
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Andra Sabina Neculai-Valeanu | -- | 4715 | 2022-10-25 08:13:17 | | | |
| 2 | Sirius Huang | Meta information modification | 4715 | 2022-10-26 02:38:17 | | |
Video Upload Options
To maximize milk production, efficiency, and profits, modern dairy cows are genetically selected and bred to produce more and more milk and are fed copious quantities of high-energy feed to support ever-increasing milk volumes. As demands for increased milk yield and milking efficiency continue to rise to provide for the growing world population, more significant stress is placed on the dairy cow’s productive capacity. In this climate, which is becoming increasingly hotter, millions of people depend on the capacity of cattle to respond to new environments and to cope with temperature shocks as well as additional stress factors such as solar radiation, animal crowding, insect pests, and poor ventilation, which are often associated with an increased risk of mastitis, resulting in lower milk quality and reduced production.
1. Introduction
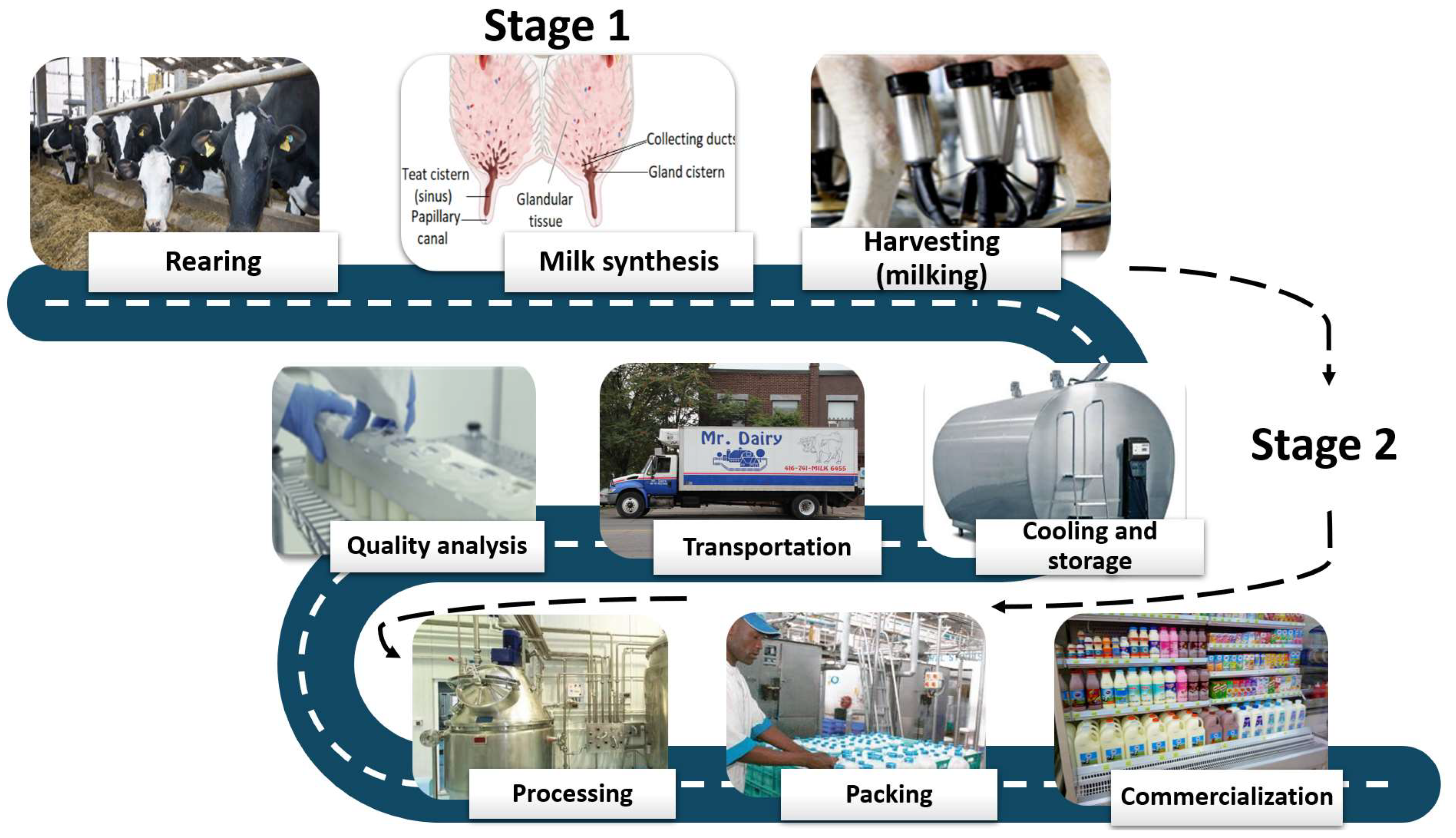
2. Conventional Methods for Monitoring Udder Health
| Method | Type (on Farm/ on Laboratory) |
Principle | Advantages | Drawbacks |
|---|---|---|---|---|
| Teat-end scoring | On-farm | Assessment of teat hyperkeratosis using a four-grade chart: Normal—normal appearance, with no ring around the teat canal; Smooth—slightly visible ring with no keratin strands; Rough—a thickened ring that extends between one and three millimeters from the orifice. Scattered fragments of old keratin and disintegrated epithelial cells are visible; Very Rough—a high ring with scattered fragments of old keratin reaching more than 4 mm is visible. The edge of the ring is uneven and shattered, creating a look similar to that of a flower. |
|
|
| Electric conductivity |
On-farm | Changes in the ionic content of milk caused by tissue injury induced by mastitis are measured. |
|
|
| pH | Lab | The pH in milk samples assessed. Normal milk has a pH of 6.8, while in udder inflammation, the pH tends to become alkaline. |
|
|
| California Mastitis test | On-farm | The number of somatic cells (SC) in milk is estimated using an indirect indicator. The test reagent (Bromocresol-purple in the detergent used as a reagent) forms a gel by reacting with the DNA of the cell. The gel viscosity is linked to the amount of SC in the milk sample. The thicker the gel, the larger the number of cells in the milk sample |
|
|
| Detection of enzymatic activity |
Lab/ On-farm |
Detects variations in color as a means of determining the level of LDH activity. |
|
|
| Direct microscopic determination of somatic cells |
Laboratory | Identifying epithelial and leucocyte cells discharged into the milk by specific staining |
|
|
| Automatic determination of somatic cells (fluoro optoelectronic method) | Laboratory | The nuclear DNA of somatic cells is stained using a fluorescent dye |
|
|
3. Methods Based on the Detection of the Pathogen Agent Causing Mastitis
4. Emergent Methods for Monitoring Udder Health: Infrared Thermography, Biosensors, and Lab-on-Chip Devices
5. Discussions
References
- Azooz, M.F.; El-Wakeel, S.A.; Yousef, H.M. Financial and Economic Analyses of the Impact of Cattle Mastitis on the Profitability of Egyptian Dairy Farms. Vet. World 2020, 13, 1750–1759.
- Neculai-Valeanu, A.S.; Ariton, A.M.; Mădescu, B.M.; Rîmbu, C.M.; Creangă, Ş. Nanomaterials and Essential Oils as Candidates for Developing Novel Treatment Options for Bovine Mastitis. Animals 2021, 11, 1625.
- Ruegg, P.L. A 100-Year Review: Mastitis Detection, Management, and Prevention. J. Dairy Sci. 2017, 100, 10381–10397.
- Chakraborty, S.; Dhama, K.; Tiwari, R.; Mohd, I.Y.; Khurana, S.K.; Khandia, R.; Munjal, A.; Munuswamy, P.; Kumar, M.A.; Singh, M.; et al. Technological Interventions and Advances in the Diagnosis of Intramammary Infections in Animals with Emphasis on Bovine Population—A Review. Vet. Q. 2019, 39, 76–94.
- Manzat, R.M. Strategy, Test and Apparatus for Improving Milk Quality through Detection and Elimination of Mastitis (Strategie, Test Și Aparat, Pentru Creșterea Calității Laptelui, Prin Detectarea Și Eliminarea Mastitelor- Ro Title). Available online: https://www.radumogamanzat.ro/profesional-stiintific/strategie-test-cow-side-si-aparat-pentru-un-lapte-de-inalta-calitate-cu-minimum-de-efort-si-cheltuieli/ (accessed on 22 August 2022).
- Robles, I.; Kelton, D.F.; Barkema, H.W.; Keefe, G.P.; Roy, J.P.; von Keyserlingk, M.A.G.; DeVries, T.J. Bacterial Concentrations in Bedding and Their Association with Dairy Cow Hygiene and Milk Quality. Animal 2020, 14, 1052–1066.
- Alhussien, M.N.; Dang, A.K. Milk Somatic Cells, Factors Influencing Their Release, Future Prospects, and Practical Utility in Dairy Animals: An Overview. Vet. World 2018, 11, 562–577.
- Petzer, I.-M.; Karzis, J.; Donkin, E.F.; Webb, E.C.; Etter, E.M.C. Somatic Cell Count Thresholds in Composite and Quarter Milk Samples as Indicator of Bovine Intramammary Infection Status. Onderstepoort J. Vet. Res. 2017, 84, 1–10.
- Kelly, A.L.; Leitner, G.; Merin, U. Milk Quality and Udder Health: Test Methods and Standards. In Reference Module in Food Science; Elsevier: Amsterdam, The Netherlands, 2018.
- The European Parliament and The Council of the European Union Regulation (EC) No 853/2004 of the European Parliament and of the Council of 29 April 2004 Laying Down Specific Hygiene Rules for on the Hygiene of Foodstuffs. Available online: https://eur-lex.europa.eu/LexUriServ/LexUriServ.do?uri=OJ:L:2004:139:0055:0205:en:PDF (accessed on 11 October 2022).
- The Commission Of The European Communities Commission Regulation (EC) No 1664/2006 of 6 November 2006. Available online: https://eur-lex.europa.eu/legal-content/EN/TXT/PDF/?uri=CELEX:32006R1664&from=en (accessed on 11 October 2022).
- Blum, S.E.; Heller, D.E.; Jacoby, S.; Krifuks, O.; Merin, U.; Silanikove, N.; Lavon, Y.; Edery, N.; Leitner, G. Physiological Response of Mammary Glands to Escherichia Coli Infection: A Conflict between Glucose Need for Milk Production and Immune Response. Sci. Rep. 2020, 10, 9602.
- Halasa, T.; Kirkeby, C. Differential Somatic Cell Count: Value for Udder Health Management. Front. Vet. Sci. 2020, 7, 609055.
- Markusson, H. Total Bacterial Count as an Attribute for Raw Milk Quality; SLU, Department of Molecular Sciences: Uppsala, Sweden, 2021.
- Riveros-Galán, D.S.; Obando-Chaves, M. Mastitis, Somatic Cell Count, and Its Impact on Dairy-Product Quality… An Omission in Colombia? A Review. Rev. Colomb. Cienc. Pecu. 2020, 34, 241–253.
- Bezerra, J.d.S.; Sales, D.C.; de Oliviera, J.P.F.; Silva, Y.M.d.O.; Urbano, S.A.; de Lima, D.M., Jr.; Borba, L.H.F.; Macedo, C.S.; Anaya, K.; Rangel, A.H.d.N. Effect of High Somatic Cell Counts on the Sensory Acceptance and Consumption Intent of Pasteurized Milk and Coalho Cheese. Food Sci. Technol. 2021, 41, 423–431.
- Ivanov, G.Y.; Bilgucu, E.; Dimitrova, M.R.; Ivanova, I.v. Microbiological and Sensory Quality of Farmers Cheese Produced from Milk with Different Somatic Cells Count. IOP Conf. Ser. Mater. Sci. Eng. 2021, 1031, 012104.
- Abebe, R.; Hatiya, H.; Abera, M.; Megersa, B.; Asmare, K. Bovine Mastitis: Prevalence, Risk Factors and Isolation of Staphylococcus Aureus in Dairy Herds at Hawassa Milk Shed, South Ethiopia. BMC Vet. Res. 2016, 12, 270.
- Naing, Y.W.; Wai, S.S.; Lin, T.N.; Thu, W.P.; Htun, L.L.; Bawm, S.; Myaing, T.T. Bacterial Content and Associated Risk Factors Influencing the Quality of Bulk Tank Milk Collected from Dairy Cattle Farms in Mandalay Region. Food Sci. Nutr. 2019, 7, 1063–1071.
- Velázquez-Ordoñez, V.; Valladares-Carranza, B.; Tenorio-Borroto, E.; Talavera-Rojas, M.; Varela-Guerrero, J.A.; Acosta-Dibarrat, J.; Puigvert, F.; Grille, L.; Revello, Á.G.; Pareja, L. Microbial Contamination in Milk Quality and Health Risk of the Consumers of Raw Milk and Dairy Products. In Nutrition in Health and Disease—Our Challenges Now and Forthcoming Time; IntechOpen: London, UK, 2019.
- Berhe, G.; Wasihun, A.G.; Kassaye, E.; Gebreselasie, K. Milk-Borne Bacterial Health Hazards in Milk Produced for Commercial Purpose in Tigray, Northern Ethiopia. BMC Public Health 2020, 20, 894.
- de Klerk, J.N.; Robinson, P.A. Drivers and Hazards of Consumption of Unpasteurised Bovine Milk and Milk Products in High-Income Countries. PeerJ 2022, 10, e13426.
- Ohnstad, I. Teat Condition Scoring as a Management Tool. Livestock 2012, 17, 34–40.
- Zigo, F.; Vasil’, M.; Ondrašovičová, S.; Výrostková, J.; Bujok, J.; Pecka-Kielb, E. Maintaining Optimal Mammary Gland Health and Prevention of Mastitis. Front. Vet. Sci. 2021, 8, 607311.
- Odorčić, M.; Rasmussen, M.D.; Paulrud, C.O.; Bruckmaier, R.M. Review: Milking Machine Settings, Teat Condition and Milking Efficiency in Dairy Cows. Animal 2019, 13, s94–s99.
- Okkema, C.; Grandin, T. Graduate Student Literature Review: Udder Edema in Dairy Cattle—A Possible Emerging Animal Welfare Issue. J. Dairy Sci. 2021, 104, 7334–7341.
- Ohnstad, I. Teat Condition Scoring. Available online: https://www.nadis.org.uk/disease-a-z/cattle/teat-condition-scoring/ (accessed on 10 October 2022).
- Pantoja, J.C.F.; Correia, L.B.N.; Rossi, R.S.; Latosinski, G.S. Association between Teat-End Hyperkeratosis and Mastitis in Dairy Cows: A Systematic Review. J. Dairy Sci. 2020, 103, 1843–1855.
- Dohoo, I.R.; Smith, J.; Andersen, S.; Kelton, D.F.; Godden, S. Diagnosing Intramammary Infections: Evaluation of Definitions Based on a Single Milk Sample. J. Dairy Sci. 2011, 94, 250–261.
- Inzaghi, V.; Zucali, M.; Thompson, P.D.; Penry, J.F.; Reinemann, D.J. Changes in Electrical Conductivity, Milk Production Rate and Milk Flow Rate Prior to Clinical Mastitis Confirmation. Ital. J. Anim. Sci. 2021, 20, 1554–1561.
- Khatun, M.; Thomson, P.C.; García, S.C.; Bruckmaier, R.M. Suitability of Milk Lactate Dehydrogenase and Serum Albumin for Pathogen-Specific Mastitis Detection in Automatic Milking Systems. J. Dairy Sci. 2022, 105, 2558–2571.
- Kandeel, S.A.; Megahed, A.A.; Constable, P.D. Evaluation of Hand-held Sodium, Potassium, Calcium, and Electrical Conductivity Meters for Diagnosing Subclinical Mastitis and Intramammary Infection in Dairy Cattle. J. Vet. Intern. Med. 2019, 33, 2343–2353.
- Tiwari, S.; Mohanty, T.; Patbandha, T.; Kumaresan, A.; Bhakat, M.; Kumar, N.; Baithalu, R. Critical Thresholds of Milk SCC, EC and PH for Detection of Sub-Clinical Mastitis in Crossbred Cows Reared under Subtropical Agroclimatic Condition. Int. J. Livest. Res. 2018, 8, 152.
- Mollenhorst, H.; Rijkaart, L.J.; Hogeveen, H. Mastitis Alert Preferences of Farmers Milking with Automatic Milking Systems. J. Dairy Sci. 2012, 95, 2523–2530.
- Kamphuis, C.; Sherlock, R.; Jago, J.; Mein, G.; Hogeveen, H. Automatic detection of clinical mastitis is improved by in-line monitoring of somatic cell count. J. Dairy Sci. 2008, 91, 4560–4570.
- Hovinen, M.; Aisla, A.-M.; Pyörälä, S. Accuracy and Reliability of Mastitis Detection with Electrical Conductivity and Milk Colour Measurement in Automatic Milking. Acta Agric. Scand. A Anim. Sci. 2006, 56, 121–127.
- Khatun, M.; Thomson, P.C.; Kerrisk, K.L.; Lyons, N.A.; Clark, C.E.F.; Molfino, J.; García, S.C. Development of a New Clinical Mastitis Detection Method for Automatic Milking Systems. J. Dairy Sci. 2018, 101, 9385–9395.
- Galfi, A.; Radinovic, M.; Davidov, I.; Erdeljan, M.; Kovacevic, Z. Detection of Subclinical Mastitis in Dairy Cows Using California and Draminski Mastitis Test. Biotechnol. Anim. Husb. 2017, 33, 465–473.
- Martins, S.A.M.; Martins, V.C.; Cardoso, F.A.; Germano, J.; Rodrigues, M.; Duarte, C.; Bexiga, R.; Cardoso, S.; Freitas, P.P. Biosensors for On-Farm Diagnosis of Mastitis. Front. Bioeng. Biotechnol. 2019, 7, 186.
- Hovinen, M.; Simojoki, H.; Pösö, R.; Suolaniemi, J.; Kalmus, P.; Suojala, L.; Pyörälä, S. N-Acetyl-β-D-Glucosaminidase Activity in Cow Milk as an Indicator of Mastitis. J. Dairy Res. 2016, 83, 219–227.
- Iraguha, B.; Hamudikuwanda, H.; Mushonga, B.; Kandiwa, E.; Mpatswenumugabo, J.P. Comparison of Cow-Side Diagnostic Tests for Subclinical Mastitis of Dairy Cows in Musanze District, Rwanda. J. S. Afr. Vet. Assoc. 2017, 88, 1–6.
- Rowe, S.; Godden, S.; Nydam, D.v.; Gorden, P.; Lago, A.; Vasquez, A.; Royster, E.; Timmerman, J.; Thomas, M. Evaluation of Rapid Culture, a Predictive Algorithm, Esterase Somatic Cell Count and Lactate Dehydrogenase to Detect Intramammary Infection in Quarters of Dairy Cows at Dry-Off. Prev. Vet. Med. 2020, 179, 104982.
- Thomas, F.C.; Geraghty, T.; Simões, P.B.A.; Mshelbwala, F.M.; Haining, H.; Eckersall, P.D. A Pilot Study of Acute Phase Proteins as Indicators of Bovine Mastitis Caused by Different Pathogens. Res. Vet. Sci. 2018, 119, 176–181.
- Akhtar, M.; Guo, S.; Guo, Y.; Zahoor, A.; Shaukat, A.; Chen, Y.; Umar, T.; Deng, P.G.; Guo, M. Upregulated-Gene Expression of pro-Inflammatory Cytokines (TNF-α, IL-1β and IL-6) via TLRs Following NF-ΚB and MAPKs in Bovine Mastitis. Acta Trop. 2020, 207, 105458.
- Dalanezi, F.M.; Schmidt, E.M.S.; Joaquim, S.F.; Guimarães, F.F.; Guerra, S.T.; Lopes, B.C.; Cerri, R.L.A.; Chadwick, C.; Langoni, H. Concentrations of Acute-Phase Proteins in Milk from Cows with Clinical Mastitis Caused by Different Pathogens. Pathogens 2020, 9, 706.
- El-Deeb, W.; Fayez, M.; Alhumam, N.; Elsohaby, I.; Quadri, S.A.; Mkrtchyan, H. The Effect of Staphylococcal Mastitis Including Resistant Strains on Serum Procalcitonin, Neopterin, Acute Phase Response and Stress Biomarkers in Holstein Dairy Cows. PeerJ 2021, 9, e11511.
- Raj, A.; Kulangara, V.; Vareed, T.P.; Melepat, D.P.; Chattothayil, L.; Chullipparambil, S. Variations in the Levels of Acute-Phase Proteins and Lactoferrin in Serum and Milk during Bovine Subclinical Mastitis. J. Dairy Res. 2021, 88, 321–325.
- Giagu, A.; Penati, M.; Traini, S.; Dore, S.; Addis, M.F. Milk Proteins as Mastitis Markers in Dairy Ruminants—A Systematic Review. Vet. Res. Commun. 2022, 46, 329–351.
- Antanaitis, R.; Juozaitienė, V.; Jonike, V.; Baumgartner, W.; Paulauskas, A. Milk Lactose as a Biomarker of Subclinical Mastitis in Dairy Cows. Animals 2021, 11, 1736.
- Oliver, S.P. How Milk Quality Is Assessed. Available online: https://dairy-cattle.extension.org/how-milk-quality-is-assessed/ (accessed on 25 July 2022).
- Costa, A.; de Marchi, M.; Neglia, G.; Campanile, G.; Penasa, M. Milk Somatic Cell Count-Derived Traits as New Indicators to Monitor Udder Health in Dairy Buffaloes. Ital. J. Anim. Sci. 2021, 20, 548–558.
- Kaskous, S. Physiological Aspects of Milk Somatic Cell Count in Dairy Cattle. Int. J. Livest. Res. 2021, 1, 1–12.
- Rychtarova, J.; Krupova, Z.; Brzakova, M.; Borkova, M.; Elich, O.; Dragounova, H.; Seydlova, R.; Sztankoova, Z. Milk Quality, Somatic Cell Count, and Economics of Dairy Goats Farm in the Czech Republic. In Goat Science—Environment, Health and Economy ; IntechOpen: London, UK, 2021.
- Ferronatto, J.A.; Ferronatto, T.C.; Schneider, M.; Pessoa, L.F.; Blagitz, M.G.; Heinemann, M.B.; Libera, A.M.M.P.d.; Souza, F.N. Diagnosing Mastitis in Early Lactation: Use of Somaticell ®, California Mastitis Test and Somatic Cell Count. Ital. J. Anim. Sci. 2018, 17, 723–729.
- Schukken, Y.H.; Wilson, D.J.; Welcome, F.; Garrison-Tikofsky, L.; Gonzalez, R.N. Monitoring Udder Health and Milk Quality Using Somatic Cell Counts. Vet. Res. 2003, 34, 579–596.
- Tomanić, D.; Božin, B.; Kladar, N.; Stanojević, J.; Čabarkapa, I.; Stilinović, N.; Apić, J.; Božić, D.D.; Kovačević, Z. Environmental Bovine Mastitis Pathogens: Prevalence, Antimicrobial Susceptibility, and Sensitivity to Thymus vulgaris L., Thymus serpyllum L., and Origanum vulgare L. Essent. Oils. Antibiot. 2022, 11, 1077.
- Gomes, F.; Saavedra, M.J.; Henriques, M. Bovine Mastitis Disease/Pathogenicity: Evidence of the Potential Role of Microbial Biofilms. Pathog. Dis. 2016, 74, ftw006.
- Dufour, S.; Labrie, J.; Jacques, M. The Mastitis Pathogens Culture Collection. Microbiol Resour. Announc. 2019, 8, e00133-19.
- Zaragoza, C.S.; Olivares, R.A.C.; Watty, A.E.D.; de la Peña Moctezuma, A.; Tanaca, L.V. Yeasts Isolation from Bovine Mammary Glands under Different Mastitis Status in the Mexican High Plateu. Rev. Iberoam. Micol. 2011, 28, 79–82.
- Dworecka-Kaszak, B.; Krutkiewicz, A.; Szopa, D.; Kleczkowski, M.; Biegańska, M. High Prevalence of Candida Yeast in Milk Samples from Cows Suffering from Mastitis in Poland. Sci. World J. 2012, 2012, 196347.
- Pachauri, S.; Varshney, P.; Dash, S.; Gupta, M. Involvement of Fungal Species in Bovine Mastitis in and around Mathura, India. Vet. World 2013, 6, 393.
- Gao, J.; Liu, Y.-C.; Wang, Y.; Li, H.; Wang, X.-M.; Wu, Y.; Zhang, D.-R.; Gao, S.; Qi, Z. Impact of Yeast and Lactic Acid Bacteria on Mastitis and Milk Microbiota Composition of Dairy Cows. AMB Express 2020, 10, 22.
- Milanov, D.; Petrović, T.; Polaček, V.; Suvajdžić, L.; Bojkovski, J. Mastitis Associated with Prototheca Zopfii—An Emerging Health and Economic Problem on Dairy Farms. J. Vet. Res. 2016, 60, 373–378.
- Jagielski, T.; Roeske, K.; Bakuła, Z.; Piech, T.; Wlazło, Ł.; Bochniarz, M.; Woch, P.; Krukowski, H. A Survey on the Incidence of Prototheca Mastitis in Dairy Herds in Lublin Province, Poland. J. Dairy Sci. 2019, 102, 619–628.
- Shahid, M.; Cobo, E.R.; Chen, L.; Cavalcante, P.A.; Barkema, H.W.; Gao, J.; Xu, S.; Liu, Y.; Knight, C.G.; Kastelic, J.P.; et al. Prototheca Zopfii Genotype II Induces Mitochondrial Apoptosis in Models of Bovine Mastitis. Sci. Rep. 2020, 10, 698.
- Ashraf, A.; Imran, M. Diagnosis of Bovine Mastitis: From Laboratory to Farm. Trop. Anim. Health Prod. 2018, 50, 1193–1202.
- University of Minnesota. Veterinary Diagnostic Lab Minnesota Easy®Culture SystemUser’s Guide. Available online: https://dairyknow.umn.edu/topics/milk-quality/minnesota-easy-culture-system-user-s-guide/ (accessed on 30 August 2022).
- LabMediaServis s.r.o. ClearMilk Test. Available online: https://www.clearmilk.cz/ (accessed on 30 August 2022).
- Ferreira, J.C.; Gomes, M.S.; Bonsaglia, E.C.R.; Canisso, I.F.; Garrett, E.F.; Stewart, J.L.; Zhou, Z.; Lima, F.S. Comparative Analysis of Four Commercial On-Farm Culture Methods to Identify Bacteria Associated with Clinical Mastitis in Dairy Cattle. PLoS ONE 2018, 13, e0194211.
- Sipka, A.; Wieland, M.; Biscarini, F.; Rossi, R.M.; Roman, N.; Santisteban, C.; Moroni, P.; Nydam, D.V. Short Communication: Comparative Performance of 3 on-Farm Culture Systems for Detection of Mastitis Pathogens Interpreted by Trained and Untrained Observers. J. Dairy Sci. 2021, 104, 4936–4941.
- Keane, O.M.; Budd, K.E.; Flynn, J.; McCoy, F. Increased Detection of Mastitis Pathogens by Real-Time PCR Compared to Bacterial Culture. Vet. Rec. 2013, 173, 268.
- Hiitiö, H.; Riva, R.; Autio, T.; Pohjanvirta, T.; Holopainen, J.; Pyörälä, S.; Pelkonen, S. Performance of a Real-Time PCR Assay in Routine Bovine Mastitis Diagnostics Compared with in-Depth Conventional Culture. J. Dairy Res. 2015, 82, 200–208.
- Ashraf, A.; Imran, M.; Yaqub, T.; Tayyab, M.; Shehzad, W.; Thomson, P.C. A Novel Multiplex PCR Assay for Simultaneous Detection of Nine Clinically Significant Bacterial Pathogens Associated with Bovine Mastitis. Mol. Cell. Probes 2017, 33, 57–64.
- Ding, T.; Suo, Y.; Zhang, Z.; Liu, D.; Ye, X.; Chen, S.; Zhao, Y. A Multiplex RT-PCR Assay for S. Aureus, L. Monocytogenes, and Salmonella spp. Detection in Raw Milk with Pre-Enrichment. Front. Microbiol. 2017, 8, 989.
- Klaas, I.C.; Zadoks, R.N. An Update on Environmental Mastitis: Challenging Perceptions. Transbound Emerg. Dis. 2018, 65, 166–185.
- Chauhan, K.; Aly, S.S.; Lehenbauer, T.W.; Tonooka, K.H.; Glenn, K.; Rossitto, P.; Marco, M.L. Development of a Multiplex QPCR Assay for the Simultaneous Detection of Mycoplasma Bovis, Mycoplasma Species, and Acholeplasma Laidlawii in Milk. PeerJ 2021, 9, e11881.
- Pascual-Garrigos, A.; Maruthamuthu, M.K.; Ault, A.; Davidson, J.L.; Rudakov, G.; Pillai, D.; Koziol, J.; Schoonmaker, J.P.; Johnson, T.; Verma, M.S. On-Farm Colorimetric Detection of Pasteurella Multocida, Mannheimia Haemolytica, and Histophilus Somni in Crude Bovine Nasal Samples. Vet. Res. 2021, 52, 126.
- Khangembam, R.; Tóth, M.; Vass, N.; Várady, M.; Czeglédi, L.; Farkas, R.; Antonopoulos, A. Point of Care Colourimetric and Lateral Flow LAMP Assay for the Detection of Haemonchus Contortus in Ruminant Faecal Samples. Parasite 2021, 28, 82.
- Li, Y.; Fan, P.; Zhou, S.; Zhang, L. Loop-Mediated Isothermal Amplification (LAMP): A Novel Rapid Detection Platform for Pathogens. Microb. Pathog. 2017, 107, 54–61.
- Wong, Y.-P.; Othman, S.; Lau, Y.-L.; Radu, S.; Chee, H.-Y. Loop-Mediated Isothermal Amplification (LAMP): A Versatile Technique for Detection of Micro-Organisms. J. Appl. Microbiol. 2018, 124, 626–643.
- Lakshmi, B.A.; Kim, S. Recent Trends in the Utilization of LAMP for the Diagnosis of Viruses, Bacteria, and Allergens in Food. In Recent Developments in Applied Microbiology and Biochemistry; Elsevier: Amsterdam, The Netherlands, 2021; pp. 291–297.
- Garg, N.; Ahmad, F.J.; Kar, S. Recent Advances in Loop-Mediated Isothermal Amplification (LAMP) for Rapid and Efficient Detection of Pathogens. Curr. Res. Microb. Sci. 2022, 3, 100120.
- Moon, Y.-J.; Lee, S.-Y.; Oh, S.-W. A Review of Isothermal Amplification Methods and Food-Origin Inhibitors against Detecting Food-Borne Pathogens. Foods 2022, 11, 322.
- Bosward, K.L.; House, J.K.; Deveridge, A.; Mathews, K.; Sheehy, P.A. Development of a Loop-Mediated Isothermal Amplification Assay for the Detection of Streptococcus Agalactiae in Bovine Milk. J. Dairy Sci. 2016, 99, 2142–2150.
- Lee, P.L.M. DNA Amplification in the Field: Move over PCR, Here Comes LAMP. Mol. Ecol. Resour. 2017, 17, 138–141.
- Cornelissen, J.B.W.J.; de Greeff, A.; Heuvelink, A.E.; Swarts, M.; Smith, H.E.; van der Wal, F.J. Rapid Detection of Streptococcus Uberis in Raw Milk by Loop-Mediated Isothermal Amplification. J. Dairy Sci. 2016, 99, 4270–4281.
- Sheet, O.H.; Grabowski, N.T.; Klein, G.; Abdulmawjood, A. Development and Validation of a Loop Mediated Isothermal Amplification (LAMP) Assay for the Detection of Staphylococcus Aureus in Bovine Mastitis Milk Samples. Mol. Cell. Probes 2016, 30, 320–325.
- Griffioen, K.; Cornelissen, J.; Heuvelink, A.; Adusei, D.; Mevius, D.; van der Wal, F.J. Development and Evaluation of 4 Loop-Mediated Isothermal Amplification Assays to Detect Mastitis-Causing Bacteria in Bovine Milk Samples. J. Dairy Sci. 2020, 103, 8407–8420.
- Anis, E.; Hawkins, I.K.; Ilha, M.R.S.; Woldemeskel, M.W.; Saliki, J.T.; Wilkes, R.P. Evaluation of Targeted Next-Generation Sequencing for Detection of Bovine Pathogens in Clinical Samples. J. Clin. Microbiol. 2018, 56, e00399-18.
- Miura, A.; Kurumisawa, T.; Kano, R.; Ito, T.; Suzuki, K.; Kamata, H. Next-Generation Sequencing Analysis of Bacterial Flora in Bovine Protothecal Mastitic Milk and Feces. J. Vet. Med. Sci. 2019, 81, 1547–1551.
- Bi, J.; Li, Z.; Lin, X.; Li, F.; Xu, H.; Yu, X.; Liu, L.; Liang, Y.; Xu, Z.; Wang, J.; et al. Etiology of Granulomatous Lobular Mastitis Based on Metagenomic Next-Generation Sequencing. Int. J. Infect. Dis. 2021, 113, 243–250.
- Sharun, K.; Dhama, K.; Tiwari, R.; Gugjoo, M.B.; Mohd, I.Y.; Patel, S.K.; Pathak, M.; Karthik, K.; Khurana, S.K.; Singh, R.; et al. Advances in Therapeutic and Managemental Approaches of Bovine Mastitis: A Comprehensive Review. Vet. Q. 2021, 41, 107–136.
- Duarte, C.M.; Freitas, P.P.; Bexiga, R. Technological Advances in Bovine Mastitis Diagnosis. J. Vet. Diagn. Investig. 2015, 27, 665–672.
- Neethirajan, S.; Tuteja, S.K.; Huang, S.-T.; Kelton, D. Recent Advancement in Biosensors Technology for Animal and Livestock Health Management. Biosens. Bioelectron. 2017, 98, 398–407.
- Paudyal, S.; Melendez, P.; Manriquez, D.; Velasquez-Munoz, A.; Pena, G.; Roman-Muniz, I.N.; Pinedo, P.J. Use of Milk Electrical Conductivity for the Differentiation of Mastitis Causing Pathogens in Holstein Cows. Animal 2020, 14, 588–596.
- Porter, A.V.; Xercavins, A. Biosensors: The Future of Sensors for Dairy Health Management? Available online: https://4d4f.eu/content/biosensors-future-sensors-dairy-health-management (accessed on 27 July 2022).
- Alhadrami, H.A. Biosensors: Classifications, Medical Applications, and Future Prospective. Biotechnol. Appl. Biochem. 2018, 65, 497–508.
- Rotariu, L.; Lagarde, F.; Jaffrezic-Renault, N.; Bala, C. Electrochemical Biosensors for Fast Detection of Food Contaminants—Trends and Perspective. TrAC Trends Anal. Chem. 2016, 79, 80–87.
- Wu, Q.; Zhang, Y.; Yang, Q.; Yuan, N.; Zhang, W. Review of Electrochemical DNA Biosensors for Detecting Food Borne Pathogens. Sensors 2019, 19, 4916.
- Mungroo, N.A.; Neethirajan, S. Optical Biosensors for the Detection of Food Borne Pathogens. In Nanobiosensors for Personalized and Onsite Biomedical Diagnosis; Institution of Engineering and Technology: London, UK, 2016; pp. 181–208.
- Khansili, N.; Rattu, G.; Krishna, P.M. Label-Free Optical Biosensors for Food and Biological Sensor Applications. Sens. Actuators B Chem. 2018, 265, 35–49.
- Zhou, C.; Zou, H.; Li, M.; Sun, C.; Ren, D.; Li, Y. Fiber Optic Surface Plasmon Resonance Sensor for Detection of E. coli O157:H7 Based on Antimicrobial Peptides and AgNPs-RGO. Biosens. Bioelectron. 2018, 117, 347–353.
- Balbinot, S.; Srivastav, A.M.; Vidic, J.; Abdulhalim, I.; Manzano, M. Plasmonic Biosensors for Food Control. Trends Food Sci. Technol. 2021, 111, 128–140.
- Park, J.-H.; Cho, Y.-W.; Kim, T.-H. Recent Advances in Surface Plasmon Resonance Sensors for Sensitive Optical Detection of Pathogens. Biosensors 2022, 12, 180.
- Narita, F.; Wang, Z.; Kurita, H.; Li, Z.; Shi, Y.; Jia, Y.; Soutis, C. A Review of Piezoelectric and Magnetostrictive Biosensor Materials for Detection of COVID-19 and Other Viruses. Adv. Mater. 2021, 33, 2005448.
- Valderrama, W.B.; Dudley, E.G.; Doores, S.; Cutter, C.N. Commercially Available Rapid Methods for Detection of Selected Food-Borne Pathogens. Crit. Rev. Food Sci. Nutr. 2016, 56, 1519–1531.
- Umesha, S.; Manukumar, H.M. Advanced Molecular Diagnostic Techniques for Detection of Food-Borne Pathogens: Current Applications and Future Challenges. Crit. Rev. Food Sci. Nutr. 2018, 58, 84–104.
- Pérez-López, B.; Merkoçi, A. Nanoparticles for the Development of Improved (Bio)Sensing Systems. Anal. Bioanal. Chem. 2011, 399, 1577–1590.
- Duarte, C.M.; Fernandes, A.C.; Cardoso, F.A.; Bexiga, R.; Cardoso, S.F.; Freitas, P.J.P. Magnetic Counter for Group B Streptococci Detection in Milk. IEEE Trans Magn. 2015, 51, 1–4.
- Deb, R.; Pal, P.; Chaudhary, P.; Bhadsavle, S.; Behera, M.; Parmanand; Gautam, D.; Roshan, M.; Vats, A.; Ludri, A.; et al. Development of Gold Nanoparticle-Based Visual Assay for Rapid Detection of Escherichia Coli Specific DNA in Milk of Cows Affected with Mastitis. LWT 2022, 155, 112901.
- Coatrini-Soares, A.; Coatrini-Soares, J.; Neto, M.P.; de Mello, S.S.; Pinto, D.D.S.C.; Carvalho, W.A.; Gilmore, M.S.; Piazzetta, M.H.O.; Gobbi, A.L.; Brandão, H.d.M.; et al. Microfluidic E-Tongue to Diagnose Bovine Mastitis with Milk Samples Using Machine Learning with Decision Tree Models. Chem. Eng. J. 2023, 451, 138523.
- Jones, G.; Bork, O.; Ferguson, S.A.; Bates, A. Comparison of an On-Farm Point-of-Care Diagnostic with Conventional Culture in Analysing Bovine Mastitis Samples. J. Dairy Res. 2019, 86, 222–225.
- Malcata, F.B.; Pepler, P.T.; Zadoks, R.N.; Viora, L. Laboratory-Based Evaluation of a Simplified Point-of-Care Test Intended to Support Treatment Decisions in Non-Severe Bovine Clinical Mastitis. J. Dairy Res. 2021, 88, 170–175.
- Era-Learn. 1st Icrad Joint Cofund Call Project: Channel-Based Biosensors to Support a Precision Agriculture Approach for Improved Bovine Mastitis Management. Available online: https://www.era-learn.eu/network-information/networks/icrad/1st-icrad-call-2019/channel-based-biosensors-to-support-a-precision-agriculture-approach-for-improved-bovine-mastitis-management (accessed on 28 August 2022).
- Ngo Milk Cluster MAVAS—Biosensor Development for the Detection of Mastitis. Available online: https://enrd.ec.europa.eu/projects-practice/mavas-biosensor-development-detection-mastitis_en (accessed on 10 August 2022).
- McManus, C.; Tanure, C.B.; Peripolli, V.; Seixas, L.; Fischer, V.; Gabbi, A.M.; Menegassi, S.R.O.; Stumpf, M.T.; Kolling, G.J.; Dias, E.; et al. Infrared Thermography in Animal Production: An Overview. Comput. Electron. Agric. 2016, 123, 10–16.
- Sathiyabarathi, M.; Jeyakumar, S.; Manimaran, A.; Jayaprakash, G.; Pushpadass, H.A.; Sivaram, M.; Ramesha, K.P.; Das, D.N.; Kataktalware, M.A.; Prakash, M.A.; et al. Infrared Thermography: A Potential Noninvasive Tool to Monitor Udder Health Status in Dairy Cows. Vet. World 2016, 9, 1075–1081.
- Singh, A.K.; Bhakat, C.; Ghosh, M.K.; Dutta, T.K. Technologies Used at Advanced Dairy Farms for Optimizing the Performance of Dairy Animals: A Review. Span. J. Agric. Res. 2021, 19, e05R01.
- Zaninelli, M.; Redaelli, V.; Luzi, F.; Bronzo, V.; Mitchell, M.; Dell’Orto, V.; Bontempo, V.; Cattaneo, D.; Savoini, G. First Evaluation of Infrared Thermography as a Tool for the Monitoring of Udder Health Status in Farms of Dairy Cows. Sensors 2018, 18, 862.
- Sathiyabarathi, M.; Jeyakumar, S.; Manimaran, A.; Pushpadass, H.A.; Sivaram, M.; Ramesha, K.P.; Das, D.N.; Kataktalware, M.A.; Jayaprakash, G.; Patbandha, T.K. Investigation of Body and Udder Skin Surface Temperature Differentials as an Early Indicator of Mastitis in Holstein Friesian Crossbred Cows Using Digital Infrared Thermography Technique. Vet. World 2016, 9, 1386–1391.
- Neculai-Valeanu, A.-S.; Ariton, A.-M. Applications of Infrared Thermography for Bovine Mastitis Prevention and Sustainable Dairy Cattle Production in Romania. In Proceedings of the The 2nd International Electronic Conference on Animals—Global Sustainability and Animals: Welfare, Policies and Technologies, Online, 29 November–13 December 2021; Phillips, J.C., Pezzuolo, A., Eds.; MDPI: Basel, Switzerland.
- Machado, N.A.F.; da Costa, L.B.S.; Barbosa-Filho, J.A.D.; de Oliveira, K.P.L.; de Sampaio, L.C.; Peixoto, M.S.M.; Damasceno, F.A. Using Infrared Thermography to Detect Subclinical Mastitis in Dairy Cows in Compost Barn Systems. J. Biol. 2021, 97, 102881.
- Velasco-Bolaños, J.; Ceballes-Serrano, C.C.; Velásquez-Mejía, D.; Riaño-Rojas, J.C.; Giraldo, C.E.; Carmona, J.U.; Ceballos-Márquez, A. Application of Udder Surface Temperature by Infrared Thermography for Diagnosis of Subclinical Mastitis in Holstein Cows Located in Tropical Highlands. J. Dairy Sci. 2021, 104, 10310–10323.





