
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | George Saitakis | -- | 4142 | 2022-09-17 10:36:05 | | | |
| 2 | Vivi Li | Meta information modification | 4142 | 2022-09-19 03:24:14 | | |
Video Upload Options
Optic neuritis (ON) is an inflammatory condition involving the optic nerve. Several important typical and atypical ON variants are now recognized. Typical ON has a more favorable prognosis; it can be idiopathic or represent an early manifestation of demyelinating diseases, mostly multiple sclerosis (MS). The atypical spectrum includes entities such as antibody-driven ON associated with neuromyelitis optica spectrum disorder (NMOSD) and myelin oligodendrocyte glycoprotein antibody disease (MOGAD), chronic/relapsing inflammatory optic neuropathy (CRION), and sarcoidosis-associated ON. Appropriate and timely diagnosis is essential to rapidly decide on the appropriate treatment, maximize visual recovery, and minimize recurrences.
1. Introduction
2. Typical Optic Neuritis
3. Pathophysiology of ON
4. Acute Treatment of Typical Optic Neuritis/Clinically Isolated Syndrome
5. Long-Term Treatment: Immune Prophylaxis against Optic Neuritis Relapses/Progression to Multiple Sclerosis
5.1. Mechanisms of Action in Interferon β in MS and Optic Neuritis
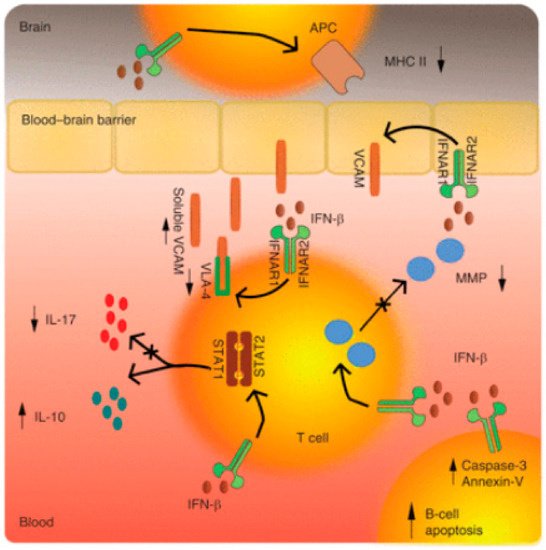

5.2. Glatiramer Acetate (GA)
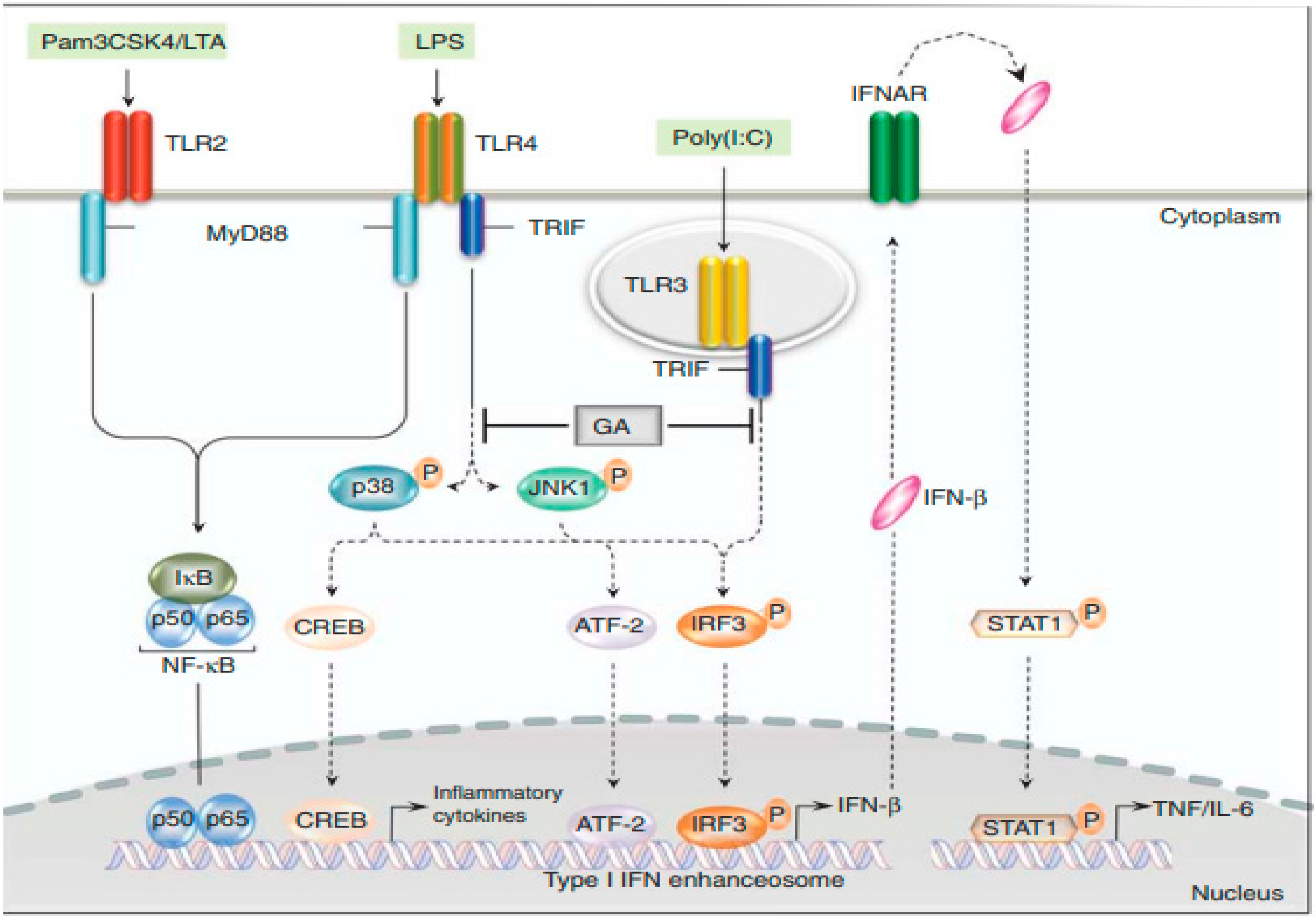

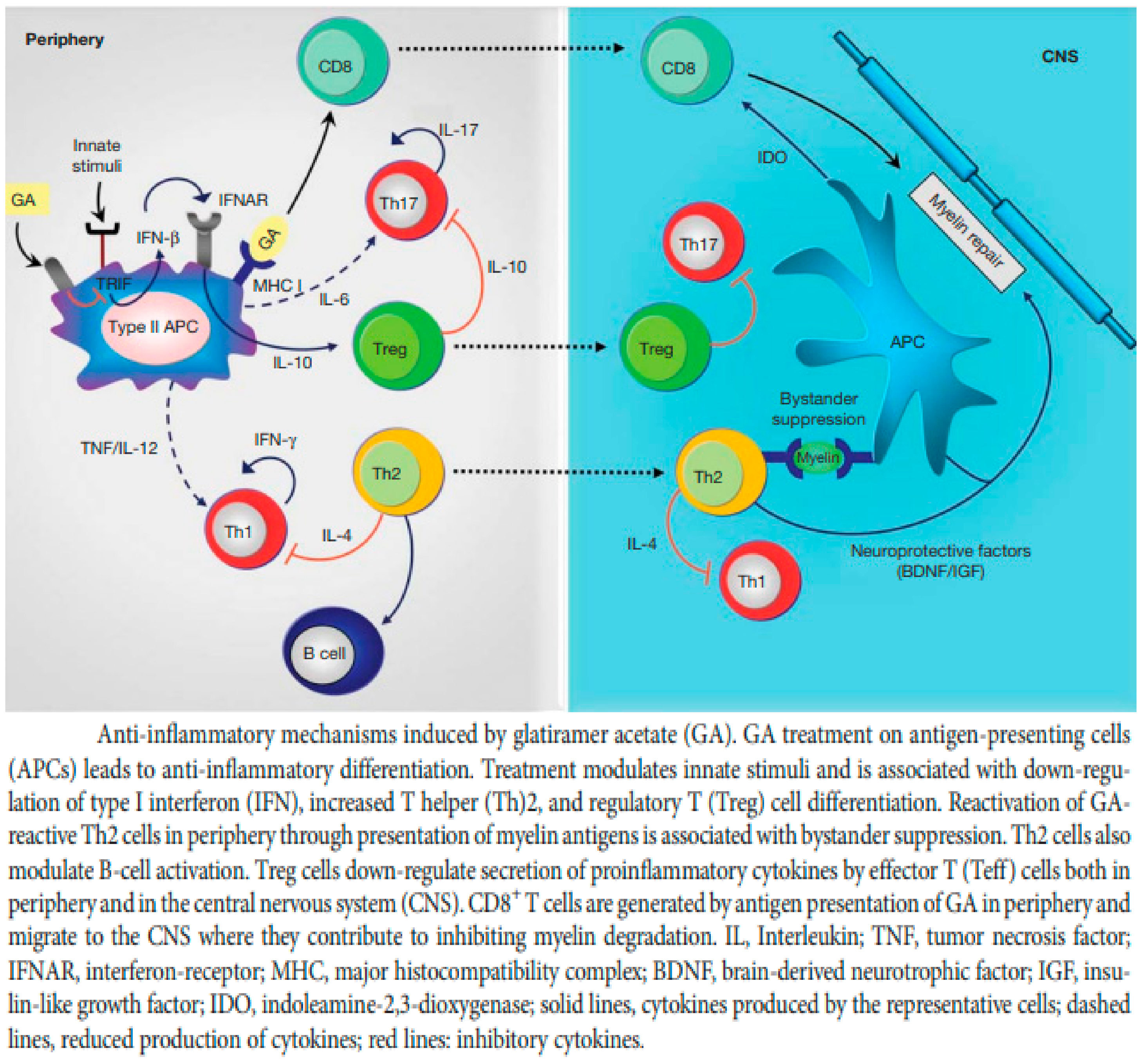
5.3. Treatment of Clinically Isolated Syndromes
References
- Menon, V.; Saxena, R.; Misra, R.; Phuljhele, S. Management of optic neuritis. Indian J. Ophthalmol. 2011, 59, 117–122.
- Morrow, S.A.; Fraser, J.A.; Day, C.; Bowman, D.; Rosehart, H.; Kremenchutzky, M.; Nicolle, M. Effect of treating acute optic neuritis with bioequivalent oral vs intravenous corticosteroids—A randomized clinical trial. JAMA Neurol. 2018, 75, 690–696.
- Huang, W.J.; Chen, W.W.; Zhang, X. Multiple sclerosis: Pathology, diagnosis and treatments. Exp. Ther. Med. 2017, 13, 3163–3166.
- Compston, A.; Coles, A. Multiple sclerosis. Lancet 2008, 372, 1502–1517.
- Wingerchuk, D.; Lucchinetti, C.; Noseworthy, J. Multiple Sclerosis: Current Pathophysiological Concepts. Lab. Investig. 2001, 81, 263–281.
- Lucchinetti, C.; Brück, W.; Parisi, J.; Scheithauer, B.; Rodriguez, M.; Lassmann, H. Heterogeneity of multiple sclerosis lesions: Implications for the pathogenesis of demyelination. Ann. Neurol. 2000, 47, 707–717.
- Traugott, U.; Reinherz, E.L.; Raine, C.S. Multiple sclerosis. Distribution of T cells, T cell subsets and Ia-positive macrophages in lesions of different ages. J. Neuroimmunol. 1983, 4, 201–221.
- Ferguson, B.; Matyszak, M.K.; Esiri, M.M.; Perry, V.H. Axonal damage in acute multiple sclerosis lesions. Brain 1997, 120, 393–399.
- Bitsch, A.; Schuchardt, J.; Bunkowski, S.; Kuhlmann, T.; Brück, W. Acute axonal injury in multiple sclerosis. Correlation with demyelination and inflammation. Brain 2000, 123, 1174–1183.
- Hu, D.; Ikizawa, K.; Lu, L.; Sanchirico, M.E.; Shinohara, M.L.; Cantor, H. Analysis of regulatory CD8 T cells in Qa-1-deficient mice. Nat. Immunol. 2004, 5, 516–523.
- Toosy, A.T.; Mason, D.F.; Miller, D.H. Optic neuritis. Lancet Neurol. 2014, 13, 83–99.
- Babbe, H.; Roers, A.; Waisman, A.; Lassmann, H.; Goebels, N.; Hohlfeld, R.; Friese, M.; Schröder, R.; Deckert, M.; Schmidt, S.; et al. Clonal expansions of CD8(+) T cells dominate the T cell infiltrate in active multiple sclerosis lesions as shown by micromanipulation and single cell polymerase chain reaction. J. Exp. Med. 2000, 192, 393–404.
- Carlström, K.E.; Zhu, K.; Ewing, E.; Krabbendam, I.E.; Harris, R.A.; Falcão, A.M.; Jagodic, M.; Castelo-Branco, G.; Piehl, F. Gsta4 controls apoptosis of differentiating adult oligodendrocytes during homeostasis and remyelination via the mitochondria-associated Fas-Casp8-Bid-axis. Nat. Commun. 2020, 11, 4071.
- Chamberlain, K.A.; Chapey, K.S.; Nanescu, S.E.; Huang, J.K. Creatine enhances mitochondrial-mediated oligodendrocyte survival after demyelinating injury. J. Neurosci. 2017, 37, 1479–1492.
- Jin, J.; Smith, M.D.; Kersbergen, C.J.; Kam, T.-I.; Viswanathan, M.; Martin, K.; Dawson, T.M.; Dawson, V.L.; Zack, D.J.; Whartenby, K.; et al. Glial pathology and retinal neurotoxicity in the anterior visual pathway in experimental autoimmune encephalomyelitis. Acta Neuropathol. Commun. 2019, 7, 125.
- Cannella, B.; Raine, C.S. The adhesion molecule and cytokine profile of multiple sclerosis lesions. Ann. Neurol. 1995, 37, 424–435.
- Kuhlmann, T.; Miron, V.; Cui, Q.; Wegner, C.; Antel, J.; Brück, W. Differentiation block of oligodendroglial progenitor cells as a cause for remyelination failure in chronic multiple sclerosis. Brain 2008, 131 Pt 7, 1749–1758.
- Goodkin, D.E. The Natural History of Multiple Sclerosis. In Treatment of Multiple Sclerosis; Clinical Medicine and the Nervous System; Rudick, R.A., Goodkin, D.E., Eds.; Springer: London, UK, 1992.
- Koopmans, R.A.; Li, D.K.B.; Oger, J.J.F.; Kastrukoff, L.F.; Jardine, C.; Costley, L.; Hall, S.; Grochowski, E.W.; Paty, D.W. Chronic progressive multiple sclerosis: Serial magnetic resonance brain imaging over six months. Ann. Neurol. 1989, 26, 248–256.
- Prineas, J.W.; Barnard, R.O.; Kwon, E.E.; Sharer, L.R.; Cho, E.S. Multiple sclerosis: Remyelination of nascent lesions. Ann. Neurol. 1993, 33, 137–151.
- Lotan, I.; Hellmann, M.A.; Benninger, F.; Stiebel-Kalish, H.; Steiner, I. Recurrent optic neuritis—Different patterns in multiple sclerosis, neuromyelitis optica spectrum disorders and MOG-antibody disease. J. Neuroimmunol. 2018, 324, 115–118.
- Burman, J.; Raininko, R.; Fagius, J. Bilateral and recurrent optic neuritis in multiple sclerosis. Acta Neurol. Scand. 2011, 123, 207–210.
- Quintana, F.J.; Patel, B.; Yeste, A.; Nyirenda, M.; Kenison, J.; Rahbari, R.; Fetco, D.; Hussain, M.; O’Mahony, J.; Magalhaes, S.; et al. Canadian Pediatric Demyelinating Disease Network. Epitope spreading as an early pathogenic event in pediatric multiple sclerosis. Neurology 2014, 83, 2219–2226.
- Frohman, E.M.; Racke, M.K.; Raine, C.S. Multiple sclerosis—The plaque and its pathogenesis. N. Engl. J. Med. 2006, 354, 942–955.
- Qin, Y.; Duquette, P.; Zhang, Y.; Talbot, P.; Poole, R.; Antel, J. Clonal expansion and somatic hypermutation of V (H) genes of B cells from cerebrospinal fluid in multiple sclerosis. J. Clin. Investig. 1998, 102, 1045–1050.
- Noseworthy, J.H.; Lucchinetti, C.; Rodriguez, M.; Weinshenker, B.G. Multiple sclerosis. N. Engl. J. Med. 2000, 343, 938–952.
- Sriram, S.; Steiner, I. Experimental allergic encephalomyelitis: A misleading model of multiple sclerosis. Ann. Neurol. 2005, 58, 939–945.
- Hellmann, M.A.; Steiner, I.; Mosberg-Galili, R. Sudden sensorineural hearing loss in multiple sclerosis: Clinical course and possible pathogenesis. Acta Neurol. Scand. 2011, 124, 245–249.
- Lemus, H.N.; Warrington, A.E.; Rodriguez, M. Multiple Sclerosis: Mechanisms of Disease and Strategies for Myelin and Axonal Repair. Neurol. Clin. 2018, 36, 1–11.
- Nancy, J.; Newman, M.D. Atlanta, Georgia, the Optic Neuritis Treatment Trial. Commentary, AAO. Available online: https://www.aaojournal.org/article/S0161-6420(19)32364-4/pdf (accessed on 2 February 2022).
- Beck, R.W.; Gal, R.L. Treatment of acute optic neuritis: A summary of findings from the optic neuritis treatment trial. Arch. Ophthalmol. 2008, 126, 994–995.
- Beck, R.W.; Cleary, P.A.; Anderson, M.M., Jr.; Keltner, J.L.; Shults, W.T.; Kaufman, D.I.; Buckley, E.G.; Corbett, J.J.; Kupersmith, M.J.; Miller, N.R.; et al. A randomized controlled trail of corticosteroids in the treatment of acute optic neuritis. N. Engl. J. Med. 1992, 326, 581–588.
- Optic Neuritis Study Group. Visual function 5 years after optic neuritis: Experience of the Optic Neuritis Treatment Trial. Arch. Ophthalmol. 1997, 115, 1545–1552.
- Gal, R.L.; Vedula, S.S.; Beck, R. Corticosteroids for treating optic neuritis. Cochrane Database Syst. Rev. 2015, 2015, CD001430.
- Petzold, A.; Braithwaite, T.; van Oosten, B.W.; Balk, L.; Martinez-Lapiscina, E.H.; Wheeler, R.; Wiegerinck, N.; Waters, C.; Plant, G.T. Case for a new corticosteroid treatment trial in optic neuritis: Review of updated evidence. J. Neurol. Neurosurg. Psychiatry 2020, 91, 9–14.
- Stiebel-Kalish, H.; Hellmann, M.A.; Mimouni, M.; Paul, F.; Bialer, O.; Bach, M.; Lotan, I. Does time equal vision in the acute treatment of a cohort of AQP4 and MOG optic neuritis? Neurol. Neuroimmunol. Neuroinflamm. 2019, 6, e572.
- Bsteh, G.; Berek, K.; Hegen, H.; Teuchner, B.; Buchmann, A.; Voortman, M.M.; Auer, M.; Zinganell, A.; Di Pauli, F.; Deisenhammer, F.; et al. Serum neurofilament levels correlate with retinal nerve fiber layer thinning in multiple sclerosis. Mult. Scler. J. 2019, 26, 1682–1690.
- Osinga, E.; van Oosten, B.; de Vries-Knoppert, W.; Petzold, A. Time is vision in recurrent optic neuritis. Brain Res. 2017, 1673, 95–101.
- Phuljhele, S.; Kedar, S.; Saxena, R. Approach to optic neuritis: An update. Indian J. Ophthalmol. 2021, 69, 2266–2276.
- Horton, L.; Bennett, J.L. Acute Management of Optic Neuritis: An Evolving Paradigm. J. Neuroophthalmol. 2018, 38, 358–367.
- Wilhelm, H.; Schabet, M. Continuing medical education the diagnosis and treatment of optic neuritis. Dtsch. Arztebl. Int. 2015, 112, 616–626.
- Le Page, E.; Veillard, D.; Laplaud, D.A.; Hamonic, S.; Wardi, R.; Lebrun, C.; Zagnoli, F.; Wiertlewski, S.; Deburghgraeve, V.; Coustans, M.; et al. Oral versus intravenous high-dose methylprednisolone for treatment of relapses inpatients with multiple sclerosis (COPOUSEP): A randomised, controlled, double-blind, non-inferiority trial. Lancet 2015, 386, 974–981.
- Bonnan, M.; Cabre, P. Plasma Exchange in Severe Attacks of Neuromyelitis Optica. Mult. Scler. Int. 2012, 2012, 787630.
- Bennett, J.L. Optic Neuritis. Continuum 2019, 25, 1236–1264.
- Jakimovski, D.; Kolb, C.; Ramanathan, M.; Zivadinov, R.; Weinstock-Guttman, B. Interferon β for Multiple Sclerosis. Cold Spring Harb. Perspect. Med. 2018, 8, a032003.
- Dhib-Jalbut, S.; Marks, S. Interferon-beta mechanisms of action in multiple sclerosis. Neurology 2010, 74 (Suppl. S1), S17–S24.
- Rudick, R.A.; Ransohoff, R.M.; Lee, J.C.; Peppler, R.; Yu, M.; Mathisen, P.M.; Tuohy, V.K. In vivo effects of interferon beta-1a on immunosuppressive cytokines in multiple sclerosis. Neurology 1998, 50, 1294–1300.
- Hartrich, L.; Weinstock-Guttman, B.; Hall, D.; Badgett, D.; Baier, M.; Patrick, K.; Feichter, J.; Hong, J.; Ramanathan, M. Dynamics of immune cell trafficking in interferon-β treated multiple sclerosis patients. J. Neuroimmunol. 2003, 139, 84–92.
- Rizzo, F.; Giacomini, E.; Mechelli, R.; Buscarinu, M.C.; Salvetti, M.; Severa, M.; Coccia, E.M. Interferon-β therapy specifically reduces pathogenic memory B cells in multiple sclerosis patients by inducing a FAS-mediated apoptosis. Immunol. Cell. Biol. 2016, 94, 886–894.
- Molnarfi, N.; Prod’homme, T.; Schulze-Topphoff, U.; Spencer, C.M.; Weber, M.S.; Patarroyo, J.C.; Lalive, P.H.; Zamvil, S.S. Glatiramer acetate treatment negatively regulates type I interferon signaling. Neurol. Neuroimmunol. Neuroinflamm. 2015, 2, e179.
- Weber, M.S.; Hohlfeld, R.; Zamvil, S.S. Mechanism of action of glatiramer acetate in treatment of multiple sclerosis. Neurotherapeutics 2007, 4, 647–653.
- Prod’homme, T.; Zamvil, S.S. The Evolving Mechanisms of Action of Glatiramer Acetate. Cold Spring Harb. Perspect. Med. 2019, 9, a029249.
- Polman, C.H.; Reingold, S.C.; Banwell, B.; Clanet, M.; Cohen, J.A.; Filippi, M.; Fujihara, K.; Havrdova, E.; Hutchinson, M.; Kappos, L.; et al. Diagnostic criteria for multiple sclerosis: 2010 revisions to the McDonald criteria. Ann. Neurol. 2011, 69, 292–302.
- Kieseier, B.C.; Benamor, M. Pregnancy outcomes following maternal and paternal exposure to teriflunomide during treatment for relapsing-remitting multiple sclerosis. Neurol. Ther. 2014, 3, 133–138.
- Andersen, J.B.; Moberg, J.Y.; Spelman, T.; Magyari, M. Pregnancy Outcomes in Men and Women Treated with Teriflunomide. A Population-Based Nationwide Danish Register Study. Front. Immunol. 2018, 9, 2706.
- Fazekas, F.; Lublin, F.D.; Li, D.; Freedman, M.S.; Hartung, H.P.; Rieckmann, P.; Sørensen, P.S.; Maas-Enriquez, M.; Sommerauer, B.; Hanna, K.; et al. Intravenous immunoglobulin in relapsing-remitting multiple sclerosis: A dose-finding trial. Neurology 2008, 71, 265–271.
- Sørensen, P.S.; Sellebjerg, F.; Lycke, J.; Färkkilä, M.; Créange, A.; Lund, C.G.; Schluep, M.; Frederiksen, J.L.; Stenager, E.; Pfleger, C.; et al. Minocycline added to subcutaneous interferon β-1a in multiple sclerosis: Randomized Recycline study. Eur. J. Neurol. 2016, 23, 861–870.
- Metz, L.M.; Li, D.K.B.; Traboulsee, A.L.; Duquette, P.; Eliasziw, M.; Cerchiaro, G.; Greenfield, J.; Riddehough, A.; Yeung, M.; Kremenchutzky, M.; et al. Trial of Minocycline in a Clinically Isolated Syndrome of Multiple Sclerosis. N. Engl. J. Med. 2017, 376, 2122–2133.
- Jacobs, L.D.; Beck, R.W.; Simon, J.H.; Kinkel, R.P.; Brownscheidle, C.M.; Murray, T.J.; Simonian, N.A.; Slasor, P.J.; Sandrock, A.W.; The CHAMPS Study Group. Intramuscular interferon beta-1a therapy initiated during a first demyelinating event in multiple sclerosis. N. Engl. J. Med. 2000, 343, 898–904.
- Goodin, D.S.; Bates, D. Treatment of early multiple sclerosis: The value of treatment initiation after a first clinical episode. Mult. Scler. 2009, 15, 1175–1182.
- Comi, G.; Filippi, M.; Barkhof, F.; Durelli, L.; Edan, G.; Fernández, O.; Hartung, H.; Seeldrayers, P.; Sørensen, P.S.; Rovaris, M.; et al. Effect of early interferon treatment on conversion to definite multiple sclerosis: A randomised study. Lancet 2001, 357, 1576–1582.
- Efendi, H. Clinically Isolated Syndromes: Clinical Characteristics, Differential Diagnosis, and Management. Noro Psikiyatr. Ars. 2015, 52 (Suppl. S1), S1–S11.
- Marcus, J.F.; Waubant, E.L. Updates on Clinically Isolated Syndrome and Diagnostic Criteria for Multiple Sclerosis. Neurohospitalist 2013, 3, 65–80.
- Gold, R.; Wolinsky, J.S. Pathophysiology of multiple sclerosis and the place of teriflunomide. Acta Neurol. Scand. 2011, 124, 75–84.
- Comi, G.; De Stefano, N.; Freedman, M.S.; Barkhof, F.; Polman, C.H.; Uitdehaag, B.M.J.; Casset-Semanaz, F.; Hennessy, B.; Moraga, M.S.; Rocak, S.; et al. Comparison of two dosing frequencies of subcutaneous interferon beta-1a in patients with a first clinical demyelinating event suggestive of multiple sclerosis (REFLEX): A phase 3 randomised controlled trial. Lancet Neurol. 2012, 11, 33.
- Comi, G.; De Stefano, N.; Freedman, M.S.; Barkhof, F.; Uitdehaag, B.M.; de Vos, M.; Marhardt, K.; Chen, L.; Issard, D.; Kappos, L. Subcutaneous interferon β-1a in the treatment of clinically isolated syndromes: 3-year and 5-year results of the phase III dosing frequency-blind multicentre REFLEXION study. J. Neurol. Neurosurg. Psychiatry 2017, 88, 285–294.
- Miller, A.E.; Wolinsky, J.S.; Kappos, L.; Comi, G.; Freedman, M.S.; Olsson, T.P.; Bauer, D.; Benamor, M.; Truffinet, P.; O’Connor, P.W.; et al. Oral teriflunomide for patients with a first clinical episode suggestive of multiple sclerosis (TOPIC): A randomised, double-blind, placebo-controlled, phase 3 trial. Lancet Neurol. 2014, 13, 977–986.





