Video Upload Options
Locusts have been devastating vegetation and agriculture since ancient times. The formation of huge swarms that migrate long distances is a hallmark of the locust phenomenon. An important aspect of locusts’ natural history is that of their interaction with the bacteria they harbor in and on their body. These locust-associated bacteria have been shown to affect certain physiological traits (such as immunity and swarm cohesion), as well as possibly also being affected by different
factors in the locust biology. This review summarizes our current understanding of the locust-bacterial interaction. We focus on identifying the relevant bacterial strains and their locations within the insects; the role of the bacteria and their importance to their host’s life; the mechanism of transmitting important bacteria across locust generations; and more. Finally, we offer some new perspectives and research directions that could broaden our understanding of the locust-associated bacteria and their tentative instrumental role in locust outbreaks.
1. Introduction
Locusts are short-horned grasshoppers (Orthoptera) belonging to the family Acrididae. A hallmark of locust biology is their density-dependent phase-plasticity, in which individuals will express vastly different behavioral, morphological, and physiological phenotypes according to the different population density levels they experience [1][2][3]. Locusts are probably best known, however, for their devastating potential to cause immense damage to natural vegetation and crops, as they are able to aggregate and migrate in swarms so large and destructive that they are commonly termed a plague. Consequently, locusts have been the focus of extensive research efforts, mostly seeking to better understand the mechanisms underlying the locust swarming phenomenon.
Although not part of the major emphasis of previous locust research, data concerning the characterization of locusts’ bacterial symbionts were published already in the late 1950s [4][5][6]; and since then a multitude of important observations have accumulated regarding the nature of locust–bacteria interactions [7][8][9][10][11][12][13][14][15][16][17][18]. Some of these findings were already summarized, nearly 20 years ago, by Dillon and Charnley [10], but ever since their extensive review, many new data have been collected, and vast improvements in sequencing technology have allowed researchers to ask and answer questions previously beyond our reach. The wealth of new findings in recent years calls for an update, incorporating the essential details of our current knowledge of locust-bacteria associations.
In this concise review, we provide an updated summary of the nature of locust bacterial symbionts and their distribution in the host, as well as hypothesized effects on the locust biology and life history. We mainly focus on the desert locust (Schistocerca gregaria) and the migratory locust (Locusta migratoria), which most of the studies conducted to date have explored. However, the main ideas presented will be largely applicable to other locust species, albeit perhaps with different bacterial players.
2. Locust Bacterial Symbionts and Their Location
Similar to most vertebrates and invertebrates, locusts harbor bacterial cells both within their internal body environment and externally on their cuticle [19][20]. Below we briefly present major data that have been gathered concerning the bacterial symbionts present in the locust digestive tract, as well as some more recent reports describing the bacterial symbionts in other parts of the locust’s body.
2.1. The Digestive Tract
The bacterial composition of the locust gut has been thoroughly described for the desert locust [21]. Some research has also been conducted on several other locust species, including the migratory locust [22][23], the brown locust (Locustana pardalina), the Moroccan locust (Dociostaurus maroccanus), and the Italian locust (Calliptamus italicus).
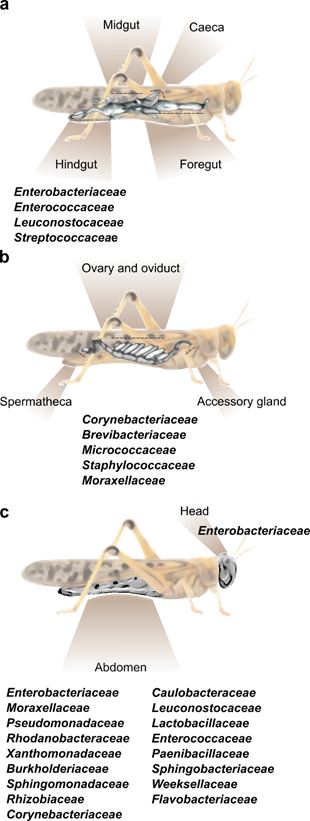
The composition of the bacterial community in any insect alimentary tract is largely affected by its specific anatomy. The locust gut is relatively simple and consists of three main parts: foregut, midgut, and hindgut (Figure 1a). In brief, the cuticle-lined foregut is where the ingested food is mixed with salivary and regurgitated enzymes to initiate the food breakdown process; the food is then transferred to the midgut, where the digestion is completed and nutrients (mainly protein and carbohydrates derivatives, i.e., amino acids and monosaccharides) are absorbed, while a peritrophic membrane envelops the digested food. Six caeca invaginations are connected to the anterior part of the midgut, each opens individually into the alimentary canal (Figure 1a). The main role of the hindgut is the absorption of water, salts, amino acids, and small metabolites. Six large lobes, called “rectal pads”, maximize the surface area of the hindgut to enable efficient absorption [24][25].
Hunt and Charnley [8] used microscopy and cultivation-dependent methods to identify the bacterial residents in different parts of the S. gregaria alimentary canal. They observed that the bacterial load was small in the anterior parts of the digestive system, and that this load increased significantly in the posterior parts. They further reported that no bacterial cells were found on the lining of the foregut or on the epithelial cells of the midgut (including caeca), where bacteria were limited to the lumen and to the inner section of the peritrophic membrane. Furthermore, they noted that the bacteria found in the foregut were roughly similar to those found in the locust’s food, while the hindgut contained a high load of Enterobacteriaceae and Streptococcaceae. These important findings served to focus subsequent studies on those bacteria residing in the locust hindgut.
Figure 1. The main bacterial families known to inhabit different body parts of the desert locust. (a) A depiction of the locust’s alimentary canal and the common bacterial families residing within the hindgut. Foregut and hindgut (including caeca) do not host a constant fraction of specific bacterial strains. [11][15]. (b) A depiction of the female locust reproductive system divided into three main sections, all harboring the same core bacterial families. (c) External bacteria found on the head and abdomen cuticle of the desert locust [26].
Several reports on the hindgut bacteria of S. gregaria have revealed a relatively simple bacterial composition, with a consistent presence of members of the families Enterobacteriaceae (mostly the genera Enterobacter and Klebsiella) and Enterocococeaea reviewed in [10] and described in [26]. Enterobacteriaceae and Enterocococeae members were also found in the hindgut of healthy L. migratoria, as well as in the gut of field-caught Italian locusts, Moroccan locusts and brown locusts.
Based on the fact that desert locusts were shown to be able to live and reproduce without their gut bacterial symbionts, at least under laboratory conditions [27], it is accepted that locusts have developed a tight but not obligatory association with these mutualistic bacteria. Yet, some bacteria seem to play an important role in the fitness of locusts by conferring colonization resistance and by means of their volatile secretions (to be discussed below). This tight but not obligatory interaction may be routed in the function that these bacteria execute for their host. If more than one bacteria can produce the same essential product for the locust, this will push towards facultative, rather than obligatory, locust–bacteria interaction [28]. For example, if both Enterobacter and Klebsiella can produce cohesion promoting volatiles and to confer gut-colonization resistance, harboring one strain or the other will not affect the locust’s fitness, thus promoting the non-obligatory interaction, where the gut niche is colonized by the first suitable bacteria to be ingested by the locust.
The genus Weissella (Firmicutes: family Leuconostocaceae), was repeatedly found in the gut of L. migratoria , as well as following high-throughput sequencing of the desert locust’s gut microbiota. The fact that this genus was absent from previous reports of the bacterial composition of locusts, may be attributed to difficulties in growing it in culture, which can be circumvented by advanced sequencing methods. Thus, it is highly plausible that additional important locust symbionts have not been cultivated to date and thus their effect on the host may be currently underappreciated.
2.2. The Female Reproductive System
As already noted, the organ at the spotlight of locust–bacteria research to date has been almost exclusively the gut. However, a recent report by Lavy et al. focused on the reproductive system of the female desert locust and revealed the unique bacterial composition of different organs in this system. That study divided the female gonads into three main parts—accessory glands, ovaries, and spermatheca (Figure 1b), and used high throughput amplicon sequencing to analyze their bacterial composition. This resulted in the observation that the reproductive organs contained a consistent fraction of Corynebacterium, Brevibacterium and Micrococcus (Actinobacteria), along with Staphylococcus (Firmicutes) and Acinetobacter (Gammaproteobacteria) (Figure 1b). This unique bacterial composition of the female reproductive organs did not seem to be affected by mating, or by oviposition (during which the female extends her abdomen into the ground and oviposits at a depth of ~10 cm; [29]). The presence of the genus Brevibacterium in locusts had been reported only once prior to the aforementioned study: Bucher and Stephens [5] used a whole-body homogenate to examine the locust’s gut bacterial composition. Some bacteria isolated by them belonged to the genus Brevibacterium, leading these authors to consider it to be a gut-residing bacterium. However, later research, employing an isolated-gut approach, failed to find this bacterium in the locust gut. It is, therefore, possible that the origin of Brevibacterium, as described by Bucher and Stephens , was the locust reproductive system, and it was mistakenly described as a gut bacterium due to the experimental procedure.
2.3. The Cuticle
Our group recently sampled the abdominal cuticle of laboratory-reared S. gregaria in order to characterize its bacterial composition, as part of a larger study examining also the bacterial content of the fecal pellets of the same individuals [18]. It was then observed that the locust cuticle harbors a large fraction of what might be gut-associated bacteria, such as Weissella and other members of Firmicutes and Proteobacteria, which were also found in the feces (and which could have been amplified due to the crowded conditions in the cage). However, the bacteria residing on the locust integument also featured members of the phyla Bacteroidetes (Flavobacteriaceae and Weeksellaceae) and Actinobacteria (Corynebacteriaceae), which were unique to the external cuticle and were not present in the locust feces. In another recent study , cultivation-dependent methods identified a species of Salmonella (Proteobacteria) externally on the head and body of S. gregaria, in agreement with our own observations noted above (Figure 1c).
3. Locust–Bacteria Mutualism
Bacterial symbionts have been repeatedly shown to play an important role in the biology of diverse insect species [30][31][32][33]. In a number of studies, bacteria were reported to augment aphid resistance to various stressors [31], to protect the gardens from Attine ants from pathogens [34], to protect wasp-larvae from potential pathogens [35], and more. The symbiotic nature of the locust–bacteria interaction has also been studied extensively, mostly in the desert locust and to some extent also in the migratory locust.
As noted above, the bacterial composition of the desert locust foregut was found to be roughly similar to that of the food ingested by the locusts. The bacterial community of the hindgut, however, differed from that in the food and the foregut, demonstrating a higher prevalence of Enterobacteriaceae cells. This finding and the multiple subsequent confirmations of the stable presence of Enterobacteriaceae species in the locust hindgut suggested a selective basis for the interaction of S. gregria and bacteria from the Enterobacteriaceae family. Dillon and Charnley hypothesized that the neutral pH of the hindgut (along with the presence of organic acids and plant remains) supports the proliferation of Enterobacteriaceae. This hypothesis was confirmed to some extent in L. migratoria by Shi et al., who observed that acidification of the locust’s hindgut by entomopathogenic fungus reduced the bacterial colony-forming units (CFU) isolated from this organ. Those authors also demonstrated experimentally, by mimicking the acidification effect of the fungus on the hindgut, that the growth of locust-isolated Enterobacter sp. (Enterobacteriaceae) was almost completely inhibited when pH dropped from 6.3 (as in the hindgut of healthy L. migratoria) to 5.6 (reflecting the fungus impact). The growth of locust-isolated Weissella, Enterococcus, and Microbacterium was inhibited by the low pH of 5.6, though not to the same extent as the tested Enterobacter. Consequently, although the locusts had clearly not developed obligatory interactions with any specific bacteria, the specific conditions in their hindgut seem to have been selected for the proliferation of certain bacterial groups, including the Enterobacteriaceae family.
Native bacteria of the desert locust’s hindgut have been shown to augment host immunity through colonization resistance (CR), thereby making it less susceptible to pathogenic infections . This is largely due to the bacterial synthesis of antifungal and antimicrobial phenolic compounds [35][36][37]. The antimicrobial nature of these compounds not only protects the locust from the establishment of pathogens, but also probably contributes to the selective conditions in the locust hindgut, creating favorable conditions for the anti-microbial tolerant native bacteria of the locust gut.
Two of the reported bacteria-secreted phenolic compounds, phenol and guaiacol, were also suggested as a dominant fraction of a cohesion pheromone, considered to help in maintaining the integrity of S. gregaria swarms [38]. Both phenol and guaiacol were shown to be produced by three bacterial species in the locust hindgut: Pantoea agglomerans, Klebsiella pneumoniae pneumoniae, and Enterobacter cloacae (all from the Enterobacteriaceae family), when incubated on axenic (i.e., bacteria-free) locust fecal pellets. The precursor for these phenolic compounds in the gut is the lignin-derived vanillic acid present in the locust’s food. Serratia marcescens a member of the Enterobacteriaceae which is not a regular inhabitant of the locust gut, did not produce the same phenolic volatiles, reinforcing the hypothesis of a mutual symbiosis between the host and specific bacterial species. Additional information on the phenolic compounds produced by the desert locust’s gut bacteria can be found in the excellent review by Dillon and Charnley.
References
- Pener, M.P.; Simpson, S.J. Locust phase polyphenism: An update. Adv. Insect Phys. 2009, 36, 1–272.
- Cullen, D.A.; Cease, A.J.; Latchininsky, A.V.; Ayali, A.; Berry, K.; Buhl, J.; De Keyser, R.; Foquet, B.; Hadrich, J.C.; Matheson, T.; et al. From Molecules to Management: Mechanisms and Consequences ofLocust Phase Polyphenism, 1st ed.; Elsevier Ltd.: Amsterdam, The Netherlands, 2017; Volume 53.
- Ayali, A. The puzzle of locust density-dependent phase polyphenism. Curr. Opin. Insect Sci. 2019, 35, 41–47. [CrossRef] [PubMed]
- Bucher, G.; Stephens, J.M. Bacteria of grasshoppers of Western Canada. I. The Enterobacteriaceae. J. Insect Pathol. 1959, 1, 356–373.
- Bucher, G.; Stephens, J.M. Bacteria of grasshoppers of Western Canada. II. The Pseudomonadaceae, Achromobacteraceae, Micrococcaceae, Brevibacteriaceae, Lactobacillaceae, and less important families. J. Insect Pathol. 1959, 1, 374–390.
- Bucher, G. Bacteria of grasshoppers of Western Canada. III. Frequency of occurrence, pathogenicity. J. Insect Pathol. 1959, 1, 391–405.
- Stevenson, J.P. The Normal of Bacterial of the the Alimentary Desert Forskiil Locust. J. Invertebr. Pathol. 1966, 211, 205–211. [CrossRef]
- Hunt, J.; Charnley, A.K. Abundance and distribution of the gut flora of the desert locust, Schistocerca gregaria. J. Invertebr. Pathol. 1981, 38, 378–385. [CrossRef]
- Mead, L.J.; Khachatourians, G.G.; Jones, G. A Microbial Ecology of the Gut in Laboratory Stocks of the Migratory Grasshopper, Melanoplus sanguinipes (Fab.) (Orthoptera: Acrididae). Appl. Environ. Microbiol. 1988, 54, 1174–1181. [CrossRef] [PubMed]
- Dillon, R.; Charnley, A. Mutualism between the desert locust Schistocerca gregaria and its gut microbiota. Res. Microbiol. 2002, 153, 503–509. [CrossRef]
- Dillon, R.J.; Vennard, C.T.; Charnley, A.K. A Note: Gut bacteria produce components of a locust cohesion pheromone. J. Appl. Microbiol. 2002, 92, 759–763. [CrossRef]
- Dillon, R.J.; Vennard, C.T.; Buckling, A.; Charnley, A.K. Diversity of locust gut bacteria protects against pathogen invasion. Ecol. Lett. 2005, 8, 1291–1298. [CrossRef]
- Dillon, R.J.; Webster, G.; Weightman, A.J.; Dillon, V.M.; Blanford, S.; Charnley, A.K. Composition of Acridid gut bacterial communities as revealed by 16S rRNA gene analysis. J. Invertebr. Pathol. 2008, 97, 265–272. [CrossRef]
- Shi, W.; Guo, Y.; Xu, C.; Tan, S.; Miao, J.; Feng, Y.; Zhao, H.; St Leger, R.J.; Fang, W. Unveiling the mechanism by which microsporidian parasites prevent locust swarm behavior. Proc. Natl. Acad. Sci. USA 2014, 111, 1343–1348. [CrossRef] [PubMed]
- Lavy, O.; Gophna, U.; Gefen, E.; Ayali, A. The effect of density-dependent phase on the locust gut bacterial composition. Front. Microbiol. 2019, 10, 1–8. [CrossRef]
- Lavy, O.; Gophna, U.; Gefen, E.; Ayali, A. Dynamics of bacterial composition in the locust reproductive tract are affected by the density-dependent phase. FEMS Microbiol. Ecol. 2020, 96, 1–10. [CrossRef]
- Lavy, O.; Gophna, U.; Ayali, A.; Gihaz, S.; Fishman, A.; Gefen, E. The maternal foam plug constitutes a reservoir for the desert locust’s bacterial symbionts. bioRxiv 2020. [CrossRef]
- Lavy, O.; Lewin-Epstine, O.; Gophna, U.; Gefen, E.; Hadany, L.; Ayali, A. Microbiome-related aspects of locust density-dependent phase transition. bioRxiv 2020. [CrossRef]
- Murdoch, C.C.; Rawls, J.F. Commensal Microbiota Regulate Vertebrate Innate Immunity-Insights from the Zebrafish. Front. Immunol. 2019, 10, 1–14. [CrossRef]
- Douglas,A.E.Multiorganismal Insects: DiversityandFunctionofResidentMicroorganisms.Annu. Rev. Entomol. 2015, 60, 17–34. [CrossRef]
- Dillon, R.J.; Webster, G.; Weightman, A.J.; Keith, C. Diversity of gut microbiota increases with aging and starvation in the desert locust. Antonie Leeuwenhoek 2010, 97, 69–77. [CrossRef]
- Stoops, J.; Crauwels, S.; Waud, M.; Claes, J.; Lievens, B.; Van Campenhout, L. Microbial community assessment of mealworm larvae (Tenebrio molitor) and grasshoppers (Locusta migratoria migratorioides) sold for human consumption. Food Microbiol. 2016, 53, 122–127. [CrossRef]
- Garofalo, C.; Osimani, A.; Milanovi´c, V.; Taccari, M.; Cardinali, F.; Aquilanti, L.; Riolo, P.; Ruschioni, S.; Isidoro, N.; Clementi, F. The microbiota of marketed processed edible insects as revealed by high-throughput sequencing. Food Microbiol. 2017, 62, 15–22. [CrossRef]
- Albrecht, F.O. The Anatomy of the Migratory Locust; University of London: London, UK; Athlone Press: London, UK; Western Printing Services Ltd.: London, UK, 1953.
- Dillon, R.J.; Charnley, A.K. The Fate of Fungal Spores in the Insect Gut. Fungal Spore Dis. Initiat. Plants Anim. 1991, 129–156. [CrossRef]
- Galal, F.H.; Seufi, A.M. Molecular characterization of cultivable bacteria associated with adult Schistocerca gregaria, using 16S rRNA. J. Taibah Univ. Sci. 2020, 3655. [CrossRef]
- Charnley, A.K.; Hunt, J.; Dillon, R.J. The germ-free culture of desert locusts, Schistocerca gregaria. J. Insect. Physiol 1985, 31, 477–485. [CrossRef]
- Furman, O.; Shenhav, L.; Sasson, G.; Kokou, F.; Honig, H.; Jacoby, S.; Hertz, T.; Cordero, O.X.; Halperin, E.; Mizrahi, I. Stochasticity constrained by deterministic effects of diet and age drive rumen microbiome assembly dynamics. Nat. Commun. 2020, 11, 1–13. [CrossRef]
- Symmons, P.M.; Cressman, K. Desert Locust Guidelines; Food and Agriculture Organization (FAO): Rome, Italy, 2001; p. 42.
- Moran, N.A.; Baumann, P. Bacterial endosymbionts in animals. Curr. Opin. Microbiol. 2000, 3, 270–275. [CrossRef]
- Oliver, K.M.; Degnan, P.H.; Burke, G.R.; Moran, N.A. Facultative symbionts in aphids and the horizontal transfer of ecologically important traits. Annu. Rev. Entomol. 2010, 55, 247–266. [CrossRef]
- Bennett, G.M.; Moran, N.A. Small, smaller, smallest: The origins and evolution of ancient dual symbioses in a phloem-feeding insect. Genome Biol. Evol. 2013, 5, 1675–1688. [CrossRef]
- Salem, H.; Florez, L.; Gerardo, N.; Kaltenpoth, M. An out-of-body experience: The extracellular dimension for the transmission of mutualistic bacteria in insects. Proc. R. Soc. B Biol. Sci. 2015, 282, 20142957. [CrossRef]
- Currie, C.R.; Poulsen, M.; Mendenhall, J.; Boomsma, J.J.; Billen, J. Coevolved crypts and exocrine glands support mutualistic bacteria in fungus-growing ants. Science 2006, 311, 81–83. [CrossRef]
- Kaltenpoth, M.; Wolfgang, G.; Gudrun, H.; Erhard, S. Symbiotic Bacteria Protect Wasp Larvaefrom Fungal Infestation. Curr. Biol. 2005, 15, 475–479. [CrossRef]
- Dillon, R.J.; Charnley, A.K. Inhibition of Metarhizium anisopliae by the gut bacterial flora of the desert locust, Schistocerca gregaria: Evidence for an antifungal toxin. J. Invertebr. Pathol. 1986, 47, 350–360. [CrossRef]
- Dillon, R.J.; Charnley, A.K. Chemical barriers to gut infection in the desert locust—In-vivo production of antimicrobial phenols associated with the bacterium Pantoea agglomerans. J. Invertebr. Pathol. 1995, 66, 72–75. [CrossRef]
- Dillon, R.J.; Vennard, C.T.; Charnley, A.K. Exploitation of gut bacteria in the locust. Nature 2000, 403, 851–852. [CrossRef] [PubMed]