
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Chun Him Nathanael LAI | -- | 4711 | 2022-09-21 10:28:38 | | | |
| 2 | Beatrix Zheng | Meta information modification | 4711 | 2022-09-22 05:10:51 | | | | |
| 3 | Beatrix Zheng | Meta information modification | 4711 | 2022-09-22 05:12:16 | | |
Video Upload Options
An ideal medical implant requires optimized properties on both bulk and microscopic scale that can hardly be accomplished by using a single material. Metallic implants such as titanium-based implants possess excellent mechanical properties in general but suffer from corrosion; polymeric implants can be multifunctional and biodegradable, however, difficult to provide some crucial mechanical properties like ductility. With the advance in polymer science and metallurgy, the polymer–metal composite materials serve as an emerging class of healthcare device with optimized bulk and microscopic properties, such polymer–metal composite devices provide good mechanical support, good bio-integration, good hygiene, and minimize bacterial infection and reduced hypersensitivity reactions.
1. Polymer–Metal Composite Materials for Dental Implant
1.1. Polymer–Metal Composite Dental Implant with Improved Load Transfer, Osseointegration, and Osteogenesis
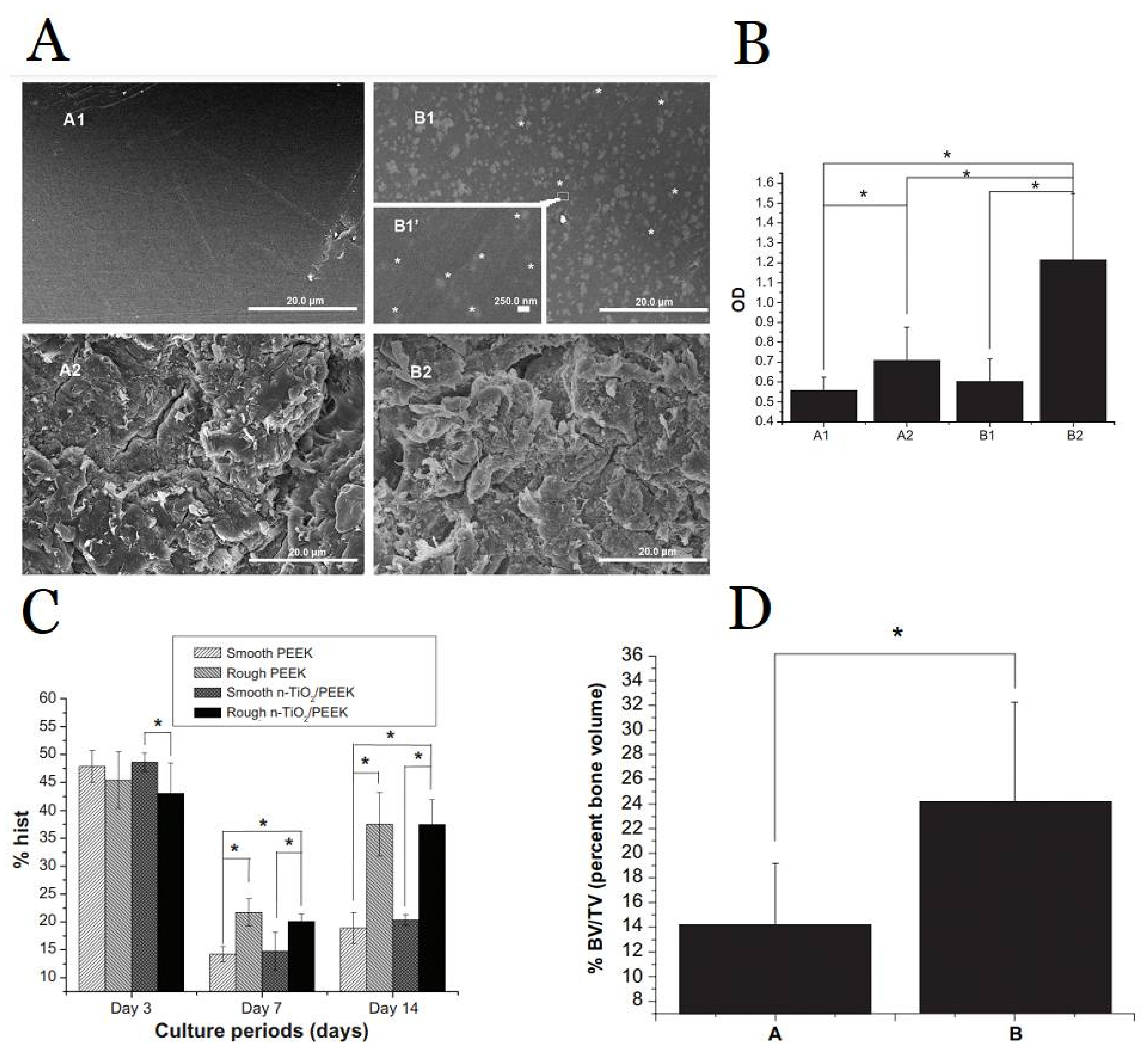
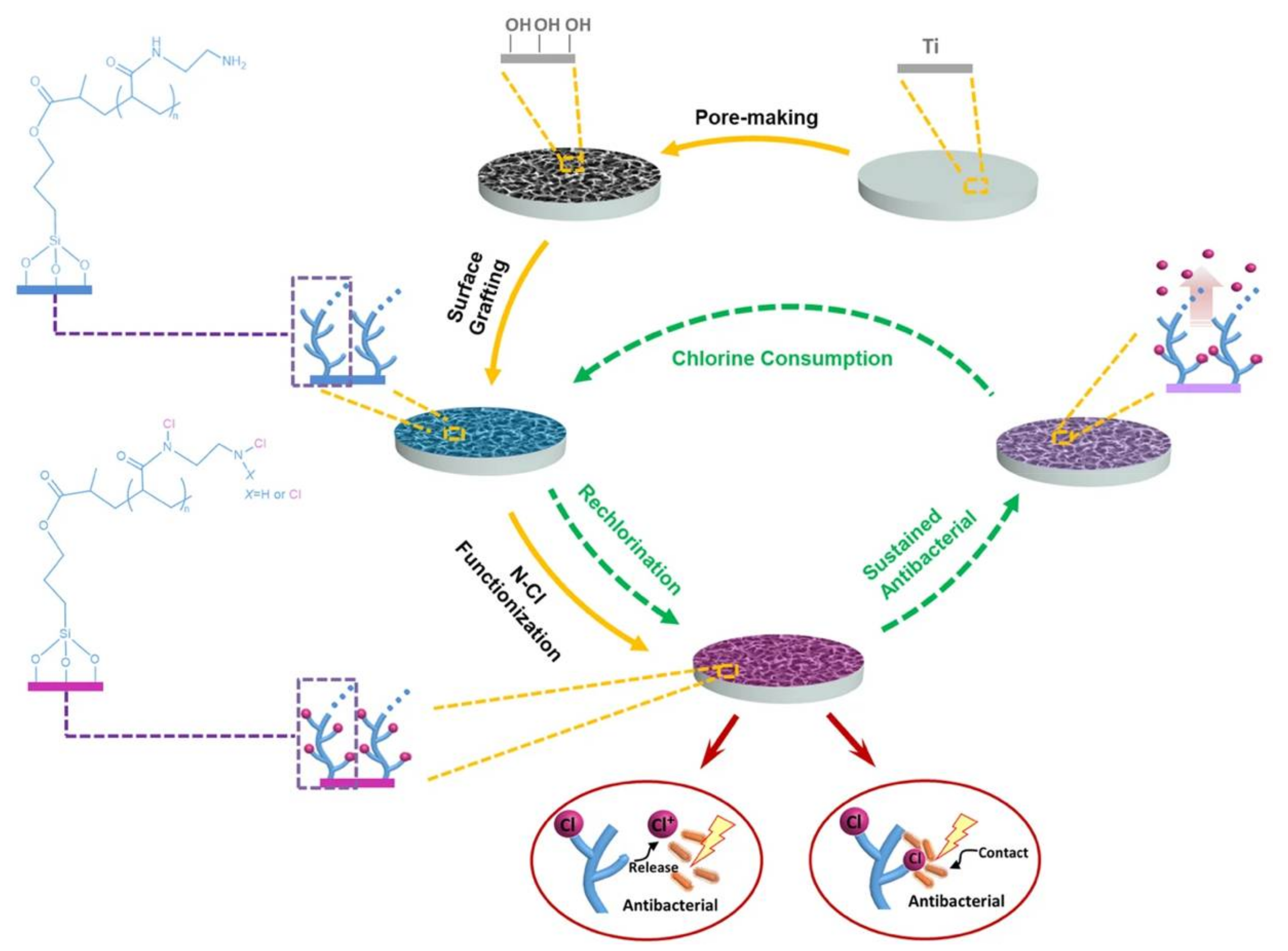
1.2. Antimicrobial Polymer-Metal Composite Dental Implant
1.3. Polymer–Metal Composite Dental Implant with Minimized Immune Reactions
2. Polymer–Metal Composite Materials for Cardiovascular Stent
2.1. Durable Polymer Metal Stent: First-Generation Drug Eluting Stent
2.2. Biodegradable Polymer Metal Stent: Second-Generation Drug Eluting Stent
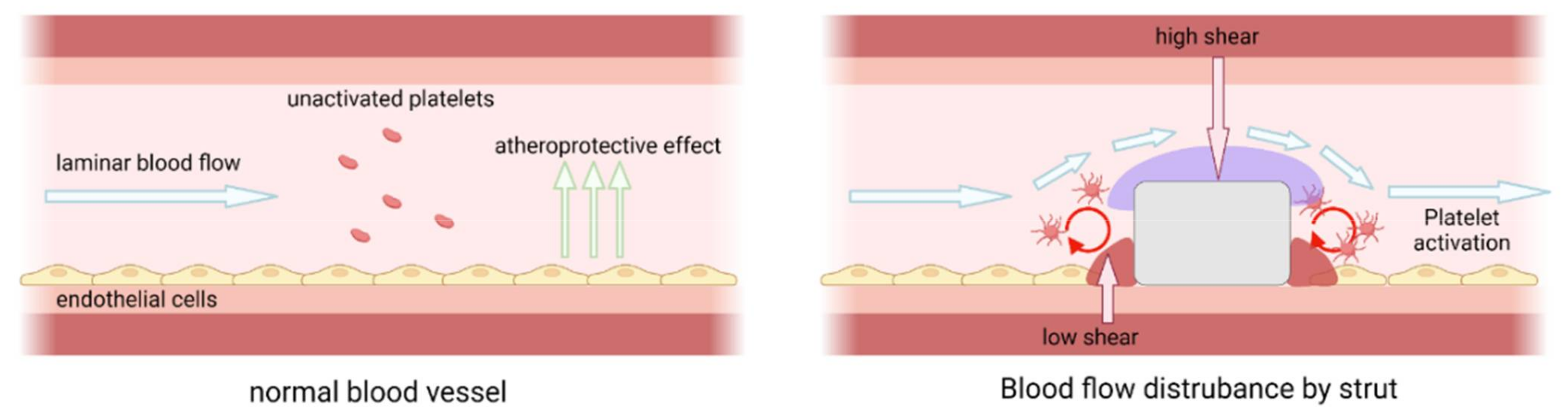
2.3. The Advance of Cardiovascular Stent: Polymer–Metal Stents Engineered with Macroscopic and Microscopic Features
| Parameters | Effects on the Performance of Cardiovascular Stents and Clinical Performance | Refs. |
|---|---|---|
| Stent Geometry | ||
| Distance between strut | Further the distance elicit less blood flow disturbance | [134] |
| Strut width | Narrower the strut elicit less blood flow disturbance | [148] |
| Strut sizing (oversize/undersize) | Slightly oversize (10% than vessel diameter) is beneficial to reducing risk of ST and VLST | [149][150] |
| Properties of metal/alloy | ||
| Metal/Alloy selection | Alloy with improved mechanical properties render thinner struts design possible CoCr, PtCr (81–91 µm) vs. 316 L stainless steel (141 µm) | [153] |
| Diameter/thickness of metal | Thin metal stent (<100 μm) → less blood flow distrubance that reduce the risk of stent malapposition and evaginations Thick metal stent (>100 μm) → generation of circulatory blood flow and promote platelet activation |
[147][152] |
| Properties of polymer | ||
| Selection of Amorphous/Semicrystalline/Crystalline polymer | Enhanced Polymer Crystallinity
|
[111][119][154][155][156] |
| Polymer thickness | Increasing the thickness of the polymer coating
|
[157][158][159] |
| Polymer degradation kinetics | No significant difference in target vessel revascularization (TVR), MI, ST, and VLST between fast (<6 months) and slow (>6 months) polymer degradation | [160] |
2.4. Key Microscopic Features of Polymer—Metal Stent
References
- Jivraj, S.; Chee, W. Rationale for dental implants. Br. Dent. J. 2006, 200, 661–665.
- Gupta, R.; Gupta, N.; Weber, K.K.; Weber, K.K. Dental Implants. In StatPearls; StatPearls Publishing: Treasure Island, FL, USA, 2022.
- Koc, D.; Dogan, A.; Bek, B. Bite force and influential factors on bite force measurements: A literature review. Eur. J. Dent. 2010, 4, 223–232.
- Robinson, D.; Aguilar, L.; Gatti, A.; Abduo, J.; Lee, P.V.S.; Ackland, D. Load response of the natural tooth and dental implant: A comparative biomechanics study. J. Adv. Prosthodont. 2019, 11, 169–178.
- Abe, I.; Milezewski, M.S.; Souza, M.A.; Kalinowski, H.J.; Machuca, O.F.; Marin, G.C.; Camargo, E.S. The force magnitude of a human bite measured at the molar intercuspidation using fiber Bragg gratings. J. Microw. Optoelectron. Electromagn. Appl. 2017, 16, 434–444.
- Wang, G.; Wan, Y.; Wang, T.; Liu, Z. Corrosion Behavior of Titanium Implant with different Surface Morphologies. Procedia Manuf. 2017, 10, 363–370.
- Souza, J.C.M.; Apaza-Bedoya, K.; Benfatti, C.A.M.; Silva, F.S.; Henriques, B. A Comprehensive Review on the Corrosion Pathways of Titanium Dental Implants and Their Biological Adverse Effects. Metals 2020, 10, 1272.
- Liu, X.; Chu, P.K.; Ding, C. Surface modification of titanium, titanium alloys, and related materials for biomedical applications. Mater. Sci. Eng. R Rep. 2004, 47, 49–121.
- Insua, A.; Monje, A.; Wang, H.L.; Miron, R.J. Basis of bone metabolism around dental implants during osseointegration and peri-implant bone loss. J. Biomed. Mater. Res. Part A 2017, 105, 2075–2089.
- Sumner, D.R.; Turner, T.M.; Igloria, R.; Urban, R.M.; Galante, J.O. Functional adaptation and ingrowth of bone vary as a function of hip implant stiffness. J. Biomech. 1998, 31, 909–917.
- Brizuela, A.; Herrero-Climent, M.; Rios-Carrasco, E.; Rios-Santos, J.V.; Pérez, R.A.; Manero, J.M.; Gil Mur, J. Influence of the Elastic Modulus on the Osseointegration of Dental Implants. Materials 2019, 12, 980.
- Knaus, J.; Schaffarczyk, D.; Cölfen, H. On the Future Design of Bio-Inspired Polyetheretherketone Dental Implants. Macromol. Biosci. 2020, 20, 1900239.
- Isidor, F. Loss of osseointegration caused by occlusal load of oral implants. A clinical and radiographic study in monkeys. Clin. Oral Implant. Res. 1996, 7, 143–152.
- Flanagan, D. Bite force and dental implant treatment: A short review. Med. Devices (Auckl) 2017, 10, 141–148.
- Anderson, J.M.; Rodriguez, A.; Chang, D.T. Foreign body reaction to biomaterials. Semin. Immunol. 2008, 20, 86–100.
- Albrektsson, T.A.-O.; Becker, W.A.-O.; Coli, P.; Jemt, T.A.-O.; Mölne, J.; Sennerby, L. Bone loss around oral and orthopedic implants: An immunologically based condition. Clin. Implant. Dent. Relat. Res. 2019, 21, 786–795.
- Albrektsson, T.A.-O.; Jemt, T.; Mölne, J.; Tengvall, P.; Wennerberg, A.A.-O. On inflammation-immunological balance theory-A critical apprehension of disease concepts around implants: Mucositis and marginal bone loss may represent normal conditions and not necessarily a state of disease. Clin. Implant. Dent. Relat. Res. 2019, 21, 183–189.
- Trindade, R.; Albrektsson, T.; Tengvall, P.; Wennerberg, A. Foreign Body Reaction to Biomaterials: On Mechanisms for Buildup and Breakdown of Osseointegration. Clin. Implant. Dent. Relat. Res. 2016, 18, 192–203.
- Trindade, R.; Albrektsson, T.; Galli, S.; Prgomet, Z.; Tengvall, P.; Wennerberg, A. Osseointegration and foreign body reaction: Titanium implants activate the immune system and suppress bone resorption during the first 4 weeks after implantation. Clin. Implant. Dent. Relat. Res. 2018, 20, 82–91.
- Hanke, M.L.; Angle, A.; Kielian, T.; Kielian, T. MyD88-dependent signaling influences fibrosis and alternative macrophage activation during Staphylococcus aureus biofilm infection. PLoS ONE 2012, 7, e42476.
- Trindade, R.A.-O.; Albrektsson, T.; Galli, S.A.-O.; Prgomet, Z.; Tengvall, P.; Wennerberg, A. Bone Immune Response to Materials, Part I: Titanium, PEEK and Copper in Comparison to Sham at 10 Days in Rabbit Tibia. J. Clin. Med. 2018, 7, 526.
- Schwarz, F.; Derks, J.; Monje, A.; Wang, H.-L. Peri-implantitis. J. Periodontol. 2018, 89, S267–S290.
- Parithimarkalaignan, S.; Padmanabhan, T.V. Osseointegration: An update. J. Indian Prosthodont. Soc. 2013, 13, 2–6.
- Irandoust, S.; Müftü, S. The interplay between bone healing and remodeling around dental implants. Sci. Rep. 2020, 10, 4335.
- Choi, J.-Y.; Sim, J.-H.; Yeo, I.-S.L. Characteristics of contact and distance osteogenesis around modified implant surfaces in rabbit tibiae. J. Periodontal Implant Sci. 2017, 47, 182–192.
- Toledano-Serrabona, J.; Sánchez-Garcés, M.Á.; Sánchez-Torres, A.; Sánchez-Torres, A.; Gay-Escoda, C.; Gay-Escoda, C. Alveolar distraction osteogenesis for dental implant treatments of the vertical bone atrophy: A systematic review. Med. Oral Patol. Oral Y Cir. Bucal 2019, 24, e70.
- Marco, F.; Milena, F.; Gianluca, G.; Gianluca, G.; Vittoria, O.; Vittoria, O. Peri-implant osteogenesis in health and osteoporosis. Micron 2005, 36, 630–644.
- Villar, C.C.; Huynh-Ba, G.; Mills, M.P.; Cochran, D.L. Wound healing around dental implants. Endod. Top. 2011, 25, 44–62.
- Rokaya, D.; Srimaneepong, V.; Sapkota, J.; Qin, J.; Siraleartmukul, K.; Siriwongrungson, V. Polymeric materials and films in dentistry: An overview. J. Adv. Res. 2018, 14, 25–34.
- Al-Rabab’ah, M.; Hamadneh, W.; Alsalem, I.; Khraisat, A.; Abu Karaky, A. Use of High Performance Polymers as Dental Implant Abutments and Frameworks: A Case Series Report. J. Prosthodont. 2019, 28, 365–372.
- Schwitalla, A.D.; Abou-Emara, M.; Zimmermann, T.; Spintig, T.; Beuer, F.; Lackmann, J.; Müller, W.D. The applicability of PEEK-based abutment screws. J. Mech. Behav. Biomed. Mater. 2016, 63, 244–251.
- Sandler, J.; Werner, P.; Shaffer, M.S.P.; Demchuk, V.; Altstädt, V.; Windle, A.H. Carbon-nanofibre-reinforced poly(ether ether ketone) composites. Compos. Part A Appl. Sci. Manuf. 2002, 33, 1033–1039.
- Bathala, L.; Majeti, V.; Rachuri, N.; Singh, N.; Gedela, S. The Role of Polyether Ether Ketone (Peek) in Dentistry—A Review. J. Med. Life 2019, 12, 5–9.
- Kurtz, S.M.; Devine, J.N. PEEK biomaterials in trauma, orthopedic, and spinal implants. Biomaterials 2007, 28, 4845–4869.
- Stawarczyk, B.; Schmid, P.; Roos, M.; Eichberger, M.; Schmidlin, P.R. Spectrophotometric Evaluation of Polyetheretherketone (PEEK) as a Core Material and a Comparison with Gold Standard Core Materials. Materials 2016, 9, 491.
- Lee, W.T.; Koak, J.-Y.; Lim, Y.-J.; Kim, S.-K.; Kwon, H.-B.; Kim, M.-J. Stress shielding and fatigue limits of poly-ether-ether-ketone dental implants. J. Biomed. Mater. Res. Part B Appl. Biomater. 2012, 100, 1044–1052.
- Najeeb, S.; Bds, Z.K.; Bds, S.Z.; Bds, M.S. Bioactivity and Osseointegration of PEEK Are Inferior to Those of Titanium: A Systematic Review. J. Oral Implantol. 2016, 42, 512–516.
- Walsh, W.R.; Bertollo, N.; Christou, C.; Schaffner, D.; Mobbs, R.J. Plasma-sprayed titanium coating to polyetheretherketone improves the bone-implant interface. Spine J. 2015, 15, 1041–1049.
- Han, C.M.; Lee, E.J.; Kim, H.-E.; Koh, Y.-H.; Kim, K.N.; Ha, Y.; Kuh, S.-U. The electron beam deposition of titanium on polyetheretherketone (PEEK) and the resulting enhanced biological properties. Biomaterials 2010, 31, 3465–3470.
- Wu, X.; Liu, X.; Wei, J.; Ma, J.; Deng, F.; Wei, S. Nano-TiO2/PEEK bioactive composite as a bone substitute material: In vitro and in vivo studies. Int. J. Nanomed. 2012, 7, 1215.
- Han, C.M.; Jang, T.S.; Kim, H.-E.; Koh, Y.-H. Creation of nanoporous TiO2 surface onto polyetheretherketone for effective immobilization and delivery of bone morphogenetic protein. J. Biomed. Mater. Res. Part A 2014, 102, 793–800.
- Lu, T.; Liu, X.; Qian, S.; Cao, H.; Qiao, Y.; Mei, Y.; Chu, P.K.; Ding, C. Multilevel surface engineering of nanostructured TiO2 on carbon-fiber-reinforced polyetheretherketone. Biomaterials 2014, 35, 5731–5740.
- Sun, Y.; Liu, X.; Tan, J.; Lv, D.; Song, W.; Su, R.; Li, L.; Liu, X.; Ouyang, L.; Liao, Y. Strontium ranelate incorporated 3D porous sulfonated PEEK simulating MC3T3-E1 cell differentiation. Regen. Biomater. 2021, 8, rbaa043.
- Bianchi, M.; Degli Esposti, L.; Ballardini, A.; Liscio, F.; Berni, M.; Gambardella, A.; Leeuwenburgh, S.C.G.; Sprio, S.; Tampieri, A.; Iafisco, M. Strontium doped calcium phosphate coatings on poly(etheretherketone) (PEEK) by pulsed electron deposition. Surf. Coat. Technol. 2017, 319, 191–199.
- Najeeb, S.; Khurshid, Z.; Matinlinna, J.P.; Siddiqui, F.; Nassani, M.Z.; Baroudi, K. Nanomodified Peek Dental Implants: Bioactive Composites and Surface Modification—A Review. Int. J. Dent. 2015, 2015, 381759.
- Gu, X.; Sun, X.; Sun, Y.; Wang, J.; Liu, Y.; Yu, K.; Wang, Y.; Zhou, Y. Bioinspired Modifications of PEEK Implants for Bone Tissue Engineering. Front. Bioeng. Biotechnol. 2021, 8, 631616.
- Cook, S.D.; Rust-Dawicki, A.M. Preliminary evaluation of titanium-coated PEEK dental implants. J. Oral Implantol. 1995, 21, 176–181.
- Delgado-Ruiz, R.; Romanos, G. Potential Causes of Titanium Particle and Ion Release in Implant Dentistry: A Systematic Review. Int. J. Mol. Sci. 2018, 19, 3585.
- Coelho, P.G.; Jimbo, R.; Tovar, N.; Bonfante, E.A. Osseointegration: Hierarchical designing encompassing the macrometer, micrometer, and nanometer length scales. Dent. Mater. 2015, 31, 37–52.
- Li, J.; Jansen, J.A.; Walboomers, X.F.; van den Beucken, J.J.J.P. Mechanical aspects of dental implants and osseointegration: A narrative review. J. Mech. Behav. Biomed. Mater. 2020, 103, 103574.
- Johansson, P.; Jimbo, R.; Kjellin, P.; Currie, F.; Chrcanovic, B.R.; Wennerberg, A. Biomechanical evaluation and surface characterization of a nano-modified surface on PEEK implants: A study in the rabbit tibia. Int. J. Nanomed. 2014, 9, 3903.
- Elawadly, T.; Radi, I.A.W.; El Khadem, A.; Osman, R.B. Can PEEK Be an Implant Material? Evaluation of Surface Topography and Wettability of Filled Versus Unfilled PEEK with Different Surface Roughness. J. Oral Implantol. 2017, 43, 456–461.
- Kubo, K.; Tsukimura, N.; Iwasa, F.; Ueno, T.; Saruwatari, L.; Aita, H.; Chiou, W.-A.; Ogawa, T. Cellular behavior on TiO2 nanonodular structures in a micro-to-nanoscale hierarchy model. Biomaterials 2009, 30, 5319–5329.
- Brammer, K.S.; Oh, S.; Cobb, C.J.; Bjursten, L.M.; van der Heyde, H.; Jin, S. Improved bone-forming functionality on diameter-controlled TiO2 nanotube surface. Acta Biomater. 2009, 5, 3215–3223.
- De Pablo, P.; Chapple, I.L.; Buckley, C.D.; Dietrich, T. Periodontitis in systemic rheumatic diseases. Nat. Rev. Rheumatol. 2009, 5, 218–224.
- Nair, S.; Faizuddin, M.; Dharmapalan, J. Role of autoimmune responses in periodontal disease. Autoimmune Dis. 2014, 2014, 596824.
- Moutsopoulos, N.M.; Konkel, J.E. Tissue-Specific Immunity at the Oral Mucosal Barrier. Trends Immunol. 2018, 39, 276–287.
- Hajishengallis, G.; Darveau, R.P.; Curtis, M.A. The keystone-pathogen hypothesis. Nat. Rev. Microbiol. 2012, 10, 717–725.
- Pan, W.; Wang, Q.; Chen, Q. The cytokine network involved in the host immune response to periodontitis. Int. J. Oral Sci. 2019, 11, 30.
- Tsukasaki, M.; Komatsu, N.; Nagashima, K.; Nitta, T.; Pluemsakunthai, W.; Shukunami, C.; Iwakura, Y.; Nakashima, T.; Okamoto, K.; Takayanagi, H. Host defense against oral microbiota by bone-damaging T cells. Nat. Commun. 2018, 9, 701.
- Prathapachandran, J.; Suresh, N. Management of peri-implantitis. Dent. Res. J. 2012, 9, 516–521.
- Ter Boo, G.J.; Grijpma, D.W.; Moriarty, T.F.; Richards, R.G.; Eglin, D. Antimicrobial delivery systems for local infection prophylaxis in orthopedic- and trauma surgery. Biomaterials 2015, 52, 113–125.
- Souza, J.G.S.; Bertolini, M.M.; Costa, R.C.; Nagay, B.E.; Dongari-Bagtzoglou, A.; Barão, V.A.R. Targeting implant-associated infections: Titanium surface loaded with antimicrobial. IScience 2021, 24, 102008.
- Lucke, M.; Schmidmaier, G.; Sadoni, S.; Wildemann, B.; Schiller, R.; Haas, N.P.; Raschke, M. Gentamicin coating of metallic implants reduces implant-related osteomyelitis in rats. Bone 2003, 32, 521–531.
- Lucke, M.; Wildemann, B.; Sadoni, S.; Surke, C.; Schiller, R.; Stemberger, A.; Raschke, M.; Haas, N.P.; Schmidmaier, G. Systemic versus local application of gentamicin in prophylaxis of implant-related osteomyelitis in a rat model. Bone 2005, 36, 770–778.
- Kälicke, T.; Schierholz, J.; Schlegel, U.; Frangen, T.M.; Köller, M.; Printzen, G.; Seybold, D.; Klöckner, S.; Muhr, G.; Arens, S. Effect on infection resistance of a local antiseptic and antibiotic coating on osteosynthesis implants: An in vitro and in vivo study. J. Orthop. Res. 2006, 24, 1622–1640.
- Zhang, B.; Braun, B.M.; Skelly, J.D.; Ayers, D.C.; Song, J. Significant Suppression of Staphylococcus aureus Colonization on Intramedullary Ti6Al4V Implants Surface-Grafted with Vancomycin-Bearing Polymer Brushes. ACS Appl. Mater. Interfaces 2019, 11, 28641–28647.
- Ghimire, A.; Skelly, J.D.; Song, J. Micrococcal-Nuclease-Triggered On-Demand Release of Vancomycin from Intramedullary Implant Coating Eradicates Staphylococcus aureus Infection in Mouse Femoral Canals. ACS Cent. Sci. 2019, 5, 1929–1936.
- Croes, M.; Bakhshandeh, S.; van Hengel, I.A.J.; Lietaert, K.; van Kessel, K.P.M.; Pouran, B.; van der Wal, B.C.H.; Vogely, H.C.; Van Hecke, W.; Fluit, A.C.; et al. Antibacterial and immunogenic behavior of silver coatings on additively manufactured porous titanium. Acta Biomater. 2018, 81, 315–327.
- Grohmann, S.; Menne, M.; Hesse, D.; Bischoff, S.; Schiffner, R.; Diefenbeck, M.; Liefeith, K. Biomimetic multilayer coatings deliver gentamicin and reduce implant-related osteomyelitis in rats. Biomed. Eng. /Biomed. Tech. 2019, 64, 383–395.
- Ma, K.; Cai, X.; Zhou, Y.; Wang, Y.; Jiang, T. In Vitro and In Vivo Evaluation of Tetracycline Loaded Chitosan-Gelatin Nanosphere Coatings for Titanium Surface Functionalization. Macromol. Biosci. 2017, 17, 1600130.
- Yang, L.; Li, L.; Li, H.; Wang, T.; Ren, X.; Cheng, Y.; Li, Y.; Huang, Q. Layer-by-Layer Assembled Smart Antibacterial Coatings via Mussel-Inspired Polymerization and Dynamic Covalent Chemistry. Adv. Healthc. Mater. 2022, 11, 2200112.
- Garg, U.; Chauhan, S.; Nagaich, U.; Jain, N. Current Advances in Chitosan Nanoparticles Based Drug Delivery and Targeting. Adv. Pharm. Bull. 2019, 9, 195–204.
- Stavrakis, A.I.; Zhu, S.; Hegde, V.; Loftin, A.H.; Ashbaugh, A.G.; Niska, J.A.; Miller, L.S.; Segura, T.; Bernthal, N.M. In Vivo Efficacy of a "Smart" Antimicrobial Implant Coating. J. Bone Joint Surg. Am. 2016, 98, 1183–1189.
- Stavrakis, A.I.; Zhu, S.; Loftin, A.H.; Weixian, X.; Niska, J.; Hegde, V.; Segura, T.; Bernthal, N.M. Controlled Release of Vancomycin and Tigecycline from an Orthopaedic Implant Coating Prevents Staphylococcus aureus Infection in an Open Fracture Animal Model. Biomed. Res. Int. 2019, 2019, 1638508.
- Lorenzetti, M.; Dogša, I.; Stošicki, T.; Stopar, D.; Kalin, M.; Kobe, S.; Novak, S. The Influence of Surface Modification on Bacterial Adhesion to Titanium-Based Substrates. ACS Appl. Mater. Interfaces 2015, 7, 1644–1651.
- Cao, Y.; Su, B.; Chinnaraj, S.; Jana, S.; Bowen, L.; Charlton, S.; Duan, P.; Jakubovics, N.S.; Chen, J. Nanostructured titanium surfaces exhibit recalcitrance towards Staphylococcus epidermidis biofilm formation. Sci. Rep. 2018, 8, 1071.
- Hizal, F.; Zhuk, I.; Sukhishvili, S.; Busscher, H.J.; van der Mei, H.C.; Choi, C.H. Impact of 3D Hierarchical Nanostructures on the Antibacterial Efficacy of a Bacteria-Triggered Self-Defensive Antibiotic Coating. ACS Appl. Mater. Interfaces 2015, 7, 20304–20313.
- Kong, M.; Chen, X.G.; Xing, K.; Park, H.J. Antimicrobial properties of chitosan and mode of action: A state of the art review. Int. J. Food Microbiol. 2010, 144, 51–63.
- Yuan, Y.; Chesnutt, B.M.; Haggard, W.O.; Bumgardner, J.D. Deacetylation of Chitosan: Material Characterization and in vitro Evaluation via Albumin Adsorption and Pre-Osteoblastic Cell Cultures. Materials 2011, 4, 1399–1416.
- Li, B.; Xia, X.; Guo, M.; Jiang, Y.; Li, Y.; Zhang, Z.; Liu, S.; Li, H.; Liang, C.; Wang, H. Biological and antibacterial properties of the micro-nanostructured hydroxyapatite/chitosan coating on titanium. Sci. Rep. 2019, 9, 14052.
- Park, S.; Kim, H.; Choi, K.S.; Ji, M.-K.; Kim, S.; Gwon, Y.; Park, C.; Kim, J.; Lim, H.-P. Graphene–Chitosan Hybrid Dental Implants with Enhanced Antibacterial and Cell-Proliferation Properties. Appl. Sci. 2020, 10, 4888.
- Hancock, R.E.; Sahl, H.G. Antimicrobial and host-defense peptides as new anti-infective therapeutic strategies. Nat. Biotechnol. 2006, 24, 1551–1557.
- Wisdom, C.; Chen, C.; Yuca, E.; Zhou, Y.; Tamerler, C.; Snead, M.L. Repeatedly Applied Peptide Film Kills Bacteria on Dental Implants. JOM (1989) 2019, 71, 1271–1280.
- Yazici, H.; O’Neill, M.B.; Kacar, T.; Wilson, B.R.; Oren, E.E.; Sarikaya, M.; Tamerler, C. Engineered Chimeric Peptides as Antimicrobial Surface Coating Agents toward Infection-Free Implants. ACS Appl. Mater. Interfaces 2016, 8, 5070–5081.
- Zhang, S.; Zhou, X.; Liu, T.; Huang, Y.; Li, J. The effects of Peptide Mel4-coated titanium plates on infection rabbits after internal fixation of open fractures. Arch. Orthop. Trauma Surg. 2022, 142, 729–734.
- Acosta, S.; Ibañez-Fonseca, A.; Aparicio, C.; Rodríguez-Cabello, J.C. Antibiofilm coatings based on protein-engineered polymers and antimicrobial peptides for preventing implant-associated infections. Biomater. Sci. 2020, 8, 2866–2877.
- Wu, S.; Xu, J.; Zou, L.; Luo, S.; Yao, R.; Zheng, B.; Liang, G.; Wu, D.; Li, Y. Long-lasting renewable antibacterial porous polymeric coatings enable titanium biomaterials to prevent and treat peri-implant infection. Nat. Commun. 2021, 12, 3303.
- Brånemark, P.I. Osseointegration and its experimental background. J. Prosthet. Dent. 1983, 50, 399–410.
- Barrak, F.N.; Li, S.; Muntane, A.M.; Jones, J.R. Particle release from implantoplasty of dental implants and impact on cells. Int. J. Implant Dent. 2020, 6, 50.
- Zhang, X.; Zhang, Y.; Jin, Z. A review of the bio-tribology of medical devices. Friction 2022, 10, 4–30.
- Kwon, H.J.; Cha, M.-Y.; Kim, D.; Kim, D.K.; Soh, M.; Shin, K.; Hyeon, T.; Mook-Jung, I. Mitochondria-Targeting Ceria Nanoparticles as Antioxidants for Alzheimer’s Disease. ACS Nano 2016, 10, 2860–2870.
- Cline, E.N.; Bicca, M.A.; Viola, K.L.; Klein, W.L. The Amyloid-beta Oligomer Hypothesis: Beginning of the Third Decade. J. Alzheimer’s Dis. 2018, 64, S567–S610.
- Messous, R.; Henriques, B.; Bousbaa, H.; Silva, F.S.; Teughels, W.; Souza, J.C.M. Cytotoxic effects of submicron- and nano-scale titanium debris released from dental implants: An integrative review. Clin. Oral Investig. 2021, 25, 1627–1640.
- Zhang, L.; Haddouti, E.M.; Welle, K.; Burger, C.; Wirtz, D.C.; Schildberg, F.A.; Kabir, K. The Effects of Biomaterial Implant Wear Debris on Osteoblasts. Front. Cell Dev. Biol. 2020, 8, 352.
- Sananez, A.; Lefebvre, C.; Looney, S.; Baker, P.; Mettenburg, D.; Rueggeberg, F.A. In vitro mechanical analysis of complete-arch mandibular implant-supported fixed prostheses abutment screws after cyclic loading. J. Prosthet. Dent. 2015, 113, 432–439.
- Łępicka, M.; Barros-Silva, S.; Licciardello, N.; Cortez, A.; Gobbo, P.; Sampaio, M.; Cortez, J.; Cortez, H.; Alves, F.; Lipowicz, P.; et al. Silane-based coating charged with TiO2 NPs for dental implant applications. Surf. Coat. Technol. 2021, 413, 127066.
- Kruk, A.; Zimowski, S.; Łukaszczyk, A.; Cieniek, Ł.; Moskalewicz, T. The influence of heat treatment on the microstructure, surface topography and selected properties of PEEK coatings electrophoretically deposited on the Ti-6Al-4V alloy. Prog. Org. Coat. 2019, 133, 180–190.
- Yadav, S.; Gangwar, S. A critical evaluation of tribological interaction for restorative materials in dentistry. Int. J. Polym. Mater. Polym. Biomater. 2019, 68, 1005–1019.
- Kan, W.H.; Chang, L. The mechanisms behind the tribological behaviour of polymer matrix composites reinforced with TiO2 nanoparticles. Wear 2021, 474–475, 203754.
- Thygesen, K.; Alpert, J.S.; Jaffe, A.S.; Chaitman, B.R.; Bax, J.J.; Morrow, D.A.; White, H.D.; Null, N. Fourth Universal Definition of Myocardial Infarction (2018). Circulation 2018, 138, e618–e651.
- Libby, P.; Buring, J.E.; Badimon, L.; Hansson, G.K.; Deanfield, J.; Bittencourt, M.S.; Tokgözoğlu, L.; Lewis, E.F. Atherosclerosis. Nat. Rev. Dis. Primers 2019, 5, 56.
- Gruntzig, A. Transluminal dilatation of coronary-artery stenosis. Lancet 1978, 311, 263.
- Sigwart, U.; Puel, J.; Mirkovitch, V.; Joffre, F.; Kappenberger, L. Intravascular stents to prevent occlusion and restenosis after transluminal angioplasty. N. Engl. J. Med. 1987, 316, 701–706.
- Torrado, J.; Buckley, L.; Durán, A.; Trujillo, P.; Toldo, S.; Valle Raleigh, J.; Abbate, A.; Biondi-Zoccai, G.; Guzmán, L.A. Restenosis, Stent Thrombosis, and Bleeding Complications: Navigating Between Scylla and Charybdis. J. Am. Coll. Cardiol. 2018, 71, 1676–1695.
- Tanner, F.C.; Yang, Z.Y.; Duckers, E.; Gordon, D.; Nabel, G.J.; Nabel, E.G. Expression of cyclin-dependent kinase inhibitors in vascular disease. Circ. Res. 1998, 82, 396–403.
- Acharya, G.; Park, K. Mechanisms of controlled drug release from drug-eluting stents. Adv. Drug Deliv. Rev. 2006, 58, 387–401.
- Dimitrova, G.; Tulman, D.B.; Bergese, S.D. Perioperative management of antiplatelet therapy in patients with drug-eluting stents. HSR Proc. Intensive Care Cardiovasc. Anesth. 2012, 4, 153–167.
- Farooq, V.; Gogas, B.D.; Serruys, P.W. Restenosis. Circ. Cardiovasc. Interv. 2011, 4, 195–205.
- Kohn, J.; Zeltinger, J. Degradable, drug-eluting stents: A new frontier for the treatment of coronary artery disease. Expert Rev. Med. Devices 2005, 2, 667–671.
- Onuma, Y.; Serruys, P.W. Bioresorbable scaffold: The advent of a new era in percutaneous coronary and peripheral revascularization? Circulation 2011, 123, 779–797.
- Yamaji, K.; Ueki, Y.; Souteyrand, G.; Daemen, J.; Wiebe, J.; Nef, H.; Adriaenssens, T.; Loh, J.P.; Lattuca, B.; Wykrzykowska, J.J.; et al. Mechanisms of Very Late Bioresorbable Scaffold Thrombosis: The INVEST Registry. J. Am. Coll. Cardiol. 2017, 70, 2330–2344.
- Onuma, Y.; Serruys, P.W.; Muramatsu, T.; Nakatani, S.; van Geuns, R.J.; de Bruyne, B.; Dudek, D.; Christiansen, E.; Smits, P.C.; Chevalier, B.; et al. Incidence and imaging outcomes of acute scaffold disruption and late structural discontinuity after implantation of the absorb Everolimus-Eluting fully bioresorbable vascular scaffold: Optical coherence tomography assessment in the ABSORB cohort B Trial (A Clinical Evaluation of the Bioabsorbable Everolimus Eluting Coronary Stent System in the Treatment of Patients with De Novo Native Coronary Artery Lesions). JACC Cardiovasc. Interv. 2014, 7, 1400–1411.
- Montone, R.A.; Niccoli, G.; De Marco, F.; Minelli, S.; D’Ascenzo, F.; Testa, L.; Bedogni, F.; Crea, F. Temporal Trends in Adverse Events After Everolimus-Eluting Bioresorbable Vascular Scaffold Versus Everolimus-Eluting Metallic Stent Implantation: A Meta-Analysis of Randomized Controlled Trials. Circulation 2017, 135, 2145–2154.
- Ang, H.Y.; Huang, Y.Y.; Lim, S.T.; Wong, P.; Joner, M.; Foin, N. Mechanical behavior of polymer-based vs. metallic-based bioresorbable stents. J. Thorac. Dis. 2017, 9, S923–S934.
- Gori, T.; Weissner, M.; Gönner, S.; Wendling, F.; Ullrich, H.; Ellis, S.; Anadol, R.; Polimeni, A.; Münzel, T. Characteristics, Predictors, and Mechanisms of Thrombosis in Coronary Bioresorbable Scaffolds: Differences Between Early and Late Events. JACC Cardiovasc. Interv. 2017, 10, 2363–2371.
- Stone, G.W.; Ellis, S.G.; Cox, D.A.; Hermiller, J.; O’Shaughnessy, C.; Mann, J.T.; Turco, M.; Caputo, R.; Bergin, P.; Greenberg, J.; et al. One-year clinical results with the slow-release, polymer-based, paclitaxel-eluting TAXUS stent: The TAXUS-IV trial. Circulation 2004, 109, 1942–1947.
- Stefanini, G.G.; Holmes, D.R. Drug-Eluting Coronary-Artery Stents. N. Engl. J. Med. 2013, 368, 254–265.
- Rizas, K.D.; Mehilli, J. Stent Polymers: Do They Make a Difference? Circ. Cardiovasc. Interv. 2016, 9, e002943.
- Joner, M.; Finn, A.V.; Farb, A.; Mont, E.K.; Kolodgie, F.D.; Ladich, E.; Kutys, R.; Skorija, K.; Gold, H.K.; Virmani, R. Pathology of drug-eluting stents in humans: Delayed healing and late thrombotic risk. J. Am. Coll. Cardiol. 2006, 48, 193–202.
- Camenzind, E.; Steg, P.G.; Wijns, W. Stent thrombosis late after implantation of first-generation drug-eluting stents: A cause for concern. Circulation 2007, 115, 1440–1455, discussion 1455.
- Varenhorst, C.; Lindholm, M.; Sarno, G.; Olivecrona, G.; Jensen, U.; Nilsson, J.; Carlsson, J.; James, S.; Lagerqvist, B. Stent thrombosis rates the first year and beyond with new- and old-generation drug-eluting stents compared to bare metal stents. Clin. Res. Cardiol. 2018, 107, 816–823.
- Byrne, R.A.; Joner, M.; Kastrati, A. Polymer coatings and delayed arterial healing following drug-eluting stent implantation. Minerva Cardioangiol. 2009, 57, 567–584.
- Yonetsu, T.; Kato, K.; Kim, S.-J.; Xing, L.; Jia, H.; McNulty, I.; Lee, H.; Zhang, S.; Uemura, S.; Jang, Y.; et al. Predictors for Neoatherosclerosis. Circ. Cardiovasc. Imaging 2012, 5, 660–666.
- Virmani, R.; Guagliumi, G.; Farb, A.; Musumeci, G.; Grieco, N.; Motta, T.; Mihalcsik, L.; Tespili, M.; Valsecchi, O.; Kolodgie, F.D. Localized hypersensitivity and late coronary thrombosis secondary to a sirolimus-eluting stent: Should we be cautious? Circulation 2004, 109, 701–705.
- Valgimigli, M.; Frigoli, E.; Heg, D.; Tijssen, J.; Jüni, P.; Vranckx, P.; Ozaki, Y.; Morice, M.-C.; Chevalier, B.; Onuma, Y.; et al. Dual Antiplatelet Therapy after PCI in Patients at High Bleeding Risk. N. Engl. J. Med. 2021, 385, 1643–1655.
- Rodriguez, F.; Harrington, R.A. Management of Antithrombotic Therapy after Acute Coronary Syndromes. N. Engl. J. Med. 2021, 384, 452–460.
- Palmerini, T.; Benedetto, U.; Biondi-Zoccai, G.; Della Riva, D.; Bacchi-Reggiani, L.; Smits, P.C.; Vlachojannis, G.J.; Jensen, L.O.; Christiansen, E.H.; Berencsi, K.; et al. Long-Term Safety of Drug-Eluting and Bare-Metal Stents: Evidence From a Comprehensive Network Meta-Analysis. J. Am. Coll. Cardiol. 2015, 65, 2496–2507.
- Kim, Y.H.; Her, A.Y.; Jeong, M.H.; Kim, B.K.; Hong, S.J.; Kim, S.; Ahn, C.M.; Kim, J.S.; Ko, Y.G.; Choi, D.; et al. Comparison of First- and Second-Generation Drug-Eluting Stents in Patients with ST-Segment Elevation Myocardial Infarction Based on Pre-Percutaneous Coronary Intervention Thrombolysis in Myocardial Infarction Flow Grade. J. Clin. Med. 2021, 10, 367.
- Kim, M.C.; Jeong, M.H.; Ahn, Y.; Kim, J.H.; Chae, S.C.; Kim, Y.J.; Hur, S.H.; Seong, I.W.; Hong, T.J.; Choi, D.H.; et al. What is optimal revascularization strategy in patients with multivessel coronary artery disease in non-ST-elevation myocardial infarction? Multivessel or culprit-only revascularization. Int. J. Cardiol. 2011, 153, 148–153.
- Hsieh, M.J.; Chen, C.C.; Lee, C.H.; Wang, C.Y.; Chang, S.H.; Chen, D.Y.; Yang, C.H.; Tsai, M.L.; Yeh, J.K.; Ho, M.Y.; et al. Complete and incomplete revascularization in non-ST segment myocardial infarction with multivessel disease: Long-term outcomes of first- and second-generation drug-eluting stents. Heart Vessel. 2019, 34, 251–258.
- Claessen, B.E.; Henriques, J.P.S.; Jaffer, F.A.; Mehran, R.; Piek, J.J.; Dangas, G.D. Stent Thrombosis. JACC Cardiovasc. Interv. 2014, 7, 1081–1092.
- Iakovou, I.; Schmidt, T.; Bonizzoni, E.; Ge, L.; Sangiorgi, G.M.; Stankovic, G.; Airoldi, F.; Chieffo, A.; Montorfano, M.; Carlino, M.; et al. Incidence, predictors, and outcome of thrombosis after successful implantation of drug-eluting stents. JAMA 2005, 293, 2126–2130.
- Gori, T.; Polimeni, A.; Indolfi, C.; Räber, L.; Adriaenssens, T.; Münzel, T. Predictors of stent thrombosis and their implications for clinical practice. Nat. Rev. Cardiol. 2019, 16, 243–256.
- Armstrong, E.J.; Waltenberger, J.; Rogers, J.H. Percutaneous coronary intervention in patients with diabetes: Current concepts and future directions. J. Diabetes Sci. Technol. 2014, 8, 581–589.
- Capodanno, D.; Bhatt, D.L.; Gibson, C.M.; James, S.; Kimura, T.; Mehran, R.; Rao, S.V.; Steg, P.G.; Urban, P.; Valgimigli, M.; et al. Bleeding avoidance strategies in percutaneous coronary intervention. Nat. Rev. Cardiol. 2022, 19, 117–132.
- Timmins, L.H.; Miller, M.W.; Clubb, F.J.; Moore, J.E. Increased artery wall stress post-stenting leads to greater intimal thickening. Lab. Investig. 2011, 91, 955–967.
- Xu, J.; Yang, J.; Huang, N.; Uhl, C.; Zhou, Y.; Liu, Y. Mechanical response of cardiovascular stents under vascular dynamic bending. BioMed. Eng. OnLine 2016, 15, 21.
- Conway, C.; Nezami, F.R.; Rogers, C.; Groothuis, A.; Squire, J.C.; Edelman, E.R. Acute Stent-Induced Endothelial Denudation: Biomechanical Predictors of Vascular Injury. Front. Cardiovasc. Med. 2021, 8, 1312.
- Rogers, C.; Edelman, E.R. Endovascular Stent Design Dictates Experimental Restenosis and Thrombosis. Circulation 1995, 91, 2995–3001.
- Rogers, C.; Tseng, D.Y.; Squire, J.C.; Edelman, E.R. Balloon-Artery Interactions During Stent Placement. Circ. Res. 1999, 84, 378–383.
- Han, Y.; Lu, W. Optimizing the deformation behavior of stent with nonuniform Poisson’s ratio distribution for curved artery. J. Mech. Behav. Biomed. Mater. 2018, 88, 442–452.
- Traub, O.; Berk, B.C. Laminar Shear Stress. Arterioscler. Thromb. Vasc. Biol. 1998, 18, 677–685.
- Cooke, J.P. Flow, NO, and atherogenesis. Proc. Natl. Acad. Sci. USA 2003, 100, 768–770.
- Jimenez, J.M.; Davies, P.F. Hemodynamically driven stent strut design. Ann. Biomed. Eng. 2009, 37, 1483–1494.
- Wei, L.; Leo, H.L.; Chen, Q.; Li, Z. Structural and Hemodynamic Analyses of Different Stent Structures in Curved and Stenotic Coronary Artery. Front. Bioeng. Biotechnol. 2019, 7, 366.
- Kolandaivelu, K.; Swaminathan, R.; Gibson, W.J.; Kolachalama, V.B.; Nguyen-Ehrenreich, K.-L.; Giddings, V.L.; Coleman, L.; Wong, G.K.; Edelman, E.R. Stent Thrombogenicity Early in High-Risk Interventional Settings Is Driven by Stent Design and Deployment and Protected by Polymer-Drug Coatings. Circulation 2011, 123, 1400–1409.
- Nakatani, S.; Nishino, M.; Taniike, M.; Makino, N.; Kato, H.; Egami, Y.; Shutta, R.; Tanouchi, J.; Yamada, Y. Initial findings of impact of strut width on stent coverage and apposition of sirolimus-eluting stents assessed by optical coherence tomography. Catheter. Cardiovasc. Interv. 2013, 81, 776–781.
- Ellis, S.G.; Gori, T.; Serruys, P.W.; Nef, H.; Steffenino, G.; Brugaletta, S.; Munzel, T.; Feliz, C.; Schmidt, G.; Sabaté, M.; et al. Clinical, Angiographic, and Procedural Correlates of Very Late Absorb Scaffold Thrombosis: Multistudy Registry Results. JACC Cardiovasc. Interv. 2018, 11, 638–644.
- Kitahara, H.; Okada, K.; Kimura, T.; Yock, P.G.; Lansky, A.J.; Popma, J.J.; Yeung, A.C.; Fitzgerald, P.J.; Honda, Y. Impact of Stent Size Selection on Acute and Long-Term Outcomes After Drug-Eluting Stent Implantation in De Novo Coronary Lesions. Circ. Cardiovasc. Interv. 2017, 10, e004795.
- Foin, N.; Sen, S.; Allegria, E.; Petraco, R.; Nijjer, S.; Francis, D.P.; Di Mario, C.; Davies, J.E. Maximal expansion capacity with current DES platforms: A critical factor for stent selection in the treatment of left main bifurcations? EuroIntervention 2013, 8, 1315–1325.
- Abreu Filho, L.M.; Forte, A.A.; Sumita, M.K.; Favarato, D.; Meireles, G.C. Influence of metal alloy and the profile of coronary stents in patients with multivessel coronary disease. Clinics (Sao Paulo) 2011, 66, 985–989.
- Nikam, N.; Steinberg, T.B.; Steinberg, D.H. Advances in stent technologies and their effect on clinical efficacy and safety. Med. Devices (Auckl) 2014, 7, 165–178.
- Livingston, M.; Tan, A. Coating Techniques and Release Kinetics of Drug-Eluting Stents. J. Med. Device 2019, 10, 010801.
- Jain, R.A. The manufacturing techniques of various drug loaded biodegradable poly(lactide-co-glycolide) (PLGA) devices. Biomaterials 2000, 21, 2475–2490.
- Bao, L.; Dorgan, J.R.; Knauss, D.; Hait, S.; Oliveira, N.S.; Maruccho, I.M. Gas permeation properties of poly(lactic acid) revisited. J. Membr. Sci. 2006, 285, 166–172.
- Pinchuk, L.; Wilson, G.J.; Barry, J.J.; Schoephoerster, R.T.; Parel, J.-M.; Kennedy, J.P. Medical applications of poly(styrene-block-isobutylene-block-styrene) (“SIBS”). Biomaterials 2008, 29, 448–460.
- Hopkins, C.; Sweeney, C.A.; O’Connor, C.; McHugh, P.E.; McGarry, J.P. Webbing and Delamination of Drug Eluting Stent Coatings. Ann. Biomed. Eng. 2016, 44, 419–431.
- Basalus, M.W.Z.; Tandjung, K.; van Westen, T.; Sen, H.; van der Jagt, P.K.N.; Grijpma, D.W.; van Apeldoorn, A.A.; von Birgelen, C. Scanning electron microscopic assessment of coating irregularities and their precursors in unexpanded durable polymer-based drug-eluting stents. Catheter. Cardiovasc. Interv. 2012, 79, 644–653.
- McKittrick, C.M.; Cardona, M.J.; Black, R.A.; McCormick, C. Development of a Bioactive Polymeric Drug Eluting Coronary Stent Coating Using Electrospraying. Ann. Biomed. Eng. 2020, 48, 271–281.
- Van Belle, E.; Tio, F.O.; Couffinhal, T.; Maillard, L.; Passeri, J.; Isner, J.M. Stent endothelialization. Time course, impact of local catheter delivery, feasibility of recombinant protein administration, and response to cytokine expedition. Circulation 1997, 95, 438–448.
- Ishiwata, S.; Tukada, T.; Nakanishi, S.; Nishiyama, S.; Seki, A. Postangioplasty restenosis: Platelet activation and the coagulation-fibrinolysis system as possible factors in the pathogenesis of restenosis. Am. Heart J. 1997, 133, 387–392.
- Davies, M.G.; Hagen, P.O. Pathobiology of intimal hyperplasia. Br. J. Surg. 1994, 81, 1254–1269.
- Jana, S. Endothelialization of cardiovascular devices. Acta Biomater. 2019, 99, 53–71.
- Rajendran, P.; Rengarajan, T.; Thangavel, J.; Nishigaki, Y.; Sakthisekaran, D.; Sethi, G.; Nishigaki, I. The vascular endothelium and human diseases. Int. J. Biol. Sci. 2013, 9, 1057–1069.
- Lee, Y.H.; Mei, F.; Bai, M.Y.; Zhao, S.; Chen, D.R. Release profile characteristics of biodegradable-polymer-coated drug particles fabricated by dual-capillary electrospray. J. Control. Release 2010, 145, 58–65.
- Mitchell, M.J.; Billingsley, M.M.; Haley, R.M.; Wechsler, M.E.; Peppas, N.A.; Langer, R. Engineering precision nanoparticles for drug delivery. Nat. Rev. Drug Discov. 2021, 20, 101–124.
- Cherian, A.M.; Nair, S.V.; Maniyal, V.; Menon, D. Surface engineering at the nanoscale: A way forward to improve coronary stent efficacy. APL Bioeng. 2021, 5, 021508.
- Liang, C.; Tian, Y.; Zou, X.; Hu, Y.; Zhou, H.; Yang, L.; Wang, H. Improve endothelialization of metallic cardiovascular stent via femtosecond laser induced micro/nanostructure dependent cells proliferation and drug delivery control. Colloids Surf. B Biointerfaces 2022, 212, 112376.
- Bedair, T.M.; ElNaggar, M.A.; Joung, Y.K.; Han, D.K. Recent advances to accelerate re-endothelialization for vascular stents. J. Tissue Eng. 2017, 8, 2041731417731546.
- Du, R.; Wang, Y.; Huang, Y.; Zhao, Y.; Zhang, D.; Du, D.; Zhang, Y.; Li, Z.; McGinty, S.; Pontrelli, G.; et al. Design and testing of hydrophobic core/hydrophilic shell nano/micro particles for drug-eluting stent coating. NPG Asia Mater. 2018, 10, 642–658.
- Carpenter, A.W.; Schoenfisch, M.H. Nitric oxide release: Part II. Therapeutic applications. Chem. Soc. Rev. 2012, 41, 3742–3752.
- Rao, J.; Pan Bei, H.; Yang, Y.; Liu, Y.; Lin, H.; Zhao, X. Nitric Oxide-Producing Cardiovascular Stent Coatings for Prevention of Thrombosis and Restenosis. Front. Bioeng. Biotechnol. 2020, 8, 578.
- Yang, Z.; Yang, Y.; Zhang, L.; Xiong, K.; Li, X.; Zhang, F.; Wang, J.; Zhao, X.; Huang, N. Mussel-inspired catalytic selenocystamine-dopamine coatings for long-term generation of therapeutic gas on cardiovascular stents. Biomaterials 2018, 178, 1–10.
- Zhang, F.; Zhang, Q.; Li, X.; Huang, N.; Zhao, X.; Yang, Z. Mussel-inspired dopamine-Cu(II) coatings for sustained in situ generation of nitric oxide for prevention of stent thrombosis and restenosis. Biomaterials 2019, 194, 117–129.
- Tu, Q.; Shen, X.; Liu, Y.; Zhang, Q.; Zhao, X.; Maitz, M.F.; Liu, T.; Qiu, H.; Wang, J.; Huang, N.; et al. A facile metal–phenolic–amine strategy for dual-functionalization of blood-contacting devices with antibacterial and anticoagulant properties. Mater. Chem. Front. 2019, 3, 265–275.
- Ashikari-Hada, S.; Habuchi, H.; Kariya, Y.; Kimata, K. Heparin Regulates Vascular Endothelial Growth Factor165-dependent Mitogenic Activity, Tube Formation, and Its Receptor Phosphorylation of Human Endothelial Cells. Comparison of the effects of heparin and modified heparins. J. Biol. Chem. 2005, 280, 31508–31515.
- Kastana, P.; Choleva, E.; Poimenidi, E.; Karamanos, N.; Sugahara, K.; Papadimitriou, E. Insight into the role of chondroitin sulfate E in angiogenesis. Febs. J. 2019, 286, 2921–2936.
- Zemani, F.; Benisvy, D.; Galy-Fauroux, I.; Lokajczyk, A.; Colliec-Jouault, S.; Uzan, G.; Fischer, A.M.; Boisson-Vidal, C. Low-molecular-weight fucoidan enhances the proangiogenic phenotype of endothelial progenitor cells. Biochem. Pharmacol. 2005, 70, 1167–1175.
- Genasetti, A.; Vigetti, D.; Viola, M.; Karousou, E.; Moretto, P.; Rizzi, M.; Bartolini, B.; Clerici, M.; Pallotti, F.; De Luca, G.; et al. Hyaluronan and human endothelial cell behavior. Connect. Tissue Res. 2008, 49, 120–123.
- Kam, A.; Li, K.M.; Razmovski-Naumovski, V.; Nammi, S.; Chan, K.; Li, G.Q. Gallic acid protects against endothelial injury by restoring the depletion of DNA methyltransferase 1 and inhibiting proteasome activities. Int. J. Cardiol. 2014, 171, 231–242.
- Yang, Y.; Gao, P.; Wang, J.; Tu, Q.; Bai, L.; Xiong, K.; Qiu, H.; Zhao, X.; Maitz, M.F.; Wang, H.; et al. Endothelium-Mimicking Multifunctional Coating Modified Cardiovascular Stents via a Stepwise Metal-Catechol-(Amine) Surface Engineering Strategy. Research 2020, 2020, 9203906.
- Chen, Y.; Gao, P.; Huang, L.; Tan, X.; Zhou, N.; Yang, T.; Qiu, H.; Dai, X.; Michael, S.; Tu, Q.; et al. A tough nitric oxide-eluting hydrogel coating suppresses neointimal hyperplasia on vascular stent. Nat. Commun. 2021, 12, 7079.
- Metavarayuth, K.; Sitasuwan, P.; Zhao, X.; Lin, Y.; Wang, Q. Influence of Surface Topographical Cues on the Differentiation of Mesenchymal Stem Cells in Vitro. ACS Biomater. Sci. Eng. 2016, 2, 142–151.
- Wang, S.; Li, J.; Zhou, Z.; Zhou, S.; Hu, Z. Micro-/Nano-Scales Direct Cell Behavior on Biomaterial Surfaces. Molecules 2018, 24, 75.
- Palmaz, J.C.; Benson, A.; Sprague, E.A. Influence of surface topography on endothelialization of intravascular metallic material. J. Vasc. Interv. Radiol. 1999, 10, 439–444.
- Ding, Y.; Yang, Z.; Bi, C.W.; Yang, M.; Xu, S.L.; Lu, X.; Huang, N.; Huang, P.; Leng, Y. Directing vascular cell selectivity and hemocompatibility on patterned platforms featuring variable topographic geometry and size. ACS Appl. Mater. Interfaces 2014, 6, 12062–12070.
- Peng, L.; Eltgroth, M.L.; LaTempa, T.J.; Grimes, C.A.; Desai, T.A. The effect of TiO2 nanotubes on endothelial function and smooth muscle proliferation. Biomaterials 2009, 30, 1268–1272.
- Cao, Y.; Desai, T.A. TiO2-Based Nanotopographical Cues Attenuate the Restenotic Phenotype in Primary Human Vascular Endothelial and Smooth Muscle Cells. ACS Biomater. Sci. Eng. 2020, 6, 923–932.
- Junkar, I.; Kulkarni, M.; Bencina, M.; Kovac, J.; Mrak-Poljsak, K.; Lakota, K.; Sodin-Semrl, S.; Mozetic, M.; Iglic, A. Titanium Dioxide Nanotube Arrays for Cardiovascular Stent Applications. ACS Omega 2020, 5, 7280–7289.





