Enzymes are mainly proteins that act as catalysts in biological processes, reducing the activation energy required for various chemical transformations. Unlike chemical catalysts, enzymes operate under moderate conditions of temperature and pH. Due to their increased stability and mild conditions of use, low toxicity, and low environmental impact, enzymes have a great industrial potential. The applications of enzymes cover a large range of fields, such as food (winemaking, cheeses, splitting proteins, clarification of juices, etc.), medical and pharmaceutical (classic drugs and systems with controlled release of active compounds, synthesis of biologically active compounds, protecting implants from the action of microorganisms etc.), cosmetics (manufacturing of hair dyes, skin care products), textile industry (cellulose splitting, natural fiber dyeing and processing technologies, fiber bleaching), paper manufacturing industry (bleaching, pulp processing), obtaining cleaning products (stain removal), chemical synthesis (favoring some chemical processes), environmental protection (degradation of some pollutants, discoloration of wastewater), and biosensors
[1][2][3][4]. The reduced stability of enzymes with industrial process parameters (high temperatures, extreme pH, and the presence of organic solvents) and their difficult separation have led to numerous techniques’ development to improve the performance of enzyme-catalyzed reactions. Therefore, enzyme immobilization technologies using different types of support materials have been implemented. The immobilization of enzymes can be achieved by chemical or physical binding on a specific support material and lead to the fabrication of composite catalysts with improved stability over a large range of conditions. The support material used for enzyme immobilization is of capital interest since it influences the properties of the obtained biocatalyst, determining the stability, enzymatic activity, and level of reusability of the biocatalyst
[5]. There are various organic and inorganic support materials that can be employed in the immobilization of enzymes, starting with metal oxides, silica, or natural and synthetic polymers, and culminating with hybrid materials based on metal nanoparticles, graphene, or carbon nanotubes. Among them, polymers are of particular importance because they can provide a large number of functional groups capable of interacting with enzymes, ensuring their binding and, at the same time, maintaining the substrate accessibility to the active site of the enzyme. The use of polymeric materials allows for obtaining composite materials with a large spectrum of properties, such as hydrophilic or hydrophobic character, porosity, and the flexibility of the support matrix. The use of natural polymers ensures a series of additional properties, such as biocompatibility, biodegradability, and lack of toxicity, leading to obtaining polymer/enzyme composite materials that can be used in a large number of applications, including in the medical and food fields, while the use of synthetic polymers is more economically feasible and ensures a better control of the properties of the polymer/enzyme composite materials thus obtained. These materials are characterized by increased stability at high temperatures and extreme pH or, in the presence of organic solvents, can be reused in a large number of reaction cycles without a significant loss of catalytic activity, and offer a plethora of potential applications. Their properties can be easily tuned by selecting a certain polymeric matrix or material morphology. Additionally, secondary treatments, such as chemical cross-linking, can be used to enhance the materials’ stability. Here, the methods for obtaining polymer/enzyme composites are classified according to material shape/morphology. Recent examples from the last 10 years’ literature on the fabrication of composite materials by enzyme immobilization, as well as their potential applications, are presented, with a special interest in subjects of concern, such as water treatment or chemical catalysis.
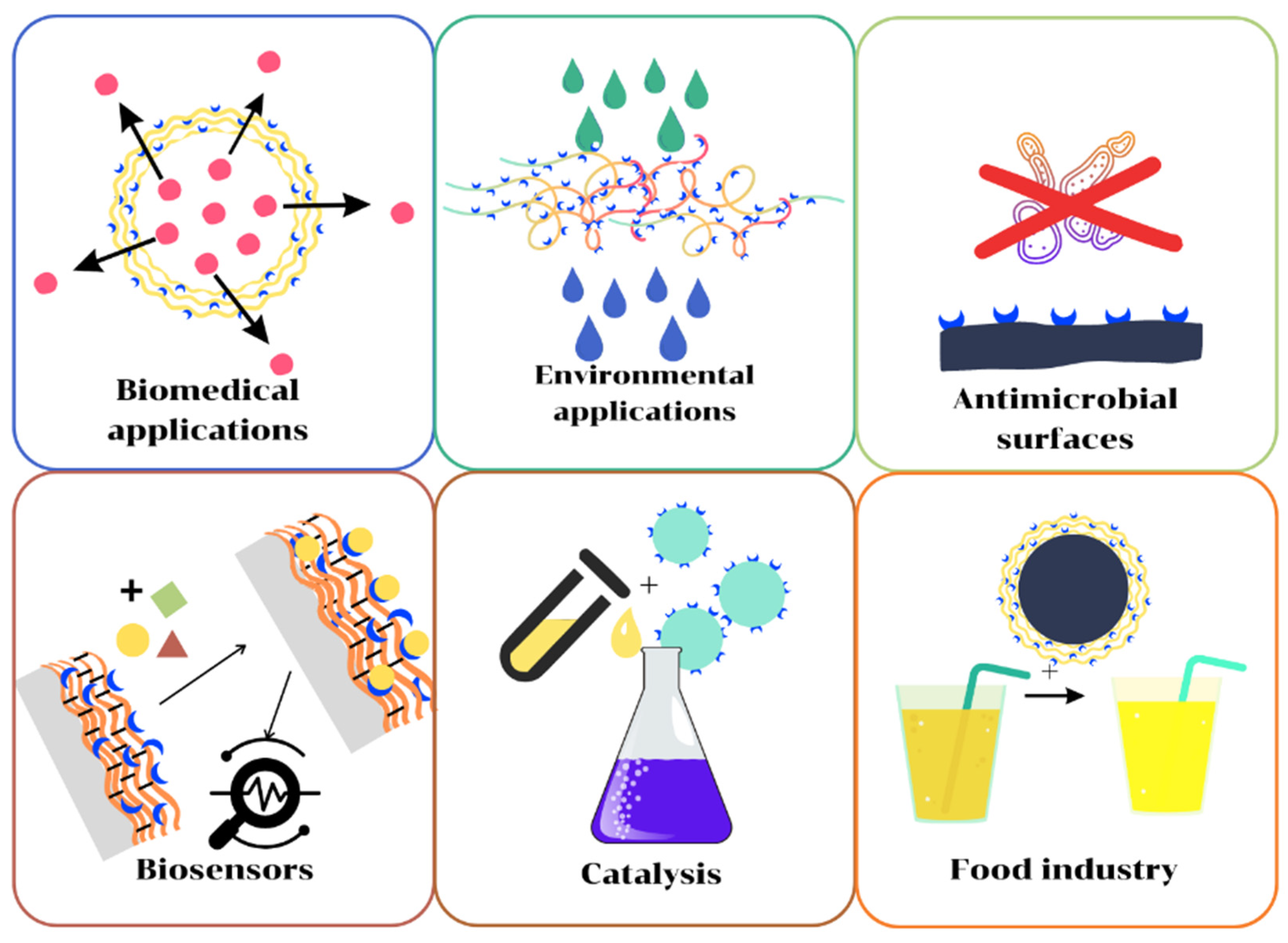
3. Applications of Polymer/Enzyme Composite Materials
Polymer/enzyme composite materials are distinguished by high stability, specificity, and selectivity, as well as easier separation and reusability. These are biocatalysts with industrial applicability, ensuring the sustainability and efficiency of industrial processes due to the advantages they offer. Unlike chemical catalysts, biocatalysts operate at mild temperature and pH conditions, have higher selectivity, are nontoxic, and can be used in a greater number of applications (Figure 2).
Figure 2. Applications of enzyme/polymer composites.
Although limited by technological barriers, the use of polymer/enzyme materials on an industrial scale is beginning to increase; these biocatalysts are successfully used in applications, such as juice clarification, paper bleaching, and the degradation of some polluting agents. A critical examination is necessary to determine the type of composite material suitable for a particular application, but examples in the literature show that polymer/enzyme composite materials find applications in areas of interest, such as the pharmaceutical industry, food industry, chemical synthesis, and environment protection.
3.1. Applications in Environmental Protection
One of the most important problems our society is facing is the increasing level of water, air, and soil pollution. While pollution is generally hard to mitigate, novel techniques for the efficient removal of pollutants are deeply investigated. A special interest is raised by water pollution since water is a very good solvent for a lot of dangerous chemicals, such as dyes, pesticides, and metal ions, which can pass from water sources to plants, animals, and even humans and raise important health concerns due to their toxicity, bioaccumulation, and carcinogenic effects
[8]. The use of polymer/enzyme composite materials in environmental applications has attracted increased interest in recent years due to their versatility and stability, satisfactory catalytic activity, and the possibility of reusing the material without an important loss of catalytic activity
[9]. Several studies have reported the use of composite polymer/enzyme systems for the removal of organic compounds found in wastewater, such as phenols, pesticides, and dyes.
Bilal and collaborators immobilized manganese peroxidase in composite microspheres obtained from PVA and ALGNa, which they subsequently used in sorption studies of some dyes from simulated and real polluted water samples
[10]. Studies in simulated polluted water revealed that composite particles with immobilized peroxidase were able to discolor water samples by 78–92%, depending on the type of dye dissolved in the water, as compared with only 57–74% discoloration when treating water samples with the free enzyme. The materials were later used in the treatment of polluted water samples taken from textile factories and printing houses, containing mixtures of dyes, obtaining satisfactory results. In another study, Aricov and collaborators used laccase/CHI composite microspheres for the removal of indigo carmine, attaining the full discoloration of the sample after 14 min
[11].
Enzymes immobilized in polymer fibers can also be successfully used in the retention and degradation of some pollutants in wastewater. An example in this regard is proposed by Maryšková and collaborators, who obtained composite fibers based on polyamide and PEI in which they immobilized laccase and subsequently used it to treat polluted water samples with different emerging contaminants
[12]. The authors used three types of water samples: deionized water, polluted river water treated by ultrafiltration, and polluted water to which citrate buffer solution was added. A mixture of pollutants consisting of bisphenol A, 17 α-ethynyl estradiol, triclosan, and diclofenac was added to these samples at an initial concentration of 10 mg/L. Following the treatment with the composite fibers with laccase, the retention of all four contaminants was noticed in a proportion of at least 10–20%. The best results were recorded when using deionized water, with the percentage of destroyed pollutants varying between 73.6% for triclosan and 17.5% for diclofenac.
Leontieș and collaborators studied the discoloration of water samples employing laccase/CHI/PAA composite microparticles
[13]. The degradation efficiency of these composites was 90% for both naphthol green and indigo carmine, achieved in about 9 and 10 min, respectively. Vera and collaborators used composite materials based on laccase and poly(glycidyl methacrylate) for the enzymatic degradation of diazinon, an insecticidal compound from the class of organophosphorus compounds
[14]. The use of the biocatalyst in the presence of 2,2-azino-bis-(3-ethylbenzothiazoline-6-sulfonic) acid allowed the total degradation of the pollutant after 48 h of contact and about 88% of the amount of the pollutant, in the absence of the mediator. A study by Lassouane and collaborators used composite materials based on CHI and laccase for the degradation of bisphenol A
[15]. After optimizing the degradation conditions, the authors achieved a pollutant degradation efficiency of over 99%, demonstrating at the same time that the biocatalyst can be successfully used in at least 10 reaction cycles, with about 70% of the activity maintained.
3.2. Catalysts for Chemical Industry
Enzymes are biological catalysts distinguished by increased specificity. Due to their versatility, they can catalyze a large number of chemical transformations of interest, such as the digestion of proteins or carbohydrates, the degradation of organic pollutants to nontoxic small molecular compounds, or the transformation of substrates into products of interest, such as drug intermediates, antibiotics, or organic acids. The increased use of polymer/enzyme composites as catalysts is the result of their stability, easy separation, and reusability, making them suitable candidates for industrial processes.
In a recent study, Pereira Cipolatti and collaborators used composite microparticles based on polyurethane and lipase, coated with trehalose to obtain two organic acids (eicosapentaenoic acid and docosahexaenoic acid) that are used in the food industry and some mandelic acid derivatives with pharmaceutical applications
[16]. The authors demonstrated that lipase-based composite microparticles can be successfully used to obtain compounds of interest, showing increased catalytic activity, as well as stability at temperature, in a wide pH range, and storage time. In another study, Pan and collaborators used composite biocatalysts obtained from CHI-coated magnetic microparticles covalently decorated with lipase
[17]. The biocatalysts thus obtained were used for the enantioselective acylation of a racemic mixture of L-phenylethylamine. The authors observed that the use of the composite biocatalyst allows the acylation reaction to take place at an elevated temperature without denaturation of the enzyme. Additionally, the biocatalyst could be reused in up to 7 reaction cycles, maintaining about 60% of the activity.
Polymer/enzyme composites also find applications in the cosmetic and perfume industry, especially as catalysts for obtaining chemical compounds of interest. An example in this sense was proposed by Melo and collaborators, who tested the catalytic activity of some composite materials obtained by encapsulating lipase B isolated from
Candida antarctica in a composite matrix consisting of CHI and sodium polyphosphate
[18]. The biocatalyst was used to obtain benzyl acetate (synthetic jasmine flavor) under different reaction conditions. The authors demonstrated that the obtained composite biocatalyst shows increased stability under the tested process conditions, being stable in a wide pH range (pH = 4–10), at high temperatures (maximum activity at around 55–60 °C and total inactivation at temperatures higher than 80 °C), but also in the presence of different organic solvents, the best catalytic activity being recorded when using a mixture of dimethyl sulfoxide and phosphate buffer solution (pH = 7). Additionally, this biocatalyst was successfully reused in 5 reaction cycles with the maintenance of about 86% of the enzyme activity. Tudorache and collaborators reported the fabrication of lipase/ALGCa and lipase/κ-carrageenan composite beads that can be used as catalysts for the conversion of α-pinene to oxy-derivatives, followed by isomerization to campholenic aldehyde and trans-carenol, compounds of interest for the cosmetic and fragrance industry
[19].
3.3. Biosensors
Biosensors find applications in the detection of molecules of interest in various fields, including the medicine, cosmetic, and food industries, but also pollution control and environmental protection. Biosensors based on immobilized enzymes are characterized by a high specificity for the molecule of interest, have the ability to mediate a chemical reaction between the receptor and the analyte, and allow the conversion of the chemical response into an electrical response
[20]. Additionally, biosensors have increased stability and low environmental impact, in recent years, the use of polymer/enzyme composites being established as one of the most versatile and economical detection methods
[20].
In a recent study, Tutunaru and collaborators developed a composite biosensor based on acetylcholinesterase grafted on carboxyl-modified single-walled carbon nanotubes and poly(3,4-ethylenedioxythiophene), which was subsequently used for the detection of organophosphate insecticides in apple juice
[21]. The biosensor thus obtained was able to successfully detect and recover dichlorvos, with detection limits of 0.447 and 5.54 ppb, depending on the detection method.
Recently, Cano-Raya and collaborators reported the fabrication of a biosensor for pyrocatechol by immobilizing laccase in polyamide 6 microparticles subsequently deposited on a semipermeable support material allowing substrate diffusion
[22]. To determine the selectivity of the biosensor, the authors tested three substrates: pyrocatechol and two of its isomers, resorcinol and hydroquinone, observing an extremely good selectivity of the biosensor for the target analyte. In addition to high selectivity and satisfactory analytical response under controlled conditions, the authors also observed that the obtained composite biosensor can be successfully used in the detection of pyrocatechol from natural water samples. Thus, the authors tested river water samples collected from various areas and in different seasons, to which they added well-determined amounts of pyrocatechol. The authors obtained pyrocatechol recovery rates of over 97% for all samples tested. A recent example was proposed by Aldea and collaborators, who fabricated a biosensor based on glucose oxidase and PMMA electrospun fibers, coated with a gold layer and deposited on a poly(ethylene terephthalate) film
[23]. The sensor exhibited a sensitivity of 3.1 µA·cm
−2·mM
−1 and a detection limit of 0.22 mM.
3.4. Antimicrobial Applications
Based on the antimicrobial activity of some natural polymers, such as CHI, but also of some enzymes, polymer/enzyme composites can be used to inhibit the development of some microorganism strains. Among the most significant enzymes with antimicrobial activity, lysozyme exhibits a growing interest based on its ability to lyse a cell wall peptidoglycan layer of Gram-positive bacteria.
In a recent study, Bayazidi and collaborators showed that cellulose fibers with immobilized lysozyme show antimicrobial activity against
Staphylococcus aureus,
Escherichia coli,
Listeria monocytogenes,
Yersinia enterocolitica,
Aspergillus niger, and
Saccharomyces cerevisiae [24]. Li and collaborators obtained composite nanoparticles based on CHI and alginate lyase and tested their antimicrobial activity on a
Pseudomonas aeruginosa strain, observing that the immobilized enzyme showed higher antimicrobial activity than the free enzyme
[25]. Thus, the biofilm formed by
Pseudomonas aeruginosa in the presence of the CHI/alginate-lyase composite was only 21 µm thick, as compared with ~49 µm observed in the presence of the free enzyme and ~87 µm in the absence of any inhibitory agent. In a recent study, Yuan and collaborators demonstrated the potential biomedical applications of composite membranes obtained by layer-by-layer deposition of lysozyme and collagen on a fibroin and nylon 6 support material
[26]. The antimicrobial activity of the composite membranes was demonstrated on a
Staphylococcus aureus strain. The authors observed a decrease of about 98% in the proliferation capacity of the bacterial culture compared with the unmodified membrane. Moreover, these composite membranes with embedded lysozyme showed increased biocompatibility, allowing the development of fibroblast cultures. Baidamshina and collaborators recently reported the fabrication of composite materials with antimicrobial properties by immobilizing papain in a CHI matrix
[27]. About 90% of papain’s enzymatic activity was maintained in the microparticles, with an extended stability at higher temperature and pH range. The authors demonstrated that the obtained materials can act efficiently on a bacterial biofilm formed by
Staphylococcus aureus and
Staphylococcus epidermidis species.
3.5. Biomedical Applications
Another area of interest for the use of polymer/enzyme composites is represented by the fabrication of materials with biomedical applications. This field can include materials for tissue engineering, implants, systems with controlled release of active substances, and biosensors of medical interest, but also catalysts to obtain biologically active substances. In a 2020 research study, Filho Moreira and collaborators reported the fabrication of a composite material with applications in tissue engineering by immobilizing papain in alginate microparticles
[28]. The use of this composite is based on the biomedical properties of the two-component materials: alginate, recognized for its properties of stimulating cell regeneration, and papain, involved in the depletion of necrotic tissues. In brief, the enzyme is released through a controlled diffusion, about 64% released for 24 h.
Another potential biomedical application of polymer/enzyme composites is related to the prevention of microbial contamination of some implants. Recently, Teske and collaborators reported novel composite films based on papain and poly(L-lactic acid) with antimicrobial properties
[29]. The composite material was customized by cross-linking papain, under the action of 1-(3-dimethylaminopropyl)-3-ethylcarbodiimide hydrochloride, on the polymer surface. The antimicrobial activity was tested against a
Clostridioides difficile strain, one of the common pathogens responsible for the occurrence of nosocomial infections, observing a reduction of about 20% of the biofilm formed on the surface of a cardiovascular implant. Another example was proposed by Visan and collaborators, who used a composite coating based on lysozyme, polycaprolactone, and PEG to prevent the microbial contamination of some Ti implants
[30]. The composite film exhibited increased antibacterial activity against both Gram-positive and Gram-negative microbial strains. The results highlighted that the composite film inhibited the growth of
Escherichia coli,
Bacillus subtilis,
Enterococcus faecalis, and
Staphylococcus aureus strains.
Numerous enzymes successfully catalyzed the transformation of some compounds into pharmacological species, this category including antibiotics
[31], analgesics
[32], and some anti-inflammatory drugs
[33]. An example is presented by Gilani and collaborators, who studied the production of (S)-naproxen under the action of three biocatalysts: CHI/lipase composite microparticles, CHI/lipase microparticles cross-linked with GA, and microparticles obtained by lipase immobilization in a commercial resin of Amberlite XAD7 type
[33]. The authors studied the influence of various parameters on the hydrolysis process of the naproxen methyl ester racemic mixture, determining the optimal conditions for obtaining (S)-naproxen. The optimal parameters were a reaction time of 24 h, in the presence of isooctane and 2-ethoxyethanol as solvent/cosolvent, at 35 °C and pH = 7, successfully reused in about 10 reaction cycles.
3.6. Applications for Food Industry
Composite materials with immobilized enzymes are finding increasing use in the food industry. One of their most important applications is in the clarification and discoloration of fruit juices, in order to increase their appearance, stability, and shelf life. The most used types of enzymes for juice clarification are peroxidases and laccases that are able to oxidize phenolic compounds in juices, responsible for sediment formation and changes in their appearance, taste, and smell
[34].
Irshad and collaborators used composite materials obtained by immobilizing pectinase in CHI microspheres for the clarification of some fruit juices (apple, mango, peach, and apricot), the use of the biocatalyst leading to a considerable reduction of turbidity, viscosity, and discoloration of fruit juice samples
[35]. Bilal and collaborators used gelatin spheres with manganese peroxidase for the clarification of some fruit juices
[34]. Following the experiments, a 36.6% decrease in juice turbidity was observed, as well as a 42.7% reduction in juice color intensity. The proposed method has a number of important economic advantages, such as the possibility of reusing the composite material, easy separation, and increased stability over time and with environmental factors.
Benucci and coworkers reported papain immobilization on CHI-clay nanocomposite films. One of these films was efficiently used to reduce haze potential and the protein content of white wines
[36]. Fernandez-Pacheco and collaborators immobilized a commercially available β-glucosidase on ALGCa beads in order to improve white wine aroma by the depletion of flavorless glycosides. The immobilized enzyme displays a superior hydrolytic activity, their activity maintained at 96.5% after reutilization for seven times
[37].
Another area of interest for the food industry is represented by the prevention of microbiological contamination of some food products. In this regard, Wang and collaborators tested the antimicrobial activity of some composites obtained by immobilizing lysozyme and ALGNa on cellulose acetate nanofibers
[38]. The composite materials showed increased stability and reusability, with the maintenance of about 70% of the enzyme activity after 4 cycles of use. At the same time, the authors showed the antibacterial activity of these composites against a
Staphylococcus aureus strain from UTH milk, at two temperature values, 4 and 25 °C, respectively. Additionally, it was observed that the inhibitory action of composite fibers depends on the number of layers of lysozyme and ALGNa deposited on the support material, the best results being recorded for composites with nine composite layers.
Another application for the food industry is represented by the decontamination of some food products. In this sense, Bedade and collaborators studied the degradation of acrylamide from coffee samples, under the action of a composite biocatalyst obtained by immobilizing acrylamidase on ALGNa microparticles coated with CHI
[39]. The authors studied the retention of the contaminant under the action of the enzyme under static and dynamic conditions, demonstrating the retention of 100% of acrylamide in the studies performed using a batch column under optimal conditions. At the same time, the biocatalyst could be used in 4 reaction cycles with the maintenance of about 80% of the activity.