Closed reduction followed by spica casting is a conservative treatment for developmental dysplasia of the hip (DDH). Magnetic resonance imaging (MRI) can verify proper closed reduction of the dysplastic hip. The abduction angle is the only influencing factor for improved development of the DDH. No other prognostic factors in MRI measurements, such as gender, age at time of the first spica cast, or treatment involving overhead extension were found to be predictive of mid-term outcome. Mild residual dysplasia in the first follow-up X-ray warrants further observation.
1. Introduction
The estimated incidence of developmental dysplasia of the hip (DDH) in infants varies geographically from 0.1–30 per 1000 newborns, depending on the population
[1][2][3]. Unrecognized and untreated DDH can lead to premature osteoarthritis and is responsible for up to one-third of hip replacements in adults younger than 60 years
[1]. The exact etiology of DDH remains unknown and is likely multifactorial, with its pathophysiology reflecting a combination of primary abnormal acetabular development and secondary abnormal interaction between the femoral head and the acetabulum during perinatal life
[4]. Specifically, previous studies have shown that breech intrauterine position, family history of DDH and female gender are the most important risk factors for DDH
[1][5]. Other risk factors such as first-born, oligohydramnios, overly restrictive swaddling practices and foot abnormalities have been linked to DDH, but the evidence is weaker
[2][5].
Detected at an early age by ultrasound, developmental hip dysplasia (DDH) can be very effectively treated by conservative means, even in severe cases. Success rates of 90.4–99.8% have been shown for the Tübinger splint, the Pavlik harness, or casts, and as such are considered first-line therapy in neonates
[6][7][8][9]. These splints or casts exploit the huge potential for growth and remodeling of the newborn hip
[10]. The goal of treatment in these cases is to achieve and maintain concentric reduction of the hip joint to promote femoral head coverage, and congruent acetabular and femoral head development. Treatment is guided by the age at presentation and the severity of the disease
[11]. Later, once the ossification center has formed in the femoral head, the sonographic possibilities become limited and AP X-ray of the pelvis provides superior diagnostic information
[12]. Here, the acetabular index is the relevant parameter up to the age of 4. Beginning at the age of 4, the centre-edge angle (CE angle), according to Wiberg, becomes increasingly relevant
[13].
In German-speaking countries, conservative therapy is usually guided by ultrasound of the hip. In these regions, the Graf technique has prevailed.
The algorithm varies in the case of spica casting. Some prefer leaving window in the cast to perform a transinguinal ultrasound
[14][15][16]. This additional window reduces the stability of the cast, which is why others prefer magnetic resonance imaging (MRI) to evaluate the position of the femoral head after closed reduction and cast application
[17][18][19][20][21]. Further available imaging methods are X-ray or computer tomography (CT) scans
[22][23][24]. Reported rates of recurrent dislocation identified on cross-sectional imaging after closed reduction range from 6% to 15%
[12]. Although the sensitivity and specificity of CT and MRI for detecting dislocation have been found to be equivalent
[25], MRI has the advantage of not exposing the sensitive developing tissues to ionizing radiation and providing superior soft-tissue resolution and enhancement profiles. Specifically, in cases of abnormal post-reduction hips, MRI can identify obstacles to reduce and detect unexpected complications
[12]. Due to the unnecessary radiation exposure and no proper display of the cartilaginous hip,
scholars prefer MRI after closed reduction and spica cast application. Even though the correct centre of the femoral head is the main concern, several indices can be measured in MRI. Jaremko et al. evaluated several of these for dysplasia on infant hip MRI
[26]. Indices of hip dysplasia and adequacy of reduction differ between modalities, including ultrasound, radiography, CT, MRI and arthrography with limited cross-correlation. Jaremko comprehensively adapted all available DDH indices from CT and other modalities to MRI, and tested which could be feasibly measured on MRI, assessed interobserver variability, and correlated indices to each other
[26].
2. Prognostic Factors in Spica Cast MRI
2.1. Sonographic Evaluation
The sonographic alpha- and beta-angle measured at the beginning and end of therapy separated by sex are summarized in Table 1. The difference of both angles (alpha and beta) was highly significant with p < 0.001. No significant correlation was noted between the first alpha-angle and the first radiologic measurements (CE-angle, AI, ACM, HAS). The initial Graf Type had no influence on the duration of the therapy in total, nor on further surgical treatment. However, the duration of spica cast therapy positively influences the alpha angle at the end of therapy (p = 0.003, beta = 0.869). No other measured parameter was shown to have a significant positive correlation. An alpha angle of 64° or more was rated as excellent, 60–63.9 as good, and below as poor.
Table 1. Shows ultrasound parameters at the beginning and end of therapy in total and separated by sex and age in weeks at time of beginning therapy. Alpha = alpha angle defined by Graf. Beta = beta angle defined by Graf.
| |
|
Mean Start Cast |
Standard Deviation |
Mean End Therapy |
Standard Deviation |
| alpha |
In total |
52.2 |
9.8 |
65.6 |
3.3 |
| female |
52.2 |
9.6 |
65.7 |
3.4 |
| male |
51.8 |
11.4 |
64.5 |
1.8 |
| beta |
In total |
74.2 |
9.3 |
63.9 |
6.8 |
| female |
74.0 |
9.9 |
64.0 |
7.0 |
| male |
75.1 |
7.9 |
63.7 |
5.8 |
No significant difference in alpha-angle or Graf Type was noted between the male and female patients. There was a difference in age at time of starting the therapy between both sexes (p = 0.003). Girls were treated on average 3 weeks later than boys. No significant differences were found in duration of the therapy (p = 0.646) or the alpha angle at the end of therapy.
2.2. Magnetic Resonance Imaging Measurements
Table 2 gives a summary of the measurements shown in the MRI. 31/48 (64.6%) patients had a second MRI, which was carried out after receiving the second spica cast. All of the MRI scans showed a concentric reduction of the femoral head within the acetabulum.
Table 2. Summary of the magnetic resonance imaging (MRI) measurements in the first and last MRI. Abd = hip abduction angle. AxAcet = axial anterior acetabular angle. AxPAcet = axial posterior acetabular angle. CorAcet = coronal acetabular angle. PelvWid = pelvic width.
| |
Mean First MRI |
Standard Deviation |
Mean Second MRI |
Standard Deviation |
| Abd |
53.5 (39.3–66.2) |
5.2 |
53.4 (29.0–62.1) |
6.7 |
| AxAcet |
49.6 (32.8–68.2) |
7.1 |
50.0 (32.8–62.4) |
6.6 |
| AxPAcet |
46.8 (36.1–63.5) |
6.0 |
46.8 (4.6–69.1) |
9.3 |
| CorAcet |
25.6 (11.8–44.9) |
6.6 |
24.2 (11.7–47.6) |
7.8 |
| PelvWid |
35.8 (28.7–51.0) |
5.0 |
38.2 (32.0–52.0) |
5.1 |
2.3. Radiographic Evaluation
Two patients had no initial ultrasound examination because of age at time of presentation and an already ossified femoral head (25 and 33 weeks). During further follow-up after Spica cast and subsequent additional Tübinger splint scholars performed anterior-posterior X-rays of the total pelvis after 1.5, 3, 5 and 10 years. Means are shown in Table 3. Moreover, 42/48 patients had at least one follow-up X-ray. Table 3 shows radiographic parameters of AI and CE angle during follow-up separated by the result of the last sonography. Figure 1 shows development of AI and CE-angle over time of follow-up. Factors influencing the outcome of congenital hip dysplasia were found at 3-year follow-up, but not at the others, perhaps due to loss to follow-up after 5 and 10 years. Abduction had a positive effect of the development of AI measured at 3 years age in the cohort (p = 0.044, beta = −0.609). No other positive effects were found to be statistically significant.
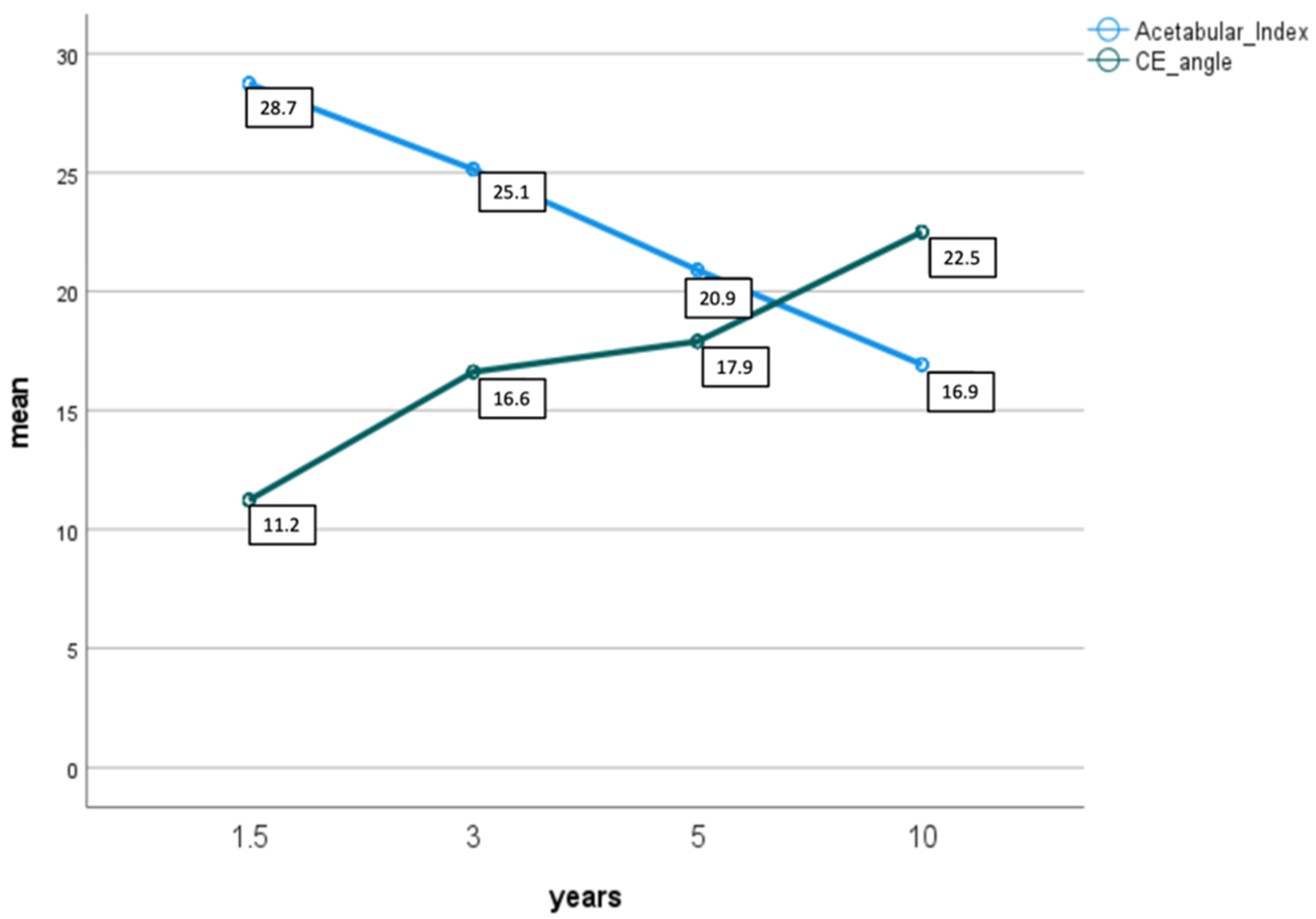 Figure 1.
Figure 1. Shows mean development of acetabular index (light blue) and CE angle (green) during follow-up after 1.5, 3, 5 and 10 years.
Table 3. Shows radiographic measurements with mean and standard deviation of X-ray measurements of AI and CE angle after 1.5, 3, 5 and 10 years of follow-up separated by the result of the last sonography. CE-angle = centre edge angle by Wiberg. AI-angle = acetabular index. n = number.
| Years |
|
Excellent (α ≥ 64°) |
Good (α < 64°) |
Poor (α < 60°) |
| n |
|
n |
|
n |
|
| 1.5–2 |
AI |
65 |
28.6 (s = 4.0) |
10 |
29.5 (s = 4.2) |
1 |
26.0 |
| CE |
11.3 (s = 8.3) |
8.8 (s = 8.8) |
18.0 |
| 3 |
AI |
46 |
25.1 (s = 4.8) |
9 |
24.11 (s = 5.6) |
1 |
22.0 |
| CE |
16.4 (s = 5.6) |
17.8 (s = 5.6) |
21.0 |
| 5 |
AI |
26 |
19.2 (s = 4.3) |
8 |
21.5 (s = 5.2) |
1 |
17.0 |
| CE |
18.4 (s = 4.0) |
18.38 (s = 3.8) |
20.0 |
| 10 |
AI |
7 |
16.3 (s = 3.9) |
3 |
15.33 (s = 5.9) |
0 |
|
| CE |
22.6 (s = 5.4) |
24.33 (s = 4.7) |
|

 Figure 1. Shows mean development of acetabular index (light blue) and CE angle (green) during follow-up after 1.5, 3, 5 and 10 years.
Figure 1. Shows mean development of acetabular index (light blue) and CE angle (green) during follow-up after 1.5, 3, 5 and 10 years.




