
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Farid Menaa | + 1741 word(s) | 1741 | 2020-10-27 07:09:49 | | | |
| 2 | Peter Tang | -383 word(s) | 1358 | 2020-10-28 04:58:34 | | |
Video Upload Options
Spectro-Fluor technology is an unique, innovative as well as a versatile and green spectroscopic method that allows to detect very sensitively, specifically and rapidly C-F bonds in any solutions, gas, materials, organisms (e.g. human/animal cells, bacteria, virus, fungi, body tissues) and molecules (e.g. drugs, polymers, nano and smaller molecules).
1. Introduction
The concept of Spectro-Fluor™ technology is mainly based on the discovery of characteristic C-F fingerprint(s) in the spectral area of 500–900 cm−1 allowing detection, characterization, imaging, monitoring, screening and measurement of fluoro-nanomaterials, fluoro-compounds, fluoro-molecules, fluoroorganic impurities or fluoro-degradation products [1][2][3]. Spectro-Fluor™ aka Carbon-Fluorine Spectroscopy™ (CFS™) is one of the key tools of our in-house technological platform called PLIRFA™ (Pulsed Laser Isochronic Raman and Fluorescence Apparatus™), which provides alternative solutions and applications across the biomedical (e.g., molecular/cell/tissue imaging), life (e.g., unravelling of molecular interactions, structures, conformations), pharmaceutical (e.g., drug design, delivery, tracing, discovery), environmental (e.g., characterization of pesticides, other contaminants), and nano(bio)technological (e.g., characterization of nano-molecules/materials) fields [1][2]. Thereby, Spectro-Fluor™ represents an innovative, disruptive, green, affordable, cost-effective, reliable and flexible analytical tool of high resolution, sensitivity and specificity (Figure 1) [1][2][3]. Interestingly, this technology constitutes a major advance over conventional spectroscopy technologies (e.g., conventional Raman tools) because of its capability to: (i) highly specific and ultrasensitive detect (ppm-ppb level or <10 cells) C-F bonds (–C-F, =CF2, –CF3); (ii) detect and characterize any halo-organic bonds or unlabeled molecules, regardless their physical state (solid, liquid or gas), via glass and polymer containers, quartz vials; (iii) acquire data in real-time (0.1 second per data point from 1000 average pulses); (iv) quantitatively determine any fluorinated substance since the emitted C-F bond(s) signal is directly proportional to the analyte concentration; (v) unravel chemical structures (e.g., molecular length determination, molecular analogs differentiation with as low as one carbon resolution); (vi) preserve the sample integrity (i.e., non-destructive and non-invasive technology) allowing re-using of the sample; (vii) analyze any unprepared sample in a solvent-less/green fashion; (viii) easily provide data interpretation thanks to its interconnection with powerful software and in-house developed databases; (ix) be used with low maintenance/calibration requirements; (x) operate using adequate time-gating and time-delays with a single laser source functioning in three modes: continuous detection, pulsed detection and two photons excitation/time resolved fluorescence; (xi) be conveniently employed anywhere due to new generation of portable/compact devices incorporating fiber optics; (xii) be versatile, flexible, progressive and hyphenated opening possibilities to be coupled/hybridized to diverse technology platforms such as high-throughput screening (HTS) (e.g., arrays, multiplex assays systems), microscopy and other imaging systems (e.g., confocal laser scanning microscopy, atomic force microscopy (AFM), positron emission tomography (PET), magnetic resonance imaging (MRI), computed tomography (CT)), chromatography (e.g., high-performance liquid chromatography (HPLC), mass spectrometry (MS)), sorting instruments (e.g., fluorescence-activated cell sorting (FACS) or magnetic-activated cell sorting (MACS)).
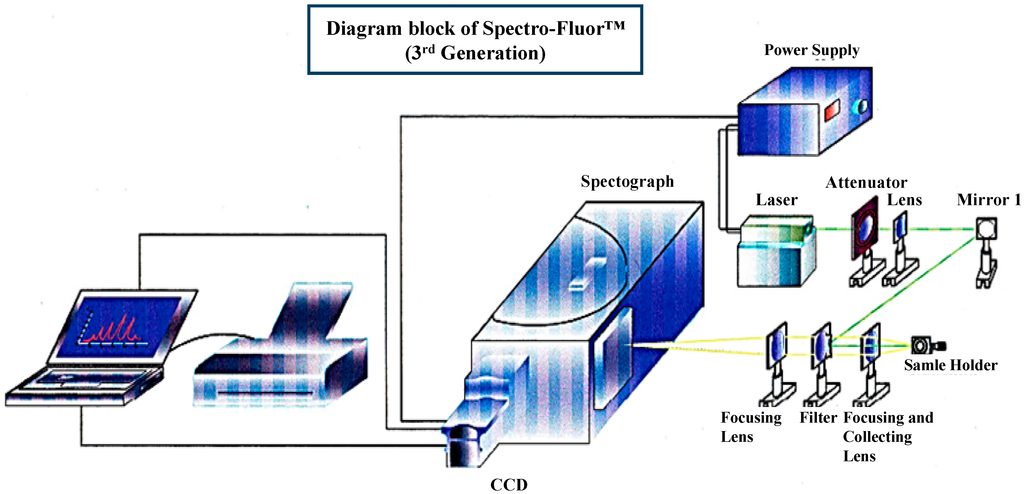
Figure 1. 3D-diagram of the Spectro-Fluor™ technology. This patented apparatus represents the third generation of the device, and includes a laser with power unit, a spectrograph, an ICCD camera with image intensifier, a desktop computer with appropriate software for data analysis. The next generation will represent a compact and portable device with focusing lenses replaced by fiber optics.
Besides, the specific features of fluorine in the formation of fluorocarbons make it attractive in the design of non-viscous but polar organic compounds, with a polarity limited to influencing the intra-molecular nature and inter-molecular interactions with the microenvironment [4][5]. Interestingly, carbon-fluorination displays the additional following properties [6][7]: (i) enhancement of thermal stability (107 Kcal/mol); (ii) increase of lipophilicity, hydrophobicity and solubility; (iii) improvement of the molecular bioavailability (e.g., diminution of the basicity of neighboring amines); and (iv) size comparable to H (i.e., 1.47 Å versus 1.20 Å) capable of mimicking enzyme substrates. Importantly, C-F bonds are [1][2][3][4]: (i) unique in nature; (ii) smaller and more stable than fluorescent dyes due to their covalent interactions; (iii) much less toxic than radioisotopes avoiding handling and logistic problems; (iv) less harmful at long-term exposure than radio-waves; and (v) inexpensively incorporated into molecules, compounds, materials or cells. Therefore, C-F bond(s) can be used in a number of industrial and academic areas including pharmacy, medicine and nanotechnology to characterize them, monitor them or confer some enhanced physicochemical features [1][2][3][4]. In terms of applications, a C-F bond can be employed as [1][2][3][4][8][9][10][11]: (i) a pharmaceutical security label to enhance the drug safety, thereby preventing and banning illegitimate drugs (i.e., counterfeited or sub-standard drugs); (ii) a nano-label to enable drug trace during the whole pharmaceutical development process (i.e., discovery, synthesis and production cycles, clinical and post-approval stages), subsequently bringing the product faster to the market; (iii) an ex vivo, in situ or in vivo label for biologics (e.g., peptides, proteins including antibodies, nucleic acids) and cells/tissues (e.g., stem cells, tumor cells); (iv) a functional chemical group for nanomaterials (e.g., nanoparticles, nanocarbon tubes, nanogels, nanoemulsions, nanoporous silica glasses).
2. Results
We have developed an innovative photonic biotool called Spectro-Fluor™ Technology (Fluorotronics, Inc.) as well as a new in vitro method based on ex vivo incorporation of fluorinated amino acids into cells in order to detect lowly-expressed molecules such as biomarkers of disease. (Figure 1 and Figure 2). Indeed, the advantages of Spectro-Fluor™ Technology (aka Carbon-Fluorine spectroscopy) over routinely used laboratory methods (e.g., Western blots, ELISA, protein dot blots) are represented by the rapid, safe, reliable, highly sensitive and specifique detection of C-F bond(s) formed into molecules [1].
We have established a valuable ex vivo protein labeling protocol that consisted of three steps (Figure 2): (i) addition in excess (e.g., in this study, 10 mg/mL as non-limiting concentration) of a selected aromatic fluoro-aminoacid (e.g., in this study, 4-fluoro-l-phenylalanine) into an adequate medium-containing cells (e.g., in this study, human colon carcinoma HCT-116 cells). In parallel, the corresponding non-modified amino-acid (e.g., in this study, l-phenylalanine) was used as control; (ii) immediately after the amino-acid addition (control) and after a convenient period of time (e.g., in this study, 24 h), simultaneous isolation of both F-p21 and p21 (control) was performed by immune-precipitation (IP). IP is a procedure by which proteins or peptides that react specifically with an antibody are removed from solution and examined for quantity or physical characteristics. The expression of the two p21 protein forms (i.e., F-p21 and p21) was routinely checked by Western blot, a widely used biochemical technique albeit long and expensive (data not shown due to low basal expression detectable with Western blot); (iii) comparative examination of the respective spectra obtained by Spectro-Fluor™ technology obtained in same experimental conditions.
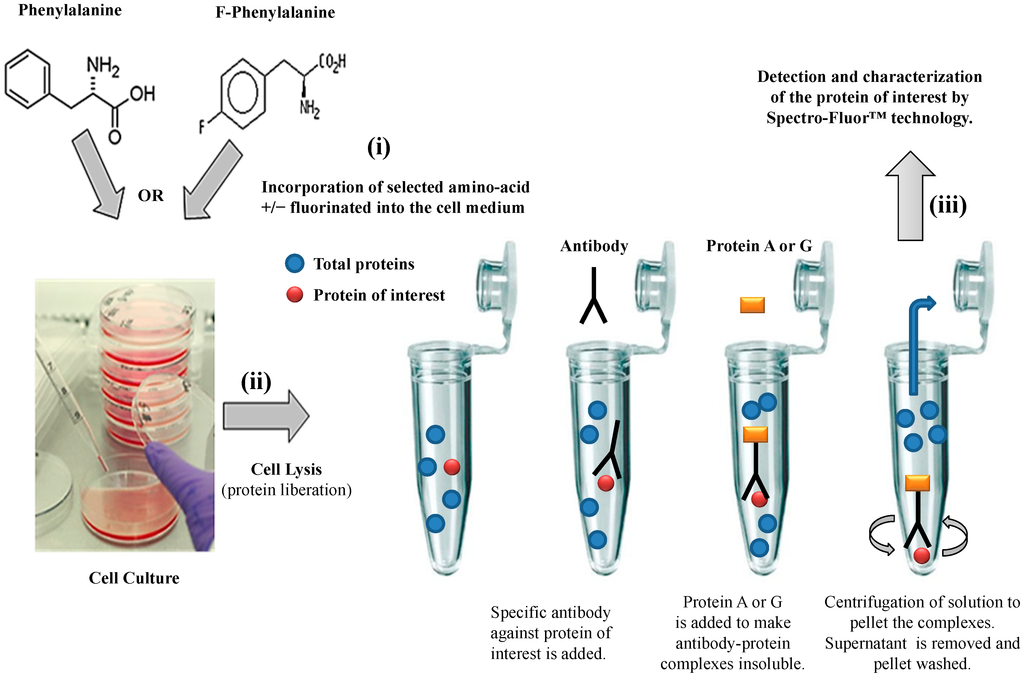
Figure 2. Flow chart of the experimental methodology. The three-step process is resumed by: (i) Ex-vivo protein labeling by incorporation of either 4-fluoro-l-phenylalanine. l-phenylalanine is used as a control; (ii) immune-precipitation by using only one antibody against the antigen/epitope of the protein of interest; (iii) detection and characterization of the protein of interest by Spectro-Fluor™ technology.
3. Conclusions
Fluorinated molecules, widely used in medicinal chemistry (e.g., drug design), emerge in the biomedical field (e.g., fluorinated bio-probes and theranostics™ such as F-nucleic acids, F-peptides) because of their special features (e.g., enhanced bioavailability and stability). Pharmacological studies suggest that fluorine modification may significantly reduce undesirable drug side-effects meantime offering superior efficacy. Therefore, we postulated that fluorinated proteins (e.g., biomarkers of disease, antibodies) should also be beneficial. Within the PLIRFA™ technology platform, which includes progressive analytical devices, we developed a becoming gold-standard Spectro-Fluor™ technology as well as a panel of green methods, enabling rapid, cost-effective, sensitive and specific detection and characterization of F-molecules, F-compounds, F-cells, F- tissues or F-(nano-)biomaterials. In this study, we successively show the possibility to detect a fluorinated protein (i.e., p53-downstream target p21) by Spectro-Fluor™ technology. This strongly suggests that this innovative biotechnology could significantly offer alternative solutions for a wide number of applications (e.g., molecular design, molecular imaging, development of F-antibodies) and technologies routinely used in laboratories and hospitals (e.g., western-blot, enzyme-linked immune sorbent assay, mass-spectrometry, MRI, PET). Eventually, we report that Spectro-Fluor™ technology is able to reduce the troublesome detection associated with low-expressed molecules (e.g., p21).
References
- Menaa, F.; Menaa, B.; Sharts, O. Development of carbon-fluorine spectroscopy for pharmaceutical and biomedical applications. Faraday Discuss. 2011, 149, 269–278; discussion 333–356.
- Menaa, F.; Menaa, B.; Sharts, O.N. Innovative Biotechnology: Carbon-fluorine spectroscopy for a broad range of applications. AAPS J. 2012, 14, 345.
- Menaa, F.; Menaa, B.; Kundu, P.P.; Narayana, C.; Sharts, O.N. Physical characterization of blood substitutes by carbon-fluorine spectroscopy. Pharm. Anal. Acta 2013, 4, 235.
- Menaa, F.; Menaa, B.; Sharts, O.N. Importance of fluorine and fluorocarbons in medicinal chemistry and oncology. J. Mol. Pharm. Org. Process. Res. 2013, 1, 104.
- O'Hagan, D. Organofluorine chemistry: Synthesis and conformation of vicinal fluoromethylene motifs. J. Org. Chem. 2012, 77, 3689–3699.
- Park, B.K.; Kitteringham, N.R.; O’Neill, P.M. Metabolism of fluorine-containing drugs. Annu. Rev. Pharmacol. Toxicol. 2001, 41, 443–470.
- Dalvi, V.H.; Rossky, P.J. Molecular origins of fluorocarbon hydrophobicity. Proc. Natl. Acad. Sci. USA 2010, 107, 13603–13607.
- Song, Z.; Zhang, Q. Design, synthesis, and incorporation of fluorous 5-methylcytosines into oligonucleotides. J. Org. Chem. 2011, 76, 10263–10268.
- Menaa, B.; Montoneri, C.; Menaa, F.; Montoneri, E.; Boffa, V.; Sharts, O. Protein helical structure enhancement in biocompatiblefluoro-phosphonate based nanoporous silica glasses assessed by circular dichroism spectroscopy. Int. J. Nanotechnol. 2011, 8, 471–491.
- O’Hanlon, C.E.; Amede, K.G.; O’Hear, M.R.; Janjic, J.M. NIR-labeled perfluoropolyether nanoemulsions for drug delivery and imaging. J. Fluor. Chem. 2012, 137, 27–33.
- Müller, K.; Faeh, C.; Diederich, F. Fluorine in pharmaceuticals: Looking beyond intuition. Science 2007, 317, 1881–1886.





