Your browser does not fully support modern features. Please upgrade for a smoother experience.

Submitted Successfully!
Thank you for your contribution! You can also upload a video entry or images related to this topic.
For video creation, please contact our Academic Video Service.
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Jose Luiz de Medeiros | -- | 2388 | 2022-09-19 07:05:40 | | | |
| 2 | Sirius Huang | Meta information modification | 2388 | 2022-09-19 08:09:07 | | |
Video Upload Options
We provide professional Academic Video Service to translate complex research into visually appealing presentations. Would you like to try it?
Cite
If you have any further questions, please contact Encyclopedia Editorial Office.
Cunha, G.P.D.; Medeiros, J.L.D.; Araújo, O.D.Q.F. Gas-Liquid Membrane Contactors with Aqueous-Amine Solvents. Encyclopedia. Available online: https://encyclopedia.pub/entry/27286 (accessed on 20 May 2026).
Cunha GPD, Medeiros JLD, Araújo ODQF. Gas-Liquid Membrane Contactors with Aqueous-Amine Solvents. Encyclopedia. Available at: https://encyclopedia.pub/entry/27286. Accessed May 20, 2026.
Cunha, Guilherme Pereira Da, José Luiz De Medeiros, Ofélia De Queiroz F. Araújo. "Gas-Liquid Membrane Contactors with Aqueous-Amine Solvents" Encyclopedia, https://encyclopedia.pub/entry/27286 (accessed May 20, 2026).
Cunha, G.P.D., Medeiros, J.L.D., & Araújo, O.D.Q.F. (2022, September 19). Gas-Liquid Membrane Contactors with Aqueous-Amine Solvents. In Encyclopedia. https://encyclopedia.pub/entry/27286
Cunha, Guilherme Pereira Da, et al. "Gas-Liquid Membrane Contactors with Aqueous-Amine Solvents." Encyclopedia. Web. 19 September, 2022.
Copy Citation
Gas–liquid membrane contactor (GLMC) is a promising process intensification technology for offshore natural gas conditioning in which weight and footprint constraints impose severe limitations. Thanks to its potential for substituting conventional packed/trayed columns for acid-gas absorption and acid-gas solvent regeneration, gas-liquid membrane contactors have been investigated experimentally and theoretically, wherein aqueous-amine solvents and their blends are the most employed solvents for carbon dioxide removal from natural gas in gas-liquid membrane contactors.
gas–liquid membrane contactor
natural gas conditioning
carbon capture
1. Introduction
The global population is continuously increasing, and it is foreseen that it will exceed 10 billion by 2050 [1]. In this scenario, natural gas (NG) exploration and production attract interest in meeting the growing world energy demand [2]. Moreover, NG plays a key role in the transition to renewable energy sources [3] as NG is the cleanest fossil fuel due to its high hydrogen/carbon ratio [4].
Proven NG reserves around the world are approximately 196.9 trillion cubic meters [4], but onshore NG reserves—with simpler production that is closer to consumers—are decreasing, while offshore NG reserves far from the coast and in deep waters are growing [5]. In this regard, oil production with associated NG in deep-waters and ultra-deep-waters usually presents high carbon dioxide (CO2) content and gas-to-oil ratio, which entail difficulties for gas processing in offshore rigs where there are weight/footprint limitations [6]. Due to complex logistics for NG production/transportation in the context of deep-water offshore rigs, high costs lead to significant offshore CO2-rich NG reserves being left unexplored. Albeit the existence of efficient technologies for onshore NG production, innovative low-cost technologies are necessary for deep-waters processing of CO2-rich NG [5].
The concept of carbon capture utilization and storage (CCUS) is necessary to allow a smooth transition from fossil energy to renewable energy to keep a profitable industrial operation. In CO2-rich NG processing, CO2 removed from NG is transported to a storage site [7]. Promising CO2 utilizations are enhanced oil recovery (EOR) in oil-and-gas reservoirs [8] or enhanced gas recovery (EGR) in gas reservoirs [9]. In CCUS-EOR or CCUS-EGR, CO2 is injected into reservoirs to increase oil and gas production. If the increase is high enough, the extra revenues can offset CO2 capture/transportation costs entailing profits [7].
1.1. Natural Gas Purification Requirements
Raw NG primarily contains methane (CH4) and secondarily ethane, propane, butanes, and heavier hydrocarbons. Other common constituents are CO2, hydrogen sulfide (H2S), water (H2O), nitrogen (N2), and trace components. Raw NG purification is necessary to meet gas pipeline specifications and market/environment regulations [10].
NG processing usually comprises: (i) H2S removal [11]; (ii) water dew-point adjustment (WDPA) via dehydration [12]; (iii) hydrocarbon dew-point adjustment (HCDPA) [10]; and (iv) CO2 removal. H2S causes pipeline/equipment corrosion, it is undesirable in combustion, and threatens human health and the environment [11]. WDPA is necessary mainly because water condensed from NG may form solid gas-hydrates under high-pressure and low-temperature typically found in subsea pipelines [12]. HCDPA is necessary to avoid hydrocarbon condensation in pipelines (safety and efficiency issues) and to recover valuable NG liquids [13]. Besides, HCDPA removes hydrocarbons to meet the heating value specification (Wobbe Indexes), ensuring adequate gas–turbine and combustion equipment operations minimizing emissions [5]. Finally, CO2 content is reduced to 2–3 mol%, increasing NG heating value, preventing solid CO2-hydrates, avoiding corrosion, and avoiding occupying pipeline capacity with inert [14].
1.2. Carbon Capture from CO2-Rich NG
Several technologies exist for carbon capture from CO2-rich NG. Pressure-swing adsorption (PSA) prescribes selective CO2 adsorption onto solid adsorbents at high-pressure conjugated with low-pressure CO2 desorption [15]. PSA issues comprehend the methane purity-recovery tradeoff [16] and the low-pressure CO2 release entailing compression costs [17].
Cryogenic distillation is considered for CO2 ≥ 10 mol% due to lesser energy consumption and footprint compared to chemical absorption [18]. However, since cryogenic distillation operates at temperatures below the CO2 triple-point [19], there is a risk of CO2 freeze-out (dry-ice) inside the equipment, causing blockage [18]. Moreover, high-pressure cryogenic columns are protected against pressure surges by blowdown valves, which under cryogenic conditions can lead to dry-ice formation via the Joule–Thomson effect [20].
In offshore CO2-rich NG processing, membrane permeation (MP) is adopted due to its low footprint, modularity, and flexibility to feed composition changes [21] and is recommended for high CO2 content NG [22]. However, thanks to its capacity-selectivity tradeoff [23], permeate methane losses are remarkable, and the low-pressure CO2-rich permeate imposes high compression costs [24].
Physical absorption (PhA) prescribes a physical solvent that dissolves CO2 at high-pressure [25], requiring high CO2 fugacity for high CO2 loadings [26]. PhA is much less energy-intense for solvent regeneration compared to chemical absorption at the expense of a low-pressure CO2 stripping [6]. PhA drawbacks comprise (i) low CO2/CH4 selectivity entailing CH4 losses in the stripped gas; (ii) low-pressure stripped gas entailing compression costs for EOR destination.
The most mature technology for CO2 and H2S removal from CO2-rich NG is chemical absorption (ChA), which is based on CO2 reactions with aqueous-alkanolamine solvents [27]. Aqueous-monoethanolamine (aqueous-MEA) and aqueous-methyl-diethanolamine (aqueous-MDEA) are the most used chemical solvents [28]. The main advantage is the high CO2/CH4 selectivity in a wide range of operation pressures [27] and this selectivity is particularly high at low CO2 fugacity [22]. ChA generally operates with packing columns that have beds of variously-shaped packings—e.g., Pall rings or Berl saddles—providing a high specific surface area for CO2 transfer. Packing columns are fed with lean solvent at the top and with raw NG at the bottom in countercurrent flows [29]. ChA drawbacks comprise: (i) high heat-ratio (kJ/kgCO2) solvent regeneration [28]; (ii) solvent degradation (oxidation and thermal degradation) causing corrosion and efficiency loss; (iii) low-pressure CO2 stripping entailing compression costs for EOR; (iv) hydraulic issues (e.g., foaming, channeling, flooding and entrainment) [29].
The gas-liquid membrane contactor (GLMC) combines MP and ChA advantages—without the respective drawbacks—becoming a promising solution for CO2-rich NG decarbonation [30]. In GLMC, the hollow-fiber membrane (HFM) provides a non-dispersive gas-liquid contact avoiding packing-columns hydraulic problems like foaming, channeling, flooding, and entrainment [31]. Comparatively, with MP, the selective chemical solvent and thousands of HFM inside the GLMC shell ensure high CO2/CH4 selectivity with low CH4 loss, high mass transfer area per shell, compactness, modularity, and easy scale-up, all desired attributes for CO2 removal from CO2-rich NG with aqueous-amine solvents in offshore rigs [22]. Comparatively, with conventional CO2 absorption packing columns, GLMC reaches reductions up to 70% and 66% in size and weight, respectively [32]. Angular indifference is another advantage of GLMC vis-à-vis the sway of floating offshore rigs [33].
2. GLMC: Principles, Materials, and Configurations
GLMC is modular equipment configured in batteries. Each GLMC module contains thousands of hydrophobic HFM tubes fixed into a shell-and-tube arrangement [34] in Figure 1. Equipment inclination translates into GLMC angular indifference [33].
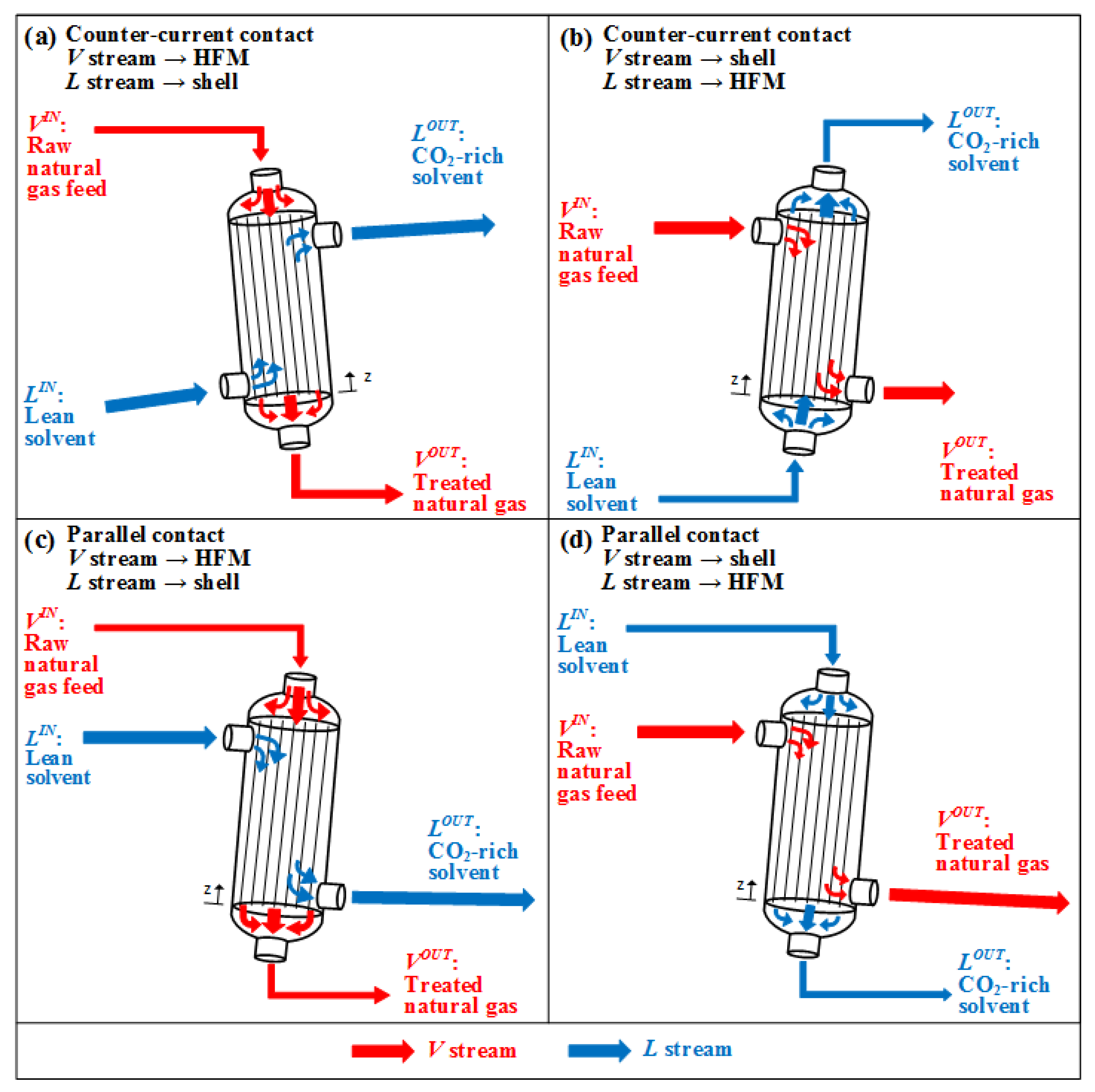
Figure 1. GLMC setups: Counter–current contact: (a) HFM:V, Shell:L; (b) HFM:L, Shell:V. Parallel contact: (c) HFM:V, Shell:L; (d) HFM:L, Shell:V.
GLMC is configurable as an absorber (for CO2 capture) or as a stripper (CO2 stripping for solvent regeneration). In both cases, GLMC has two feeds (where L is the molar flowrate of the GLMC liquid stream and V is the gas counterpart): (i) GLMC-absorber: NG feed (Vinabsorber); lean aqueous-amine solvent (Linabsorber) [35]; (ii) GLMC-stripper: Sweep-gas (nitrogen [36] or steam [37]) (Vinstripper); rich aqueous-amine solvent (Linstripper).
GLMC modules can operate with parallel V and L flows (co-current contact) [38] or opposed V and L flows (counter-current contact) [39]. Whichever the case, V flows inside HFM tubes or on the shell-side, while L respectively flows on the shell-side or inside HFM tubes. Consequently, there are four different GLMC configurations shown in Figure 1: (a,b) apply counter-current contact; (c,d) apply parallel contact; (a,c) have L in shell-side and V inside HFM tubes; (b,d) have L inside HFM tubes and V in shell-side [40]. Since HFM is a physical barrier against phase dispersion, additional phase separation equipment is not necessary [41]. HFM also increases the interfacial area reducing equipment size [42].
GLMC is based on reactive-vapor-liquid-equilibrium (RVLE) since V and L streams are kept in direct contact through the HFM pores and relatively rapid chemical reactions occur in the liquid. In GLMC-absorber with chemical solvent, high CO2/CH4 selectivity is imposed by the aqueous solvent thanks to CH4 hydrophobicity [33] and selective CO2 conversion via liquid phase chemical reactions. When crossing the membranes, CH4 rapidly reaches its low solubility in the aqueous solvent, such that CH4 bubbles as a small vapor phase may form on the liquid side. As absorption chemical reactions between the solvent and CO2 (or H2S) are exothermic, the liquid temperature continuously increases, reducing CH4 solubility and raising CH4 fugacity over the liquid. Therefore, an L stream can be a single-phase liquid or a two-phase stream. Moreover, the CH4 bubbles on the solvent side contribute to stripping some CO2/H2S from the solvent, at the same time raising trans-membrane transport via reduction of CO2/H2S fugacities in the liquid [33].
On the other hand, in GLMC-absorber with physical solvent, CO2 does not react with the solvent, so species mass transfers and CO2/CH4 selectivity depend only on gas diffusivities into the solvent and solute solubilities in the solvent. In this case, CO2 Henry’s Law is frequently used for VLE predictions [43].
In GLMC, convective heat transfers occur due to temperature differences between (i) V and L streams across HFM; (ii) shell-side phase and shell outside. As molar isobaric heat capacity (C¯¯¯P) values imply VC¯¯¯PV<<LC¯¯¯PL, the L stream controls the temperature distribution; consequently, L and V temperature difference is near zero |TV−TL|≈ 0 at a given GLMC axial position due to convective interfacial heat transfer.
Water can transfer L→V or V→L, depending on the interfacial difference of H2O fugacities fˆVH2O− fˆLH2O, influenced by the water content of feeds and absorption heat effects. For counter-current GLMC, the lowest TV, TL values occur at the L inlet, which is also the V outlet position. As V approaches its outlet at water saturation, its temperature lowers and condensation can occur, turning V into a two-phase flow [33]. Additionally, the large absorption heat release can vaporize solvent on the L side [44]. Considering all these thermal effects, both L and V can be single-phase or two-phase streams at some GLMC point [33].
GLMC-stripper regenerates physical solvents (L) with nitrogen or steam as sweep-gas (V). A vacuum-driven pump can also be employed to reduce V pressure. Since CO2/H2S fugacities in the sweep-gas are lower than in the solvent, CO2/H2S move L→V enabling separation without heat supply [37]. As Figure 2 shows, CO2/H2S move V→L in GLMC-absorber and L→V in GLMC-stripper, while H2O moves V→L or L→V whether fˆVH2O− fˆLH2O>0 or fˆVH2O− fˆLH2O<0. N2 transfer in GLMC-stripper is negligible since N2 is highly hydrophobic [36].

Figure 2. Multicomponent mass transfers: GLMC-absorber (a); GLMC-stripper (b) (H2O can move V→L or L→V).
2.1. HFM for GLMC
Contrarily to MP, which uses dense selective membranes, GLMC adopts non-selective microporous HFM to allow V-L contact and selectivity is imposed by the solvent [45]. Figure 3 depicts how HFM works as a mass transfer barrier preventing phase dispersion [46]. Figure 3a represents direct V-L contact in tray columns or packing columns with phase dispersion. Figure 3b shows microporous HFM with gas-filled pores ensuring V-L contact with low mass transfer resistance [47]. However, during GLMC operation, the solvent may fill the pores (Figure 3c), increasing mass transfer resistance drastically and reducing transfer fluxes. This undesirable phenomenon compromises GLMC performance and is called membrane pore wetting (MPW) [46]. MPW is influenced by membrane characteristics (pores diameter, tortuosity, porosity, surface roughness), solvent surface tension, and operating conditions, while some solvents may favor MPW by affecting HFM hydrophobicity, morphology, and roughness. To avoid MPW, HFM material must be hydrophobic such as polypropylene (PP), polyvinylidene fluoride (PVDF), polytetrafluorethylene (PTFE), polyether ether ketone (PEEK), and solvents should have high surface tension, negligible vapor pressure and should not chemically attack HFM [48]. Figure 3d shows another solution against MPW wherein dense HFM CO2 permeable and solvent impermeable are used. However, dense HFM presents high transfer resistance and does not fit in the original GLMC concept of non-dispersive V-L contact [46]. A compromise solution uses composite HFM (Figure 3e) consisting of an ultrathin dense skin layer onto microporous support. The ultrathin layer is generally a fluorine-based hydrophobic material, while the microporous support—made of a cheaper material like PP—should be mechanically resistant, chemically stable, and highly porous to minimize transfer resistance [46].

Figure 3. HFM as a barrier for mass transfer without phase dispersion: (a) direct V-L contact with phase dispersion; (b) microporous HFM V-L contact with gas-filled pores; (c) microporous HFM V-L contact with liquid-filled pores; (d) Dense HFM without V-L contact; (e) Composite HFM without V-L contact.
2.2. Solvents for GLMC CO2 Absorption
Hereafter, physical and chemical GLMC solvents are discussed.
2.2.1. Physical Solvents
PhA relies on CO2 solubility, which follows Henry’s law, such that the absorption loading is favored by high CO2 fugacity and low temperature. Thus, PhA can perform CO2 removal from high-pressure raw NG. Comparatively to ChA, the main PhA advantage is the lower heat ratio for solvent regeneration [49]. Physical solvents are listed in Table 1 for GLMC CO2 removal from NG.
Table 1. Physical solvents proposed for GLMC CO2 removal from NG.
Some promising GLMC physical solvents are ionic liquids, which are salts of organic cations with organic anions. Ionic liquids exhibit melting points below 100 °C, negligible vapor pressure, high CO2 solubility, and thermal stability but present high viscosity as a disadvantage [55].
2.2.2. Chemical Solvents
ChA is characterized by high CO2/CH4 selectivity and limited absorption loadings (kgCO2/kgSolvent), both due to chemical reactions of the solvent with CO2. Aqueous-amine solvents are the most used in ChA for CO2 capture from NG due to high reactivity, low cost, easy regeneration and high CO2/CH4 selectivity [56]. Aqueous-MEA, aqueous-DEA (aqueous-diethanolamine) and aqueous-MDEA are examples of aqueous-amine solvents extensively employed for NG decarbonation since 1930 [57]. However, aqueous-amines exhibit drawbacks such as high heat-ratio (kJ/kgCO2) solvent regeneration, thermal/oxidative degradation, corrosiveness and limited CO2 loading [58].
Aqueous-MEA is the most used for ChA CO2 capture, exhibiting high reactivity at low-pressure or high-pressure [59], while aqueous-MDEA is adequate at high pressure despite lower absorption rate [60], which recommends using reactivity promoters. Table 2 lists literature studies on chemical solvents for GLMC NG decarbonation.
Table 2. Chemical solvents investigated for GLMC CO2 removal from NG.
| Chemical Solvents | Reference |
|---|---|
| Primary amines | |
| Aqueous-MEA | [61] |
| Aqueous-DGA (Aqueous-Diglycolamine) | [62] |
| Secondary amines | |
| Aqueous-DEA | [63] |
| Aqueous-DIPA (Aqueous-Diisopropanolamine) | [64] |
| Aqueous-PZ (Aqueous-Piperazine) | [65] |
| Aqueous-EMEA (Aqueous-N-ethyl-monoethanolamine) | [66] |
| Tertiary amines | |
| Aqueous-TEA (Aqueous-Triethanolamine) | [65] |
| Aqueous-MDEA | [67] |
| Sterically hindered amines | |
| Aqueous-AMP (Aqueous-2-amino-2-methyl-1-propanol) | [68] |
| Alkali-salts | |
| Aqueous-K2CO3 | [69] |
| Alkali | |
| Aqueous-NaOH | [70] |
| Amino-acid salts | |
| Aqueous-AAS | [71] |
2.2.3. Solvent Blends
The literature presents CO2 capture studies (for GLMC or not) with several blended solvents [72], namely: (i) aqueous-MDEA-PZ (PZ promoter) [73]; (ii) aqueous-MEA-MDEA, aqueous-MEA-DIPA and several combinations of aqueous-amines [74]; (iii) aqueous-PZ-K2CO3 (PZ promoter) [75]. Physical and chemical solvents are also blended, such as NMP with aqueous-ethanolamine [26] and ionic-liquid with aqueous-ethanolamine [76].
References
- Sedej, O.; Mbonimpa, E. CFD Modeling of a Lab-Scale Microwave Plasma Reactor for Waste-to-Energy Applications: A Review. Gases 2021, 1, 11.
- Günther, M.; Nissen, V. Effects of the Henry Hub Price on U.S. LNG exports and on gas flows in Western Europe. Gases 2021, 1, 6.
- Chandrasekar, A.; Syron, E. Evaluation of Heat Decarbonation Strategies and Their Impact on the Irish Gas Network. Gases 2021, 1, 14.
- Shahid, M.Z.; Maulud, A.S.; Bustam, M.A.; Suleman, H.; Halim, H.N.A.; Shariff, A.M. Packed column modelling and experimental evaluation for CO2 absorption using MDEA solution at high pressure and high CO2 concentrations. J. Nat. Gas Sci. Eng. 2021, 88, 103829.
- Mokhatab, S.; Poe, W.A.; Mak, J.Y. Handbook of Natural Gas Transmission and Processing—Principles and Practices, 3rd ed.; Gulf Professional Publishing-Elsevier: Waltham, MA, USA, 2015; pp. 2–125.
- Araújo, O.Q.F.; Reis, A.C.; de Medeiros, J.L.; do Nascimento, J.F.; Grava, W.M.; Musse, A.P.S. Comparative analysis of separation technologies for processing carbon dioxide rich natural gas in ultra-deepwater oil fields. J. Clean. Prod. 2017, 155, 12–22.
- Rakhiemah, A.N.; Xu, Y. Economic viability of full-chain CCUS-EOR in Indonesia. Resour. Conserv. Recyl. 2022, 179, 106069.
- Amarasinghe, W.; Farzaneh, S.; Fjelde, I.; Sohrabi, M.; Guo, Y. A visual investigation of CO2 convective mixing in water and oil at the pore scale using a micromodel apparatus at reservoir conditions. Gases 2021, 1, 5.
- Liu, S.; Yuan, L.; Liu, W.; Zhao, C.; Zhang, Y.; Song, Y. Study on the influence of various factors on dispersion during enhance natural gas recovery with CO2 sequestration in depleted gas reservoir. J. Nat. Gas Sci. Eng. 2022, 103, 104644.
- Egging-Bratseth, R. A techno-economic perspective on natural gas and its value chain. Gases 2021, 1, 1.
- Shi, M.; Xiong, W.; Zhang, X.; Ji, J.; Hu, X.; Tu, Z.; Wu, Y. Highly efficient and selective H2S capture by task-specific deep eutectic solvents through chemical dual-site absorption. Sep. Purif. Technol. 2022, 283, 120167.
- Trueba, L., Jr.; Gaston, T.; Brackin, J.; Miller, J.; You, B.H. Effective strategies to reduce triethylene glycol consumption in natural gas processing plants. Case Stud. Chem. Environ. Eng. 2022, 5, 100196.
- Shamsi, M.; Farokhi, S.; Pourghafari, M.; Bayat, A. Tuning the natural gas dew poInt. by Joule-Thomson and Mechanical Refrigeration processes: A comparative energy and exergy analysis. J. Petrol. Sci. Eng. 2022, 212, 110270.
- Ecker, A.M.; Klein, H.; Peschel, A. Systematic and efficient optimization-based design of a process for CO2 removal from natural gas. Chem. Eng. J. 2022, 445, 136178.
- Abd, A.A.; Othman, M.R. Biogas upgrading to fuel grade methane using pressure swing adsorption: Parametric sensitivity analysis on an industrial scale. Fuel 2022, 308, 121986.
- Abdullah, A.; Idris, I.; Shamsudin, I.K.; Othman, M.R. Methane enrichment from high carbon dioxide content natural gas by pressure swing adsorption. J. Nat. Gas Sci. Eng. 2019, 69, 102929.
- Rebello, C.M.; Martins, M.A.F.; Rodrigues, A.E.; Loureiro, J.M.; Ribeiro, A.M.; Nogueira, I.B.R. A novel standpoInt. of Pressure Swing Adsorption processes multi-objective optimization: An approach based on feasible operation region mapping. Chem. Eng. Res. Des. 2022, 178, 590–601.
- Park, K.H.; Lee, J.W.; Lim, Y.; Seo, Y. Life cycle cost analysis of CO2 compression processes coupled with a cryogenic distillation unit for purifying high-CO2 natural gas. J. CO2 Util. 2022, 60, 102002.
- Baccanelli, M.; Langé, S.; Rocco, M.V.; Pellegrini, L.A.; Colombo, E. Low temperature techniques for natural gas purification and LNG production: An energy and exergy analysis. Appl. Energy 2016, 180, 546–559.
- Shafiq, U.; Shariff, A.M.; Babar, M.; Azeem, B.; Ali, A.; Bustam, A. Study of dry ice formation during blowdown of CO2-CH4 from cryogenic distillation column. J. Loss Prev. Proc. 2020, 64, 104073.
- Arinelli, L.O.; de Medeiros, J.L.; de Melo, D.C.; Teixeira, A.M.; Brigagão, G.V.; Passarelli, F.M.; Grava, W.M.; Araújo, O.Q.F. Carbon capture and high-capacity supercritical fluid processing with supersonic separator: Natural gas with ultra-high CO2 content. J. Nat. Gas Sci. Eng. 2019, 66, 265–283.
- Baker, R.W.; Lokhandwala, K. Natural Gas Processing with Membranes: An Overview. Ind. Eng. Chem. Res. 2008, 47, 2109–2121.
- Law, L.C.; Ng, J.J.Y.; Hasan, M.M.; Othman, M.R.; Helwani, Z.; Idroes, R. Effects of membrane selectivity and configuration on methane purity and recovery from high carbon dioxide content natural gas. J. Nat. Gas Sci. Eng. 2021, 89, 103882.
- Siagian, U.W.R.; Raksajati, A.; Himma, N.F.; Khoiruddin, K.; Wenten, I.G. Membrane-based carbon capture technologies: Membrane gas separation vs. membrane contactor. J. Nat. Gas Sci. Eng. 2019, 67, 172–195.
- Shokrollahi, F.; Lau, K.K.; Partoon, B.; Smith, A.M. A review on the selection criteria for slow and medium kinetic solvents used in CO2 absorption for natural gas purification. J. Nat. Gas Sci. Eng. 2022, 98, 104390.
- Tan, L.S.; Shariff, A.M.; Lau, K.K.; Bustam, M.A. Impact of high pressure on high concentration carbon dioxide capture from natural gas by monoethanolamine/N-methyl-2-pyrrolidone solvent in absorption packed column. Int. J. Greenh. Gas Control 2015, 34, 25–30.
- Araújo, O.Q.F.; de Medeiros, J.L. Carbon capture and storage technologies: Present scenario and drivers of innovation. Curr. Opin. Chem. Eng. 2017, 17, 22–34.
- Kazmi, B.; Raza, F.; Taqvi, S.A.A.; Awan, Z.U.H.; Ali, S.I.; Suleman, H. Energy, exergy and economic (3E) evaluation of CO2 capture from natural gas using pyridinium functionalized ionic liquids: A simulation study. J. Nat. Gas Sci. Eng. 2021, 90, 103951.
- Tay, W.H.; Lau, K.K.; Lai, L.S.; Shariff, A.M.; Wang, T. Current development and challenges in the intensified absorption technology for natural gas purification at offshore condition. J. Nat. Gas Sci. Eng. 2019, 71, 102977.
- Mansourizadeh, A.; Rezaei, I.; Lau, W.J.; Seah, M.Q.; Ismail, A.F. A review on recent progress in environmental applications of membrane contactor technology. J. Environ. Chem. Eng. 2022, 10, 107631.
- Sabzekar, M.; Chenar, M.P.; Khayet, M.; García-Payo, C.; Maghsoud, Z.; Pagliero, M. Cyclic olefin polymer membrane as an emerging material for CO2 capture in gas-liquid membrane contactor. J. Environ. Chem. Eng. 2022, 10, 107669.
- Amaral, R.A.; Habert, C.A.; Borges, C.P. Performance evaluation of composite and microporous gas-liquid membrane contactors for CO2 removal from a gas mixture. Chem. Eng. Process. Process Intensif. 2016, 102, 202–209.
- de Medeiros, J.L.; Nakao, A.; Grava, W.M.; Nascimento, J.F.; Araújo, O.Q.F. Simulation of an Offshore Natural Gas Purification Process for CO2 Removal with Gas-Liquid Contactors Employing Aqueous Solutions of Ethanolamines. Ind. Eng. Chem. Res. 2013, 52, 7074–7089.
- Gabelman, A.; Hwang, S.T. Hollow fiber membrane contactors. J. Membr. Sci. 1999, 159, 61–106.
- Zhang, H.; Xue, K.; Cheng, C.; Gao, D.; Chen, H. Study on the performance of CO2 capture from flue gas with ceramic membrane contactor. Sep. Purif. Technol. 2021, 265, 118521.
- Rahbari-Sisakht, M.; Rana, D.; Matsuura, T.; Emadzadeh, D.; Padaki, M.; Ismail, A.F. Study on CO2 stripping from water through novel surface modified PVDF hollow fiber membrane contactor. Chem. Eng. J. 2014, 246, 306–310.
- Scholes, C.A.; Kentish, S.E.; Qader, A. Membrane gas-solvent contactor pilot plant trials for post-combustion CO2 capture. Sep. Purif. Technol. 2020, 237, 116470.
- Zhang, H.Y.; Wang, R.; Liang, D.T.; Tay, J.H. Modeling and experimental study of CO2 absorption in a hollow fiber membrane contactor. J. Membr. Sci. 2006, 279, 301–310.
- Al-Saffar, H.B.; Ozturk, B.; Hughes, R. A comparison of porous and non-porous gas-liquid membrane contactors for gas separation. Chem. Eng. Res. Des. 1997, 75, 685–692.
- Cesari, L.; Castel, C.; Favre, E. Membrane contactors for intensified gas-liquid absorption processes with physical solvents: A critical parametric study. J. Membr. Sci. 2021, 635, 119377.
- Babin, A.; Bougie, F.; Rodrigue, D.; Iliuta, M.C. A closer look on the development and commercialization of membrane contactors for mass transfer and separation processes. Sep. Purif. Technol. 2019, 227, 115679.
- Reed, B.W.; Semmens, M.J.; Cussler, E.L. Membrane contactors. In Membrane Separations Technology: Principles and Applications; Noble, R.D., Stern, S.A., Eds.; Elsevier Science B.V.: New York, NY, USA, 1995; Volume 2, pp. 467–498.
- Tantikhajorngosol, P.; Laosiripojana, N.; Jiraratananon, R.; Assabumrungrat, S. Physical absorption of CO2 and H2S from synthetic biogas at elevated pressures. using hollow fiber membrane contactors: The effects of Henry’s constants and gas diffusivities. Int. J. Heat Mass Transf. 2019, 128, 1136–1148.
- Ghasem, N.; Al-Marzouqi, M.; Rahim, N.A. Modeling of CO2 absorption in a membrane contactor considering solvent evaporation. Sep. Purif. Technol. 2013, 110, 1–10.
- Keshavarz, P.; Fathikalajahi, J.; Ayatollahi, S. Analysis of CO2 separation and simulation of a partially wetted hollow fiber membrane contactor. J. Hazard. Mater. 2008, 152, 1237–1247.
- Nguyen, P.T.; Lasseuguette, E.; Medina-Gonzalez, Y.; Remigy, J.C.; Roizard, D.; Favre, E. A dense membrane contactor for intensified CO2 gas/liquid absorption in post-combustion capture. J. Membr. Sci. 2011, 377, 261–272.
- Karoor, S.; Sirkar, K.K. Gas Absorption Studies in Microporous Hollow Fiber Membrane Modules. Ind. Eng. Chem. Res. 1993, 32, 674–684.
- Zhao, S.; Feron, P.H.M.; Deng, L.; Favre, E.; Chabanon, E.; Yan, S.; Hou, J.; Chen, V.; Qi, H. Status and progress of membrane contactors in post-combustion carbon capture: A state-of-the-art review of new developments. J. Membr. Sci. 2016, 511, 180–206.
- Chen, W.H.; Chen, S.M.; Hung, C.I. Carbon dioxide capture by single droplet using Selexol, Rectisol and water as absorbents: A theoretical approach. Appl. Energy 2013, 111, 731–741.
- Al-Marzouqi, M.H.; El-Naas, M.H.; Marzouk, S.A.M.; Al-Zarooni, M.A.; Abdullatif, N.; Faiz, R. Modeling of CO2 absorption in membrane contactors. Sep. Purif. Technol. 2008, 59, 286–293.
- Xiang, L.; Wu, L.; Gao, L.; Chen, J.; Liu, Y.; Zhao, H. Pilot scale applied research on CO2 removal of natural gas using a rotating packed bed with propylene carbonate. Chem. Eng. Res. Des. 2019, 150, 33–39.
- Roussanaly, S.; Anantharaman, R.; Lindqvist, K. Multi-criteria analyses of two solvent and one low-temperature concepts for acid gas removal from natural gas. J. Nat. Gas Sci. Eng. 2014, 20, 38–49.
- Hughes, T.J.; Kandil, M.E.; Graham, B.F.; May, E.F. Simulating the capture of CO2 from natural gas: New data and improved models for methane + carbon dioxide + methanol. Int. J. Greenh. Gas Control 2014, 31, 121–127.
- Sohaib, Q.; Vadillo, J.M.; Gómez-Coma, L.; Albo, J.; Druon-Bocquet, S.; Irabien, A.; Sanchez-Marcano, J. CO2 capture with room temperature ionic liquids; coupled absorption/desorption and single module absorption in membrane contactor. Chem. Eng. Sci. 2020, 223, 115719.
- Swati, I.K.; Sohaib, Q.; Khan, H.; Younas, M.; Monjezi, A.H.; Li, J.; Rezakazemi, M. Non-dispersive solvent absorption of post-combustion CO2 in membrane contactors using ionic liquids. J. Mol. Liq. 2022, 351, 118566.
- Mandal, B.; Bandyopadhyay, S.S. Simultaneous Absorption of CO2 and H2S Into Aqueous Blends of N-Methyldiethanolamine and Diethanolamine. Environ. Sci. Technol. 2006, 40, 6076–6084.
- Cleeton, C.; Kvam, O.; Rea, R.; Sarkisov, L.; De Angelis, M.G. Competitive H2S–CO2 absorption in reactive aqueous methyldiethanolamine solution: Prediction with ePC-SAFT. Fluid Phase Equilibria 2020, 511, 112453.
- Khan, S.N.; Hailegiorgis, S.M.; Man, Z.; Shariff, A.M.; Garg, S. Thermophysical properties of concentrated aqueous solution of N-methyldiethanolamine (MDEA), piperazine (PZ), and ionic liquids hybrid solvent for CO2 capture. J. Mol. Liq. 2017, 229, 221–229.
- Lepaumier, H.; Picq, D.; Carrette, P.L. New Amines for CO2 Capture. I. Mechanisms of Amine Degradation in the Presence of CO2. Ind. Eng. Chem. Res. 2009, 48, 9061–9067.
- Kiani, S.; Taghizade, A.; Ramezani, R.; Di Felice, R.; Molelekwa, G.F.; Mazinani, S. Enhancement of CO2 removal by promoted MDEA solution in a hollow fiber membrane contactor: A numerical and experimental study. Carbon Capture Sci. Technol. 2022, 2, 100028.
- Faiz, R.; Al-Marzouqi, M. Mathematical modeling for the simultaneous absorption of CO2 and H2S using MEA in hollow fiber membrane contactors. J. Membr. Sci. 2009, 342, 269–278.
- Zahid, U.; Al Rowaili, F.N.; Ayodeji, M.K.; Ahmed, U. Simulation and parametric analysis of CO2 capture from natural gas using diglycolamine. Int. J. Greenh. Gas Control 2017, 57, 42–51.
- Keshavarz, P.; Fathikalajahi, J.; Ayatollahi, S. Mathematical modeling of the simultaneous absorption of carbon dioxide and hydrogen sulfide in a hollow fiber membrane contactor. Sep. Purif. Technol. 2008, 63, 145–155.
- Razavi, S.M.R.; Shirazian, S.; Najafabadi, M.S. Investigations on the Ability of Di-Isopropanol Amine Solution for Removal of CO2 From Natural Gas in Porous Polymeric Membranes. Polym. Eng. Sci. 2015, 55, 598–603.
- Abdolahi-Mansoorkhani, H.; Seddighi, S. CO2 capture by modified hollow fiber membrane contactor: Numerical study on membrane structure and membrane wettability. Fuel Process. Technol. 2020, 209, 106530.
- Sutar, P.N.; Jha, A.; Vaidya, P.D.; Kenig, E.Y. Secondary amines for CO2 capture: A kinetic investigation using N-ethylmonoethanolamine. Chem. Eng. J. 2012, 207–208, 718–724.
- Rezakazemi, M.; Niazi, Z.; Mirfendereski, M.; Shirazian, S.; Mohammadi, T.; Pak, A. CFD simulation of natural gas sweetening in a gas-liquid hollow-fiber membrane contactor. Chem. Eng. J. 2011, 168, 1217–1226.
- Boucif, N.; Favre, E.; Roizard, D. CO2 capture in HFMM contactor with typical amine solutions: A numerical analysis. Chem. Eng. Sci. 2008, 63, 5375–5385.
- Nakhjiri, A.T.; Heydarinasab, A.; Bakhtiari, O.; Mohammadi, T. Numerical simulation of CO2/H2S simultaneous removal from natural gas using potassium carbonate aqueous solution in hollow fiber membrane contactor. J. Environ. Chem. Eng. 2020, 8, 104130.
- Hatab, F.A.; Abdullatif, N.; Marzouk, S.A.M.; Al-Marzouqi, M.H. Experimental and modeling of CO2 removal from gas mixtuRes. using membrane contactors packed with glass beads. Sep. Purif. Technol. 2019, 217, 240–246.
- Rahim, N.A.; Ghasem, N.; Al-Marzouqi, M. Absorption of CO2 from natural gas using different amino acid salt solutions and regeneration using hollow fiber membrane contactors. J. Nat. Gas Sci. Eng. 2015, 26, 108–117.
- Magnone, E.; Lee, H.J.; Shin, M.C.; Park, J.H. A performance comparison study of five single and sixteen blended amine absorbents for CO2 capture using ceramic hollow fiber membrane contactors. J. Ind. Eng. Chem. 2021, 100, 174–185.
- Kang, G.; Chan, Z.P.; Saleh, S.B.M.; Cao, Y. Removal of high concentration CO2 from natural gas using high pressure membrane contactors. Int. J. Greenh. Gas Control 2017, 60, 1–9.
- Glasscock, D.A.; Critchfield, J.E.; Rochelle, G.T. CO2 absorption/desorption in mixtuRes. of methyldiethanolamine with monoethanolamine or diethanolamine. Chem. Eng. Sci. 1991, 46, 2829–2845.
- Mesbah, M.; Momeni, M.; Soroush, E.; Shahsavari, S.; Galledari, S.A. Theoretical study of CO2 separation from CO2/CH4 gaseous mixture using 2-methylpiperazine-promoted potassium carbonate through hollow fiber membrane contactor. J. Environ. Chem. Eng. 2019, 7, 102781.
- Lu, J.G.; Lu, Z.Y.; Chen, Y.; Wang, J.T.; Gao, L.; Gao, X.; Tang, Y.Q.; Liu, D.G. CO2 absorption into aqueous blends of ionic liquid and amine in a membrane contactor. Sep. Purif. Technol. 2015, 150, 278–285.
More
Information
Subjects:
Energy & Fuels
Contributors
MDPI registered users' name will be linked to their SciProfiles pages. To register with us, please refer to https://encyclopedia.pub/register
:
View Times:
1.4K
Revisions:
2 times
(View History)
Update Date:
19 Sep 2022
Notice
You are not a member of the advisory board for this topic. If you want to update advisory board member profile, please contact office@encyclopedia.pub.
OK
Confirm
Only members of the Encyclopedia advisory board for this topic are allowed to note entries. Would you like to become an advisory board member of the Encyclopedia?
Yes
No
${ textCharacter }/${ maxCharacter }
Submit
Cancel
Back
Comments
${ item }
|
More
No more~
There is no comment~
${ textCharacter }/${ maxCharacter }
Submit
Cancel
${ selectedItem.replyTextCharacter }/${ selectedItem.replyMaxCharacter }
Submit
Cancel
Confirm
Are you sure to Delete?
Yes
No





