the atomic level should also be considered. These basic structures and reactions can be investigated by using Bohr’s atomic model and can explain biological processes in ponds.
Atoms of various elements occurring on our planet are made up of an atomic nucleus and electron shells around it (Figure 4). The attraction between these two components ensures the stability of atoms. The nucleus is made up of protons and neutrons. In the electron shells, electrons are located around the nucleus, the quantity of which depends on the number of protons in the nucleus. These electrons consist of pairs. When combining elements, compounds are formed, the stability of which is established by their common pairs of electrons.
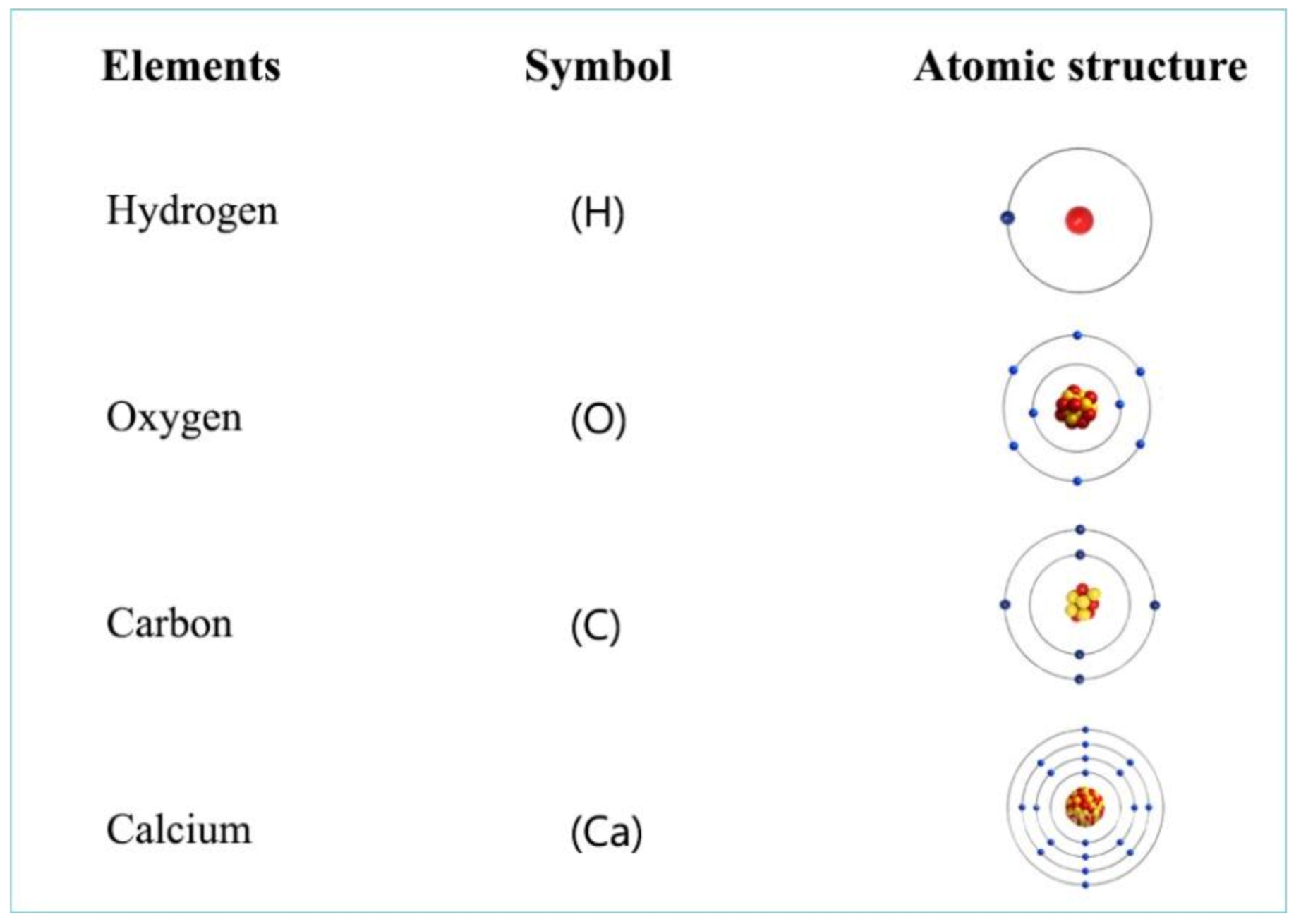
Figure 4. Atomic structures of a few biologically important elements.
Larger elements with more protons and thus more electrons have more subshells within the electron shells. The electrons on the outer layer are the so-called binding electrons. They also try to form electron pairs and aim for a filled subshell. For that reason, missing electrons are taken from or excess electrons are transferred to other elements. The origin of the electron pairs formed in the binding electron shell determines the type of chemical bond between different elements (covalent or ionic bond, etc.) and the charge of the elements and compounds. Negative ones with an electron excess relative to the proton number are called anions, while positively charged ones with an electron deficiency relative to the proton number are called cations.
Four electrons are located in the outermost electron shell (called valence shell) of a carbon atom. As it would require eight electrons for saturation, four more are needed. How can the carbon atom reach this status? The simplest stable compound of a carbon atom is gaseous carbon dioxide, the structure of the outer binding electron shell of which is shown in Figure 5. Missing electrons are received from two oxygen atoms and the type of the chemical binding is covalent, which is a very stable connection among atomic bindings.
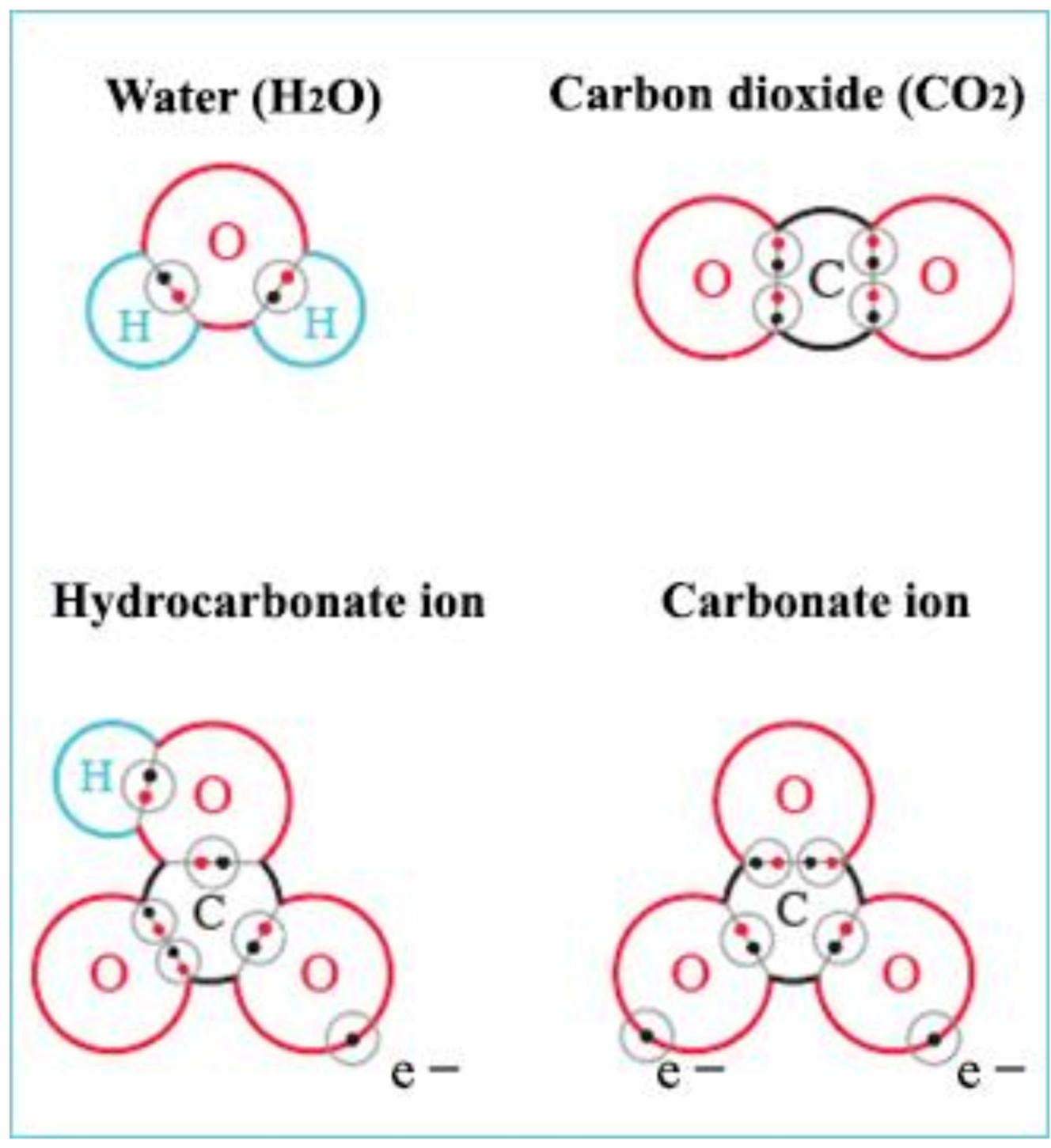
Figure 5. Binding types of certain compounds important in the biological production of ponds. Water molecules and gaseous carbon dioxide have purely covalent bonds, while hydrocarbonate and carbonate anions have both covalent and ionic ones.
Algal assimilation generates proper conditions for the production of organic matter and energy in lakes (Figure 6). The process requires carbon dioxide, water molecules, sunlight and functional chlorophyll of algae. During the daytime, algae synthesize organic matter (sugar and organic macromolecules such as starch, protein, fat, vitamins, hormones, etc.) that are incorporated into the algal biomass. Through the aquatic food chain, algae-consumers transform a small portion of this organic matter into their body material; the energy produced by the algae moves to higher trophic levels. Biocenosis (all living organisms in a pond) uses some of the energy produced during the day for metabolism and produces carbon dioxide during dissimilation. When the result of assimilation is investigated, it is recognised that the difference between production and decomposition is the net biological product.
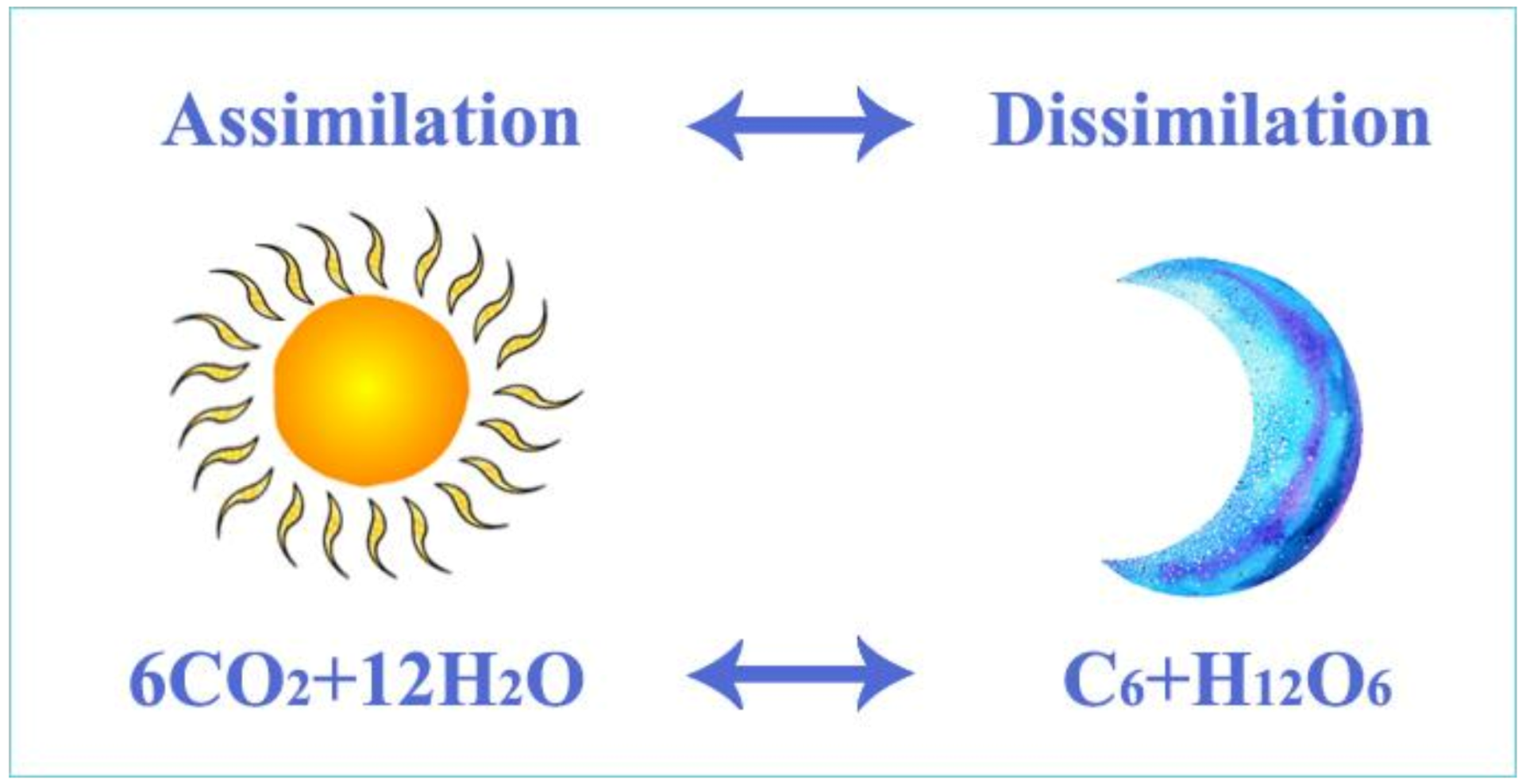
Figure 6. A basic equation of the assimilation of water plants, mainly algae. During the process of assimilation, algae synthesize high-energy organic molecules from carbon dioxide molecules by using the energy of sunlight.
In an aquatic environment, depending on the pH of water, only a very small proportion of carbon dioxide can be directly absorbed by algae. At a neutral pH, most of the inorganic carbon is present as hydrocarbon or carbonate anions. Before discussing the circulation of all carbon compounds in aqueous environments, it is necessary to define what pH is: pH and pOH specify the acidity or alkalinity of water (the presence of free hydrogen or hydroxyl ions).
Fresh waters not only contain carbon compounds and calcium and magnesium ions, but also many other elements which influence the total ionic character. Clean water can be characterized by a high solubility. From minerals and soils, different ions can be dissolved into water. For a chemical characterization of water, four main cations and anions need to be determined: these cations are Ca2+, Mg2+, Na+ and K+, and the anions are HCO3−, CO32−, Cl− and SO42−. To demonstrate these ions quantitatively in a water body, the Maucha diagram (Figure 7) can be used. Separate peaks of different ions indicate their amounts as well.
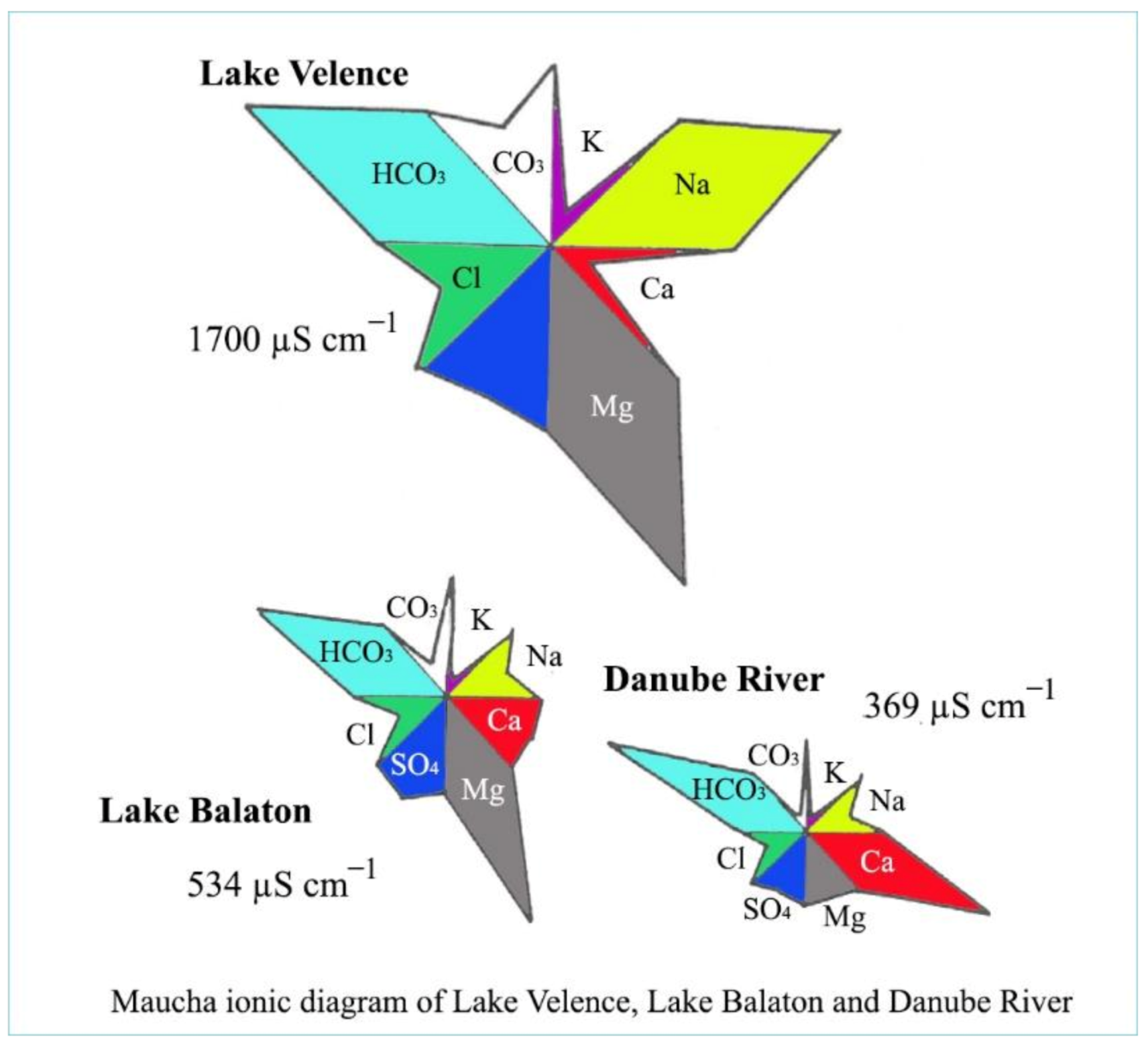
Figure 7. Maucha diagrams of some waters in the Carpathian basin.
The final value of water pH evolves from the overall amount and type of single ions in the water.
As is presented in Figure 7, the waters of the Carpathian basin are characterized by calcium/magnesium hydrocarbonate, which is the result of the ion content of soils and minerals of the surrounding hills. The pH value of calcium and magnesium hydrocarbon types of waters is around 7–8, which belongs to the neutral category. Not only is the neutrality of these waters beneficial, but so is their pH stability, as living organisms prefer a stable pH for their metabolic processes.
How can pH be stable in these types of water? To understand this phenomenon, the effect of algal assimilation on inorganic carbon circulation must be analysed (Figure 8, Figure 9, Figure 10, Figure 11 and Figure 12).
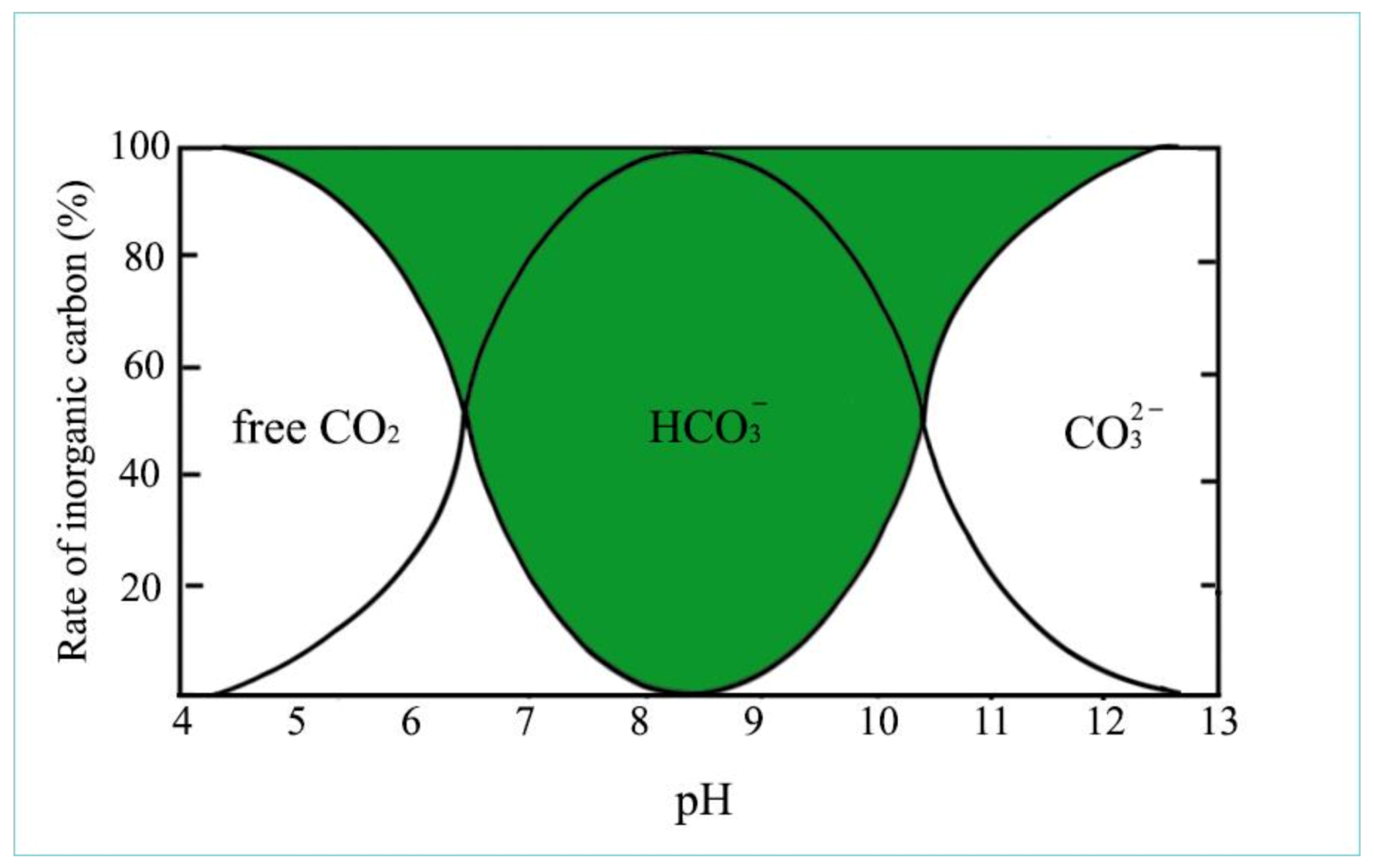
Figure 8. Forms of inorganic carbon compounds based on pH level (Padisák, 2005, modified).
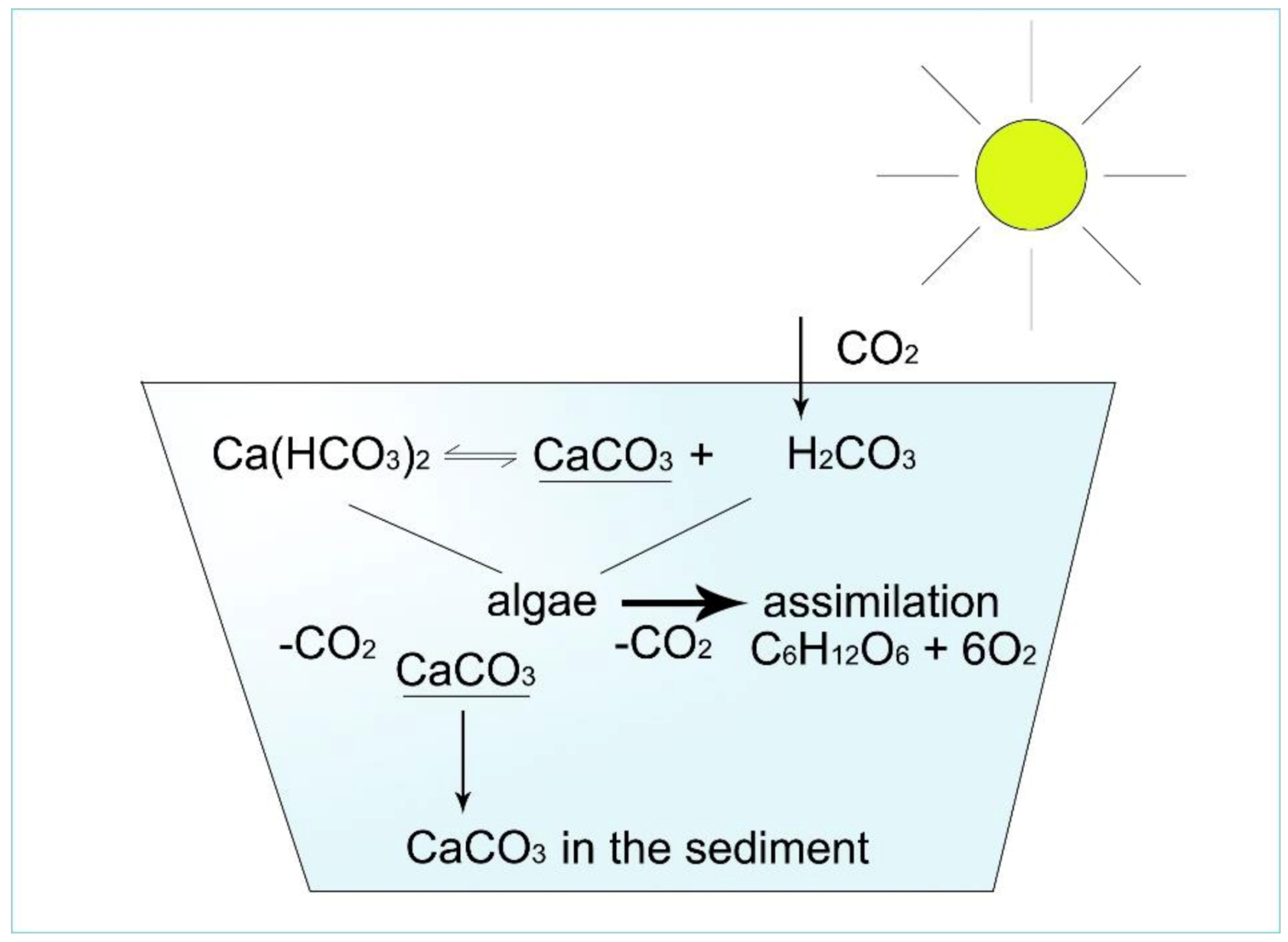
Figure 9. Well-buffered pond water contains calcium hydrogencarbonate as a carbon dioxide reservoir for algae. As a result of assimilation, daytime CaCO3 crystals are produced; thus, water pH remains stable.
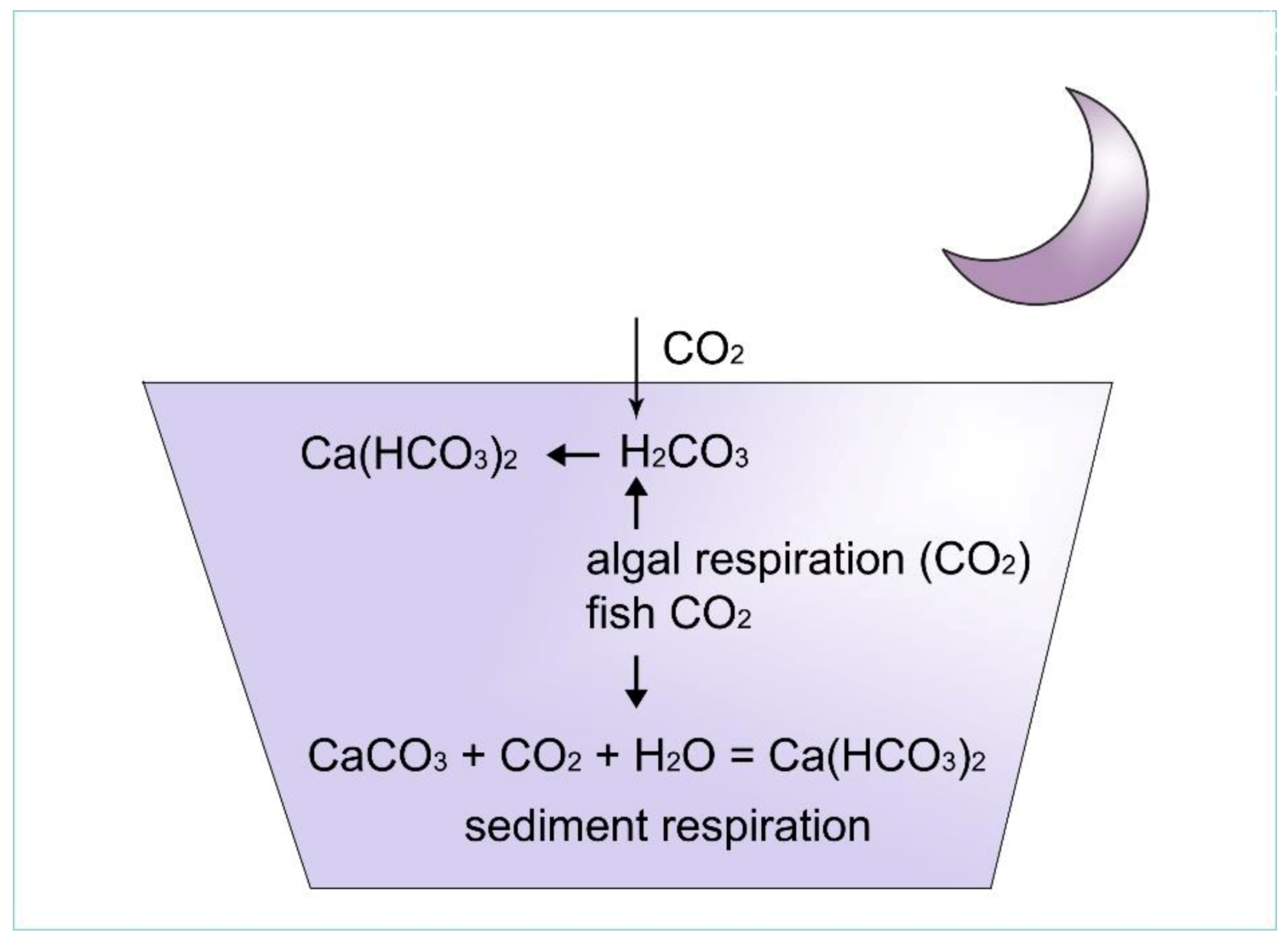
Figure 10. At night, free carbon dioxide reacts with calcium carbonate crystals and water to produce calcium hydrogen carbonate. Thus, the pH of water remains steady.
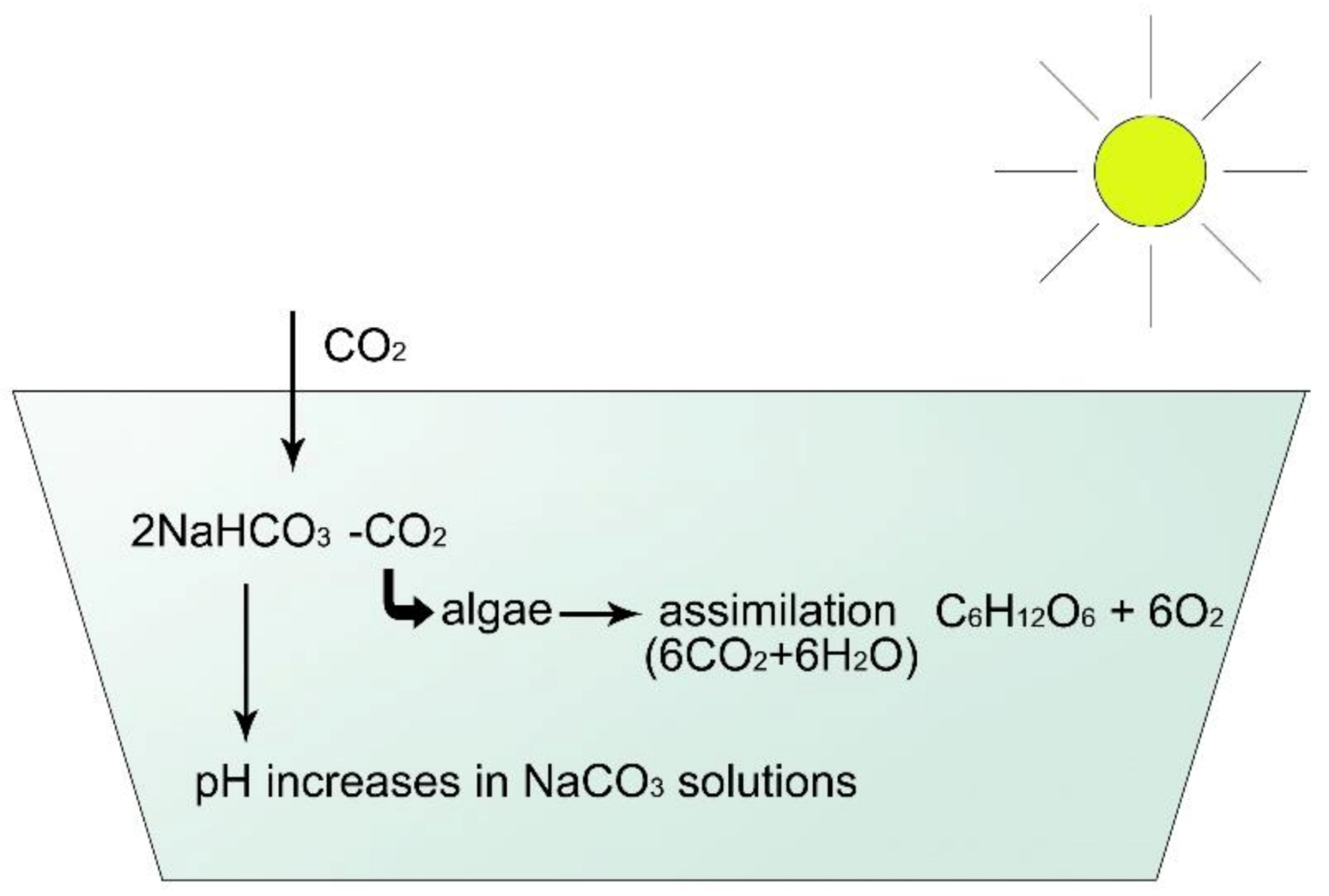
Figure 11. In sodic ponds, pH increases during the daytime because of assimilation because sodic Na2CO3 remains dissolved.
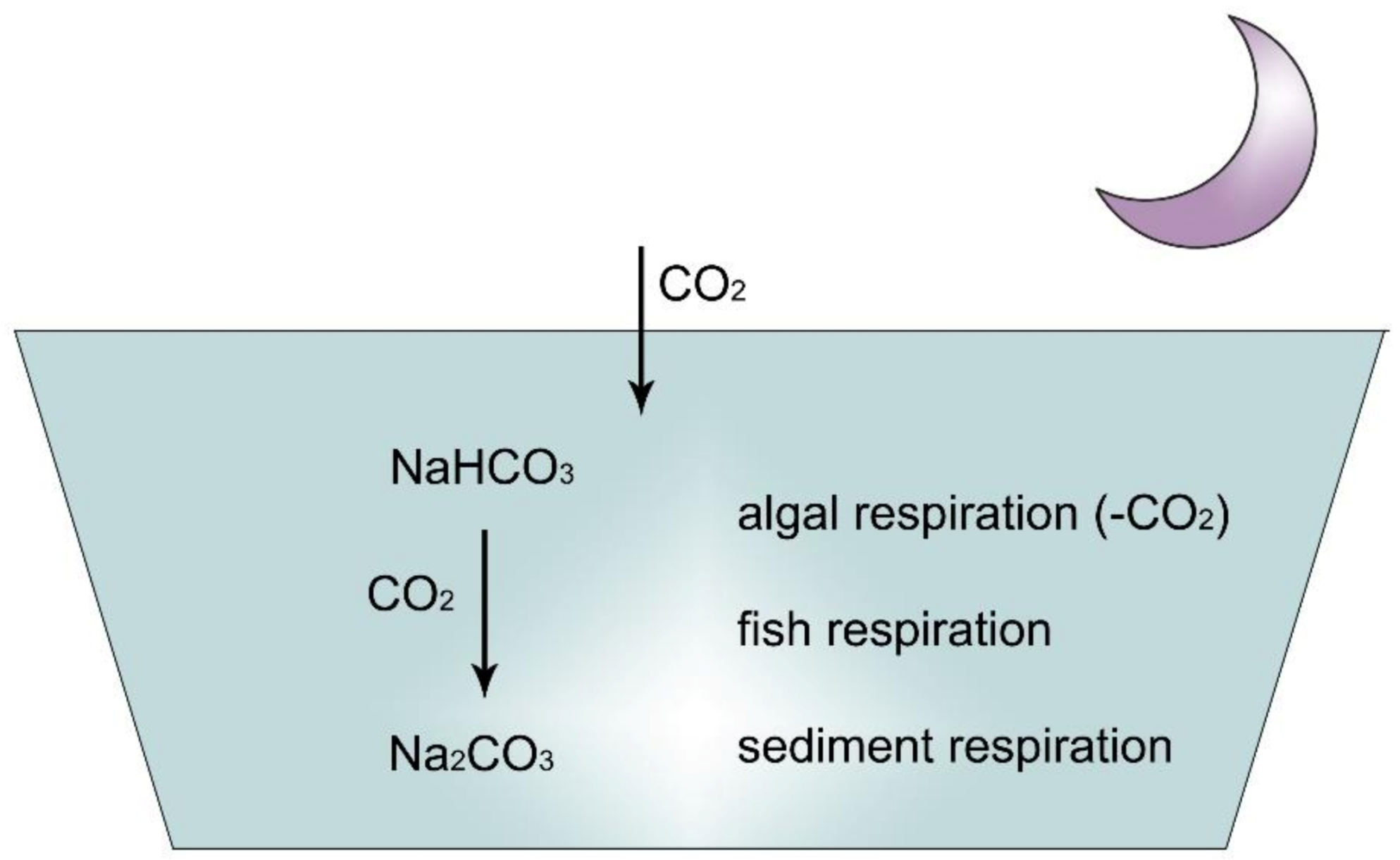
Figure 12. At night, due to carbon dioxide, pH decreases in sodic ponds.
6. Inorganic Carbon Circulation of Aquatic Ecosystems
Figure 8 provides an overview of the appearance of inorganic carbon compounds that depend on the pH of water.
As illustrated in Figure 8, in acidic conditions (pH 4–6), all inorganic carbon occurs as dissolved free CO2, which create carbonic acid (H2CO3) with H ions, while at alkalic level (pH 11–14), carbonate anions dominate. In the neutral pH range, 100% of carbon is present as bicarbonate (green field), mainly in the form of hydrocarbon anions and about 1% as so-called non-equilibrium carbon dioxide. This is the form of CO2 which can be used up by algae through osmotrophy. When algae consume this small amount of CO2, new hydrocarbon molecules dissociate into CO2 and carbonate ion for maintaining the equilibrium, as is described in the following:
Anions are connected to different cations, forming an ionic bond compound. The strength of electronegativity of cations decreases as follows: Ca, Mg, Na, K. Therefore, HCO3 anions will join first to Ca cations. During the assimilation process, algae will first ingest free equilibrium CO2 from Ca(HCO3)2 as:
Ca(HCO3)2 = CaCO3 + H2CO3
H2CO3 = H2O + CO2
Fortunately, the end product of this process is CaCO3, a crystalline compound (calcite) which immediately descends into the sediment, and the pH of water remains neutral. The process is the same with magnesium, resulting in MgCO3 (called crystalline dolomite). This is why a stable pH characterizes well-buffered Ca/Mg type of water.
In contrast, when no Ca or Mg cations are present but Na+ or K+ are available, the pH does not remain stable:
2Na(HCO3) = Na2CO3 + H2O + CO2
Na2CO3 is not a crystalline compound and remains in the solution as soda, which increases alkalinity! During the daytime, when assimilation is intensive, pH in sodic water can increase to high, toxic levels.
The high level of Ca and/or Mg in waters of the Carpathian Basin results in stability and high production efficacy of water vegetation, including algae [3].
The cycles of nitrogen and phosphorus compounds in ponds can be studied in Figure 13 and Figure 14.
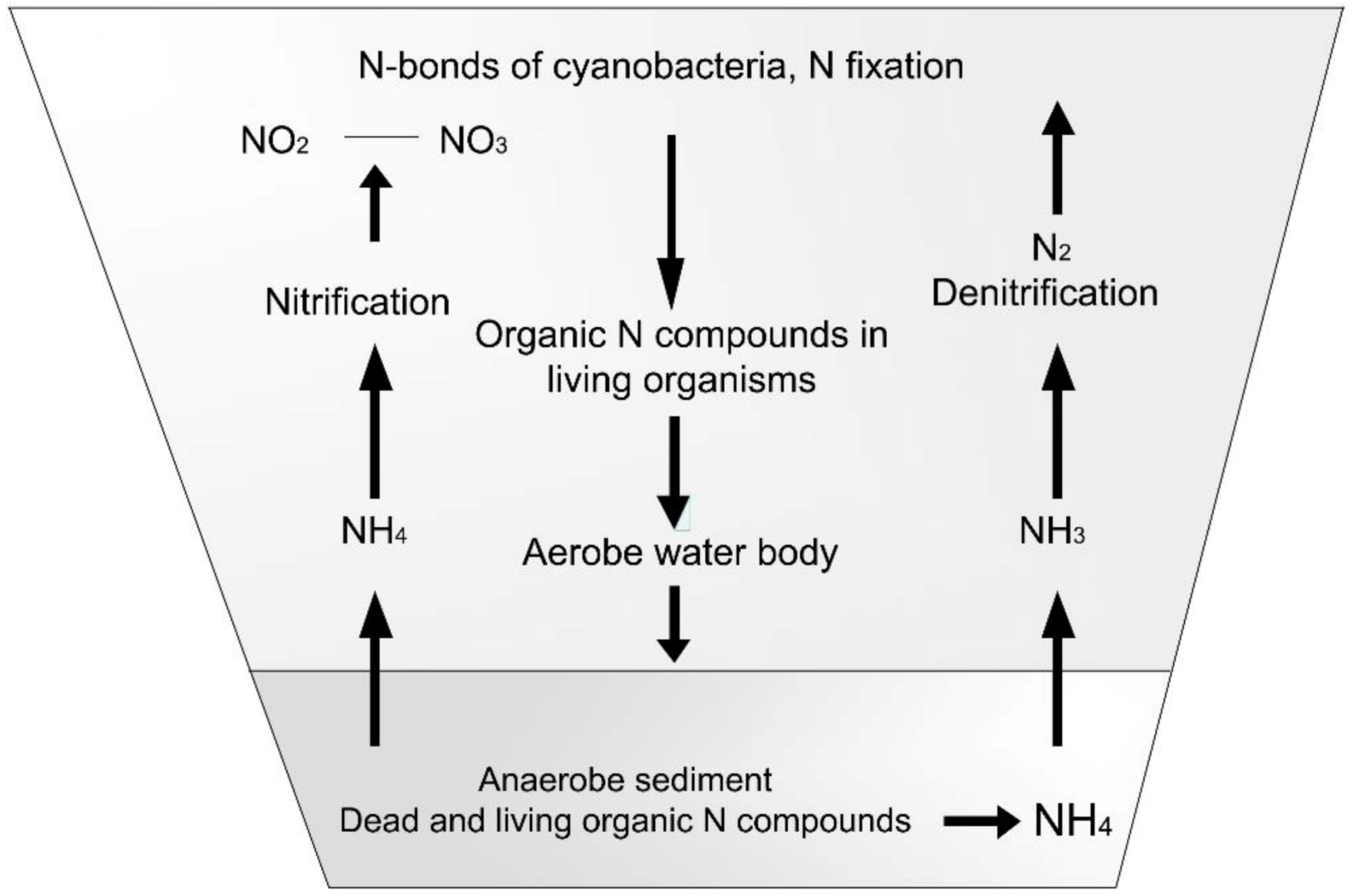
Figure 13. Nitrogen cycle in ponds.
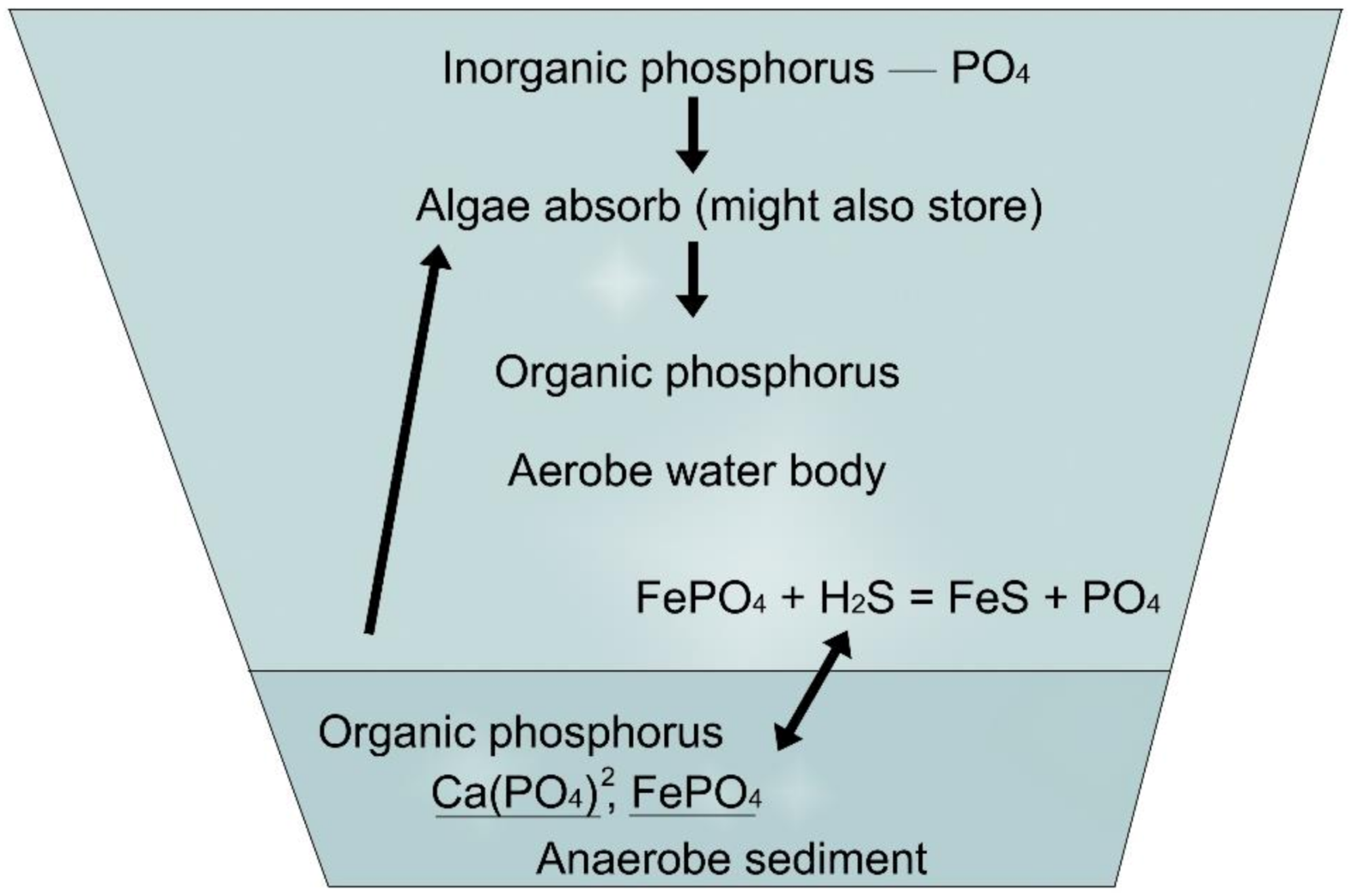
Figure 14. Phosphorus cycle.
7. Aquatic Habitats and Communities
Aquatic organisms inhabit the entire area of a water body, from the bottom to the surface, unlike organisms of terrestrial areas. The same organisms can live on different surfaces, or in open water as well (planktonic lifestyle); thus, aquatic communities and habitats occupied by them can be very diverse.
The most common freshwater habitats are:
-
Open water area, called pelagic zone, which provides an environment for (a) floating communities of plankton and (b) a rapidly swimming biocenosis called nekton, composed mainly of fish species.
-
The surface habitat, called facial, which has two communities, the moving pleuston with larger members and neuston with tiny ones.
-
The vegetation-rich lake shore zone with a shallow water depth; the phytal and littoral zones have mixed communities. Periphyton lives on the surfaces of water vegetation, but in the littoral zone, members of the plankton are generally also present.
-
Both the pelagic and littoral zones have sedimental bottom areas, which create a benthic habitat where members of a community called benthos live.
These habitats and communities (biocenosis) are present in artificially constructed fishponds as well.
8. Competition of Water Plants in Ponds
The next important and exciting question is: how the number of one-cell algae can be increased in shallow ponds, where they need to compete for carbon sources and nutrient elements with higher water weeds and cyanobacteria.
Water weeds in shallow waters can be divided into two groups: emersed and submerged types of plants (Figure 15).
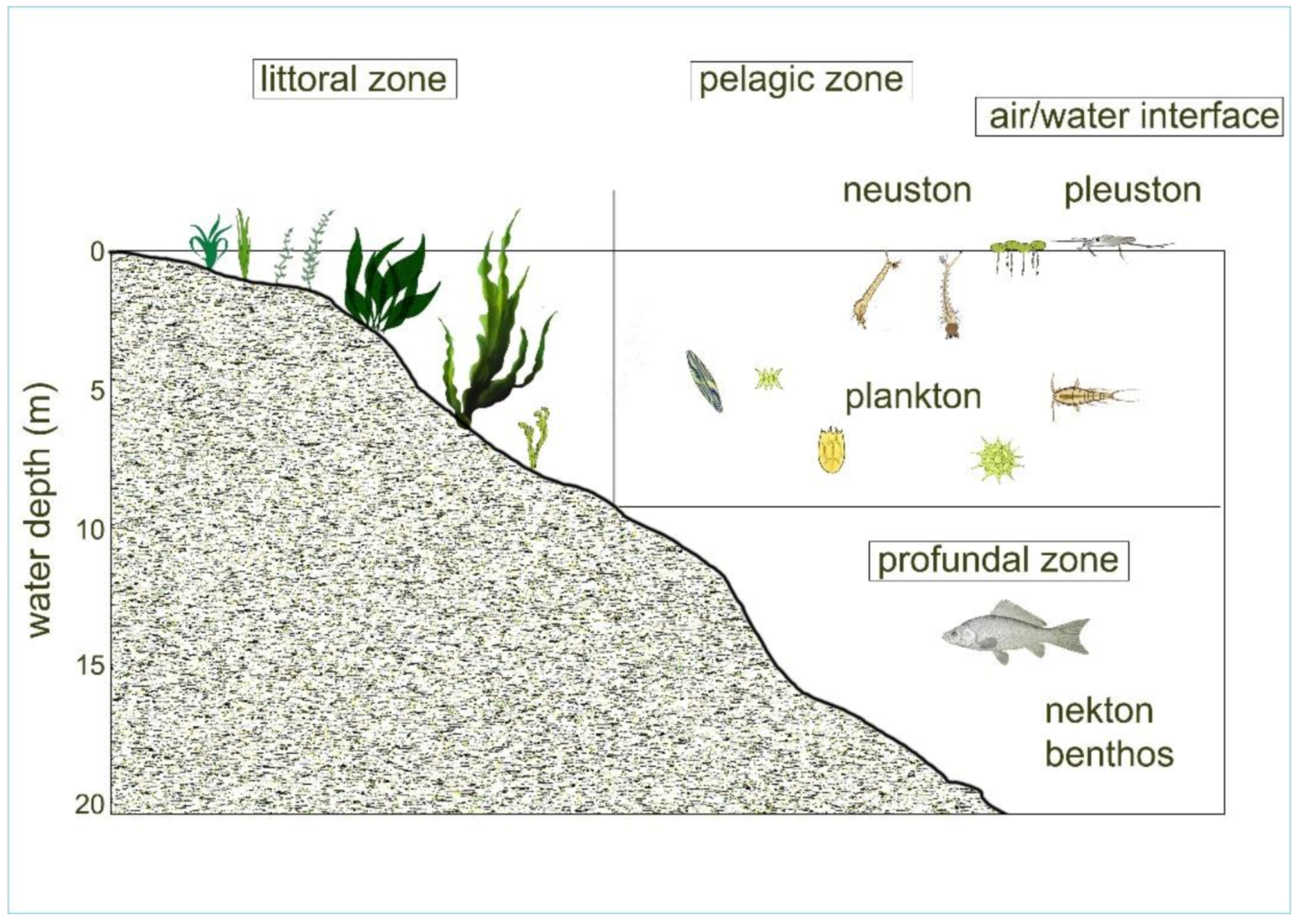
Figure 15. Most important habitats and communities in lakes/stagnant waters.
Leaves, stems, shoots, flowers and seeds of emersed plants extend above water level, while their stems remain underwater and roots in the mud. During photosynthesis, carbon dioxide is assimilated from the atmosphere, while nutrients are obtained from the water and the sediment. Some of the submersed aquatic plants have roots as well, while others float freely in the water body. Moreover, there are higher aquatic plants that live at the water–air phase. Controlling these diverse groups in ponds is possible by either mechanical or biological methods. Emersed plants can mainly be controlled mechanically, while submerged water weeds, in addition to mechanical methods, can also be controlled efficiently via biological practices (mainly by stocking large grass carp). Increasing water turbidity can also be effective, as there would not be enough solar energy in turbid waters for assimilation.
Regulating the blooming of cyanobacteria (formerly mentioned as blue-green algae) is also an important and difficult task. Some species of these tiny aquatic plants consist of a single cell, while others form colonies. Certain species are also able to bind nitrogen from the atmosphere and therefore are able to reproduce in an environment with limited nitrogen availability.
In contrast to these groups, unicellular green algae, which are important for pond production, can only multiply rapidly and synthesize organic matter in water bodies with high nutrient content, and without the competition of other plants.
If it is possible to produce turbid lake water, solar energy will be absorbed in the upper 20–30 cm, heating up this layer and creating a suitable environment for the development of green algae. This upper layer is called the trophogenic zone, where green algae can multiply and fix solar energy into synthesized organic compounds. Organic substance and energy can migrate through the food chain towards higher trophic levels. In turbid waters, neither submersed macrophytes nor dangerous cyanobacteria living on the bottom of shallow ponds receive enough solar energy.
Carp bioturbation is the most effective way to disturb pond water. Furthermore, a regular mechanical agitation of settled organic sediments and a frequent organic fertilization and liming of the pond water also have a positive effect on disturbance. Figure 16, Figure 17 and Figure 18 demonstrate the cross-section and activities of a shallow fishpond.
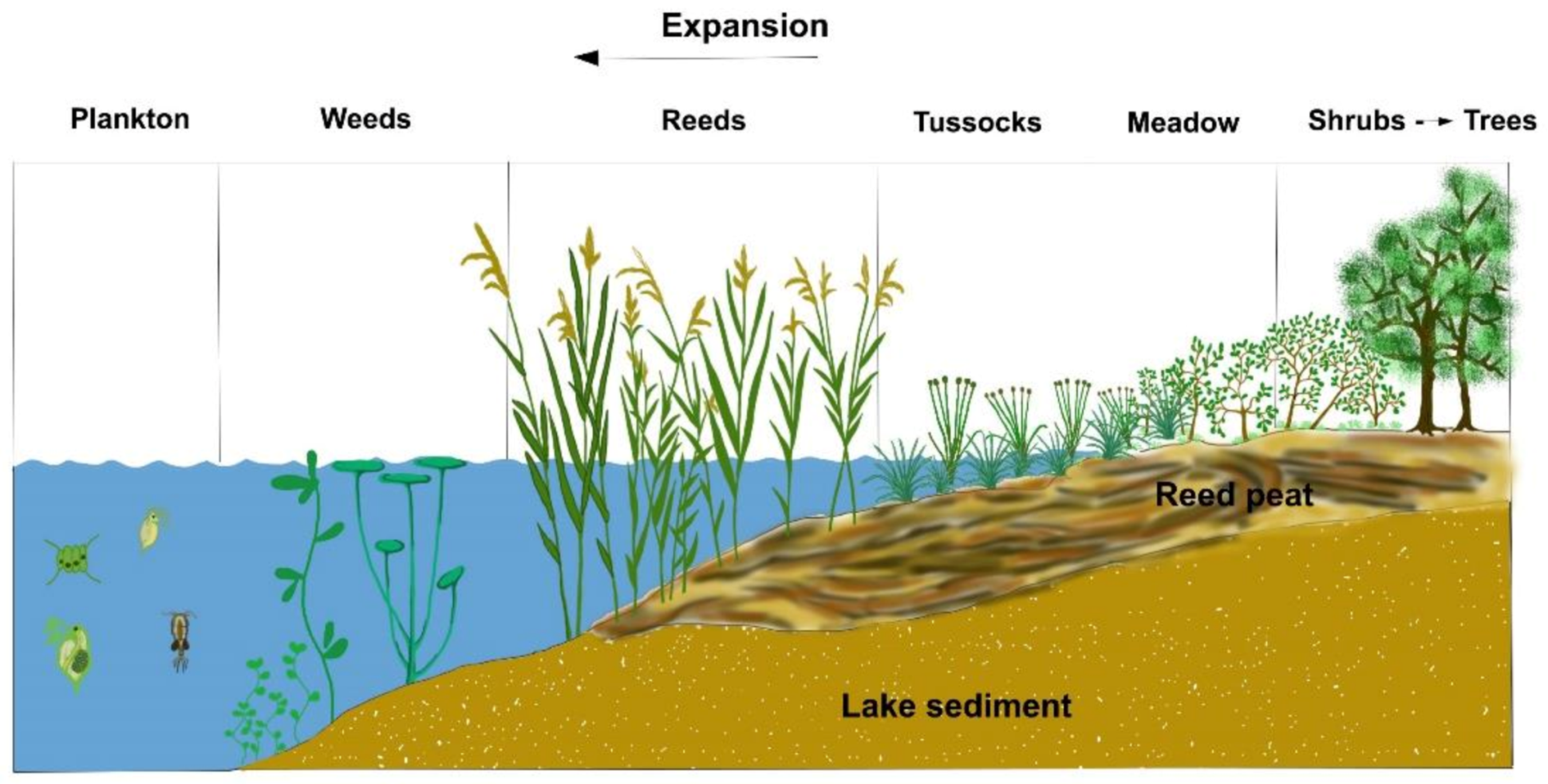
Figure 16. Zonation of macrophytes from open water bodies containing algae to forests.
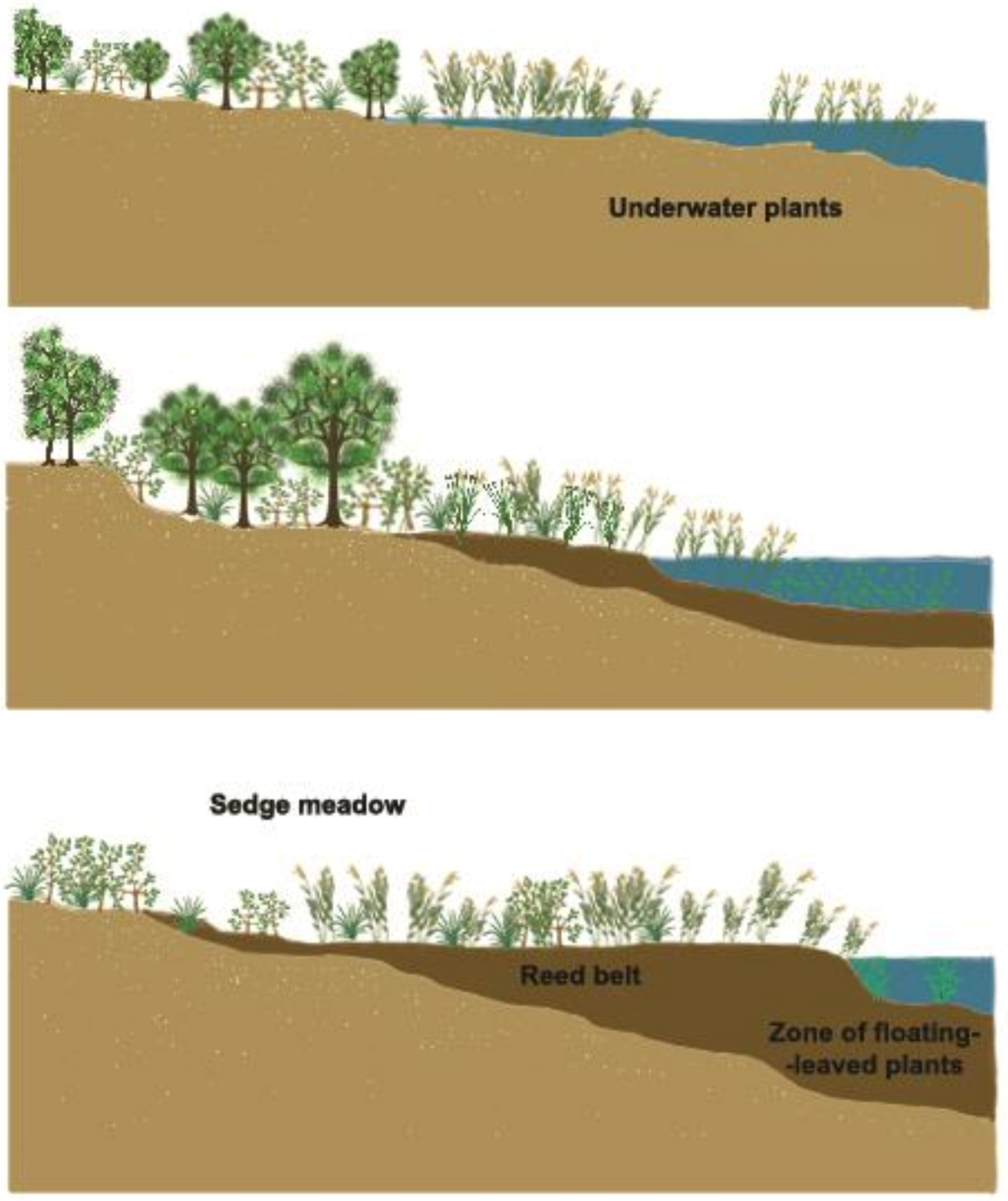
Figure 17. Proliferation of the macrovegetation enables the growth of organic sediment and the recharge of shallow fishponds.
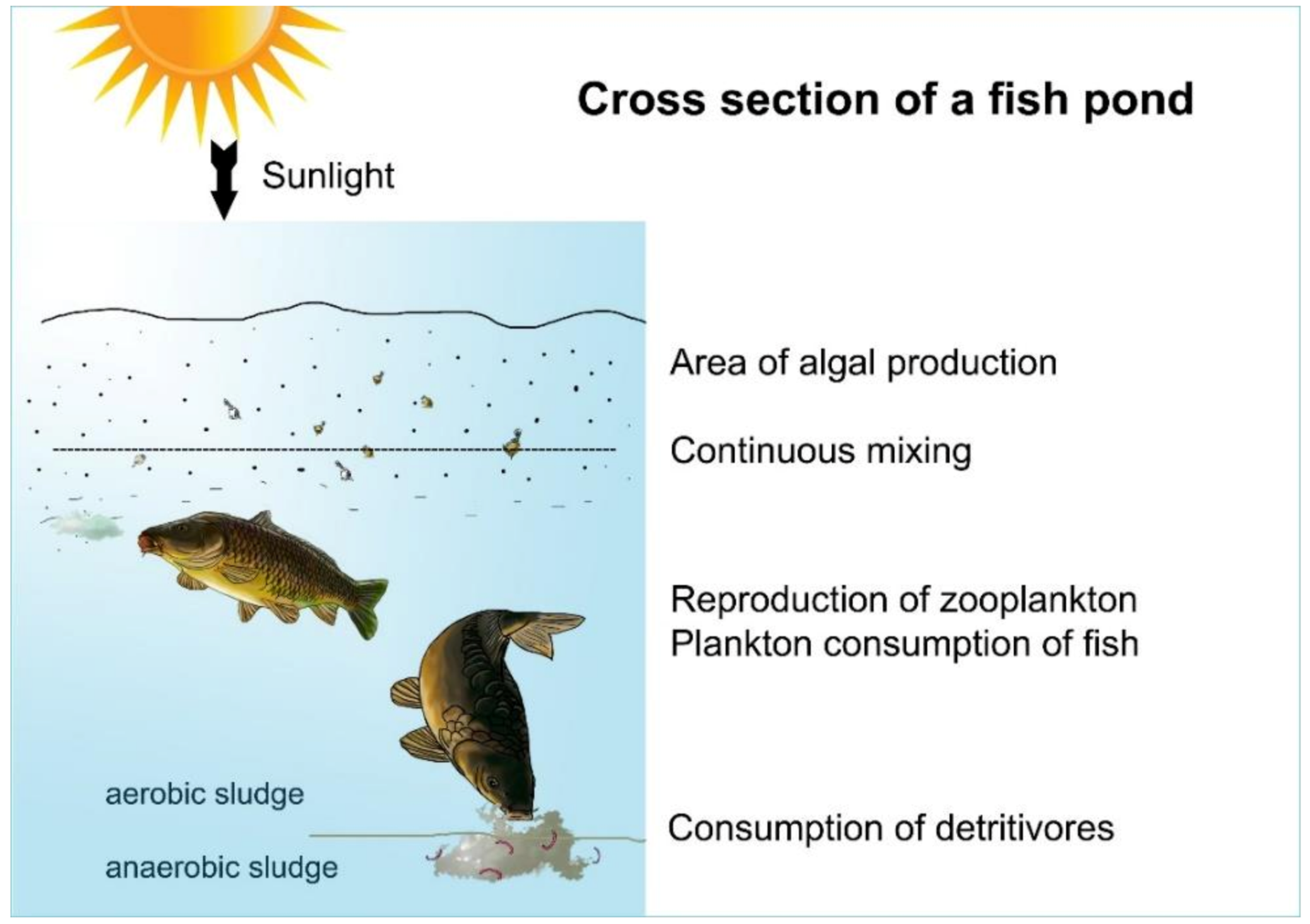
Figure 18. Cross section of a shallow pond. The upper part of the water, where intensive algal assimilation happens, is the trophogenic zone. Carp feed on zooplankton from open water or from mud sediment.
9. The Origin of Carbon Dioxide Required for the Synthesis of Organic Matter in Fishponds
Organic matter synthesized by algae requires large amounts of carbon dioxide. To form one molecule of a simple sugar, six molecules of carbon dioxide are required. Organic macromolecules such as starch or cellulose are composed of a large number, sometimes even many thousands, of simple sugar molecules. For the synthesis of proteins and fatty acids, not only carbon dioxide but also nitrogen and phosphorous compounds are needed. In addition to these basic substances, many other organic molecules, such as hormones, vitamins, enzymes, DNA and RNA macromolecules, existing in living organisms, need carbon dioxide (hydrogen carbonate within aquatic conditions). There was a theory in the past that carbon dioxide was a limiting factor for biological production. This is absolutely true for the so-called oligotrophic, nutrient-poor, cold and deep natural lakes, but not for shallow, nutrient-rich eutrophic fishponds, where carbon dioxide can emerge from many sources. A distinction can be made between carbon dioxide from endogenous/internal and exogenous/external sources.
10. Carbon Dioxide Originating from Endogenous Sources
An important endogenous source for carbon dioxide is nocturnal dissimilation or “respiration” of pond biocoenosis, another one is the metabolism of algae, cianobacteria, higher water vegetation, aerobic and anaerobic water bacteria, invertebrates and fish. The amount of metabolic of carbon dioxide can be deduced from the difference between the maximum and minimum daily levels of dissolved oxygen generated as a by-product of assimilation on the previous day. The basis of this calculation is the monitoring of oxygen production and consumption in proportion to the magnitude of assimilation and dissimilation.
Another internal source of carbon dioxide in the lake is the decomposition activities of settled organic particles (detritus) by water bacteria and tiny fungi, as well as by invertebrates (e.g., chironomids, worms and snails). After the extinction of aquatic macrovegetation (in the autumn), a lot of organic matter enters the sediment, which immediately begins to decompose. Aerobic decomposition occurs in the upper part of the sedentary organic matter, because oxygen from the oxygen-rich water body diffuses into the sediment. The result of this decomposition is carbon dioxide, which is instantly converted to a hydrogen carbonate ion which bonds ionically to a cation present, or forms carbon dioxide gas bubbles in the sediment. In deeper layers of the sediment, within reducing conditions, not only does carbon dioxide develop as a product of decomposition, but methane gas as well, which either effervesces through water or forms bubbles in the sediment. When methane is released from the sediment (as air pressure decreases), it removes dissolved oxygen from the water body while methane is oxidized to carbon dioxide.
Further important endogenous carbon dioxide sources originate in recycling, resuspending dead, sedimented organic matter accumulating in the mud into oxygen-rich water space. Aerobic decomposition of the recycled organic sediment produces a large amount of carbon dioxide. This process is initiated by the resuspending activity of pond organisms, especially of common carp and grass carp. The food search of these fish in the sediment induces a strong bioturbation. Not only fish but also many invertebrates, such as worms, snails, mussels and larvae of insect living in the mud, resuspend the sedimentary organic detritus. In addition to the bioturbation of living organisms, agrotechnical methods can also be applied (e.g., sludge blasting, sediment mixing). Processes that facilitate the carbon release from the sediment partially eliminate or diminish this “dead end” of organic matter.
11. Carbon Dioxide Originating from Exogenous Sources
The origin of exogenous carbon dioxide is even more diverse.
When inundating ponds in spring or adding extra water in summer, the water entering to the pond can transport a lot of inorganic carbon (carbon dioxide, bicarbonate, carbon anions) and dissolved or particulate organic matter where algae immediately begin to absorb the inorganic carbon via osmotrophy, and bacteria start to decompose detritus to carbon dioxide.
The next source of CO2 is the atmospheric gaseous carbon dioxide entering lake water via diffusion, as substances with different concentrations tend to level off. The concentration of carbon dioxide in the air is 0.03%, which is constantly increasing due to global warming. As discussed earlier, in well-buffered neutral (Ca/Mg rich) waters, carbon dioxide is present in the form of hydrocarbonate. There is practically no free carbon dioxide in ponds, so due to the equalization of partial atmospheric pressure, CO2 slowly diffuses from the air into the water. Calcium carbonate crystals (calcite) in water immediately react with carbon dioxide immigrating into the water and are converted to hydrocarbonate anions. Seawater (with limited Ca/Mg cations and dominating Na/K cations) only has a weak Ca/Mg hydrocarbon/carbonate buffering system. In other words, calcium carbonate crystals cannot absorb carbon dioxide; thus, they form hydrocarbon anions (see Figure 4). Carbon dioxide is bound to hydrogen ions and forms a weak acid called carbonic acid (H2CO3), which is responsible for the slow acidification of oceans. The same process can be observed in freshwater bodies on volcanic soil, which contain little to no Ca/Mg cations.
Another exogenous source of carbon dioxide can be an efficient agricultural intervention, when dry straw with a huge amount of cellulose is transferred to the ponds. This treatment (Figure 19) controls the reproduction of cyanobacteria and stops their blooming. The blocking effect of straw is the result of its polyphenol compounds diffusing into the water, which is only toxic to blue green algae (cyanobacteria). Within a few days, this effect eliminates cyanobacteria and a new controlling mechanism develops instead: a strong nutrient competition between aquatic microfungi and cyanobacteria. An aggressive microfungi population starts to develop on the high carbon content of straw which takes up all N and P components of water; therefore, cyanobacteria starve. Under these circumstances, a new food chain occurs: rotifers and ciliates feeding on microfungi. These planktonic organisms are consumed by copepods, which are then filtered by carp, thus forming a five-step food chain in ponds. The cellulose from straw practically appears as an exogenous carbon source in ponds.

Figure 19. Raschel bags filled with straw reduce the growth of cyanobacteria (Photo: A. Hegyi).
All agrotechnical treatments of the breeding protocol (fertilization with organic manure, fish feeding and pond liming) can be evaluated as large amounts of important exogenous carbon dioxide sources. Carbon dioxide is produced from organic manure through the same decomposition processes as from the sediment discussed in the previous sections.
During the decomposition process of organic manure (Figure 20), not only is carbon dioxide produced but also many other elements and compounds (molecules with N and P content) which play an essential role in the reproduction of algae; therefore, mature animal manure has a complex effect on biological production.
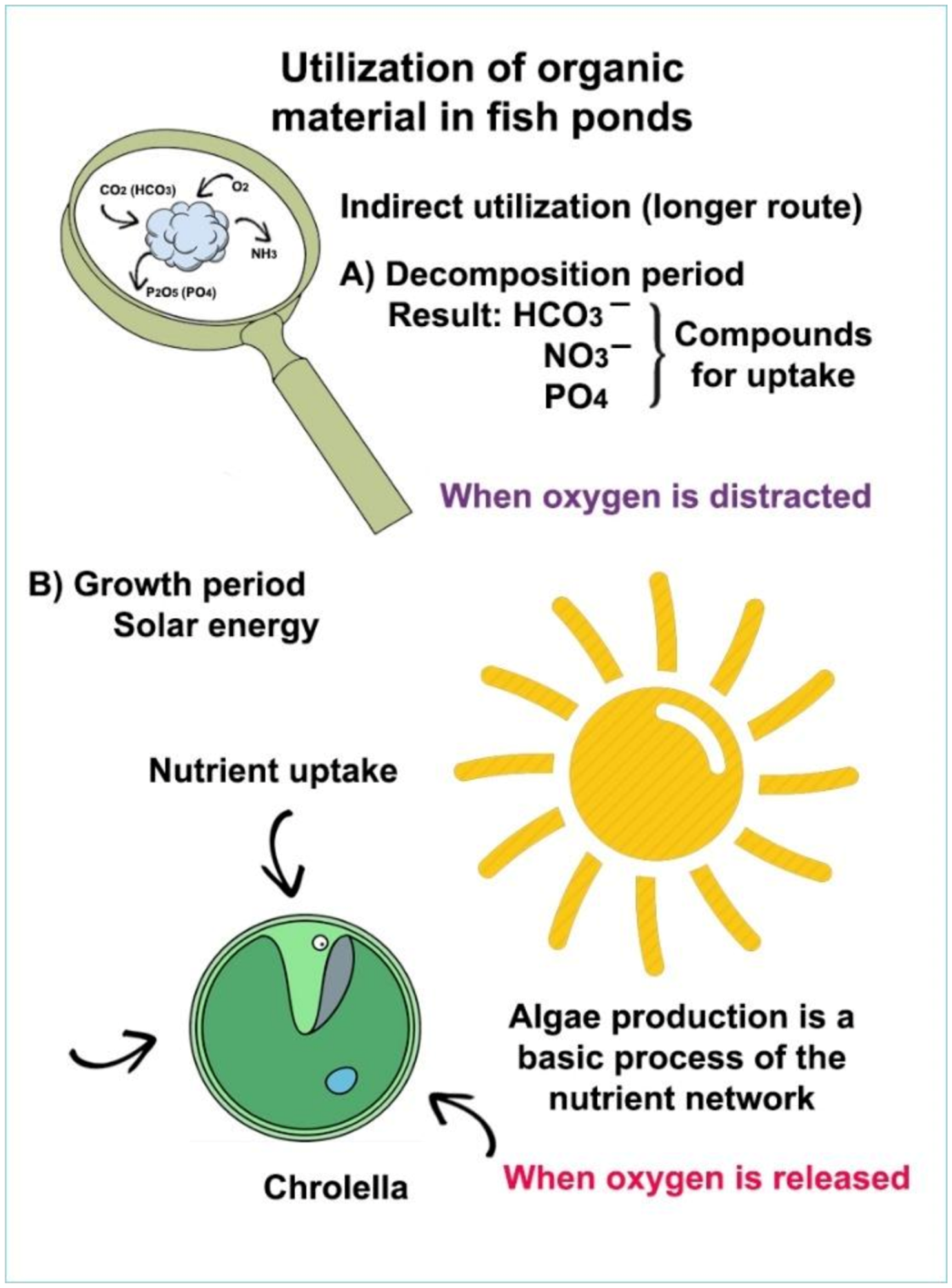
Figure 20. Two adverse processes of organic compounds in the ponds: (A) decomposition of dead organic matter to carbon dioxide; (B) production of new, energy-rich organic compounds by algal assimilation.
A continuous enabling of the uptake of carbon dioxide and other important biogenic elements for algae has created suitable conditions for continuous reproduction. However, some questions still remain open: what happens with the algal biomass? How will high-quality freshwater fish products suitable for human consumption be developed from the primary producer water algae? To answer this question, the whole process of energy transfer and trophic levels in a pond ecosystem needs to be analysed. The synthesis of inorganic carbon compounds into organic matter is the result of plant assimilation. Newly produced organic matter (biomass of algae) enters the food chain when consumed by heterotrophic organisms.
12. Trophic Levels in Terrestrial and Aquatic Ecosystems
According to a simple definition, an ecosystem is a self-regulating biotic community (biocenosis) together with its physical and chemical environment, where interactions occur between the organisms and the environment. The migration and transformation of all organic matter/energy in an ecosystem is triggered by vital activities of biological organisms. The most important activity within the interactions of living organisms of an ecosystem is consumption (trophic levels). While osmotrophy is the feeding mechanism of autotrophic plants to take up nutrient elements, animals use phagotrophy for that. The organisms use the energy of nutrients/foods for metabolisms and to increase their biomass.
Energy-rich organic matter can only be produced by autotrophic plants. The migration pathways of organic matter and stored energy via the different trophic levels were recognized by the founder of modern ecology, Charles Elton (1900–1991), an ecologist from Oxford. His discovery [4] is modelled in Figure 21.
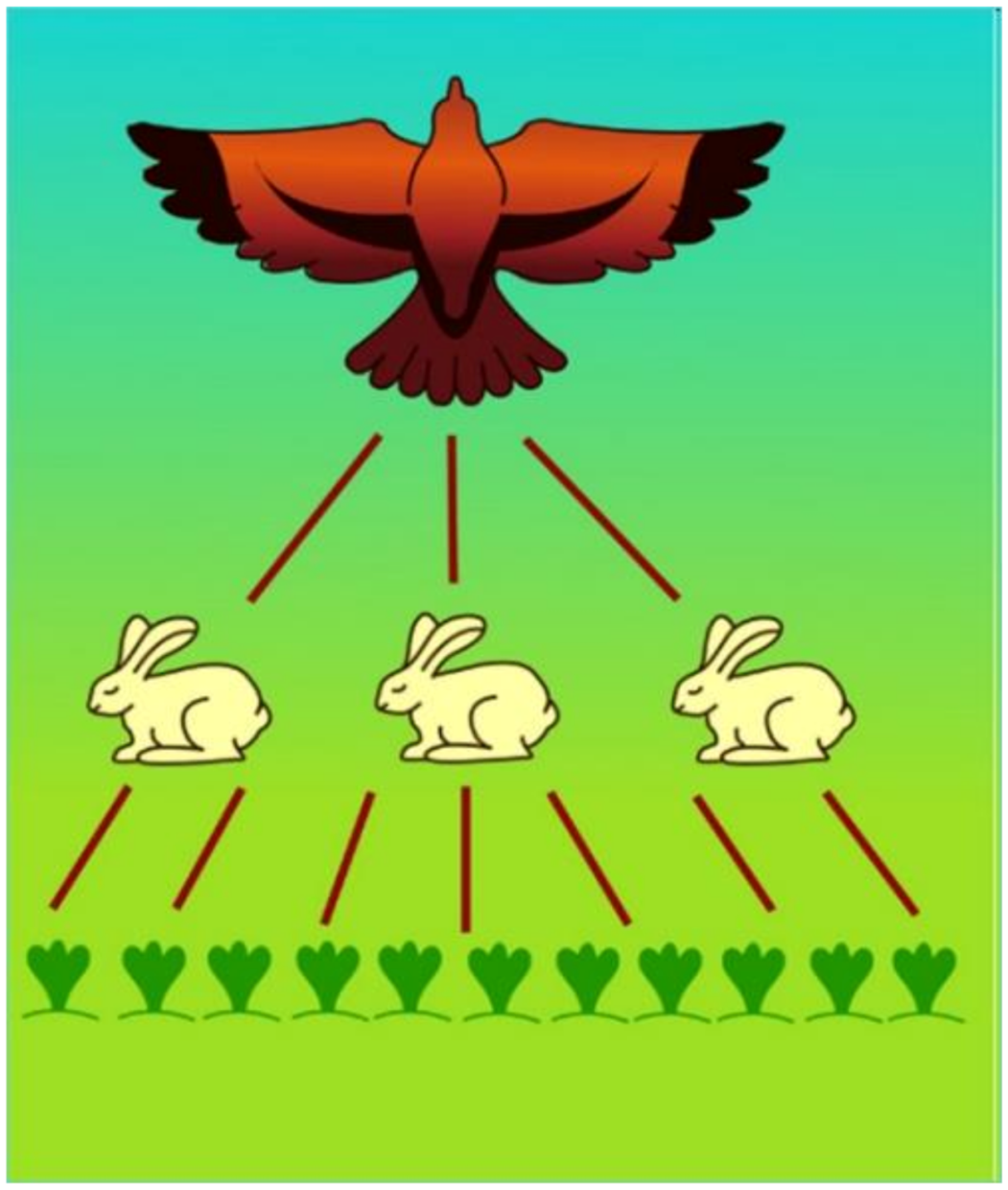
Figure 21. Progress of organic matter and its energy through trophic levels: autotrophic plants as Primary producers, plant consumers as Primary consumers, predators as Secondary consumers (in this figure: top predator).
This ecological law is not only valid for terrestrial ecosystems but for aquatic one as well.
The efficiency of biomass incorporation between different trophic levels is rather poor at no more than 10%[5]; therefore, the organic energy of primary production (algae) is dissipated via a maximum of four trophic levels (see Figure 22).

Figure 22. Quantitative relationship of different trophic levels in a lake ecosystem. From bottom to top: algae, fish consuming zooplankton and algae, fish consuming zooplankton.
Quantitative ratios of fish products based on natural (evolutionary preference) food are shown in Figure 23. In the classical Central-Eastern European production system, carp is a dominant species. It belongs to the tertiary trophic level and consumes zooplankton and zoobenthos from the secondary trophic level. In Asian multispecies cultures, which are economically more productive, carp is stocked together with herbivorous fishes. The Asian fish species of the second trophic level consume algae from the primary production level and higher water vegetation as well. Therefore, based on Figure 24, the total fish biomass produced in polycultures can theoretically be ten times higher than that of a carp monoculture. Unfortunately, the population of Central and Eastern Europe, which traditionally consume carp, is not open to accepting fish products made from herbivorous fish, which are produced much more cheaply.
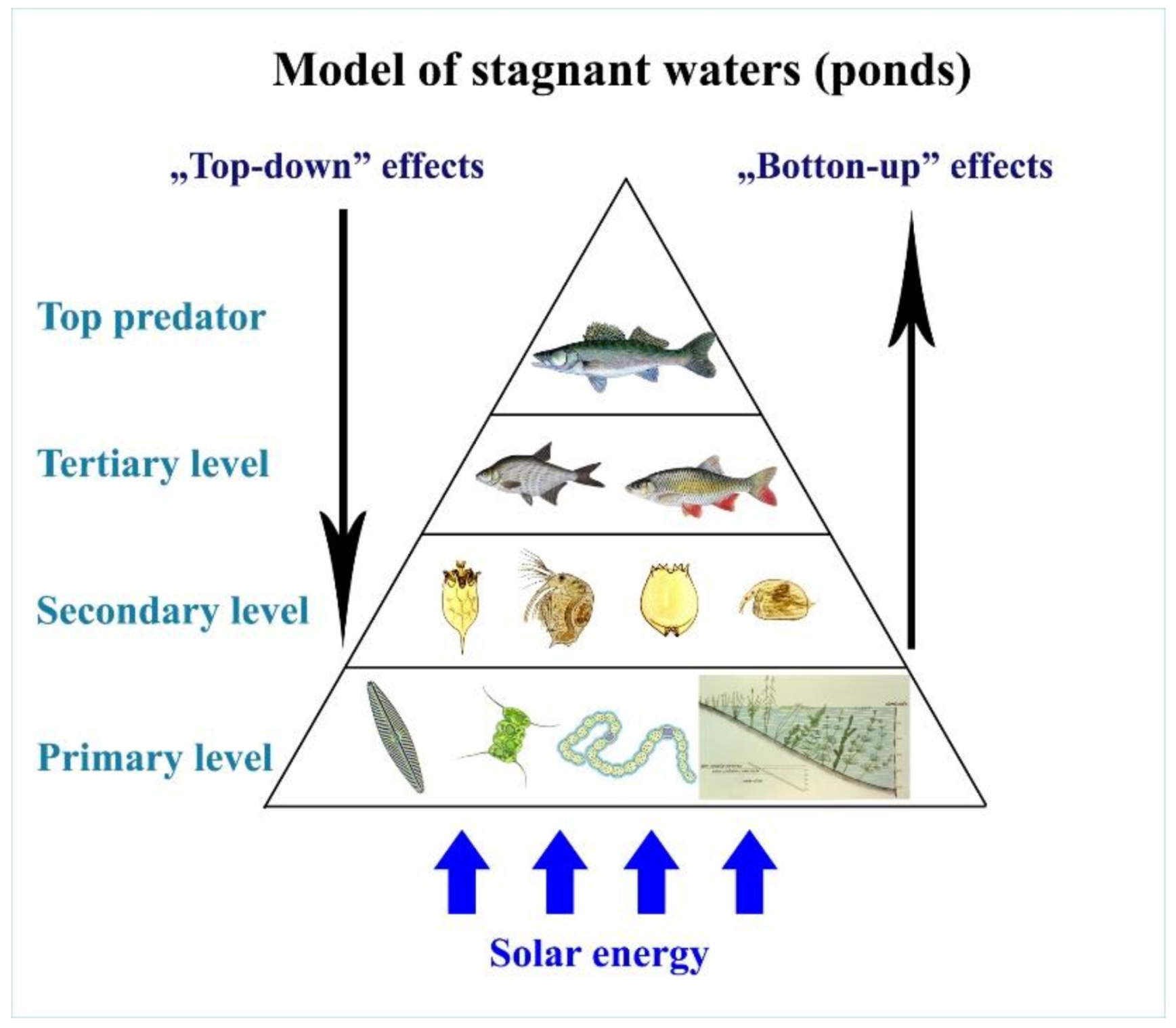
Figure 23. A food chain with four levels in a natural lake.
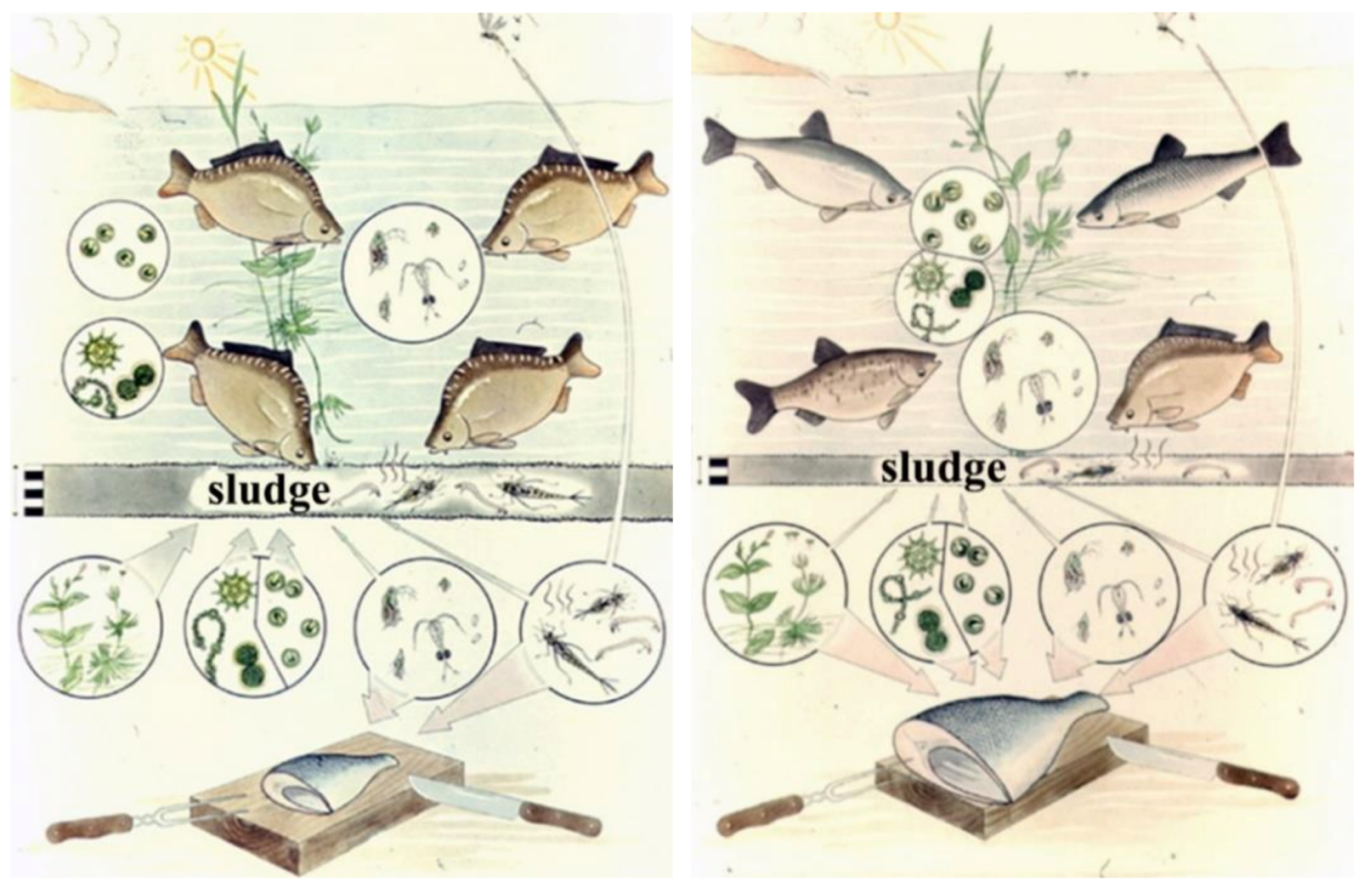
Figure 24. Fish production ratios based on natural food: monoculture vs. polyculture.
Investigating the route of the food chain and energy transfer in pond farming systems dominated by carp, at first sight it seems to be evident that the process of energy transfer is very similar to that of natural lake ecosystems (see Figure 24). The main difference is the huge number of stocked fish in fishponds. Consequently, the migrating energy and the total biological production are much higher in them. When stocking predator fish in ponds, the goal is not the consumption of carp but the elimination of invasive trash fish that are food competitors for carp.
A deeper investigation of the circulation of carbon compounds in a fishpond shows significant differences, thanks to the special feeding habit of carps stocked in large numbers. First the bottom-up regulation of energy transfer in ponds is analysed, as presented in Figure 25.
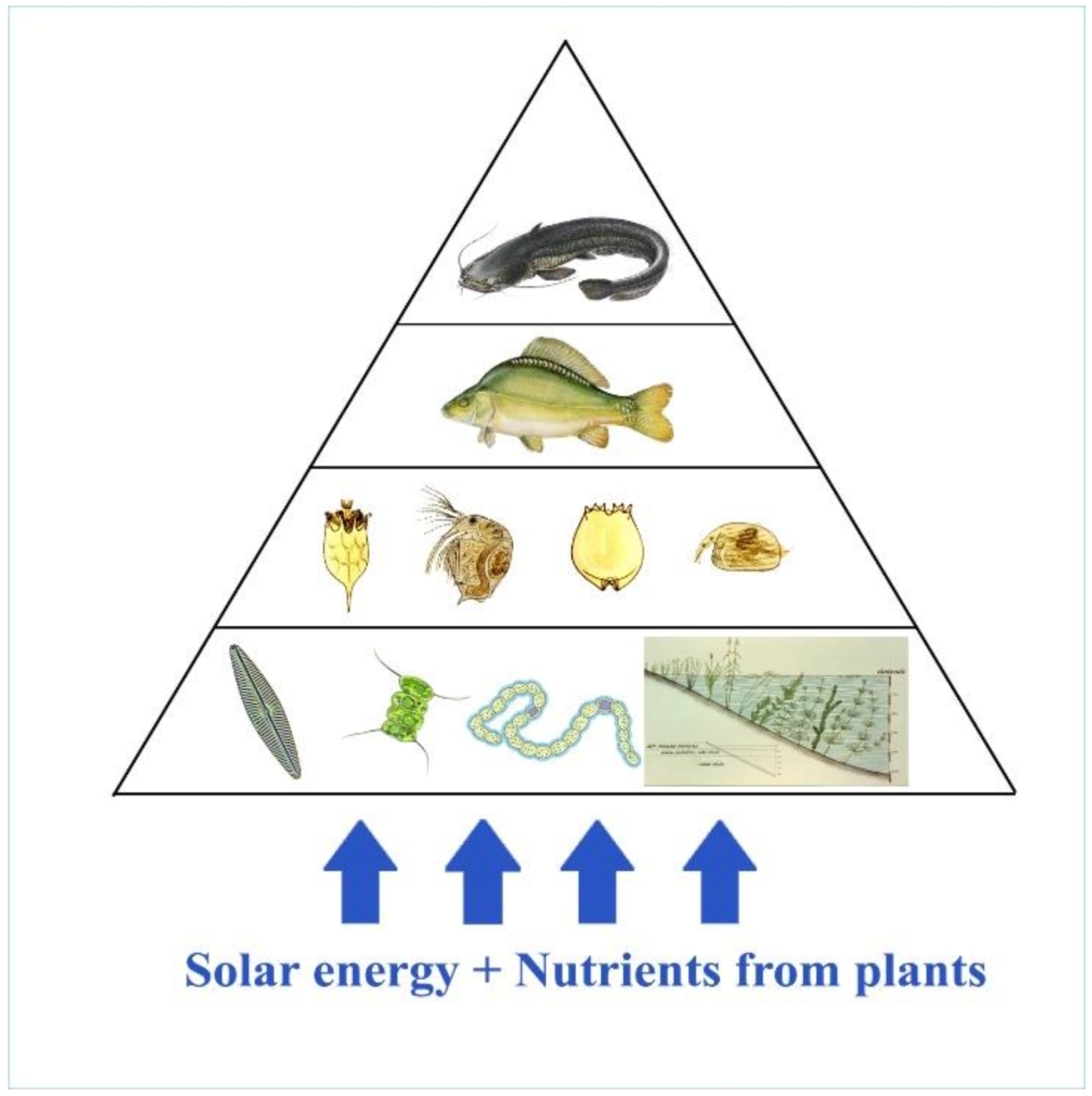
Figure 25. At first sight, trophic levels in ponds are very similar to that of natural lakes. However, huge differences can be detected due to the number of stocked fish.
At each trophic level, a small part (10%) of the energy fixed in carbon compounds by algae integrates into organisms—biomass—of the next level, while a much greater part is used to cover the metabolic energy needs of organisms of the current level. This larger part of the energy dissipates from the system. The final product of metabolic activities (dissimilation) is carbon dioxide (present in water as bicarbonate), which then returns to the first nutrient level (carbon circles 1–5), and after being reenergized by algae, the carbon migration process starts again. As Figure 26 indicates, this type of biological production has practically no carbon footprint, as carbon dioxide cannot leave the system (staying in the form of bicarbonates) but circulates in the water until being trapped either as fish biomass or as organic sediment in the mud. This migration of biological matter and energy in fishponds is very similar to the process of energy transfer in natural eutrophic lakes. However, when carp feeding habits are analysed, significant differences can be observed. The large number of carp present in ponds (and not in lakes) can modify the physical circumstances. As discussed., common carp not only filters zooplankton, but also feeds on worms, snails, etc., from sedentary mud. When a large number of carp hunt these organisms with a high protein content, it results in a resuspension of organic mud (called bioturbation). The consequence is a provoked, quick carbon circulation, eliminating or at least decreasing the amount of carbon trapped in the sediment and increasing carbon fixation in fish biomass. This is a notable advantage of carp culture systems: the large number of carp influences the production environment by developing a significant trophogenic zone for algae production and also a quick carbon circulation within a pond without any carbon footprint (see Figure 27).
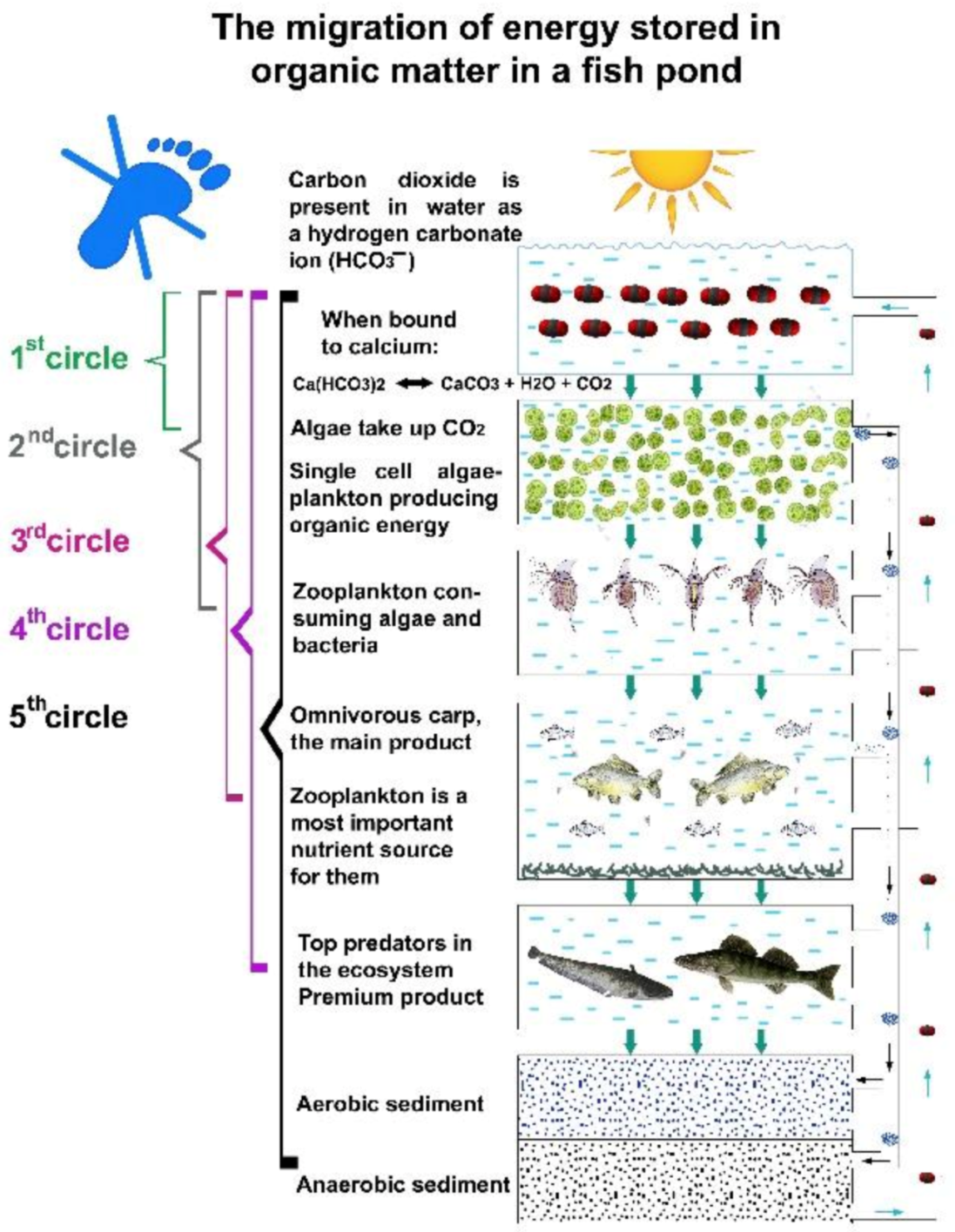
Figure 26. The migration of carbon atoms via trophic levels of pond ecosystems. Energized carbon compounds produced by algae start to move to upper trophic levels. Between two levels, the loss of energy is around 90%.
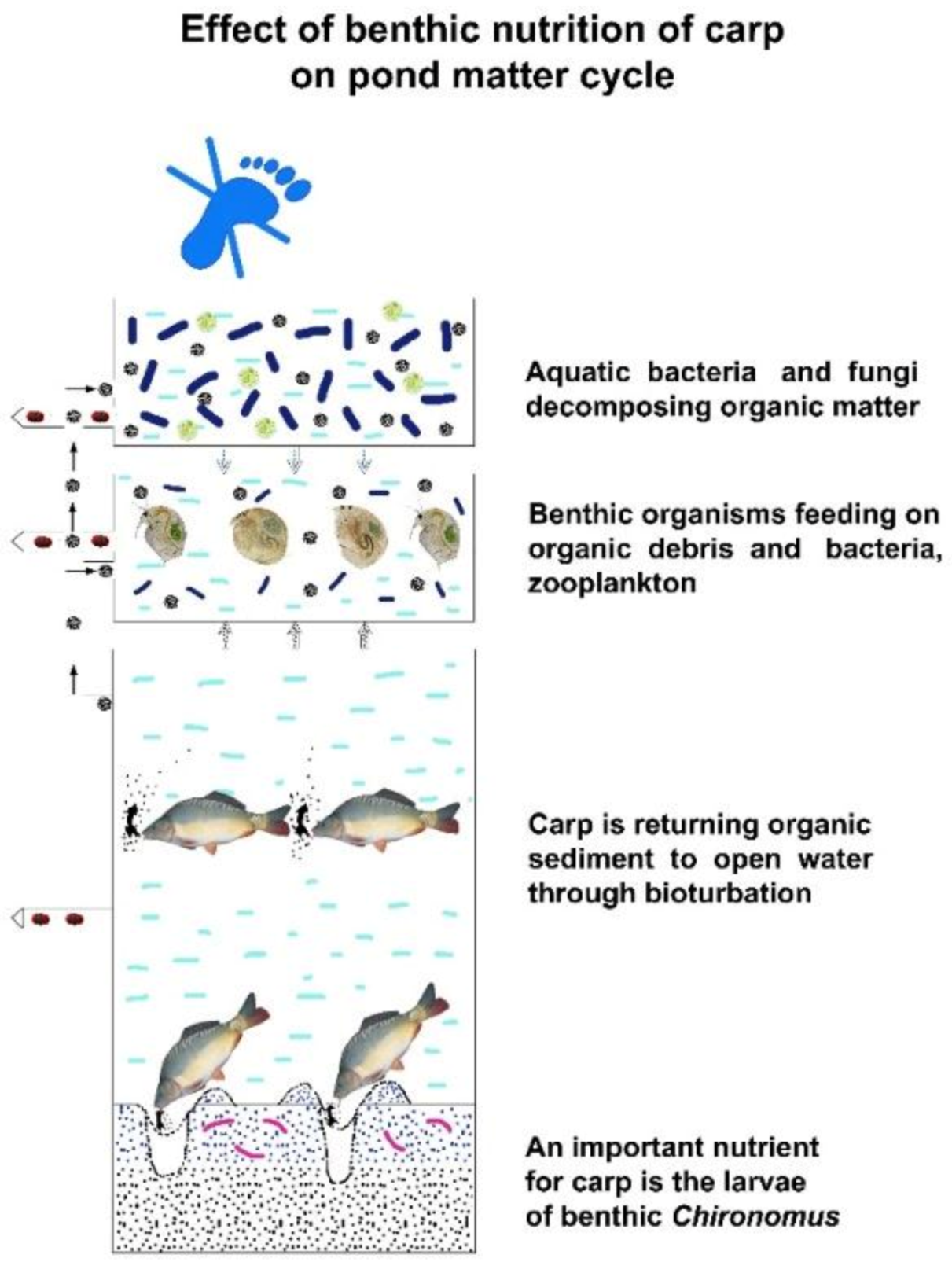
Figure 27. Bioturbation of common carp in pond culture systems. Organic mud is resuspended forming a trophogenic layer and a production environment.
To summarize the above analysed chemical and biological processes of water, it is evident that, in fishponds, electron pairs in covalent and ionic bonds keep carbon dioxide molecules in stable forms within bicarbonate (HCO3) and carbonate (CO3) anions; therefore, gaseous carbon dioxide is not able to diffuse into the atmosphere, nor can it increase the amount of atmospheric greenhouse gases, which is favourable from an environmental point of view.
References
- Padisák, J. Általános limnológia; ELTE Eötvös Kiadó: Budapest, Hungary, 2005; pp. 1–310.
- Felföldy, L. A Vizek Környezettana, Általános Hidrobiológia; Mezőgazdasági Kiadó: Budapest, Hungary, 1981; pp. 1–289.
- Ördög, V. Halastavak hidrobiológiája—A tókezelés hidrobiológiai alapjai. In Horváth L. (szerk.): Halbiológia és Haltenyésztés; Mezőgazda Kiadó: Budapest, Hungary, 2000; pp. 344–385.
- Elton, C. Animal Ecology; Wiley: Oxford, UK, 1927; pp. 1–298.
- Lindeman, R.L. The trophic-dynamic aspect of ecology. Ecology 1942, 23, 399–418.
More
Information
Subjects:
Environmental Sciences; Biology
Contributors
MDPI registered users' name will be linked to their SciProfiles pages. To register with us, please refer to https://encyclopedia.pub/register
:
View Times:
1.7K
Entry Collection:
Environmental Sciences
Revisions:
2 times
(View History)
Update Date:
03 Nov 2022
Notice
You are not a member of the advisory board for this topic. If you want to update advisory board member profile, please contact office@encyclopedia.pub.
OK
Confirm
Only members of the Encyclopedia advisory board for this topic are allowed to note entries. Would you like to become an advisory board member of the Encyclopedia?
Yes
No
${ textCharacter }/${ maxCharacter }
Submit
Cancel
Back
Comments
${ item }
|
More
No more~
There is no comment~
${ textCharacter }/${ maxCharacter }
Submit
Cancel
${ selectedItem.replyTextCharacter }/${ selectedItem.replyMaxCharacter }
Submit
Cancel
Confirm
Are you sure to Delete?
Yes
No





