
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Daniel Souza Monteiro de Araujo | -- | 5663 | 2022-08-17 11:42:23 | | | |
| 2 | Catherine Yang | Meta information modification | 5663 | 2022-08-18 03:16:02 | | | | |
| 3 | Catherine Yang | -4 word(s) | 5659 | 2022-08-18 03:17:26 | | |
Video Upload Options
Vision is an important sense for humans, and visual impairment/blindness has a huge impact in daily life. The retina is a nervous tissue that is essential for visual processing since it possesses light sensors (photoreceptors) and performs a pre-processing of visual information. Thus, retinal cell dysfunction or degeneration affects visual ability and several general aspects of the day-to-day of a person’s lives. The retina has a blood–retinal barrier, which protects the tissue from a wide range of molecules or microorganisms. However, several agents, coming from systemic pathways, reach the retina and influence its function and survival. Pesticides are still used worldwide for agriculture, contaminating food with substances that could reach the retina. Natural products have also been used for therapeutic purposes and are another group of substances that can get to the retina. Finally, a wide number of medicines administered for different diseases can also affect the retina. Here gathers recent information about the hazard of these products to the retina, which could be used to encourage the search for more healthy, suitable, or less risky agents.
1. Introduction
2. Natural Products
3. Drugs and Medicine
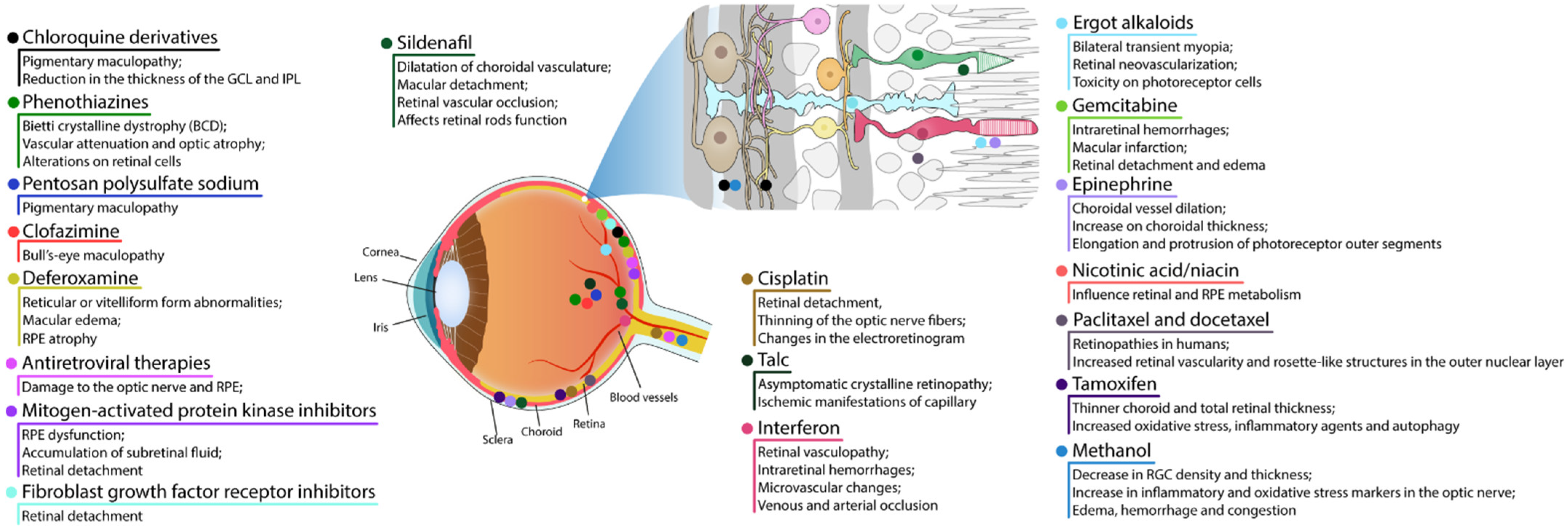
3.1. RPE and Photoreceptor Complex
3.1.1. Chloroquine Derivatives
3.1.2. Phenothiazines
3.1.3. Pentosan Polysulfate Sodium
3.1.4. Clofazimine
3.1.5. Deferoxamine
3.1.6. Antiretroviral Therapies
3.1.7. Mitogen-Activated Protein Kinase Inhibitors
3.1.8. Fibroblast Growth Factor Receptor Inhibitors
3.1.9. Sildenafil
3.1.10. Cisplatin
3.2. Retinal Vascular Damage
3.2.1. Talc
3.2.2. Interferon
3.2.3. Ergot Alkaloids
3.2.4. Gemcitabine
3.3. Cystoid Macular Edema
3.3.1. Epinephrine
3.3.2. Nicotinic Acid/Niacin
3.3.3. Paclitaxel and Docetaxel
3.4. Crystalline Retinopathy
Tamoxifen
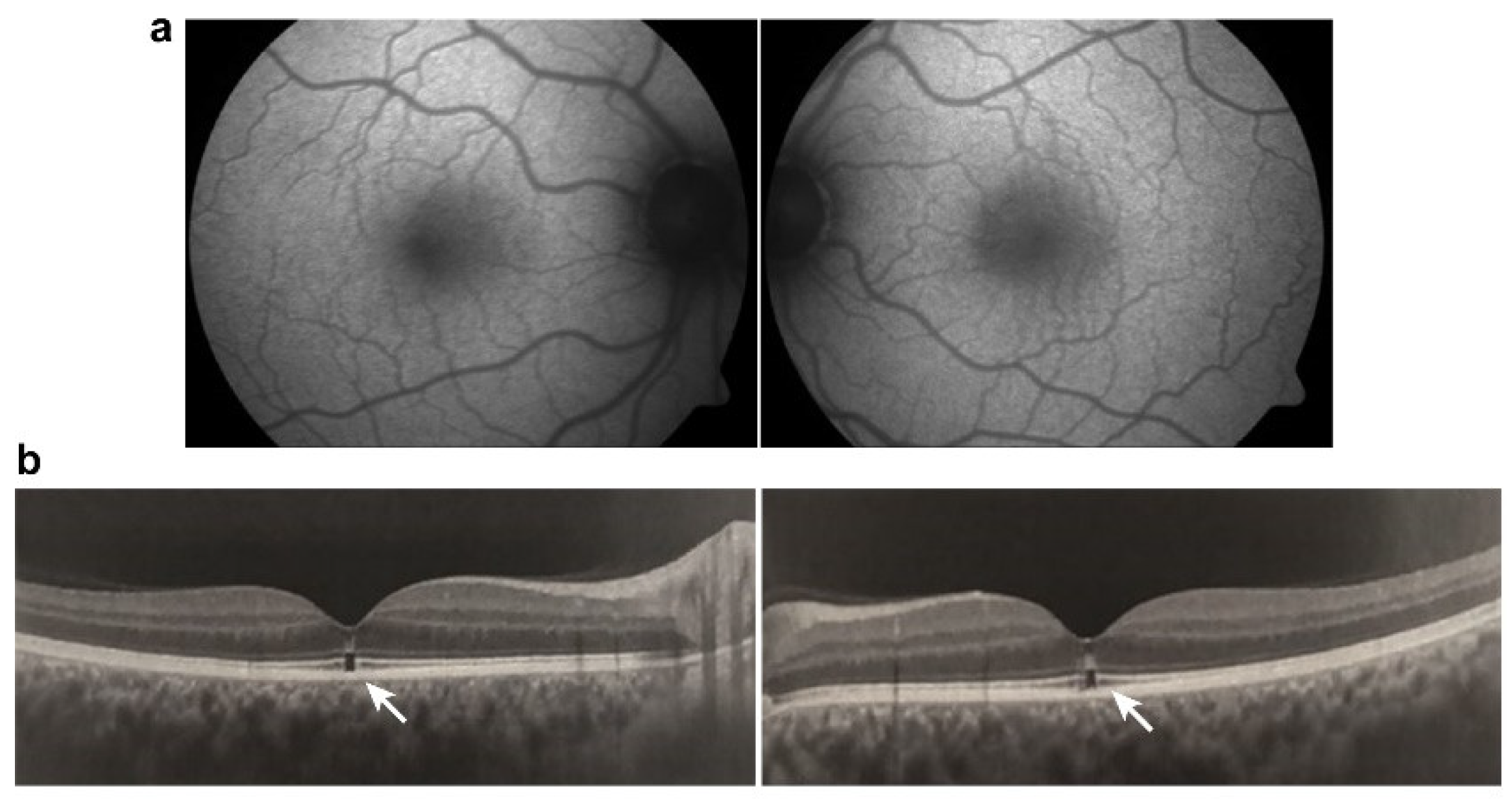
3.5. Damage to Ganglion Cell Layer or Optic Nerve
Methanol
References
- Wässle, H. Parallel processing in the mammalian retina. Nat. Rev. Neurosci. 2004, 5, 747–757.
- Hoon, M.; Okawa, H.; Della Santina, L.; Wong, R.O. Functional architecture of the retina: Development and disease. Prog. Retin. Eye Res. 2014, 42, 44–84.
- De Campos, V.S.; Calaza, K.C.; Adesse, D. Implications of TORCH Diseases in Retinal Development-Special Focus on Congenital Toxoplasmosis. Front. Cell Infect. Microbiol. 2020, 10, 585727.
- Thoreson, W.B.; Dacey, D.M. Diverse Cell Types, Circuits, and Mechanisms for Color Vision in the Vertebrate Retina. Physiol. Rev. 2019, 99, 1527–1573.
- Fain, G.; Sampath, A.P. Rod and cone interactions in the retina. F1000Research 2018, 7, 657.
- Coughlin, B.A.; Feenstra, D.J.; Mohr, S. Müller cells and diabetic retinopathy. Vis. Res. 2017, 139, 93–100.
- Newman, E.A. Glial cell regulation of neuronal activity and blood flow in the retina by release of gliotransmitters. Philos. Trans. R. Soc. B 2015, 370, 1672.
- Reichenbach, A.; Bringmann, A. Glia of the human retina. Glia 2020, 68, 768–796.
- Carpi-Santos, R.; de Melo Reis, R.A.; Gomes, F.C.A.; Calaza, K.C. Contribution of Müller Cells in the Diabetic Retinopathy Development: Focus on Oxidative Stress and Inflammation. Antioxidants 2022, 11, 617.
- Subirada, P.V.; Paz, M.C.; Ridano, M.E.; Lorenc, V.E.; Vaglienti, M.V.; Barcelona, P.F.; Luna, J.D.; Sánchez, M.C. A journey into the retina: Müller glia commanding survival and death. Eur. J. Neurosci. 2018, 47, 1429–1443.
- Vecino, E.; Rodriguez, F.D.; Ruzafa, N.; Pereiro, X.; Sharma, S.C. Glia-neuron interactions in the mammalian retina. Prog. Retin. Eye Res. 2016, 51, 1–40.
- Santos, A.M.; Calvente, R.; Tassi, M.; Carrasco, M.C.; Martín-Oliva, D.; Marín-Teva, J.L.; Navascués, J.; Cuadros, M.A. Embryonic and postnatal development of microglial cells in the mouse retina. J. Comp. Neurol. 2008, 506, 224–239.
- Li, F.; Jiang, D.; Samuel, M.A. Microglia in the developing retina. Neural. Dev. 2019, 14, 12.
- Kinuthia, U.M.; Wolf, A.; Langmann, T. Microglia and Inflammatory Responses in Diabetic Retinopathy. Front. Immunol. 2020, 11, 564077.
- Yu, C.; Roubeix, C.; Sennlaub, F.; Saban, D.R. Microglia versus Monocytes: Distinct Roles in Degenerative Diseases of the Retina. Trends Neurosci. 2020, 43, 433–449.
- Strauss, O. The retinal pigment epithelium in visual function. Physiol. Rev. 2005, 85, 845–881.
- Ye, X.; Wang, Y.; Nathans, J. The Norrin/Frizzled4 signaling pathway in retinal vascular development and disease. Trends Mol. Med. 2010, 16, 417–425.
- George, S.M.; Lu, F.; Rao, M.; Leach, L.L.; Gross, J.M. The retinal pigment epithelium: Development, injury responses, and regenerative potential in mammalian and non-mammalian systems. Prog. Retin. Eye Res. 2021, 85, 100969.
- Yamashita, H.; Hoenerhoff, M.J.; Peddada, S.D.; Sills, R.C.; Pandiri, A.R. Chemical Exacerbation of Light-induced Retinal Degeneration in F344/N Rats in National Toxicology Program Rodent Bioassays. Toxicol. Pathol. 2016, 44, 892–903.
- Yamashita, H.; Hoenerhoff, M.J.; Shockley, K.R.; Peddada, S.D.; Gerrish, K.E.; Sutton, D.; Cummings, C.A.; Wang, Y.; Julie, F.F.; Behl, M.; et al. Reduced Disc Shedding and Phagocytosis of Photoreceptor Outer Segment Contributes to Kava Kava Extract-induced Retinal Degeneration in F344/N Rats. Toxicol. Pathol. 2018, 46, 564–573.
- Dong, Q.; Hu, N.; Yue, H.; Wang, H. Inhibitory Activity and Mechanism Investigation of Hypericin as a Novel α-Glucosidase Inhibitor. Molecules 2021, 26, 4566.
- Harris, M.S.; Sakamoto, T.; Kimura, H.; He, S.; Spee, C.; Gopalakrishna, R.; Gundimeda, U.; Yoo, J.S.; Hinton, D.R.; Ryan, S.J. Hypericin inhibits cell growth and induces apoptosis in retinal pigment epithelial cells: Possible involvement of protein kinase C. Curr. Eye Res. 1996, 15, 255–262.
- Wielgus, A.R.; Chignell, C.F.; Miller, D.S.; Van Houten, B.; Meyer, J.; Hu, D.N.; Roberts, J.E. Phototoxicity in human retinal pigment epithelial cells promoted by hypericin, a component of St. John’s wort. Photochem. Photobiol. 2007, 83, 706–713.
- Devi Daimary, U.; Girisa, S.; Parama, D.; Verma, E.; Kumar, A.; Kunnumakkara, A.B. Embelin: A novel XIAP inhibitor for the prevention and treatment of chronic diseases. J. Biochem. Mol. Toxicol. 2022, 36, e22950.
- Kifle, Z.D.; Belayneh, Y.M. Antidiabetic and Anti-hyperlipidemic Effects of the Crude Hydromethanol Extract of Hagenia abyssinica (Rosaceae) Leaves in Streptozotocin-Induced Diabetic Mice. Diabetes Metab. Syndr. Obes. 2020, 13, 4085–4094.
- Kifle, Z.D.; Atnafie, S.A.; Yimer Tadesse, T.; Belachew, T.F.; Kidanu, B.B. Methanolic Crude Extract of Hagenia abyssinica Possesses Significant Antidiarrheal Effect: Evidence for In Vivo Antidiarrheal Activity. Evid. Based Complement. Alternat. Med. 2021, 2021, 9944629.
- Low, G.; Rogers, L.J.; Brumley, S.P.; Ehrlich, D. Visual deficits and retinotoxicity caused by the naturally occurring anthelmintics, Embelia ribes and Hagenia abyssinica. Toxicol. Appl. Pharmacol. 1985, 81, 220–230.
- Mandal, M.N.; Patlolla, J.M.; Zheng, L.; Agbaga, M.P.; Tran, J.T.; Wicker, L.; Kasus-Jacobi, A.; Elliott, M.H.; Rao, C.V.; Anderson, R.E. Curcumin protects retinal cells from light-and oxidant stress-induced cell death. Free Radic. Biol. Med. 2009, 46, 672–679.
- Vasireddy, V.; Chavali, V.R.; Joseph, V.T.; Kadam, R.; Lin, J.H.; Jamison, J.A.; Kompella, U.B.; Reddy, G.B.; Ayyagari, R. Rescue of photoreceptor degeneration by curcumin in transgenic rats with P23H rhodopsin mutation. PLoS ONE 2011, 6, e21193.
- Wang, L.; Li, C.; Guo, H.; Kern, T.S.; Huang, K.; Zheng, L. Curcumin inhibits neuronal and vascular degeneration in retina after ischemia and reperfusion injury. PLoS ONE 2011, 6, e23194.
- Xie, T.; Chen, X.; Chen, W.; Huang, S.; Peng, X.; Tian, L.; Wu, X.; Huang, Y. Curcumin is a Potential Adjuvant to Alleviates Diabetic Retinal Injury via Reducing Oxidative Stress and Maintaining Nrf2 Pathway Homeostasis. Front. Pharmacol. 2021, 12, 796565.
- López-Malo, D.; Villarón-Casares, C.A.; Alarcón-Jiménez, J.; Miranda, M.; Díaz-Llopis, M.; Romero, F.J.; Villar, V.M. Curcumin as a Therapeutic Option in Retinal Diseases. Antioxidants 2020, 9, 48.
- Chandrasekaran, P.R.; Madanagopalan, V.G. Role of Curcumin in Retinal Diseases-A review. Graefes. Arch. Clin. Exp. Ophthalmol. 2022, 260, 1457–1473.
- Lu, H.F.; Lai, K.C.; Hsu, S.C.; Lin, H.J.; Yang, M.D.; Chen, Y.L.; Fan, M.J.; Yang, J.S.; Cheng, P.Y.; Kuo, C.L.; et al. Curcumin induces apoptosis through FAS and FADD, in caspase-3-dependent and -independent pathways in the N18 mouse-rat hybrid retina ganglion cells. Oncol. Rep. 2009, 22, 97–104.
- Lin, H.L.; Yang, J.S.; Yang, J.H.; Fan, S.S.; Chang, W.C.; Li, Y.C.; Chung, J.G. The role of Ca2+ on the DADS-induced apoptosis in mouse-rat hybrid retina ganglion cells (N18). Neurochem. Res. 2006, 31, 383–393.
- Ekor, M. The growing use of herbal medicines: Issues relating to adverse reactions and challenges in monitoring safety. Front. Pharmacol. 2014, 4, 177.
- Zhang, H.W.; Zhang, H.; Grant, S.J.; Wan, X.; Li, G. Single herbal medicine for diabetic retinopathy. Cochrane Database Syst. Rev. 2018, 12, Cd007939.
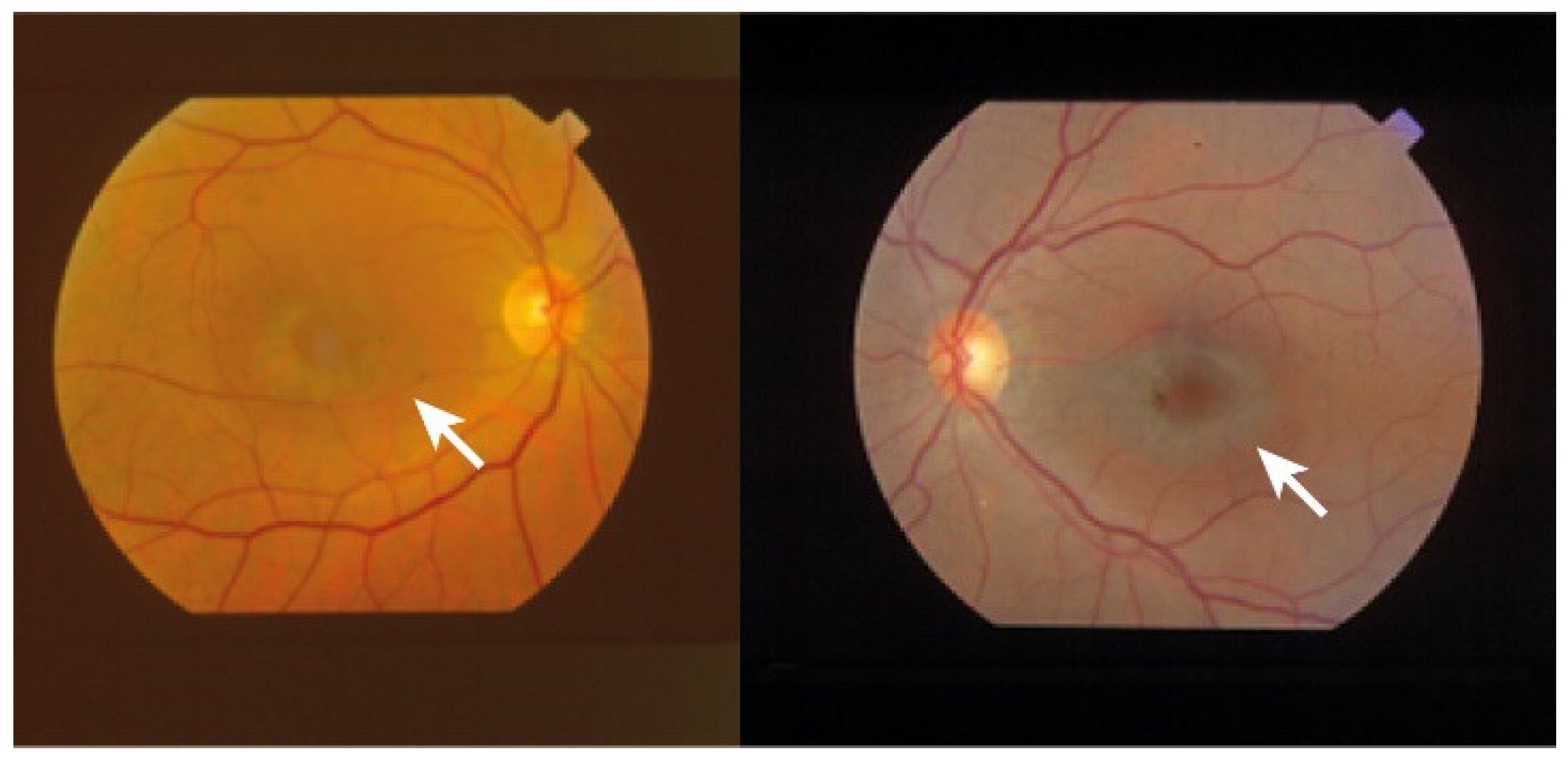
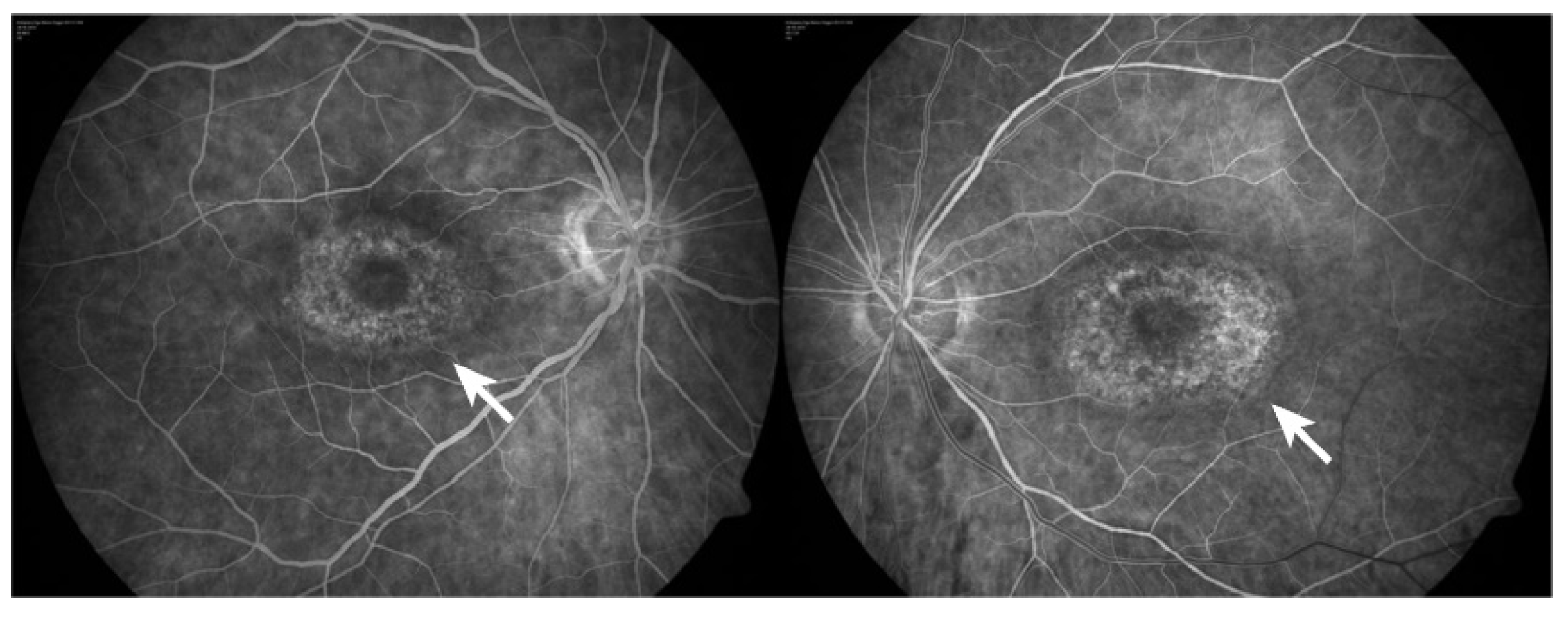
- Yusuf, I.H.; Sharma, S.; Luqmani, R.; Downes, S.M. Hydroxychloroquine retinopathy. Eye 2017, 31, 828–845.
- Godinho, G.; Madeira, C.; Falcão, M.; Penas, S.; Dinah-Bragança, T.; Brandão, E.; Carneiro, Â.; Santos-Silva, R.; Falcão-Reis, F.; Beato, J. Longitudinal Retinal Changes Induced by Hydroxychloroquine in Eyes without Retinal Toxicity. Ophthalmic. Res. 2021, 64, 290–296.
- Yang, J.; Guo, Z.; Liu, X.; Liu, Q.; Wu, M.; Yao, X.; Liu, Y.; Cui, C.; Li, H.; Song, C.; et al. Cytotoxicity Evaluation of Chloroquine and Hydroxychloroquine in Multiple Cell Lines and Tissues by Dynamic Imaging System and Physiologically Based Pharmacokinetic Model. Front. Pharmacol. 2020, 11, 574720.
- Nguyen Hoang, A.T.; Lee, H.; Lee, S.J. Casein kinase I inhibitor D4476 influences autophagy and apoptosis in chloroquine-induced adult retinal pigment epithelial-19 cells. Exp. Eye Res. 2022, 218, 109004.
- Corradetti, G.; Violanti, S.; Au, A.; Sarraf, D. Wide field retinal imaging and the detection of drug associated retinal toxicity. Int. J. Retin. Vitr. 2019, 5, 26.
- Richa, S.; Yazbek, J.C. Ocular adverse effects of common psychotropic agents: A review. CNS Drugs 2010, 24, 501–526.
- Dorgau, B.; Georgiou, M.; Chaudhary, A.; Moya-Molina, M.; Collin, J.; Queen, R.; Hilgen, G.; Davey, T.; Hewitt, P.; Schmitt, M.; et al. Human Retinal Organoids Provide a Suitable Tool for Toxicological Investigations: A Comprehensive Validation Using Drugs and Compounds Affecting the Retina. Stem. Cells Transl. Med. 2022, 11, 159–177.
- Wang, D.; Au, A.; Gunnemann, F.; Hilely, A.; Scharf, J.; Tran, K.; Sun, M.; Kim, J.H.; Sarraf, D. Pentosan-associated maculopathy: Prevalence, screening guidelines, and spectrum of findings based on prospective multimodal analysis. Can. J. Ophthalmol. 2020, 55, 116–125.
- Hanif, A.M.; Armenti, S.T.; Taylor, S.C.; Shah, R.A.; Igelman, A.D.; Jayasundera, K.T.; Pennesi, M.E.; Khurana, R.N.; Foote, J.E.; O’Keefe, G.A.; et al. Phenotypic Spectrum of Pentosan Polysulfate Sodium-Associated Maculopathy: A Multicenter Study. JAMA Ophthalmol. 2019, 137, 1275–1282.
- Leung, E.H.; Sharma, S.; Levie-Sprick, A.; Lee, G.D.; Cho, H.; Mukkamala, K. Pentosan Polysulfate Sodium-Associated Pigmentary Retinopathy: Risk Factors and Fundus Findings. Clin. Ophthalmol. 2021, 15, 4809–4816.
- Van Bergen, T.; Etienne, I.; Jia, J.; Li, J.P.; Vlodavsky, I.; Stitt, A.; Vermassen, E.; Feyen, J.H.M. Heparanase Deficiency Is Associated with Disruption, Detachment, and Folding of the Retinal Pigment Epithelium. Curr. Eye Res. 2021, 46, 1166–1170.
- Khan, M.J.; Papakostas, T.; Kovacs, K.; Gupta, M.P. Drug-induced maculopathy. Curr. Opin. Ophthalmol. 2020, 31, 563–571.
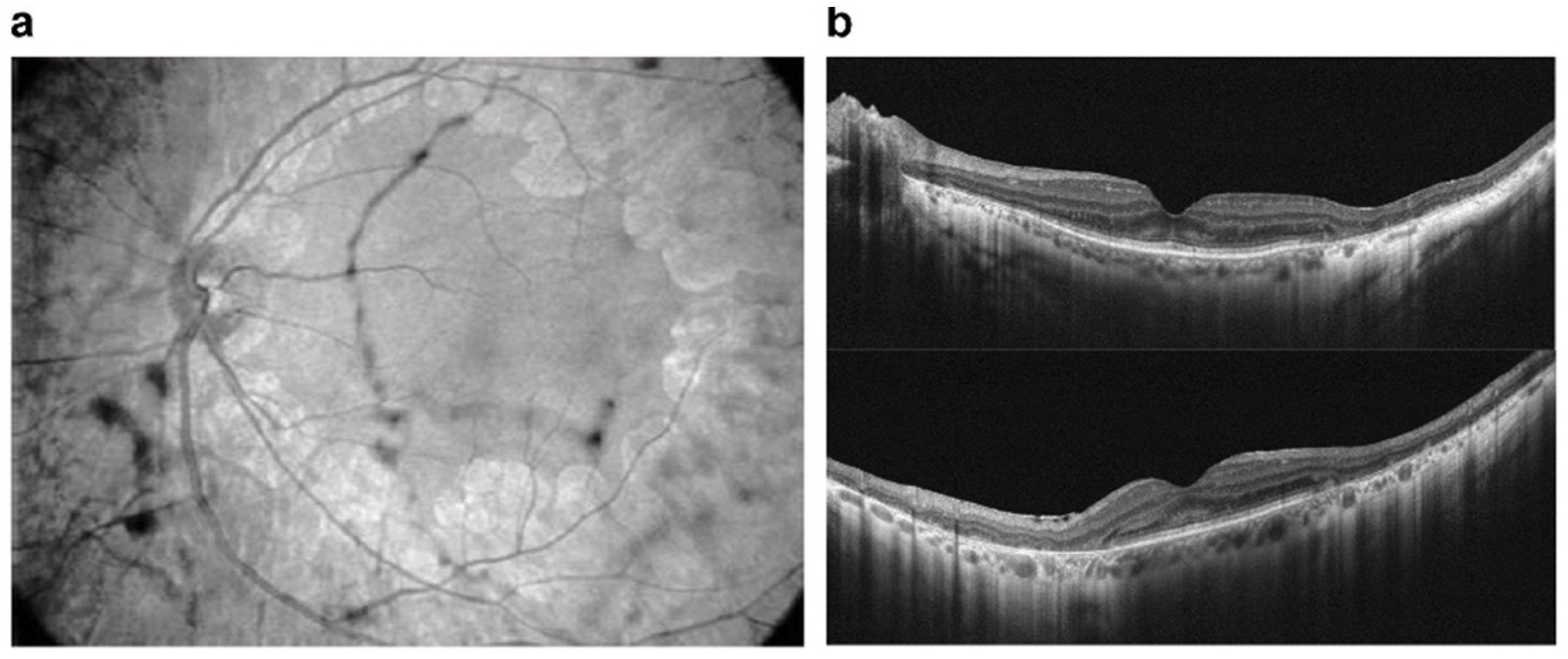
- Viola, F.; Barteselli, G.; Dell’Arti, L.; Vezzola, D.; Mapelli, C.; Villani, E.; Ratiglia, R. Multimodal imaging in deferoxamine retinopathy. Retina 2014, 34, 1428–1438.
- Sakamoto, K.; Suzuki, T.; Takahashi, K.; Koguchi, T.; Hirayama, T.; Mori, A.; Nakahara, T.; Nagasawa, H.; Ishii, K. Iron-chelating agents attenuate NMDA-Induced neuronal injury via reduction of oxidative stress in the rat retina. Exp. Eye Res. 2018, 171, 30–36.
- Totsuka, K.; Ueta, T.; Uchida, T.; Roggia, M.F.; Nakagawa, S.; Vavvas, D.G.; Honjo, M.; Aihara, M. Oxidative stress induces ferroptotic cell death in retinal pigment epithelial cells. Exp. Eye Res. 2019, 181, 316–324.
- Liu, B.; Wang, W.; Shah, A.; Yu, M.; Liu, Y.; He, L.; Dang, J.; Yang, L.; Yan, M.; Ying, Y.; et al. Sodium iodate induces ferroptosis in human retinal pigment epithelium ARPE-19 cells. Cell Death Dis. 2021, 12, 230.
- Chen, C.; Chen, J.; Wang, Y.; Liu, Z.; Wu, Y. Ferroptosis drives photoreceptor degeneration in mice with defects in all-trans-retinal clearance. J. Biol. Chem. 2021, 296, 100187.
- Abalem, M.F.; Carricondo, P.C.; Rao, R.C. Bullseye Retinopathy from Antiretroviral Therapy. Ophthalmology 2017, 124, 1539.
- Hu, X.; Calton, M.A.; Tang, S.; Vollrath, D. Depletion of Mitochondrial DNA in Differentiated Retinal Pigment Epithelial Cells. Sci. Rep. 2019, 9, 15355.
- Urner-Bloch, U.; Urner, M.; Jaberg-Bentele, N.; Frauchiger, A.L.; Dummer, R.; Goldinger, S.M. MEK inhibitor-associated retinopathy (MEKAR) in metastatic melanoma: Long-term ophthalmic effects. Eur. J. Cancer 2016, 65, 130–138.
- Van Dijk, E.H.C.; Duits, D.E.M.; Versluis, M.; Luyten, G.P.M.; Bergen, A.A.B.; Kapiteijn, E.W.; de Lange, M.J.; Boon, C.J.F.; van der Velden, P.A. Loss of MAPK Pathway Activation in Post-Mitotic Retinal Cells as Mechanism in MEK Inhibition-Related Retinopathy in Cancer Patients. Medicine 2016, 95, e3457.
- Pellino, A.; Loupakis, F.; Cadamuro, M.; Dadduzio, V.; Fassan, M.; Guido, M.; Cillo, U.; Indraccolo, S.; Fabris, L. Precision medicine in cholangiocarcinoma. Transl. Gastroenterol. Hepatol. 2018, 3, 40.
- Abou-Alfa, G.K.; Sahai, V.; Hollebecque, A.; Vaccaro, G.; Melisi, D.; Al-Rajabi, R.; Paulson, A.S.; Borad, M.J.; Gallinson, D.; Murphy, A.G.; et al. Pemigatinib for previously treated, locally advanced or metastatic cholangiocarcinoma: A multicentre, open-label, phase 2 study. Lancet Oncol. 2020, 21, 671–684.
- Alekseev, O.; Ojuok, E.; Cousins, S. Multifocal serous retinopathy with pemigatinib therapy for metastatic colon adenocarcinoma. Int. J. Retin. Vitr. 2021, 7, 34.
- Etminan, M.; Sodhi, M.; Mikelberg, F.S.; Maberley, D. Risk of Ocular Adverse Events Associated with Use of Phosphodiesterase 5 Inhibitors in Men in the US. JAMA Ophthalmol. 2022, 140, 480–484.
- Zahavi, A.; Weiss, S.; Vieyra, M.; Nicholson, J.D.; Muhsinoglu, O.; Barinfeld, O.; Zadok, D.; Goldenberg-Cohen, N. Ocular Effects of Sildenafil in Naïve Mice and a Mouse Model of Optic Nerve Crush. Investig. Ophthalmol. Vis. Sci. 2019, 60, 1987–1995.
- Berkowitz, B.A.; Podolsky, R.H.; Lins Childers, K.; Saadane, A.; Kern, T.S.; Roberts, R.; Olds, H.; Joy, J.; Richards, C.; Rosales, T.; et al. Sildenafil-evoked photoreceptor oxidative stress in vivo is unrelated to impaired visual performance in mice. PLoS ONE 2021, 16, e0245161.
- Haugnes, H.S.; Bosl, G.J.; Boer, H.; Gietema, J.A.; Brydøy, M.; Oldenburg, J.; Dahl, A.A.; Bremnes, R.M.; Fosså, S.D. Long-term and late effects of germ cell testicular cancer treatment and implications for follow-up. J. Clin. Oncol. 2012, 30, 3752–3763.
- Alkan, A.; Talaz, S. Cilioretinal artery occlusion associated with cisplatin. J. Oncol. Pharm. Pract. 2019, 25, 969–971.
- Dulz, S.; Asselborn, N.H.; Dieckmann, K.P.; Matthies, C.; Wagner, W.; Weidmann, J.; Seidel, C.; Oing, C.; Berger, L.A.; Alsdorf, W.; et al. Retinal toxicity after cisplatin-based chemotherapy in patients with germ cell cancer. J. Cancer Res. Clin. Oncol. 2017, 143, 1319–1325.
- Langevin, S.; Chang, J.S.; Chang, S. Serous retinopathy associated with cisplatin treatment. Retin. Case. Brief Rep. 2019, 13, 211–214.
- Khadka, S.; Byanju, R.; Poon, S. Chemotherapy-Induced Central Retinal Artery Occlusion in Gestational Trophoblastic Neoplasia: Case Report. Int. Med. Case. Rep. J. 2020, 13, 431–435.
- Fındık, H.; Tumkaya, L.; Yılmaz, A.; Gökhan Aslan, M.; Okutucu, M.; Akyildiz, K.; Mercantepe, T. The protective effects of astaxanthin against cisplatin-induced retinal toxicity. Cutan. Ocul. Toxicol. 2019, 38, 59–65.
- Taşlı, N.G.; Uçak, T.; Karakurt, Y.; Keskin Çimen, F.; Özbek Bilgin, A.; Kurt, N.; Süleyman, H. The effects of rutin on cisplatin induced oxidative retinal and optic nerve injury: An experimental study. Cutan. Ocul. Toxicol. 2018, 37, 252–257.
- Karakurt, Y.; Uçak, T.; Tasli, N.; Ahiskali, I.; Şipal, S.; Kurt, N.; Süleyman, H. The effects of lutein on cisplatin-induced retinal injury: An experimental study. Cutan. Ocul. Toxicol. 2018, 37, 374–379.
- Sunar, M.; Yazici, G.N.; Mammadov, R.; Kurt, N.; Arslan, Y.K.; Süleyman, H. Coenzyme Q10 effect on cisplatin-induced oxidative retinal injury in rats. Cutan. Ocul. Toxicol. 2021, 40, 312–318.
- Wu, C.M.; Su, F.H.; Muo, C.H.; Huang, J.C.; Wu, M.M.; Yeh, C.C. Analysis of Different Types of Interferon-Associated Retinopathy in Patients with Chronic Hepatitis C Virus Infection Treated with Pegylated Interferon Plus Ribavirin. Viruses 2021, 13, 475.
- Roche, S.L.; Ruiz-Lopez, A.M.; Moloney, J.N.; Byrne, A.M.; Cotter, T.G. Microglial-induced Müller cell gliosis is attenuated by progesterone in a mouse model of retinitis pigmentosa. Glia 2018, 66, 295–310.
- Kutty, R.K.; Samuel, W.; Duncan, T.; Postnikova, O.; Jaworski, C.; Nagineni, C.N.; Redmond, T.M. Proinflammatory cytokine interferon-γ increases the expression of BANCR, a long non-coding RNA, in retinal pigment epithelial cells. Cytokine 2018, 104, 147–150.
- Wei, T.T.; Zhang, M.Y.; Zheng, X.H.; Xie, T.H.; Wang, W.; Zou, J.; Li, Y.; Li, H.Y.; Cai, J.; Wang, X.; et al. Interferon-γ induces retinal pigment epithelial cell Ferroptosis by a JAK1-2/STAT1/SLC7A11 signaling pathway in Age-related Macular Degeneration. FEBS J. 2022, 289, 1968–1983.
- Taguchi, M.; Someya, H.; Inada, M.; Nishio, Y.; Takayama, K.; Harimoto, K.; Karasawa, Y.; Ito, M.; Takeuchi, M. Retinal changes in mice spontaneously developing diabetes by Th17-cell deviation. Exp. Eye Res. 2020, 198, 108155.
- Jung, I.; Jung, D.; Zha, Z.; Jeong, J.; Noh, S.; Shin, J.; Park, J.K.; Kim, K.S.; Jeong, Y.; Hur, J.; et al. Interferon-γ inhibits retinal neovascularization in a mouse model of ischemic retinopathy. Cytokine 2021, 143, 155542.
- Silberstein, S.D.; McCrory, D.C. Ergotamine and dihydroergotamine: History, pharmacology, and efficacy. Headache 2003, 43, 144–166.
- Arana, L.A.; Bach, M.B.; Vedana, G.; Volkmann, M.A.; Arana, J. Cefalium-induced bilateral transient myopia, retinal folds, and focal choroidal delay. Retin. Case. Brief Rep. 2018, 12, 118–121.
- Leinonen, H.; Choi, E.H.; Gardella, A.; Kefalov, V.J.; Palczewski, K. A Mixture of U.S. Food and Drug Administration-Approved Monoaminergic Drugs Protects the Retina from Light Damage in Diverse Models of Night Blindness. Investig. Ophthalmol. Vis. Sci. 2019, 60, 1442–1453.
- Kasica, N.; Święch, A.; Saładziak, K.; Mackiewicz, J.; Osęka, M. The Inhibitory Effect of Selected D(2) Dopaminergic Receptor Agonists on VEGF-Dependent Neovascularization in Zebrafish Larvae: Potential New Therapy in Ophthalmic Diseases. Cells 2022, 11, 1202.
- Hu, Q.D.; Xu, L.L.; Gong, Y.; Wu, G.H.; Wang, Y.W.; Wu, S.J.; Zhang, Z.; Mao, W.; Zhou, Y.S.; Li, Q.B.; et al. Lysergic acid diethylamide causes photoreceptor cell damage through inducing inflammatory response and oxidative stress. Cutan. Ocul. Toxicol. 2018, 37, 233–239.
- Chen, K.; He, X.; Li, C.; Ou, Y.; Li, Y.; Lai, J.; Lv, M.; Li, X.; Ran, P.; Li, Y. Lysergic acid diethylamide causes mouse retinal damage by up-regulating p-JAK1/p-STAT1. Cutan. Ocul. Toxicol. 2020, 39, 106–110.
- Jhaj, G.; Jhaj, R.; Shrier, E.M. Gemcitabine-Induced Retinopathy. Retina 2017, 37, e130–e131.
- Behera, U.C.; Modi, R.R.; Sheth, J.; Singh, A. Bilateral macular infarction after gemcitabine and carboplatin chemotherapy. Int. Ophthalmol. 2018, 38, 2195–2198.
- Loscalzo, F.; Balbarrey, M.; Grigera, J.D. Gemcitabine-Associated Retinopathy with Bilateral Exudative Retinal Detachment and Elschnig’s Spots. Ophthalmic. Surg. Lasers Imaging Retin. 2022, 53, 222–226.
- Martins, J.R.; Reichhart, N.; Kociok, N.; Stindl, J.; Foeckler, R.; Lachmann, P.; Todorov, V.; Castrop, H.; Strauß, O. Systemic ß adrenergic stimulation/sympathetic nerve system stimulation influences intraocular RAS through cAMP in the RPE. Exp. Eye Res. 2019, 189, 107828.
- Skarphedinsdottir, S.B.; Eysteinsson, T.; Árnason, S.S. Mechanisms of Ion Transport Across the Mouse Retinal Pigment Epithelium Measured In Vitro. Investig. Ophthalmol. Vis. Sci. 2020, 61, 31.
- Cheong, K.X.; Barathi, V.A.; Teo, K.Y.C.; Chakravarthy, U.; Tun, S.B.B.; Busoy, J.M.; Ho, C.E.H.; Agrawal, R.; Takahashi, K.; Cheung, C.M.G. Choroidal and Retinal Changes After Systemic Adrenaline and Photodynamic Therapy in Non-Human Primates. Investig. Ophthalmol. Vis. Sci. 2021, 62, 25.
- Hazim, R.A.; Volland, S.; Yen, A.; Burgess, B.L.; Williams, D.S. Rapid differentiation of the human RPE cell line, ARPE-19, induced by nicotinamide. Exp. Eye Res. 2019, 179, 18–24.
- Kaya, M.; Atas, F.; Gulsum Guc, Z.; Oztop, I.; Durak, I.; Saatci, A.O. A cross-sectional optical coherence tomography study in patients on taxane-based therapy and a case report with the literature review. Cutan. Ocul. Toxicol. 2020, 39, 287–293.
- Nghiem-Buffet, S.; Cohen, S.Y.; Giocanti-Auregan, A. Docetaxel Retinopathy: A Case Report. Case. Rep. Ophthalmol. 2017, 8, 21–25.
- Torrado, L.A.; Fivgas, G.D. Unilateral cystoid macular edema and bilateral subfoveal hyperreflectivity following docetaxel chemotherapy: A case report. Am. J. Ophthalmol. Case. Rep. 2020, 20, 100995.
- Tapia Quijada, H.E.; Quijada Fumero, E.; Mesa Lugo, F.I.; Serrano García, M.; Betancor Caro, N. Nepafenac for cystoid macular oedema secondary to paclitaxel. Arch. Soc. Esp. Oftalmol. 2021, 96, 434–437.
- Malcolm, J.; Lune Wong, C.O.; Ching, J.; Saidkasimova, S. Paclitaxel may be a risk factor for retinal phototoxicity. Am. J. Ophthalmol. Case. Rep. 2022, 25, 101292.
- Lin, P.K.; Salvador, J.; Xie, J.; Aguera, K.N.; Koller, G.M.; Kemp, S.S.; Griffin, C.T.; Davis, G.E. Selective and Marked Blockade of Endothelial Sprouting Behavior Using Paclitaxel and Related Pharmacologic Agents. Am. J. Pathol. 2021, 191, 2245–2264.
- Cinici, E.; Dilekmen, N.; Kutlu, Z.; Dincer, B.; Cinici, O.; Balta, H.; Calık, I. Carvone protects against paclitaxel-induced retinal and optic nerve cytotoxicity: A histopathological study. Cutan. Ocul. Toxicol. 2019, 38, 290–293.
- Kim, H.A.; Lee, S.; Eah, K.S.; Yoon, Y.H. Prevalence and Risk Factors of Tamoxifen Retinopathy. Ophthalmology 2020, 127, 555–557.
- Doshi, R.R.; Fortun, J.A.; Kim, B.T.; Dubovy, S.R.; Rosenfeld, P.J. Pseudocystic foveal cavitation in tamoxifen retinopathy. Am. J. Ophthalmol. 2014, 157, 1291–1298.
- Wang, X.; Zhao, L.; Zhang, Y.; Ma, W.; Gonzalez, S.R.; Fan, J.; Kretschmer, F.; Badea, T.C.; Qian, H.H.; Wong, W.T. Tamoxifen Provides Structural and Functional Rescue in Murine Models of Photoreceptor Degeneration. J. Neurosci. 2017, 37, 3294–3310.
- Liberski, S.; Kaluzny, B.J.; Kocięcki, J. Methanol-induced optic neuropathy: A still-present problem. Arch. Toxicol. 2022, 96, 431–451.
- Klein, K.A.; Warren, A.K.; Baumal, C.R.; Hedges, T.R., 3rd. Optical coherence tomography findings in methanol toxicity. Int. J. Retin. Vitr. 2017, 3, 36.
- Laksmita, Y.A.; Sidik, M.; Siregar, N.C.; Nusanti, S. Neuroprotective Effects of Citicoline on Methanol-Intoxicated Retina Model in Rats. J. Ocul. Pharmacol. Ther. 2021, 37, 534–541.
- Taşlı, N.G.; Çimen, F.K.; Karakurt, Y.; Uçak, T.; Mammadov, R.; Süleyman, B.; Kurt, N.; Süleyman, H. Protective effects of Rutin against methanol induced acute toxic optic neuropathy: An experimental study. Int. J. Ophthalmol. 2018, 11, 780–785.
- Ahiskali, I.; Pinar, C.L.; Kiki, M.; Cankaya, M.; Kunak, C.S.; Altuner, D. Effect of taxifolin on methanol-induced oxidative and inflammatory optic nerve damage in rats. Cutan. Ocul. Toxicol. 2019, 38, 384–389.





