Diabetes mellitus (DM) is a worldwide-spread chronic metabolic disease that occurs when the pancreas fails to produce enough insulin levels or when the body fails to effectively use the secreted pancreatic insulin, eventually resulting in hyperglycemia. According to the International Diabetes Federation, in 2021, 537 million adults were suffering from DM, resulting in 6.7 million deaths and a 966 billion dollars healthcare cost. Systematic glycemic control is the only procedure at the disposal to prevent diabetes long-term complications such as cardiovascular disorders, kidney diseases, nephropathy, neuropathy, and retinopathy. The gold standard for glycemic control assessment in clinics is the glycated hemoglobin (HbA1c) measurement, but glycated albumin (GA) has recently gained more and more attention as a control biomarker thanks to its shorter lifespan and wider reliability compared to HbA1c. Continuous glucose monitoring (CGM) and blood glucose monitoring (BGM) remain useful individual tools for diabetes self-management.
1. Introduction
The current management of diabetes mellitus (DM) demands rigorous and systematic monitoring of the glycemic status of the patient, whose results can be exploited to assess therapy effectiveness, as well as to adjust diet and/or medications to improve blood glucose control. In the ’70s, diabetes was predominantly monitored through urine ketone and glucose measurements
[1][2][3]. The former currently remains a valid tool to denote imminent or established diabetic ketoacidosis, i.e., a life-threatening complication induced by insulin deficiency, and hence it is recommended to all patients with diabetes, especially type 1 DM
[4][5]. Conversely, technical advances in blood glucose monitoring
[6][7][8] together with additional clinical experience and extensive research investigations, marked the progressive abandonment of urine glucose testing as the recommended approach to home diabetes monitoring. Indeed, urine glucose level was proven to be an unreliable estimator of plasma glucose concentration due to wide confidence levels and poor correlation
[9][10], significant inter-patient variability of glucose renal thresholds
[11], and drug interference
[12]. At the end of the last century, the results of two important randomized controlled trials were published. The Diabetes Control and Complications Trial (DCCT)
[13][14] was conducted on 1441 type 1 diabetic patients, randomly assigned to an intervention group administered with intensive insulin therapy (three or more daily injections) guided by frequent BGM, and a control group in which patients followed conventional therapy requiring one or two daily injections; its findings cemented the clinical importance of rigorous monitoring aimed at maintaining the glycemic status of the subject as close as possible to the normal range. The UK Prospective Diabetes Study (UKPDS)
[15] involved 3867 type 2 diabetic patients randomly split into an intervention group receiving different sulfonylureas (i.e., chlorpropamide or glibenclamide) or insulin and a control group treated with conventional diet; the study is to establishing whether intensive glucose control had an impact on lowering the risk of macro/micro-vascular complications, and whether any pharmaceutical therapy was more advantageous than the others. Its results showed that improvement in glycemic control, assessed over a 10-year temporal window with HbA1c systematic monitoring, rather than any specific therapy, was the principal factor involved in the observed risk reductions. As a consequence, nowadays, glycemic control is assessed in clinics by the HbA1c measurement, whereas continuous glucose monitoring (CGM) and BGM are useful individual tools for diabetes self-management
[16].
2. Glycated Proteins
Blood glucose and urine ketone measurements are single-point measurements that provide essential information on diabetes management on a daily basis; glycated proteins instead, such as HbA1c and glycated albumin (GA), introduce a new, complementary layer in glycemic control monitoring by reflecting the mean glucose level over longer, past periods, and are not affected by daily fluctuations induced by diet or physical activity. Glycation is a non-enzymatic mechanism also called a Maillard reaction, which consists of the covalent addition of a reducing sugar to a free amino group of amine-containing molecules such as proteins to form an unstable, reversible product (i.e., Schiff base) which is then rearranged to a more stable conformation known as Amadori product or ketoamine. This process is shown in Figure 1.
Figure 1. Reactions involved in the glycation of proteins. In particular, hemoglobin (PDB ID: 1BBB) and albumin (PDB ID: 1AO6) have been reported, and the main glycation sites for each of them have been highlighted in red: N-terminal valine of the β-chains of hemoglobin, and lysine and arginine residues of albumin.
Eventually, this process leads to the formation of irreversible compounds designated as advanced glycation end-products (AGEs)
[17]. Advanced glycation is a critical pathway involved in the development of several diabetic complications such as neuropathy, nephropathy, and retinopathy that arise from AGEs-induced oxidative stress and inflammatory processes
[18]. Protein glycation is affected by the time of exposure to glucose, and its concentration; extracellular proteins such as albumin have higher glycation rates than intracellular ones, such as hemoglobin, due to their direct exposure to blood glucose
[19].
2.1. Glycated Hemoglobin
HbA1c is the Amadori rearrangement of the adduct of glucose with the N-terminal valine of the
β-chain of hemoglobin
[4][20][21], which is the most reactive site
[22][23]. Its rate of formation is proportional to the ambient glucose concentration, and it reflects the mean glycemia over the past two to four months, correlating directly with the lifespan of erythrocytes
[4][24][25][26]. Its value is expressed in terms of percentage with respect to the total hemoglobin concentration and can be used as a diagnostic biomarker
[27] and as a monitoring tool to assess treatment effectiveness in diabetic patients
[16]. Despite being supported by large-scale clinical trials, i.e., the DCCT and the UKPDS, its employment suffers from some intrinsic disadvantages related to the breadth of the temporal window, which does not allow for accurately tracking rapid changes in glycemic control
[28][29][30], and to its reliability under certain clinical circumstances such as hematologic disorders (variant hemoglobin, different types of anemia), recent blood transfusions, use of erythropoietin-based drugs, and pregnancy, which alter the lifespan of red blood cells hence affecting HbA1c measurements
[16][31][32]. Moreover, there is evidence for inter-individual heterogeneity of glucose gradient across the membrane of red blood cells, which changes the dynamics of hemoglobin glycation hence impacting HbA1c assessment tests
[33].
2.2. Glycated Albumin
Human serum albumin (HSA) is the most abundant protein in human blood: with a normal concentration ranging from 3 to 5 g/dL, it accounts for approximately 60% of serum proteins
[19][34]. It is composed of a single, 585 amino acids-long polypeptidic chain with a molecular weight of 66.5 kDa
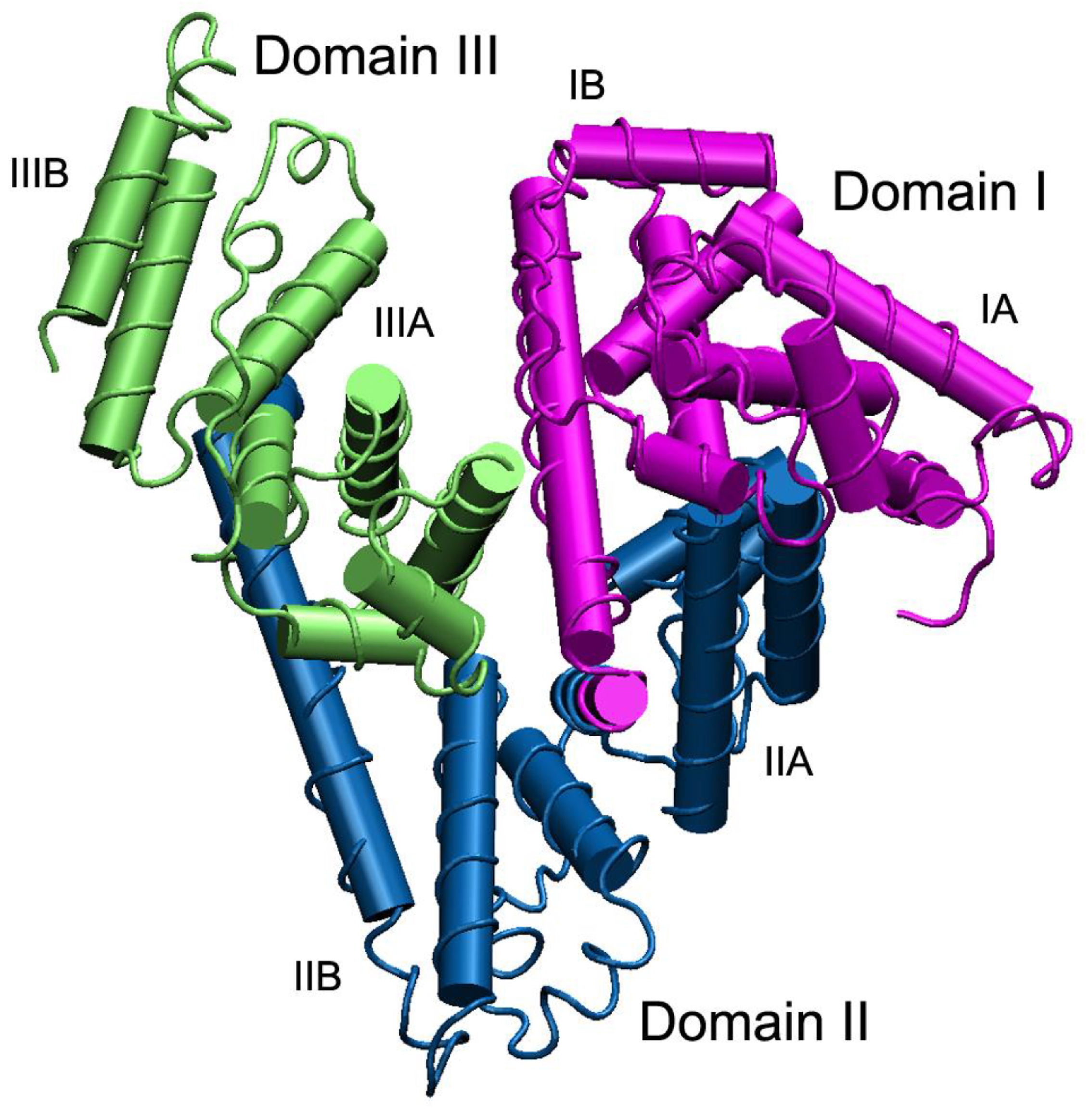
[35][36], and its three-dimensional structure is reported in
Figure 2. HSA has a half-life of approximately three weeks
[37], during which the exposure to blood glucose induces glycation processes primarily at its lysine and arginine residues
[38] that modify its spatial arrangement as well as the N-terminal region
[31]; glycation of albumin also leads to a slight increase in the polarity of the molecule
[21].
Figure 2. Three-dimensional structure of human serum albumin. The three domains I, II, and III are highlighted in purple, blue and green, respectively, and for each domain the two subdomains A and B are shown—from Belinskaia et al.
[36].
Clinically, GA has some clear advantages over HbA1c. Firstly, thanks to a higher rate of formation and shorter lifespan, it can reflect hyperglycemia earlier than HbA1c
[31], and it is a more adequate indicator to evaluate glycemic variability
[29][39]. Secondly, due to its independence from red blood cells, it offers a more robust parameter whenever the patient suffers from erythrocyte lifespan-affecting events.
Table 1 summarizes the principal clinical conditions in which GA may offer a better understanding of the glycemic status of a patient.
Table 1. List of conditions in which GA may be more reliable than HbA1c as a glycemic control biomarker.
GA, however, is not exempt from limitations, and medical operators should be aware of the conditions in which glycated albumin does not accurately reflect the glycemic status of a patient because of the involvement of other factors. GA measurements, being corrected for total albumin, should not be influenced by albumin concentration
[19][51]; nevertheless, the association between low plasma albumin levels and increased protein glycation rates, probably caused by different exposure to glucose, has been demonstrated
[52]. Indeed, disorders that impact HSA metabolism may alter GA levels; in particular, higher GA levels have been observed in patients with chronic liver disease
[53] and hypothyroidism
[54], whereas lower values have been reported for nephrotic syndrome
[55][56] and hyperthyroidism
[54] cases. Body mass index (BMI) has an impact on glycated albumin too
[57], with absolute GA values decreasing by 0.13% every 1 kg/m
2 increment in BMI
[58]. Finally, patient age should be considered when analyzing GA levels, since newborns show much lower values of GA with respect to adults
[59], and the values significantly increase as the patient’s age increases
[60][61]. According to the Japanese Diabetes Society, values of GA in non-diabetic patients should range within 11–16%, normalized to the total albumin, whereas diabetic patients generally exhibit values greater than 20%
[62]. Nevertheless, the lack of standardization in the reference method used to assess diabetes (some studies used FPG, others oral glucose tolerance test (OGTT) or HbA1c) is responsible for slight variations in the definition of the reference thresholds. Another critical aspect is related to the choice of the analytical technique used to obtain the GA measurement. Kohzuma et al.
[20] meticulously summarized the main clinical studies and their relative findings related to GA reference range and cutoff values for diabetes diagnosis and screening, whereas Roohk et al.
[63] reported the reference values employed in six US clinical laboratories, showing the discrepancies related to the different GA testing methods used.