Your browser does not fully support modern features. Please upgrade for a smoother experience.

Submitted Successfully!
Thank you for your contribution! You can also upload a video entry or images related to this topic.
For video creation, please contact our Academic Video Service.
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Chaiwat Aneklaphakij | -- | 2631 | 2022-09-24 05:48:26 | | | |
| 2 | Beatrix Zheng | + 2 word(s) | 2633 | 2022-09-26 03:56:43 | | |
Video Upload Options
We provide professional Academic Video Service to translate complex research into visually appealing presentations. Would you like to try it?
Cite
If you have any further questions, please contact Encyclopedia Editorial Office.
Aneklaphakij, C.; Chamnanpuen, P.; Bunsupa, S.; Satitpatipan, V. Stilbenoids. Encyclopedia. Available online: https://encyclopedia.pub/entry/27549 (accessed on 23 May 2026).
Aneklaphakij C, Chamnanpuen P, Bunsupa S, Satitpatipan V. Stilbenoids. Encyclopedia. Available at: https://encyclopedia.pub/entry/27549. Accessed May 23, 2026.
Aneklaphakij, Chaiwat, Phatthilakorn Chamnanpuen, Somnuk Bunsupa, Veena Satitpatipan. "Stilbenoids" Encyclopedia, https://encyclopedia.pub/entry/27549 (accessed May 23, 2026).
Aneklaphakij, C., Chamnanpuen, P., Bunsupa, S., & Satitpatipan, V. (2022, September 24). Stilbenoids. In Encyclopedia. https://encyclopedia.pub/entry/27549
Aneklaphakij, Chaiwat, et al. "Stilbenoids." Encyclopedia. Web. 24 September, 2022.
Copy Citation
Stilbenoids are well-known phytoalexins in the group of polyphenolic compounds. Because of their potent bioactivities, including antioxidant, antityrosinase, photoprotective, and antibacterial activities, stilbenoids are utilized as pharmaceutical active ingredient in cosmetic products. Thus, the demand for stilbenoids in the cosmetic industry is increasing.
bioactivity
resveratrol
oxyresveratrol
biosynthesis
polyphenols
1. Introduction
Cosmetic ingredients are generally originated from both chemical synthesis and natural sources [1][2][3][4]. Synthetic and/or semi-synthetic polymers are clear examples of chemical substances employed in cosmetics to prolong the release, improve delivery systems of each specific molecule to the target site of action, and decrease the evaporation rate of volatilizable formulations [1]. In some cases, the interaction between polymers and other compounds in the formulation can lead to hazardous effects on human health [1]. Moreover, some chemicals can act as pollutants that harm the environment, for example, parabens, a well-known preservative, from the factory contaminate the air, dust, soil, and water [5]. Thus, the cosmetic agents from nature such as plants, seaweed (macroalgae), ferns, animals, and marine creatures are preferred, and also attract customers [4][6][7][8][9]. Plants are renowned as enormous sources of pharmaceutical active ingredients since they accumulate diverse metabolites and show numerous biological activities.
Stilbenoids are one of the famous classes of the phyto-polyphenols [10]. The use of phytochemicals in cosmetic products is widespread because of their several prominent bioactivities [11][12]. For cosmetic purposes, several outstanding bioactivities of stilbenoids have been reported and have attracted the interest of customers, including antioxidant and anti-inflammatory activities, antityrosinase activity (depigmentation), antimicrobial activity, and photoprotective effect, i.e., ultraviolet (UV) protection [10][13][14][15]. Remarkably, stilbenoids are conventionally extracted from many plant species by several types of organic solvents such as methanol, ethanol, and acetone [16][17]. Nevertheless, these are not appropriate for the cosmetic industry since the procedures of extraction and purification are multifaceted, requiring high technical skills, the contamination of residual solvent, and are expensive [18]. Furthermore, the yield fluctuates greatly depending on the quality of raw material, geography, temperature, humidity, rainfall, soil type and season [19]. Although the solvent is intentionally removed from the extract, residual solvents may remain in the extract because of incomplete evaporation processes. Thus, the residual solvents should be identified since they may potentially deteriorate human health. In the United States Pharmacopeia, the classification by risk assessment, limitation, identification, quantification, analytical procedures, and control strategy of residual solvents are described under the topic no. 467. The harvest of plant materials, especially perennial trees, directly from the natural resources is an unsustainable approach and also affects the ecosystem. Furthermore, direct cultivation of plants is not appropriate for the cosmetic industry because of its high cost, and because it is time-consuming and laborious work. Hence, other alternative methods for the extraction and sustainable production of stilbenoids are necessary to be considered for utilization in the future of the cosmetic industry.
2. Stilbenoids
Plants naturally produce chemical compounds called “plant-specialized (secondary) metabolites” to survive and protect themselves from abiotic and biotic stresses [20][21]. The phytochemical compounds are generally classified into three main groups based on their chemical structures and biosynthesis, including alkaloids, terpenoids, and polyphenols [20]. Polyphenols are abundantly present in daily diets, in foods such as vegetables, fruits, and nuts, and have been associated with health-promoting benefits [20]. The typical chemical structure of polyphenols consists of more than one hydroxyl group which are bound to one or more aromatic ring systems [22]. The most commonly known polyphenols are phenolic acids, flavonoids, tannins, lignans, coumarins, and stilbenoids [20]. Here, three compounds in stilbenoids containing resveratrol, oxyresveratrol, and piceatannol are emphasized and reviewed because of their outstanding bioactivities and high possibilities for utilization in cosmeceuticals.
2.1. Chemical Structures and Biosynthesis
The core structure of stilbene compounds comprises two aromatic rings connected with an ethylene bridge (C6–C2–C6 backbone) and is commonly found as monomers and oligomers in both aglycone and glycoside forms [20]. Phenylalanine and tyrosine are the amino acid precursors for stilbenoids biosynthesis in the phenylpropanoid pathway, although the chemical reactions occur differently [23]. Phenylalanine ammonia lyase (PAL) converts phenylalanine into trans-cinnamic acid and ammonia as by-products, then, cinnamate-4-hydroxylase (C4H) catalyzed trans-cinnamic acid to produce p-coumaric acid [23]. In addition, p-coumaric acid is also synthesized from tyrosine by tyrosine ammonia lyase (TAL) [23]. Hence, p-coumaric acid synthesis from tyrosine requires one step less than phenylalanine. Next, p-coumaric acid is the substrate for 4-coumarate:coenzyme A (CoA) ligase (4CL) to generate p-coumaroyl-CoA [23]. The p-coumaroyl-CoA is the most important intermediate compound for flavonoids and stilbenoids biosynthesis. In the case of stilbenoids, stilbene synthase (STS) or resveratrol synthase, the key enzyme for stilbenoids synthesis, and three molecules of malonyl-CoA are coupled with p-coumaroyl-CoA to yield resveratrol by the aldol reaction [23].
Resveratrol (3,4′,5-trihydroxystilbene) is the parent compound for other derivatives such as hydroxylated, methylated, and prenylated derivatives. Here, the researchers focused on two hydroxylated derivatives, i.e., oxyresveratrol (2,3’,4,5’-tetrahydroxystilbene) and piceatannol (3,5,3′,4′-tetrahydroxystilbene), since these compounds have several potential bioactivities, and may be possibly used in the cosmetic industry. An overview of the biosynthesis and chemical structures of stilbenoids is shown in Figure 1.
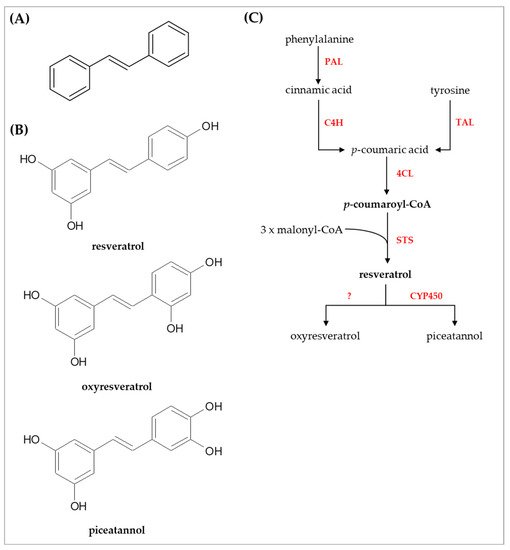
Figure 1. Chemical structures and biosynthesis of stilbenoids. (A) core structure of stilbenoids (B) chemical structures of resveratrol, oxyresveratrol, and piceatannol (C) overview of the biosynthetic pathway of stilbenoids. Abbreviations used: PAL, phenylalanine ammonia lyase; TAL, tyrosine ammonia lyase; C4H, cinnamate-4-hydroxylase; 4CL, 4-coumarate-CoA ligase; STS, stilbene synthase or resveratrol synthase; CYP450, cytochrome P450.
2.2. Plant Sources
To discover current plant sources of resveratrol, oxyresveratrol, and piceatannol, a search was conducted in the well-known phytochemical database “KNApSAcK: A Comprehensive Species-Metabolite Relationship Database”, by chemical names (http://www.knapsackfamily.com/KNApSAcK/ accessed on 5 July 2022 [24]).
It is known that stilbenoids are found ubiquitously in several plant species; however, only some of them are mentioned and discussed in this research based on quantity in the plant extract, known plant species, and potential application as a raw material for stilbenoids production.
Resveratrol was first discovered in the root of V. grandiflorum O. Loes. (white hellebore) and then detected in more than seventy plant species [25][26]. Grapes, mulberries, and peanuts are well-recognized as rich sources of resveratrol. Fruits of grapes (V. vinifera L.), especially a part of the skin and seeds contain 3.66 × 10−2 g/kg of resveratrol [27]. Numerous parts of mulberries (M. alba L.), i.e., root, fruit, aerial part, and leaves, are comprised of resveratrol in the range from 1.6 × 10−3 to 7.95 × 10−3 g/kg [27]. Resveratrol is found in peanut (A. hypogaea L.) stem and fruit from 1.1 × 10−2 to 1.5 × 10−2 g/kg. In addition, resveratrol is also detected in peanut skin and other nut species, such as whole almond seeds and pistachio kernel [20]. Apart from the KNApSAcK database, a list of plant sources containing resveratrol was also compiled by Tian and Liu in 2020 [27].
Oxyresveratrol is believed to be generated by hydroxylation at the C-2 position of resveratrol, although there is no reliable evidence to support this hypothesis [13]. Additionally, the enzyme involved in hydroxylation is still unknown (Figure 1). Mostly, oxyresveratrol is the major compound of plant species in the Moraceae family. The dried aqueous extract (so-called “Puag-Haad”) of heartwood extract of a well-known Thai medicinal plant named “Mahat” (A. lacucha Buch.-Ham.) is comprised of approximately 80% w/w oxyresveratrol [28][29][30]. Another source of oxyresveratrol is mulberries (M. alba L.). Oxyresveratrol is detected in the root, stem, and twig of mulberries [31][32][33]. In 2021, Likhitwitayawuid also well-summarized both gymnosperms and angiosperms which consist of oxyresveratrol [13].
Piceatannol is another hydroxylated derivative of resveratrol. It is formed by cytochrome P450 (CYP450) metabolism by adding a hydroxyl group to the C-3’ position [34]. Piceatannol is also found in grapes, passion fruits, and blueberries. Both red and white grapes contain piceatannol but in different quantities [34]. Piceatannol is approximately nine times more accumulated in red than white grapes (red grapes: 374 ng/g; white grapes 43 ng/g) [34][35]. Passion fruit seeds are very rich in piceatannol, containing 4.8 mg/g [34][36]. The amount of piceatannol in blueberries is reported as 138–422 ng/g at dry concentration [34][37].
2.3. Bioactivities
As mentioned above, stilbenoids show various bioactivities that are beneficial to human health. In cosmeceuticals, five bioactivities, i.e., antioxidant, anti-aging—either as photoprotective or in terms of autophagy—MMP inhibitory, antityrosinase, and antibacterial activities are mainly recognized to produce high-quality cosmetic products. Here, the researchers summarized the bioactivities of resveratrol, oxyresveratrol, and piceatannol as follows.
Recently, stilbenoids have been promoted not only as a potential antioxidant, but also for skin aging protection. Skin aging is a complex physiological and pathological process, including a series of continuous changes, which leads to wrinkles, loss of elasticity, laxity, and rough-textured appearance [38]. It is caused by both intrinsic and extrinsic factors. Exposure to UV radiation is the primary factor of extrinsic skin aging by stimulating the generation and accumulation of reactive oxygen species (ROS), impairing the skin’s antioxidant status, which causes damage to deoxyribonucleic acid (DNA), and proteins that lead to photocarcinogenesis and photoaging [39].
Resveratrol has been described as a potent antioxidant [40]. The capacity of antioxidant activity, including free radical scavenging and metal ion chelation, of resveratrol depends on the position and number of hydroxyl groups in the chemical structure [10]. Based on this activity, resveratrol is reported to protect cells from UV irradiation-induced cell death, and contains photoprotective effects [10][25]. Besides, resveratrol promotes the activity of antioxidant enzymes in the skin, i.e., glutathione S-transferase (GST), and superoxide dismutase (SOD). This activity leads to the reduction of superoxide ion production from UV-A and UV-B irradiation as well as lipid peroxidation activity [14]. Oxyresveratrol is also claimed as a potent antioxidant based on several reported models of antioxidant testing, such as DPPH (1,1-diphenyl-2-picrylhydrazyl radical), superoxide anion, hydroxyl radical, 2,2′-azino-bis-(3-ethylbenzthiazoline-6-sulfonic acid) radical, etc. [13][41]. Combinations of resveratrol and oxyresveratrol result in the synergism of antioxidant activities [42]. Free radical scavenging of piceatannol has been described as it obviously reduces intracellular reactive oxygen species (ROS) levels in human keratinocytes (HaCat) cells irradiated by UV-B. Moreover, oxyresveratrol and piceatannol mostly contain antioxidant activity stronger than that of resveratrol due to an additional hydroxyl group [10][13][43][44]. However, resveratrol exhibits stronger inhibition of peroxy oxygen radical absorbance capacity (ORAC), lipopolysaccharide (LPS)-induced production of nitric oxide (NO) in murine BV-2 microglial cells, and cyclooxygenase 1 and 2 (COX-1, COX-2) than oxyresveratrol [13].
To date, the matrix metalloproteinase (MMP) inhibitory effect of stilbenoids is interesting to investigate for its known anti-aging effects. The MMPs are induced by extrinsic factors such as UV irradiation, inflammation, or toxins. Members of the MMP group show an important role in the degradation of corneocyte desmosomes (collagen and elastin) in the extracellular matrix of skin, such as MMP-1, MMP-2, MMP-3, MMP-7, MMP-9, and MMP-13 [45]. This degradation results in the induction of wrinkle formation [46][47]. Among them, MMP-1 (collagenase) plays a major role in the specific degradation of collagen types I and III during the aging process of the human dermis, suggesting that inhibition of MMP-1 induction reduces UV-induced photoaging [48][49]. Meanwhile some MMPs are known to be involved in the degradation of elastin resulting in skin aging [50]. Resveratrol, oxyresveratrol, and its acetylated derivatives show markedly strong inhibition of UV-B-induced MMP-1 activity and expression in human dermal fibroblast cells, indicating that stilbenoids can prevent the degradation of collagens [51]. Moreover, it has been found that a reduction of UV-B-induced MMP-1 expression was inhibited via mitogen-activated protein kinases (MAPKs) and Akt/mammalian targeting of rapamycin (mTOR) signaling pathways. Chuang et al. demonstrated that autophagy is normally induced by the inhibition of the mTOR signaling pathway, which contributes to preventing the aging process [52]. There are fewer studies on anti-aging in terms of autophagy, although it has been found that resveratrol can enhance autophagy, which may be able to suppress oxidative stress and thus greatly to improve the aging process [53][54].
Resveratrol is able to inhibit melanogenesis causing skin-whitening effects or depigmentation via several mechanisms [14]. Melanogenesis is the process of melanin synthesis that utilizes L-tyrosine as a precursor [13]. Tyrosinase is the crucial enzyme for the conversion of L-tyrosine to L-DOPA (L-3,4-dihydroxyphenylalanine), dopaquinone, followed by cyclization, oxidation, and polymerization, until finally melanin is produced [13]. The metabolite from the biotransformation of resveratrol by tyrosinase inhibits dopa oxidase activity and competes with tyrosine and L-DOPA as a substrate for melanogenesis [10][14][25]. In addition, resveratrol diminishes gene expression of melanogenesis-related proteins such as tyrosinase-related protein (TRYP) 1, TRYP2, and microphthalmia-associated transcription factor (MITF) in melanoma cells [10][14][25]. Oxyresveratrol shows potent tyrosinase inhibitory effects after testing with several methods, as concluded by Likhitwitayawuid [13]. Oxyresveratrol is a prominent compound for skin-whitening since its activity in this respect is obviously higher than that of resveratrol [13]. The anti-tyrosinase activity of piceatannol has also been studied, and its activity has been shown as stronger than that of resveratrol [43][55]. The mechanisms of action of piceatannol are the reduction of ROS and increasing the glutathione/oxidized glutathione ratio [55]. In addition, molecular targets of stilbenoids in skin cell lines are presented in Table 1.
Table 1. Studies of molecular targets of stilbenoids in skin cells.
| Compounds | Biological Activities | Molecular Mechanism | Type of Cell Cultures | References |
|---|---|---|---|---|
| Resveratrol | antioxidative stress | ↑ GST and SOD activities | HaCat | [56] |
| MMP inhibition | ↓phosphorylation of MAPKs and Akt/mTOR signaling pathways | HDF | [51] | |
| antioxidant | ↑ SOD, and GSH-Px activities; ↓ lipid peroxidation | HaCat | [57] | |
| anti-tyrosinase | ↓ melanin pigmentation | B16 F10 melanoma cells | [58] | |
| Oxyresveratrol | suppression of UV-B-induced MMP-1 | ↓phosphorylation of MAPKs and Akt/mTOR signaling pathways | HDF | [51] |
| Piceatannol | antioxidant | ↑ GSH activity; ↓ intracellular ROS level | HaCat | [59] |
Abbreviation: ROS, Reactive Oxygen Species; SOD, superoxide dismutase; GSH-Px, glutathione peroxidase; GSH, Glutathione; GST, Glutathione S-transferase; Akt/mTOR, protein kinase B/mammalian target of rapamycin; MAPKs, mitogen-activated protein kinases; HaCat, human keratinocyte cell; HDF, human dermal fibroblast cells; B16 F10 melanoma cells, murine melanoma cell; ↑, increase; ↓: decrease.
Resveratrol has antimicrobial activity against numerous types of microorganisms [15][26]. One of the most concerning dermatological diseases is acne vulgaris. This disease is caused not only by sebum overproduction, hyperkeratosis of the hair follicles (epidermal hyperproliferation), but also the growth of Cutibacterium acnes (formerly Propionibacterium acnes) [25][60]. Resveratrol contains antibacterial activity against C. acnes and also decreases sebum production. The potency of resveratrol is comparable to conventional anti-C. acnes drug i.e., benzyl peroxide, as well as having none of its cytotoxicity [25][60]. In addition, resveratrol is reported to have a higher inhibitory effect on quorum-sensing of Chromobacterium violaceum, which is the motile gram-negative bacillus, than oxyresveratrol [61][62]. Piceatannol relieves C. acnes-induced HaCaT cell proliferation and migration via its antioxidant and anti-inflammatory activities [63]. Until now, there has been no study on the anti-C. acnes of oxyresveratrol. However, oxyresveratrol plays other roles in antibacterial activity, such as the inhibition of the periodontal pathogenic bacteria, Staphylococcus aureus, and Bacillus subtilis [13]. Oxyresveratrol has been associated with antifungal activity against human pathogenic fungi such as Microsporum gypseum, Microsporum canis, Trichophyton mentagrophytes, and its antistaphylococcal effects (inhibition of S. aureus) are more potent than those of resveratrol [13][64].
At present, resveratrol is extensively utilized as an ingredient in numerous cosmetic formulations, particularly moisturizing cream and serum. In Thailand, the heartwood extract of A. lacucha containing oxyresveratrol as the major active compound is used as an ingredient in moisturizing cream, serum, toner, and soap [29].
2.4. Safety
At present, there are few data on the safety of stilbenoids for use in humans. Resveratrol is non-toxic, safe, and well tolerated for oral and dermal administration since the 50% lethal dose (LD50) is high (reported as 2 g/day), and irritation of the skin and eyes have not been recorded [25]. In a study with mice, resveratrol did not induce carcinogenesis, and reproductive and developmental toxicity [25]. However, a high dose of resveratrol consumption may inhibit systemic P450 and can interact with numerous drugs [65]. Thus, additional studies of simultaneous medication with resveratrol must be investigated. Furthermore, adverse effects from the long-term application of resveratrol in both oral and dermal routes also have to be studied in order to limit usage or provide precautions to consumers. The safety information on oxyresveratrol for human use is still not reported, but has not been found to generate irritation, edema, or erythema when tested in a white guinea pig model [66]. Toxicological studies of piceatannol are very few and lacking in vivo studies. Based on current data, piceatannol does not cause any severe adverse effects; however, further in-depth studies to discover the safety of piceatannol are necessary [67].
References
- Alves, T.F.R.; Morsink, M.; Batain, F.; Chaud, M.V.; Almeida, T.; Fernandes, D.A.; da Silva, C.F.; Souto, E.B.; Severino, P. Applications of Natural, Semi-Synthetic, and Synthetic Polymers in Cosmetic Formulations. Cosmetics 2020, 7, 75.
- He, H.; Li, A.; Li, S.; Tang, J.; Li, L.; Xiong, L. Natural Components in Sunscreens: Topical Formulations with Sun Protection Factor (SPF). Biomed. Pharmacother. 2021, 134, 111161.
- Kostyuk, V.; Potapovich, A.; Albuhaydar, A.R.; Mayer, W.; De Luca, C.; Korkina, L. Natural Substances for Prevention of Skin Photoaging: Screening Systems in the Development of Sunscreen and Rejuvenation Cosmetics. Rejuvenation Res. 2018, 21, 91–101.
- Barbulova, A.; Apone, F.; Colucci, G. Plant Cell Cultures as Source of Cosmetic Active Ingredients. Cosmetics 2014, 1, 94–104.
- Juliano, C.; Magrini, G.A. Cosmetic Ingredients as Emerging Pollutants of Environmental and Health Concern. A Mini-Review. Cosmetics 2017, 4, 11.
- Guillerme, J.-B.; Couteau, C.; Coiffard, L. Applications for Marine Resources in Cosmetics. Cosmetics 2017, 4, 35.
- Morais, T.; Cotas, J.; Pacheco, D.; Pereira, L. Seaweeds Compounds: An Ecosustainable Source of Cosmetic Ingredients? Cosmetics 2021, 8, 8.
- Xi, X.; Li, J.; Guo, S.; Li, Y.; Xu, F.; Zheng, M.; Cao, H.; Cui, X.; Guo, H.; Han, C. The Potential of Using Bee Pollen in Cosmetics: A Review. J. Oleo Sci. 2018, 67, 1071–1082.
- Farràs, A.; Cásedas, G.; Les, F.; Terrado, E.M.; Mitjans, M.; López, V. Evaluation of Anti-Tyrosinase and Antioxidant Properties of Four Fern Species for Potential Cosmetic Applications. Forests 2019, 10, 179.
- Akinwumi, B.C.; Bordun, K.-A.M.; Anderson, H.D. Biological Activities of Stilbenoids. Int. J. Mol. Sci. 2018, 19, 792.
- Zillich, O.V.; Schweiggert-Weisz, U.; Eisner, P.; Kerscher, M. Polyphenols as Active Ingredients for Cosmetic Products. Int. J. Cosmet. Sci. 2015, 37, 455–464.
- de Lima Cherubim, D.J.; Buzanello Martins, C.V.; Oliveira Fariña, L.; da Silva de Lucca, R.A. Polyphenols as Natural Antioxidants in Cosmetics Applications. J. Cosmet. Dermatol. 2020, 19, 33–37.
- Likhitwitayawuid, K. Oxyresveratrol: Sources, Productions, Biological Activities, Pharmacokinetics, and Delivery Systems. Molecules 2021, 26, 4212.
- Nagapan, T.S.; Ghazali, A.R.; Basri, D.F.; Lim, W.N. Photoprotective Effect of Stilbenes and Its Derivatives Against Ultraviolet Radiation-Induced Skin Disorders. Biomed. Pharmacol. J. 2018, 11, 1199–1208.
- Mattio, L.M.; Catinella, G.; Dallavalle, S.; Pinto, A. Stilbenoids: A Natural Arsenal against Bacterial Pathogens. Antibiotics 2020, 9, 336.
- Soural, I.; Vrchotová, N.; Tříska, J.; Balík, J.; Horník, Š.; Cuřínová, P.; Sýkora, J. Various Extraction Methods for Obtaining Stilbenes from Grape Cane of Vitis Vinifera L. Molecules 2015, 20, 6093–6112.
- Kanda, H.; Oishi, K.; Machmudah, S.; Wahyudiono; Goto, M. Ethanol-Free Extraction of Resveratrol and Its Glycoside from Japanese Knotweed Rhizome by Liquefied Dimethyl Ether without Pretreatments. Asia-Pacific J. Chem. Eng. 2021, 16, e2600.
- Feng, C.; Chen, J.; Ye, W.; Liao, K.; Wang, Z.; Song, X.; Qiao, M. Synthetic Biology-Driven Microbial Production of Resveratrol: Advances and Perspectives. Front. Bioeng. Biotechnol. 2022, 10, 833920.
- Karimi, A.; Krähmer, A.; Herwig, N.; Schulz, H.; Hadian, J.; Meiners, T. Variation of Secondary Metabolite Profile of Zataria Multiflora Boiss. Populations Linked to Geographic, Climatic, and Edaphic Factors. Front. Plant Sci. 2020, 11, 969.
- Aneklaphakij, C.; Saigo, T.; Watanabe, M.; Naake, T.; Fernie, A.R.; Bunsupa, S.; Satitpatipan, V.; Tohge, T. Diversity of Chemical Structures and Biosynthesis of Polyphenols in Nut-Bearing Species. Front. Plant Sci. 2021, 12, 440.
- Tohge, T.; Wendenburg, R.; Ishihara, H.; Nakabayashi, R.; Watanabe, M.; Sulpice, R.; Hoefgen, R.; Takayama, H.; Saito, K.; Stitt, M.; et al. Characterization of a Recently Evolved Flavonol-Phenylacyltransferase Gene Provides Signatures of Natural Light Selection in Brassicaceae. Nat. Commun. 2016, 7, 12399.
- Vermerris, W.; Nicholson, R. Phenolic Compound Biochemistry; Springer Dordrecht: Dordrecht, The Netherlands, 2006; ISBN 978-1-4020-5163-0.
- Milke, L.; Aschenbrenner, J.; Marienhagen, J.; Kallscheuer, N. Production of Plant-Derived Polyphenols in Microorganisms: Current State and Perspectives. Appl. Microbiol. Biotechnol. 2018, 102, 1575–1585.
- Afendi, F.M.; Okada, T.; Yamazaki, M.; Hirai-Morita, A.; Nakamura, Y.; Nakamura, K.; Ikeda, S.; Takahashi, H.; Amin, A.U.M.; Darusman, L.K.; et al. KNApSAcK Family Databases: Integrated Metabolite–Plant Species Databases for Multifaceted Plant Research. Plant Cell Physiol. 2012, 53, e1.
- Ratz-Łyko, A.; Arct, J. Resveratrol as an Active Ingredient for Cosmetic and Dermatological Applications: A Review. J. Cosmet. Laser Ther. 2019, 21, 84–90.
- Salehi, B.; Mishra, A.P.; Nigam, M.; Sener, B.; Kilic, M.; Sharifi-Rad, M.; Fokou, P.V.; Martins, N.; Sharifi-Rad, J. Resveratrol: A Double-Edged Sword in Health Benefits. Biomedicines 2018, 6, 91.
- Tian, B.; Liu, J. Resveratrol: A Review of Plant Sources, Synthesis, Stability, Modification and Food Application. J. Sci. Food Agric. 2020, 100, 1392–1404.
- Maneechai, S.; Likhitwitayawuid, K.; Sritularak, B.; Palanuvej, C.; Ruangrungsi, N.; Sirisa-Ard, P. Quantitative Analysis of Oxyresveratrol Content in Artocarpus lakoocha and “Puag-Haad”. Med. Princ. Pract. 2009, 18, 223–227.
- Aneklaphakij, C.; Bunsupa, S.; Sirichamorn, Y.; Bongcheewin, B.; Satitpatipan, V. Taxonomic Notes on the ‘Mahat’ (Artocarpus lacucha and A. thailandicus, Moraceae) Species Complex in Thailand. Plants 2020, 9, 391.
- Wongon, M.; Limpeanchob, N. Inhibitory Effect of Artocarpus Lakoocha Roxb and Oxyresveratrol on α-Glucosidase and Sugar Digestion in Caco-2 Cells. Heliyon 2020, 6, e03458.
- Zhou, J.; Li, S.-X.; Wang, W.; Guo, X.-Y.; Lu, X.-Y.; Yan, X.-P.; Huang, D.; Wei, B.-Y.; Cao, L. Variations in the Levels of Mulberroside A, Oxyresveratrol, and Resveratrol in Mulberries in Different Seasons and during Growth. Sci. World J. 2013, 2013, 380692.
- Inyai, C.; Yusakul, G.; Komaikul, J.; Kitisripanya, T.; Likhitwitayawuid, K.; Sritularak, B.; Putalun, W. Improvement of Stilbene Production by Mulberry Morus alba Root Culture via Precursor Feeding and Co-Elicitation. Bioprocess Biosyst. Eng. 2021, 44, 653–660.
- Lu, H.-P.; Jia, Y.-N.; Peng, Y.-L.; Yu, Y.; Sun, S.-L.; Yue, M.-T.; Pan, M.-H.; Zeng, L.-S.; Xu, L. Oxyresveratrol, a Stilbene Compound from Morus alba L. Twig Extract Active Against Trichophyton Rubrum. Phyther. Res. 2017, 31, 1842–1848.
- Kershaw, J.; Kim, K.-H. The Therapeutic Potential of Piceatannol, a Natural Stilbene, in Metabolic Diseases: A Review. J. Med. Food 2017, 20, 427–438.
- Viñas, P.; Martínez-Castillo, N.; Campillo, N.; Hernández-Córdoba, M. Directly Suspended Droplet Microextraction with in Injection-Port Derivatization Coupled to Gas Chromatography-Mass Spectrometry for the Analysis of Polyphenols in Herbal Infusions, Fruits and Functional Foods. J. Chromatogr. A 2011, 1218, 639–646.
- Matsui, Y.; Sugiyama, K.; Kamei, M.; Takahashi, T.; Suzuki, T.; Katagata, Y.; Ito, T. Extract of Passion Fruit (Passiflora edulis) Seed Containing High Amounts of Piceatannol Inhibits Melanogenesis and Promotes Collagen Synthesis. J. Agric. Food Chem. 2010, 58, 11112–11118.
- Rimando, A.M.; Kalt, W.; Magee, J.B.; Dewey, A.J.; Ballington, J.R. Resveratrol, Pterostilbene, and Piceatannol in Vaccinium Berries. J. Agric. Food Chem. 2004, 52, 4713–4719.
- Kammeyer, A.; Luiten, R.M. Oxidation Events and Skin Aging. Ageing Res. Rev. 2015, 21, 16–29.
- Afaq, F.; Adhami, V.M.; Mukhtar, H. Photochemoprevention of Ultraviolet B Signaling and Photocarcinogenesis. Mutat. Res. Mol. Mech. Mutagen. 2005, 571, 153–173.
- Malhotra, A.; Bath, S.; Elbarbry, F. An Organ System Approach to Explore the Antioxidative, Anti-Inflammatory, and Cytoprotective Actions of Resveratrol. Oxidative Med. Cell. Longev. 2015, 2015, 803971.
- Lorenz, P.; Roychowdhury, S.; Engelmann, M.; Wolf, G.; Horn, T.F.W. Oxyresveratrol and Resveratrol Are Potent Antioxidants and Free Radical Scavengers: Effect on Nitrosative and Oxidative Stress Derived from Microglial Cells. Nitric Oxide 2003, 9, 64–76.
- Aftab, N.; Likhitwitayawuid, K.; Vieira, A. Comparative Antioxidant Activities and Synergism of Resveratrol and Oxyresveratrol. Nat. Prod. Res. 2010, 24, 1726–1733.
- Krambeck, K.; Oliveira, A.; Santos, D.; Pintado, M.M.; Baptista Silva, J.; Sousa Lobo, J.M.; Amaral, M.H. Identification and Quantification of Stilbenes (Piceatannol and Resveratrol) in Passiflora edulis By-Products. Pharmaceuticals 2020, 13, 73.
- Piotrowska, H.; Kucinska, M.; Murias, M. Biological Activity of Piceatannol: Leaving the Shadow of Resveratrol. Mutat. Res. Mutat. Res. 2012, 750, 60–82.
- Huertas, A.C.M.; Schmelzer, C.E.H.; Hoehenwarter, W.; Heyroth, F.; Heinz, A. Molecular-Level Insights into Aging Processes of Skin Elastin. Biochimie 2016, 128–129, 163–173.
- Varani, J.; Warner, R.L.; Gharaee-Kermani, M.; Phan, S.H.; Kang, S.; Chung, J.H.; Wang, Z.Q.; Datta, S.C.; Fisher, G.J.; Voorhees, J.J. Vitamin A Antagonizes Decreased Cell Growth and Elevated Collagen-Degrading Matrix Metalloproteinases and Stimulates Collagen Accumulation in Naturally Aged Human Skin. J. Investig. Dermatol. 2000, 114, 480–486.
- Ralf Paus, L.; Berneburg, M.; Trelles, M.; Friguet, B.; Ogden, S.; Esrefoglu, M.; Kaya, G.; Goldberg, D.J.; Mordon, S.; Calderhead, R.G.; et al. How Best to Halt and/or Revert UV-Induced Skin Ageing: Strategies, Facts and Fiction. Exp. Dermatol. 2008, 17, 228–229.
- Gelse, K.; Pöschl, E.; Aigner, T. Collagens--Structure, Function, and Biosynthesis. Adv. Drug Deliv. Rev. 2003, 55, 1531–1546.
- Brennan, M.; Bhatti, H.; Nerusu, K.C.; Bhagavathula, N.; Kang, S.; Fisher, G.J.; Varani, J.; Voorhees, J.J. Matrix Metalloproteinase-1 Is the Major Collagenolytic Enzyme Responsible for Collagen Damage in UV-Irradiated Human Skin. Photochem. Photobiol. 2003, 78, 43–48.
- Zhang, S.; Duan, E. Fighting against Skin Aging: The Way from Bench to Bedside. Cell Transplant. 2018, 27, 729–738.
- Lee, J.-E.; Oh, J.; Song, D.; Lee, M.; Hahn, D.; Boo, Y.C.; Kang, N.J. Acetylated Resveratrol and Oxyresveratrol Suppress UVB-Induced MMP-1 Expression in Human Dermal Fibroblasts. Antioxidants 2021, 10, 1252.
- Chuang, S.-Y.; Lin, C.-H.; Fang, J.-Y. Natural Compounds and Aging: Between Autophagy and Inflammasome. BioMed Res. Int. 2014, 2014, 297293.
- Yang, S.J.; Lim, Y. Resveratrol Ameliorates Hepatic Metaflammation and Inhibits NLRP3 Inflammasome Activation. Metabolism 2014, 63, 693–701.
- Pietrocola, F.; Mariño, G.; Lissa, D.; Vacchelli, E.; Malik, S.A.; Niso-Santano, M.; Zamzami, N.; Galluzzi, L.; Maiuri, M.C.; Kroemer, G. Pro-Autophagic Polyphenols Reduce the Acetylation of Cytoplasmic Proteins. Cell Cycle 2012, 11, 3851–3860.
- Yokozawa, T.; Kim, Y.J. Piceatannol inhibits melanogenesis by its antioxidative actions. Biol. Pharm. Bull. 2007, 30, 2007–2011.
- Nichols, J.A.; Katiyar, S.K. Skin Photoprotection by Natural Polyphenols: Anti-Inflammatory, Antioxidant and DNA Repair Mechanisms. Arch. Dermatol. Res. 2010, 302, 71–83.
- Liu, Y.; Chan, F.; Sun, H.; Yan, J.; Fan, D.; Zhao, D.; An, J.; Zhou, D. Resveratrol Protects Human Keratinocytes HaCaT Cells from UVA-Induced Oxidative Stress Damage by Downregulating Keap1 Expression. Eur. J. Pharmacol. 2011, 650, 130–137.
- D’Orazio, J.; Jarrett, S.; Amaro-Ortiz, A.; Scott, T. UV Radiation and the Skin. Int. J. Mol. Sci. 2013, 14, 12222–12248.
- Sirerol, J.A.; Feddi, F.; Mena, S.; Rodriguez, M.L.; Sirera, P.; Aupí, M.; Pérez, S.; Asensi, M.; Ortega, A.; Estrela, J.M. Topical Treatment with Pterostilbene, a Natural Phytoalexin, Effectively Protects Hairless Mice against UVB Radiation-Induced Skin Damage and Carcinogenesis. Free Radic. Biol. Med. 2015, 85, 1–11.
- Taylor, E.J.M.; Yu, Y.; Champer, J.; Kim, J. Resveratrol Demonstrates Antimicrobial Effects Against Propionibacterium acnes In Vitro. Dermatol. Ther. 2014, 4, 249–257.
- Sheng, J.-Y.; Chen, T.-T.; Tan, X.-J.; Chen, T.; Jia, A.-Q. The Quorum-Sensing Inhibiting Effects of Stilbenoids and Their Potential Structure-Activity Relationship. Bioorg. Med. Chem. Lett. 2015, 25, 5217–5220.
- Kumar, M.R. Chromobacterium violaceum: A Rare Bacterium Isolated from a Wound over the Scalp. Int. J. Appl. Basic Med. Res. 2012, 2, 70–72.
- Zhu, T.; Fang, F.; Sun, D.; Yang, S.; Zhang, X.; Yu, X.; Yang, L. Piceatannol Inhibits P. acnes-Induced Keratinocyte Proliferation and Migration by Downregulating Oxidative Stress and the Inflammatory Response. Inflammation 2020, 43, 347–357.
- Basset, C.; Rodrigues, A.M.S.; Eparvier, V.; Silva, M.R.R.; Lopes, N.P.; Sabatier, D.; Fonty, E.; Espindola, L.S.; Stien, D. Secondary Metabolites from Spirotropis longifolia (DC) Baill and Their Antifungal Activity against Human Pathogenic Fungi. Phytochemistry 2012, 74, 166–172.
- Shaito, A.; Posadino, A.M.; Younes, N.; Hasan, H.; Halabi, S.; Alhababi, D.; Al-Mohannadi, A.; Abdel-Rahman, W.M.; Eid, A.H.; Nasrallah, G.K.; et al. Potential Adverse Effects of Resveratrol: A Literature Review. Int. J. Mol. Sci. 2020, 21, 2084.
- Park, K.-T.; Kim, J.-K.; Lim, Y.-H. Evaluation on Skin Irritation and Sensitization of Oxyresveratrol and Oxyresveratrol-3-O-Glucoside Produced by Biotransformation of Morus Alba Extract. Korean J. Food Sci. Technol. 2012, 44, 251–256.
- Medrano-Padial, C.; Prieto, A.I.; Puerto, M.; Pichardo, S. Toxicological Evaluation of Piceatannol, Pterostilbene, and ε-Viniferin for Their Potential Use in the Food Industry: A Review. Foods 2021, 10, 592.
More
Information
Subjects:
Plant Sciences
Contributors
MDPI registered users' name will be linked to their SciProfiles pages. To register with us, please refer to https://encyclopedia.pub/register
:
View Times:
1.6K
Revisions:
2 times
(View History)
Update Date:
26 Sep 2022
Notice
You are not a member of the advisory board for this topic. If you want to update advisory board member profile, please contact office@encyclopedia.pub.
OK
Confirm
Only members of the Encyclopedia advisory board for this topic are allowed to note entries. Would you like to become an advisory board member of the Encyclopedia?
Yes
No
${ textCharacter }/${ maxCharacter }
Submit
Cancel
Back
Comments
${ item }
|
More
No more~
There is no comment~
${ textCharacter }/${ maxCharacter }
Submit
Cancel
${ selectedItem.replyTextCharacter }/${ selectedItem.replyMaxCharacter }
Submit
Cancel
Confirm
Are you sure to Delete?
Yes
No





