
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Sabri El-Saied | -- | 1710 | 2022-09-28 08:17:10 | | | |
| 2 | Catherine Yang | Meta information modification | 1710 | 2022-09-28 10:08:33 | | |
Video Upload Options
Hearing loss represents a significant handicap that gravely impacts the quality of life. Normal hearing function depends on the mechanical and physiological integrity of the middle and inner ear structures and their associated nervous system. The middle ear is composed of the tympanic membrane and the ossicles: malleus, incus and stapes. Accordingly, middle ear pathologies primarily involve the mechanical compromise of the eardrum or its associated ossicles due to, for example, an infection, a fluid accumulation or trauma. Corticosteroid therapy has been mainly effective in stopping, decreasing or correcting an auditory impairment in numerous other etiologies of hearing loss, a steroid therapy has also been encouraged in the field of surgical inner ear interventions.
1. Cochlear Implantation and Glucocorticoid Therapy
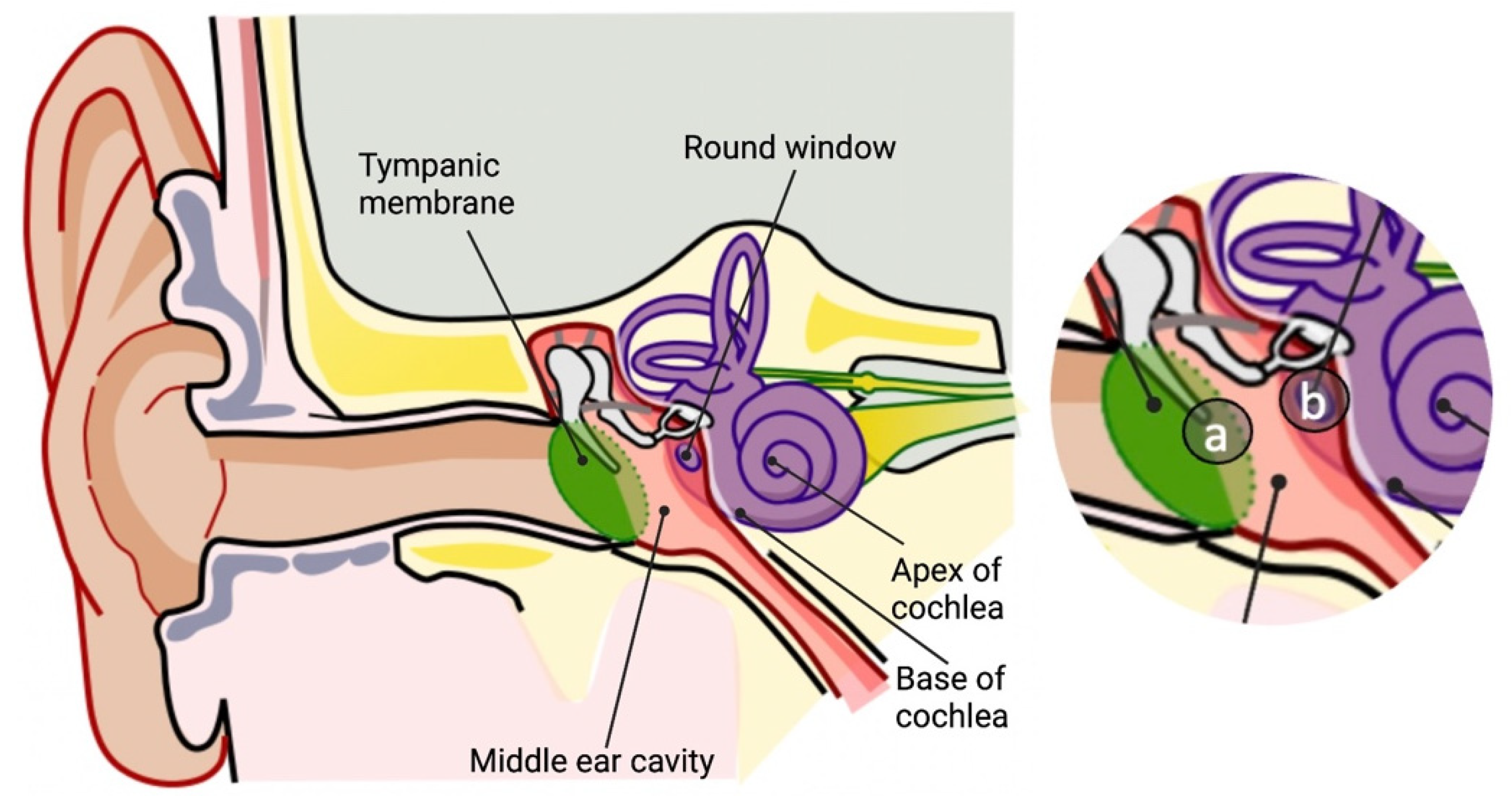
2. Stapedectomy and Glucocorticoid Therapy
3. Perilymphatic Fistula and Glucocorticoid Therapy
4. Post-Surgical Facial Paralysis and Glucocorticoid Therapy
References
- Nadol, J.B.; Eddington, D.K. Histopathology of the inner ear relevant to cochlear implantation. Adv. Otorhinolaryngol. 2006, 64, 31–49.
- Roland, P.S.; Wright, C.G. Surgical aspects of cochlear implantation: Mechanisms of insertional trauma. Adv. Otorhinolaryngol. 2006, 64, 11–30.
- Eshraghi, A.A.; He, J.; Mou, C.H.; Polak, M.; Zine, A.; Bonny, C.; Balkany, T.J.; Van De Water, T.R. D-JNKI-1 treatment prevents the progression of hearing loss in a model of cochlear implantation trauma. Otol. Neurotol. 2006, 27, 504–511.
- Dinh, C.T.; Van De Water, T.R. Blocking pro-cell-death signal pathways to conserve hearing. Audiol. Neurootol. 2009, 14, 383–392.
- Haake, S.M.; Dinh, C.T.; Chen, S.; Eshraghi, A.A.; Van De Water, T.R. Dexamethasone protects auditory hair cells against TNFalpha-initiated apoptosis via activation of PI3K/Akt and NFkappaB signaling. Hear. Res. 2009, 255, 22–32.
- Dinh, C.T.; Bas, E.; Chan, S.S.; Dinh, J.N.; Vu, L.; Van De Water, T.R. Dexamethasone treatment of tumor necrosis factor-alpha challenged organ of Corti explants activates nuclear factor kappa B signaling that induces changes in gene expression that favor hair cell survival. Neuroscience 2011, 188, 157–167.
- Van De Water, T.R.; Abi Hachem, R.N.; Dinh, C.T.; Bas, E.; Haake, S.M.; Hoosien, G.; Vivero, R.; Chan, S.; He, J.; Eshraghi, A.A.; et al. Conservation of hearing and protection of auditory hair cells against trauma-induced losses by local dexamethasone therapy: Molecular and genetic mechanisms. Cochlear Implant. Int. 2010, 11 (Suppl. S1), 42–55.
- Dhanasingh, A.; Hochmair, I. Drug delivery in cochlear implantation. Acta Otolaryngol. 2021, 141, 135–156.
- Causon, A.; Verschuur, C.; Newman, T.A. A retrospective analysis of the contribution of reported factors in cochlear implantation on hearing preservation outcomes. Otol. Neurotol. 2015, 36, 1137–1145.
- Nguyen, S.; Cloutier, F.; Philippon, D.; Côté, M.; Bussières, R.; Backous, D.D. Outcomes review of modern hearing preservation technique in cochlear implant. Auris Nasus Larynx 2016, 43, 485–488.
- Skarżyńska, M.B.; Skarżyński, P.H.; Król, B.; Kozieł, M.; Osińska, K.; Gos, E.; Skarżyński, H. Preservation of hearing following cochlear implantation using different steroid therapy regimens: A prospective clinical study. Med. Sci. Monit. 2018, 24, 2437–2445.
- O’Leary, S.J.; Choi, J.; Brady, K.; Matthews, S.; Ozdowska, K.B.; Payne, M.; McLean, T.; Rousset, A.; Lo, J.; Creber, N.; et al. Systemic methylprednisolone for hearing preservation during cochlear implant surgery: A double blinded placebo-controlled trial. Hear. Res. 2021, 404, 108224.
- Cortés Fuentes, I.A.; Videhult Pierre, P.; Engmér Berglin, C. Improving clinical outcomes in cochlear implantation using glucocorticoid therapy: A review. Ear Hear. 2020, 41, 17–24.
- Rajan, G.P.; Kuthubutheen, J.; Hedne, N.; Krishnaswamy, J. The role of preoperative, intratympanic glucocorticoids for hearing preservation in cochlear implantation: A prospective clinical study. Laryngoscope 2012, 122, 190–195.
- Ramos, B.F.; Tsuji, R.K.; Bento, R.F.; Goffi-Gomez, M.V.; Ramos, H.F.; Samuel, P.A.; Brito, R. Hearing preservation using topical dexamethasone alone and associated with hyaluronic acid in cochlear implantation. Acta Otolaryngol. 2015, 135, 473–477.
- Enticott, J.C.; Eastwood, H.T.; Briggs, R.J.; Dowell, R.C.; O’Leary, S.J. Methylprednisolone applied directly to the round window reduces dizziness after cochlear implantation: A randomized clinical trial. Audiol. Neurootol. 2011, 16, 289–303.
- Paasche, G.; Tasche, C.; Stöver, T.; Lesinski-Schiedat, A.; Lenarz, T. The long-term effects of modified electrode surfaces and intracochlear corticosteroids on postoperative impedances in cochlear implant patients. Otol. Neurotol. 2009, 30, 592–598.
- Prenzler, N.K.; Salcher, R.; Timm, M.; Gaertner, L.; Lenarz, T.; Warnecke, A. Intracochlear administration of steroids with a catheter during human cochlear implantation: A safety and feasibility study. Drug Deliv. Transl. Res. 2018, 8, 1191–1199.
- Sweeney, A.D.; Carlson, M.L.; Zuniga, M.G.; Bennett, M.L.; Wanna, G.B.; Haynes, D.S.; Rivas, A. Impact of perioperative oral steroid use on low-frequency hearing preservation after cochlear implantation. Otol. Neurotol. 2015, 36, 1480–1485.
- Kuthubutheen, J.; Joglekar, S.; Smith, L.; Friesen, L.; Smilsky, K.; Millman, T.; Ng, A.; Shipp, D.; Coates, H.; Arnoldner, C.; et al. The role of preoperative steroids for hearing preservation cochlear implantation: Results of a randomized controlled trial. Audiol. Neurootol. 2017, 22, 292–302.
- Bas, E.; Bohorquez, J.; Goncalves, S.; Perez, E.; Dinh, C.T.; Garnham, C.; Hessler, R.; Eshraghi, A.A.; Van De Water, T.R. Electrode array-eluted dexamethasone protects against electrode insertion trauma induced hearing and hair cell losses, damage to neural elements, increases in impedance and fibrosis: A dose response study. Hear. Res. 2016, 337, 12–24.
- Wilk, M.; Hessler, R.; Mugridge, K.; Jolly, C.; Fehr, M.; Lenarz, T.; Scheper, V. Impedance changes and fibrous tissue growth after cochlear implantation are correlated and can be reduced using a dexamethasone eluting electrode. PLoS ONE 2016, 11, e0147552.
- Wiet, R.J.; Harvey, S.A.; Bauer, G.P. Complications in stapes surgery. Options for prevention and management. Otolaryngol. Clin. N. Am. 1993, 26, 471–490.
- Chavanis, P.-H.; Sire, C. Virial theorem and dynamical evolution of self-gravitating Brownian particles in an unbounded domain. I. Overdamped models. Phys. Rev. E Stat. Nonlin. Soft Matter Phys. 2006, 73, 066103.
- Kaufman, R.S.; Schuknecht, H.F. Reparative granuloma following stapedectomy: A clinical entity. Ann. Otol. Rhinol. Laryngol. 1967, 76, 1008–1017.
- Shambaugh, G.E., Jr.; Causse, J.; Petrovic, A.; Chevance, L.G.; Valvassori, G.E. New concepts in management of otospongiosis. Arch Otolaryngol. 1974, 100, 419–426.
- Causse, J.B. Etiology and therapy of cochlear drops following stapedectomy. Am. J. Otol. 1980, 1, 221–224.
- Causse, J.B.; Causse, J.R. Minimizing cochlear loss during and after stapedectomy. Otolaryngol. Clin. N. Am. 1982, 15, 813–835.
- Dornhoffer, J.L.; Milewski, C. Management of the open labyrinth. Otolaryngol. Head Neck Surg. 1995, 112, 410–414.
- Riechelmann, H.; Tholen, M.; Keck, T.; Rettinger, G. Perioperative glucocorticoid treatment does not influence early post-laser stapedotomy hearing thresholds. Am. J. Otol. 2000, 21, 809–812.
- Deveze, A.; Matsuda, H.; Elziere, M.; Ikezono, T. Diagnosis and treatment of perilymphatic fistula. Adv. Otorhinolaryngol. 2018, 81, 133–145.
- Goto, F.; Ogawa, K.; Kunihiro, T.; Kurashima, K.; Kobayashi, H.; Kanzaki, J. Perilymph fistula—45 case analysis. Auris Nasus Larynx 2001, 28, 29–33.
- Vrabec, J.T. Delayed facial palsy after tympanomastoid surgery. Am. J. Otol. 1999, 20, 26–30.
- Althaus, S.R.; House, H.P. Delayed post-stapedectomy facial paralysis: A report of five cases. Laryngoscope 1973, 83, 1234–1240.





