Your browser does not fully support modern features. Please upgrade for a smoother experience.

Submitted Successfully!
Thank you for your contribution! You can also upload a video entry or images related to this topic.
For video creation, please contact our Academic Video Service.
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Ghanyah Al-Qadami | -- | 1446 | 2022-10-27 02:03:14 | | | |
| 2 | Conner Chen | Meta information modification | 1446 | 2022-10-27 04:14:21 | | |
Video Upload Options
We provide professional Academic Video Service to translate complex research into visually appealing presentations. Would you like to try it?
Cite
If you have any further questions, please contact Encyclopedia Editorial Office.
Al-Qadami, G.H.; Secombe, K.R.; Subramaniam, C.B.; Wardill, H.R.; Bowen, J.M. The Short-Chain Fatty Acids. Encyclopedia. Available online: https://encyclopedia.pub/entry/31470 (accessed on 26 May 2026).
Al-Qadami GH, Secombe KR, Subramaniam CB, Wardill HR, Bowen JM. The Short-Chain Fatty Acids. Encyclopedia. Available at: https://encyclopedia.pub/entry/31470. Accessed May 26, 2026.
Al-Qadami, Ghanyah H., Kate R. Secombe, Courtney B. Subramaniam, Hannah R. Wardill, Joanne M. Bowen. "The Short-Chain Fatty Acids" Encyclopedia, https://encyclopedia.pub/entry/31470 (accessed May 26, 2026).
Al-Qadami, G.H., Secombe, K.R., Subramaniam, C.B., Wardill, H.R., & Bowen, J.M. (2022, October 27). The Short-Chain Fatty Acids. In Encyclopedia. https://encyclopedia.pub/entry/31470
Al-Qadami, Ghanyah H., et al. "The Short-Chain Fatty Acids." Encyclopedia. Web. 27 October, 2022.
Copy Citation
Through fermentation, the gut microbiota can produce several types of metabolites, including short-chain fatty acids (SCFAs). SCFAs play an important role in maintaining epithelial barrier functions and intestinal homeostasis.
microbiota
short-chain fatty acids
cancer
chemotherapy
radiotherapy
immunotherapy
1. Introduction
The collection of bacteria and other microorganisms residing in the gastrointestinal tract, termed the gut microbiota, has emerged as an important target in improving and personalizing oncology treatment. A growing body of research has shown potential roles for specific microbial taxa in the efficacy of cancer treatment, as well as in the development of associated toxicities [1][2][3]. The richness of the microbiome has also been investigated, with higher microbial diversity shown to be a key predictor of survival in people having chemoradiation for cervical cancer [4]. A seminal study by Gopalakrishnan et al. [1] clearly showed significant differences in both the diversity and composition of the gut microbiota of people who responded to PD-1 inhibitor immunotherapy for melanoma compared to non-responders. These compositional differences may lead to altered treatment response in a variety of ways, including via changes in direct drug metabolism (for example the action of beta-glucuronidase in the intestinal toxicity of irinotecan [5]), or modulation of the host immune responses [6][7].
With the advent of more sophisticated analytical methods, there is now more appreciation for the functional aspects of the microbiota, including the production of metabolites. Short-chain fatty acids (SCFAs) are one important class of metabolites produced by the gut microbiota. SCFAs, which include butyrate, acetate, propionate, and others (discussed below), are produced in the gut by bacterial fermentation of indigestible fibers and have a variety of functions both in the gut and distal sites including the brain and kidneys [8]. SCFAs have previously been linked to colorectal cancer development, with multiple studies showing that butyrate, propionate, and acetate induce apoptosis in colorectal cancer cells but not in healthy cells. In addition, some studies have suggested that butyrate and propionate have potent anti-neoplastic effects [9]. This has led to suggestions that SCFA manipulation may be a useful preventative or therapeutic strategy in a variety of cancers [9][10][11][12][13].
Results linking the microbiota and cancer treatment outcomes have been promising but have so far failed to provide significant improvements to clinical practice [14]. It is therefore now increasingly important to move beyond simply correlating the abundance of particular bacterial taxa with a disease or phenotype, and to understand how the functional capacity of the microbiota may be critical [15]. Furthermore, in the development of microbial-based therapeutics, there is a strong rationale to investigate metabolites such as SCFAs, rather than bacteria themselves. This is because direct metabolite supplementation would remove the need to ensure microorganisms successfully colonize the host, such is the case with probiotics.
2. Short-Chain Fatty Acids
SCFAs are small organic carboxylic acids with 1 to 6 carbon atoms, and in the intestine, are the main product of anaerobic fermentation of indigestible polysaccharides such as resistant starch, inulin, cellulose, and pectin (Figure 1) [16]. Acetate, propionate, and butyrate are the most commonly produced SCFAs in the human gut, in a roughly 3:1:1 ratio [17]. Other SCFAs include formate, isobutyrate, valerate, isovalerate, and 2-methylbutanoate. SCFAs can move from the gut via the bloodstream in differing amounts. A study using stable isotopes in healthy human subjects found that systemic availability of acetate, propionate, and butyrate was 36%, 9%, and 2%, respectively, [18]. Subsequently, SCFAs have a range of functions both in the gut and elsewhere, with differences in local and systemic effects governed by their systemic availability. These include being a key energy source for colonocytes and playing roles in G-protein coupled receptor (GPCR) binding, histone deacetylation, and immune modulation [16][19]. SCFA quantification in fecal samples is commonly used to quantify SCFA production. This may however not be an accurate measure, as previous research has suggested that a majority of SCFAs produced in the colon are absorbed by the gut mucosa [16][20]. In addition, systemic SCFA levels may be affected by gut epithelial integrity [21].
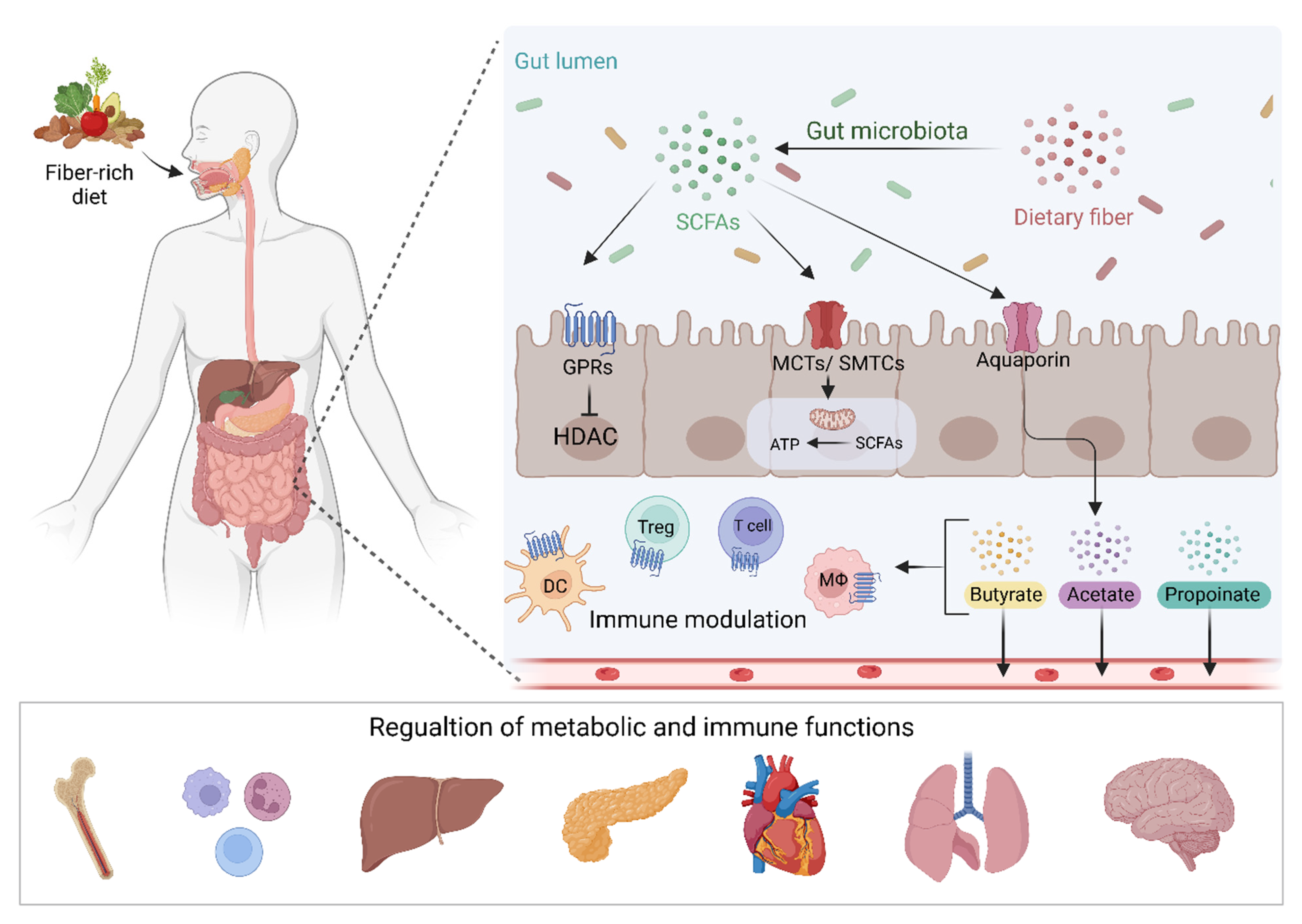
Figure 1. Dietary fiber and other fermentable substrates are fermented by the gut microbiota leading to the production of SCFAs. These metabolites interact with GPCRs or MCTs and SMTCs transporters leading to the regulation of gene transcription and energy productions in IECs. SCFAs can also passively cross the intestinal mucosa and regulate intestinal immunity and pass into the circulation to modulate metabolic and immune functions in different body organs. SCFAs; short-chain fatty acids, GPRs; G protein-coupled receptors, MCTs; Monocarboxylate transporters, SMTCs; Sodium-coupled monocarboxylate transporter; DC, dendritic cell, Treg; T regulatory cell, Mϕ; Macrophage. Figure created with BioRender.com.
A wide range of bacteria, many of which have previously been implicated in the efficacy or toxicity of cancer treatment, can produce SCFA, with the amounts and types of SCFA produced dependent on the types of bacteria present. Acetate is the SCFA produced in the highest levels in the gut. This is due to acetate production pathways being widely distributed among multiple types of bacteria, whereas other SCFAs such as butyrate and propionate production is restricted to a small group of bacterial types [22]. The phylum Firmicutes is the main butyrate-producing taxa, particularly Faecalibacterium prausnitzii (F. prausnitzii) (Ruminococcaceae) and Roseburia spp. (Lachnospiraceae). Eubacterium and Coprococcus are also important butyrate-producers [23]. These key butyrate-producing bacteria are generally anaerobes, and therefore flourish in the low-oxygen environment of the colon. In addition, Bifidobacterium species are also able to produce acetate and lactate, and Akkermansia muciniphila, among others, produce propionate and acetate, with most propionate producers in the colon belonging to the Bacteroidota phylum [8][16][24]. Luminally, SCFAs serve to acidify their environment, and in doing so restrict the growth of pathogenic microbes by preventing cellular respiration [25]. Similarly, SCFAs such as acetate are potent nutrient sources for other commensal microbes (including butyrate producers), and this cross-feeding mechanism is critical for the maintenance of a healthy and diverse microbial ecosystem.
SCFAs are absorbed by colonocytes via hydrogen or sodium-dependent monocarboxylate transporters [19], where much is used as an energy source for these cells. SCFAs, primarily butyrate and propionate, not metabolized within colonocytes are transported into the portal circulation and often then used as an energy source for hepatocytes [26][27]. Butyrate is a particularly important energy source for colonocytes, with previous research showing that colonizing germ-free mice with butyrate-producing bacteria increased oxidative phosphorylation and contained autophagy to normal levels in the gut [28]. Butyrate is also important in stabilizing gut epithelial barrier function, via the consumption of local oxygen molecules and subsequent stabilization of the barrier protecting transcription factor hypoxia-inducible factor (HIF) [29].
Aside from the above-mentioned monocarboxylate transporters, SCFAs can activate G protein-coupled receptors (GPCR), also known as free fatty acid receptors [16][30]. GPR41, GRP43, and GPR109A can be activated by multiple SCFAs and subsequently inhibit the production of cAMP [31]. These GPCRs are expressed on epithelial cells, neutrophils, and macrophages both within and outside of the gut, as well as a variety of other cell types including sympathetic neurons and pancreatic beta cells [32]. The wide range of GPCR sites shows the broad potential for SCFA-mediated effects. Enteroendocrine cells also express these receptors and respond by releasing contents that regulate neuroendocrine pathways controlling local and distant activity. Activities include appetite regulation, glucose homeostasis, and mucosal growth. See van der Hee and Wells [32] for further discussion of these effects.
Butyrate and propionate can modulate inflammatory responses in the gut mucosa via histone deacetylase (HDAC) inhibition. Previous research has shown that butyrate can suppress colonic inflammation via HDAC1-dependent Fas upregulation [33]. SCFAs can also alter the immune response in numerous other ways [34]. For example, butyrate can induce Treg differentiation [35] and increase the generation of Th1 and Th17 cells [36]. Microbiota-derived SCFA has also been shown to promote the cellular metabolism of antigen-activated CD8+ T cells and enhance their differentiation into long-term memory T cells [37]. Combined, these functions support the central role of SCFAs in the modulation of the immune system.
While it is beyond the scope of these contents to describe all individual differences in metabolism and function of different SCFAs, it is important to note that each SCFA has specific functions, which may be important in understanding their role in cancer treatment outcomes. As mentioned above, pathways for acetate production are spread amongst a range of bacterial types, leading to its high production in the gut, of which a relatively high level is able to move into the bloodstream. Acetate also has specific functions systemically, including involvement in lipid synthesis and acetylation reaction [38]. Butyrate is primarily produced via butyryl CoA:acetate CoA transferase pathways, and is one of the most researched SCFAs, due to its important role as an energy source. Propionate is produced via two pathways; the succinate pathways and the propanediol pathway. See Deleu, Machiels [8] for further description of SCFA production pathways. Propionate has also been suggested to have roles in lipogenesis in hepatocytes, as well as having antiproliferative effects in colon cancer cell lines [39].
References
- Gopalakrishnan, V.; Spencer, C.N.; Nezi, L.; Reuben, A.; Andrews, M.C.; Karpinets, T.V.; Prieto, P.A.; Vicente, D.; Hoffman, K.; Wei, S.C.; et al. Gut microbiome modulates response to anti-PD-1 immunotherapy in melanoma patients. Science 2018, 359, 97–103.
- Alexander, J.L.; Wilson, I.D.; Teare, J.; Marchesi, J.R.; Nicholson, J.K.; Kinross, J.M. Gut microbiota modulation of chemotherapy efficacy and toxicity. Nat. Rev. Gastroenterol. Hepatol. 2017, 14, 356–365.
- Chaput, N.; Lepage, P.; Coutzac, C.; Soularue, E.; Le Roux, K.; Monot, C.; Boselli, L.; Routier, E.; Cassard, L.; Collins, M.; et al. Baseline gut microbiota predicts clinical response and colitis in metastatic melanoma patients treated with ipilimumab. Ann. Oncol. 2017, 28, 1368–1379.
- Sims, T.T.; El Alam, M.B.; Karpinets, T.V.; Dorta-Estremera, S.; Hegde, V.L.; Nookala, S.; Yoshida-Court, K.; Wu, X.; Biegert, G.W.G.; Delgado Medrano, A.Y.; et al. Gut microbiome diversity is an independent predictor of survival in cervical cancer patients receiving chemoradiation. Commun. Biol. 2021, 4, 237.
- Takasuna, K.; Hagiwara, T.; Hirohashi, M.; Kato, M.; Nomura, M.; Nagai, E.; Yokoi, T.; Kamataki, T. Involvement of beta-glucuronidase in intestinal microflora in the intestinal toxicity of the antitumor camptothecin derivative irinotecan hydrochloride (CPT-11) in rats. Cancer Res. 1996, 56, 3752–3757.
- Rong, Y.; Dong, Z.; Hong, Z.; Jin, Y.; Zhang, W.; Zhang, B.; Mao, W.; Kong, H.; Wang, C.; Yang, B.; et al. Reactivity toward Bifidobacterium longum and Enterococcus hirae demonstrate robust CD8+ T cell response and better prognosis in HBV-related hepatocellular carcinoma. Exp. Cell Res. 2017, 358, 352–359.
- Atarashi, K.; Tanoue, T.; Oshima, K.; Suda, W.; Nagano, Y.; Nishikawa, H.; Fukuda, S.; Saito, T.; Narushima, S.; Hase, K.; et al. Treg induction by a rationally selected mixture of Clostridia strains from the human microbiota. Nature 2013, 500, 232–236.
- Deleu, S.; Machiels, K.; Raes, J.; Verbeke, K.; Vermeire, S. Short chain fatty acids and its producing organisms: An overlooked therapy for IBD? EBioMedicine 2021, 66, 103293.
- Gomes, S.D.; Oliveira, C.S.; Azevedo-Silva, J.; Casanova, M.R.; Barreto, J.; Pereira, H.; Chaves, S.R.; Rodrigues, L.R.; Casal, M.; Côrte-Real, M.; et al. The Role of Diet Related Short-Chain Fatty Acids in Colorectal Cancer Metabolism and Survival: Prevention and Therapeutic Implications. Curr. Med. Chem. 2020, 27, 4087–4108.
- Luu, M.; Riester, Z.; Baldrich, A.; Reichardt, N.; Yuille, S.; Busetti, A.; Klein, M.; Wempe, A.; Leister, H.; Raifer, H.; et al. Microbial short-chain fatty acids modulate CD8(+) T cell responses and improve adoptive immunotherapy for cancer. Nat. Commun. 2021, 12, 4077.
- Nomura, M.; Nagatomo, R.; Doi, K.; Shimizu, J.; Baba, K.; Saito, T.; Matsumoto, S.; Inoue, K.; Muto, M. Association of Short-Chain Fatty Acids in the Gut Microbiome With Clinical Response to Treatment With Nivolumab or Pembrolizumab in Patients With Solid Cancer Tumors. JAMA Netw. Open 2020, 3, e202895.
- Coutzac, C.; Jouniaux, J.M.; Paci, A.; Schmidt, J.; Mallardo, D.; Seck, A.; Asvatourian, V.; Cassard, L.; Saulnier, P.; Lacroix, L.; et al. Systemic short chain fatty acids limit antitumor effect of CTLA-4 blockade in hosts with cancer. Nat. Commun. 2020, 11, 2168.
- Mirzaei, R.; Afaghi, A.; Babakhani, S.; Sohrabi, M.R.; Hosseini-Fard, S.R.; Babolhavaeji, K.; Khani Ali Akbari, S.; Yousefimashouf, R.; Karampoor, S. Role of microbiota-derived short-chain fatty acids in cancer development and prevention. Biomed. Pharmacother. 2021, 139, 111619.
- Lee, K.A.; Luong, M.K.; Shaw, H.; Nathan, P.; Bataille, V.; Spector, T.D. The gut microbiome: What the oncologist ought to know. Br. J. Cancer 2021, 125, 1197–1209.
- Surana, N.K.; Kasper, D.L. Moving beyond microbiome-wide associations to causal microbe identification. Nature 2017, 552, 244–247.
- Parada Venegas, D.; De la Fuente, M.K.; Landskron, G.; González, M.J.; Quera, R.; Dijkstra, G.; Harmsen, H.J.M.; Faber, K.N.; Hermoso, M.A. Short Chain Fatty Acids (SCFAs)-Mediated Gut Epithelial and Immune Regulation and Its Relevance for Inflammatory Bowel Diseases. Front. Immunol. 2019, 10, 277.
- Cummings, J.H.; Pomare, E.W.; Branch, W.J.; Naylor, C.P.; Macfarlane, G.T. Short chain fatty acids in human large intestine, portal, hepatic and venous blood. Gut 1987, 28, 1221–1227.
- Boets, E.; Gomand, S.V.; Deroover, L.; Preston, T.; Vermeulen, K.; De Preter, V.; Hamer, H.M.; Van den Mooter, G.; De Vuyst, L.; Courtin, C.M.; et al. Systemic availability and metabolism of colonic-derived short-chain fatty acids in healthy subjects: A stable isotope study. J. Physiol. 2017, 595, 541–555.
- Silva, Y.P.; Bernardi, A.; Frozza, R.L. The Role of Short-Chain Fatty Acids From Gut Microbiota in Gut-Brain Communication. Front. Endocrinol. 2020, 11, 25.
- McNeil, N.I.; Cummings, J.H.; James, W.P. Short chain fatty acid absorption by the human large intestine. Gut 1978, 19, 819–822.
- Trend, S.; Leffler, J.; Jones, A.P.; Cha, L.; Gorman, S.; Brown, D.A.; Breit, S.N.; Kermode, A.G.; French, M.A.; Ward, N.C.; et al. Associations of serum short-chain fatty acids with circulating immune cells and serum biomarkers in patients with multiple sclerosis. Sci. Rep. 2021, 11, 5244.
- Morrison, D.J.; Preston, T. Formation of short chain fatty acids by the gut microbiota and their impact on human metabolism. Gut Microbes 2016, 7, 189–200.
- Pryde, S.E.; Duncan, S.H.; Hold, G.L.; Stewart, C.S.; Flint, H.J. The microbiology of butyrate formation in the human colon. FEMS Microbiol. Lett. 2002, 217, 133–139.
- Louis, P.; Flint, H.J. Formation of propionate and butyrate by the human colonic microbiota. Environ. Microbiol. 2017, 19, 29–41.
- den Besten, G.; van Eunen, K.; Groen, A.K.; Venema, K.; Reijngoud, D.-J.; Bakker, B.M. The role of short-chain fatty acids in the interplay between diet, gut microbiota, and host energy metabolism. J. Lipid Res. 2013, 54, 2325–2340.
- Schönfeld, P.; Wojtczak, L. Short- and medium-chain fatty acids in energy metabolism: The cellular perspective. J. Lipid Res. 2016, 57, 943–954.
- Bloemen, J.G.; Venema, K.; van de Poll, M.C.; Olde Damink, S.W.; Buurman, W.A.; Dejong, C.H. Short chain fatty acids exchange across the gut and liver in humans measured at surgery. Clin. Nutr. 2009, 28, 657–661.
- Donohoe, D.R.; Garge, N.; Zhang, X.; Sun, W.; O’Connell, T.M.; Bunger, M.K.; Bultman, S.J. The microbiome and butyrate regulate energy metabolism and autophagy in the mammalian colon. Cell Metab. 2011, 13, 517–526.
- Kelly, C.J.; Zheng, L.; Campbell, E.L.; Saeedi, B.; Scholz, C.C.; Bayless, A.J.; Wilson, K.E.; Glover, L.E.; Kominsky, D.J.; Magnuson, A.; et al. Crosstalk between Microbiota-Derived Short-Chain Fatty Acids and Intestinal Epithelial HIF Augments Tissue Barrier Function. Cell Host Microbe 2015, 17, 662–671.
- Sun, M.; Wu, W.; Liu, Z.; Cong, Y. Microbiota metabolite short chain fatty acids, GPCR, and inflammatory bowel diseases. J. Gastroenterol. 2017, 52, 1–8.
- Le Poul, E.; Loison, C.; Struyf, S.; Springael, J.Y.; Lannoy, V.; Decobecq, M.E.; Brezillon, S.; Dupriez, V.; Vassart, G.; Van Damme, J.; et al. Functional characterization of human receptors for short chain fatty acids and their role in polymorphonuclear cell activation. J. Biol. Chem. 2003, 278, 25481–25489.
- van der Hee, B.; Wells, J.M. Microbial Regulation of Host Physiology by Short-chain Fatty Acids. Trends Microbiol. 2021, 29, 700–712.
- Zimmerman, M.A.; Singh, N.; Martin, P.M.; Thangaraju, M.; Ganapathy, V.; Waller, J.L.; Shi, H.; Robertson, K.D.; Munn, D.H.; Liu, K. Butyrate suppresses colonic inflammation through HDAC1-dependent Fas upregulation and Fas-mediated apoptosis of T cells. Am. J. Physiol.-Gastrointest. Liver Physiol. 2012, 302, G1405–G1415.
- Corrêa-Oliveira, R.; Fachi, J.L.; Vieira, A.; Sato, F.T.; Vinolo, M.A.R. Regulation of immune cell function by short-chain fatty acids. Clin. Transl. Immunol. 2016, 5, e73.
- Furusawa, Y.; Obata, Y.; Fukuda, S.; Endo, T.A.; Nakato, G.; Takahashi, D.; Nakanishi, Y.; Uetake, C.; Kato, K.; Kato, T.; et al. Commensal microbe-derived butyrate induces the differentiation of colonic regulatory T cells. Nature 2013, 504, 446–450.
- Park, J.; Kim, M.; Kang, S.G.; Jannasch, A.H.; Cooper, B.; Patterson, J.; Kim, C.H. Short-chain fatty acids induce both effector and regulatory T cells by suppression of histone deacetylases and regulation of the mTOR–S6K pathway. Mucosal Immunol. 2015, 8, 80–93.
- Bachem, A.; Makhlouf, C.; Binger, K.J.; de Souza, D.P.; Tull, D.; Hochheiser, K.; Whitney, P.G.; Fernandez-Ruiz, D.; Dähling, S.; Kastenmüller, W.; et al. Microbiota-Derived Short-Chain Fatty Acids Promote the Memory Potential of Antigen-Activated CD8(+) T Cells. Immunity 2019, 51, 285–297.e5.
- Moffett, J.R.; Puthillathu, N.; Vengilote, R.; Jaworski, D.M.; Namboodiri, A.M. Acetate Revisited: A Key Biomolecule at the Nexus of Metabolism, Epigenetics, and Oncogenesis–Part 2: Acetate and ACSS2 in Health and Disease. Front. Physiol. 2020, 11, 580171.
- Hosseini, E.; Grootaert, C.; Verstraete, W.; Van de Wiele, T. Propionate as a health-promoting microbial metabolite in the human gut. Nutr. Rev. 2011, 69, 245–258.
More
Information
Subjects:
Oncology
Contributors
MDPI registered users' name will be linked to their SciProfiles pages. To register with us, please refer to https://encyclopedia.pub/register
:
View Times:
1.7K
Revisions:
2 times
(View History)
Update Date:
27 Oct 2022
Notice
You are not a member of the advisory board for this topic. If you want to update advisory board member profile, please contact office@encyclopedia.pub.
OK
Confirm
Only members of the Encyclopedia advisory board for this topic are allowed to note entries. Would you like to become an advisory board member of the Encyclopedia?
Yes
No
${ textCharacter }/${ maxCharacter }
Submit
Cancel
Back
Comments
${ item }
|
More
No more~
There is no comment~
${ textCharacter }/${ maxCharacter }
Submit
Cancel
${ selectedItem.replyTextCharacter }/${ selectedItem.replyMaxCharacter }
Submit
Cancel
Confirm
Are you sure to Delete?
Yes
No





