Your browser does not fully support modern features. Please upgrade for a smoother experience.

Submitted Successfully!
Thank you for your contribution! You can also upload a video entry or images related to this topic.
For video creation, please contact our Academic Video Service.
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Paola Lanteri | -- | 3019 | 2022-08-09 16:42:45 | | | |
| 2 | Catherine Yang | Meta information modification | 3019 | 2022-08-10 03:44:51 | | |
Video Upload Options
We provide professional Academic Video Service to translate complex research into visually appealing presentations. Would you like to try it?
Cite
If you have any further questions, please contact Encyclopedia Editorial Office.
Garbarino, S.; Garbarino, E.; Lanteri, P. Cyrcadian Rhythm, Mood, and Temporal Patterns of Chocolate. Encyclopedia. Available online: https://encyclopedia.pub/entry/25992 (accessed on 09 May 2026).
Garbarino S, Garbarino E, Lanteri P. Cyrcadian Rhythm, Mood, and Temporal Patterns of Chocolate. Encyclopedia. Available at: https://encyclopedia.pub/entry/25992. Accessed May 09, 2026.
Garbarino, Sergio, Emanuela Garbarino, Paola Lanteri. "Cyrcadian Rhythm, Mood, and Temporal Patterns of Chocolate" Encyclopedia, https://encyclopedia.pub/entry/25992 (accessed May 09, 2026).
Garbarino, S., Garbarino, E., & Lanteri, P. (2022, August 09). Cyrcadian Rhythm, Mood, and Temporal Patterns of Chocolate. In Encyclopedia. https://encyclopedia.pub/entry/25992
Garbarino, Sergio, et al. "Cyrcadian Rhythm, Mood, and Temporal Patterns of Chocolate." Encyclopedia. Web. 09 August, 2022.
Copy Citation
Taking small amounts of chocolate, in the morning at breakfast at the onset of the active phase, helps speed up resynchronization time. The high flavonoid contents in chocolate promote cardioprotection, metabolic regulation, neuroprotection, and neuromodulation with direct actions on brain function, neurogenesis, angiogenesis, and mood. Cocoa products and especially chocolate are foodstuffs originating from South America. Cocoa is obtained from the seeds of the Theobroma cacao tree, which are then dried, shelled, fermented, and ground with other substances such as sugar, fat, and other flavorings to produce the wide variety of chocolate available on the market, from dark to milk variations.
chocolate
flavonoid
brain function
mood
1. Biochemical Components and Neurobiological Impact
Chocolate contains more than 300–500 known chemicals, some of which also act on brain cells and modulate mood [1]. Of these 300 to 500 chemicals in chocolate, some play an important role in humans, influencing neurocognitive functions.
- -
-
Carbohydrates, which have known behavioral effects.
- -
-
Flavanols, which are ubiquitous in the plant kingdom. In foods normally consumed in the diet, high levels of flavonoids can be found in green and black tea, grapes, red wine, apples, and especially in cocoa and cocoa-containing products. In fact, cocoa is particularly rich in flavonoids and contains a distinct complement of flavanols (a subclass of flavonoids), flavan-3-ols, mainly present in the form of epicatechin and catechin [4], and their derivatives in high concentrations [5]. Flavan-3-ols are the building blocks for polymeric procyanidin type B-2.
- -
-
Methylxanthines (MX), such as caffeine and its highly fat-soluble derivative and metabolite theobromine, which have peak plasma levels 60–120 min after ingestion.Like caffeine, theobromine binds to adenosine receptors, exhibiting its psychoactive potential similar to that of caffeine. However, these two MX have distinct functional binding properties.
- -
-
Biogenic amines, such as serotonin, tryptophan, phenylethylamine, tyrosine, tryptamine, and tyramine, have a concentration that increases during fermentation and decreases during roasting and alkalinization.
- -
-
Anandamide, an endogenous ligand for the cannabinoid receptor that is found in low quantities, such as 0.5 mg g−1, salsolinol, and tetrahydro-b-carboline.
2. Chocolate and Brain Functions
In order to exert effects in the brain, flavanols cross the BBB by a process that is not only time-dependent but also stereoselective, which, thus, favors the passage of epicatechin more than catechin, as has been demonstrated in two cell lines, one of rat and one of human origin [6]; the permeability is proportional to the degree of lipophilicity and inversely proportional to the degree of polarity.
In humans, the main polyphenols increase cerebral blood flow (CBF) and are derived from nutrients such as cocoa, wine, grape seeds, berries, tea, tomatoes, and soy [7].
Optimal brain function requires an adequate CBF that promotes the correct and constant supply of oxygen and glucose to neurons and the excretion of deposited waste products. Increasing CBF is also a potential tool for enhancing brain function. Flavanols and their metabolites have the ability to reach and accumulate in the brain regions mainly involved in learning processes and memory, and are therefore thought to exert a direct brain action on cognitive function and neuroprotection [3]. After chronic administration of chocolate, high concentrations of tangeretin were found in the striatum nucleus, hypothalamus, and hippocampus of the rat [8].
The neurobiological impact of flavanols on the brain in the areas of learning, memory, cognitive function, and mood is thought to occur mainly in two ways. The effects of flavonoids on the brain are mediated by their function of neuroprotecting vulnerable neurons in particular, improving neuronal function and stimulating regeneration (neurogenesis) [9] by interacting with intracellular neuronal signaling pathways that control neuronal survival and differentiation, long-term potentiation (LTP), and memory.
First, flavonoids interact with a number of cellular signaling pathways by activating gene expression and protein synthesis for the maintenance of LTP, and the stabilization of long-term memories [10] are critical for neurogenesis, synaptic growth, and survival of neurons, mainly in the brain hippocampus and subventricular area related to learning and memory [11][12].
Secondly, flavonoids induce vasodilation through nitric oxide at both cardiovascular and peripheral levels through the production of nitric oxide (NO), a key regulator of vascular function, which acts as a signaling molecule by inhibiting the action of adhesion molecules in atheromatous plaque that cause inflammation [13], and, most importantly, promote and improve the function of the vascular endothelium by acting, with dilating action, on the smooth muscle tissue of blood vessels [14]. This effect on the vascular system with endothelium-dependent vasodilation, contributes to the maintenance of normal blood flow and improvement of blood pressure; it also induces a reduction in platelet aggregation [15][16][17][18][19][20]. This. in turn. results in increased CBF and blood perfusion throughout the central and peripheral nervous system [21], allowing better oxygen and glucose delivery to neurons and removal of waste metabolites in the nervous systems [22]. Among flavanols, epicatechin has the greatest ability to increase nitric oxide (NO) bioavailability, leading to improvements in vascular tone and blood pressure regulation [23]. These vascular changes occurring at the peripheral level may also extend to cerebral perfusion, leading to optimized cerebrovascular integration during neuronal activation phases, a mechanism considered crucial for the functional and structural integrity of the brain and for promoting adult neurogenesis in the hippocampus [24].
Administration of cocoa flavonoids, therefore, also stimulates angiogenesis in the hippocampus [25][26], as demonstrated by administering epicatechin, given to mice at a dose of 500 mg g−1 (daily supply of 2.5 mg). Combining epicatechin with exercise also improved the consolidation of spatial memory and the density of dendritic spines in the dentate gyrus of the hippocampus. In the same study, epicatechin treatment was shown to increase learning-associated gene expression in the hippocampus, while it did not appear to influence neurogenesis in the adult hippocampus [26].
Thus, flavonoids could also exert their neurocognitive effects both directly and indirectly by interacting with the cellular network and molecular system deputed to memory acquisition, storage, retrieval, and learning [9], also through long-term potentiation, synaptic plasticity [27], enhanced neuronal connection, and communication.
Epidemiological studies suggest that regular flavonoid intake may be associated with better cognitive function [28], to decreased risk of dementia and cognitive decline [29][30][31], better cognitive development over a 10-year period [32], and improved dose-dependent cognitive performance in physiological aging [33].
3. Chocolate and Mood
A lot of data from the literature support the hypothesis of the influence of theobromine and caffeine on mood and cognitive function [34][35][36][37], but the impact and mechanism by which flavanols affect mood remains unclear.
It is commonly believed that eating chocolate improves mood and rapidly induces a sense of well-being in people [37]. An initial rapid effect of chocolate on emotional comfort appears to be related to the ability of the carbohydrates it contains, to promote such positive feelings through the release of several gut and brain peptides [38].
In rats, the intake of cocoa-extracted polyphenols, while significantly reducing the duration of immobility in a forced swimming test, had no effect on locomotor activity in the open field, confirming its specific antidepressant effect [39]. The most likely basis for this effect may be attributable to endorphin release [40]. Indeed, sweet food intake is increased by opioid agonists and decreased by opioid antagonists [41][42]. The effect of chocolate is also exerted through interaction with neurotransmitters such as dopamine (tyrosine contained in chocolate is the precursor to dopamine), serotonin, and endorphins, which contribute to appetite, reward, and mood regulation. The dopaminergic system contributes to the desire to consume chocolate, probably by acting mainly non-specifically towards food. After carbohydrate ingestion, only when the protein component of the meal is less than 2% does it induce an increase in serotonin concentrations in the brain [38]. It should be noted that chocolate contains 5% of its caloric content in the form of protein, which would cancel out any effect of serotonin. Moreover, manipulations of tryptophan, the precursor of serotonin, also cause physiological changes that are too slow to explain the mood effects described during or immediately after eating chocolate [43]. Another area where chocolate might act could be in the area of opioids, which are known to play a role in the palatability of preferred foods [44], releasing endorphins during food intake, and, thus, justifying the increase in pleasure during food intake [45]. The mood effects of cocoa may also be partly due to opioids released in response to the ingestion of sweets and other pleasantly palatable foods [46][47]. The increase in central opioidergic activity, in turn, stimulates the immediate release of beta-endorphin in the hypothalamus, which exerts an analgesic effect. Bad mood stimulates consumption of comfort foods such as chocolate in two different ways [48]. The former is called craving and is associated with an impulsive desire for chocolate, and its compulsive consumption occurs especially when under high emotional stress, showing a clear link between the perception of a negative mood and the intense desire to consume chocolate [49]. The association between chocolate craving and consumption under emotional stress was demonstrated in a study in which subjects had to listen to music that induced a happy or sad mood. Chocolate consumption was increased by listening to sad music [48].
The second modality to be considered is the palatability of the food. The pleasure induced by palatable food is regulated by endogenous opioids that stimulate food intake in rats. The pleasure induced by palatable foods is regulated by endogenous opioids that stimulate their intake in rats. In humans, however, the critical factor in satisfying chocolate craving appears to be taste and mouthfeel [50]. Females, mainly in the perimenstrual period, seems more sensitive to chocolate. The response to satiety seems to vary by gender [51].
It is more conceivable that an important role in liking or craving chocolate is due more to the composite sensory properties of chocolate than to its role in appetite and satiety [52]. During the consumption of chocolate, different brain areas are also activated depending on the motivation to eat chocolate, based on positive/appetitive stimuli or associated with negative/adverse stimuli. Modulation of brain activity has been observed in chemosensory cortical areas such as the insula, prefrontal regions, and caudomedial and caudolateral orbitofrontal cortex, with overlapping and co-activation under contrasting motivational conditions [53]. The ability to activate images of appetizing foods involved in food motivation and hedonism in a fronto–striatal–amygdala–midbrain network appears to be dependent on individual variability in reward sensitivity. If this same neuronal circuit is stimulated in the animal, it may result in the cancellation of the sense of satiety and cause overeating of highly palatable foods [54].
The smell of chocolate itself is sufficient to modulate brain activity recorded on the electroencephalography (EEG). The smell of chocolate induces a significant reduction in theta activity compared to any other stimulus. Theta activity is considered to be closely related to attentional level, cognitive load in general, and, in this specific case, to olfactory perception, so a reduction in theta activity could be indicative of a reduced level of attention and an increased propensity to distraction [55]. In addition to olfaction, the sight of chocolate also evokes activations in the brain and especially in the medial orbitofrontal cortex and ventral striatum, particularly in subjects who crave chocolate compared to those who do not: the combination of an image of chocolate with chocolate in the mouth evoked greater brain activation than the sight of the sum of the different components in the medial orbitofrontal cortex and cingulate cortex [56].
The motivation for chocolate preference appears to be primarily, if not entirely, sensory. The origin of the liking of its sensory properties is unclear; it could be innate or acquired based on the sweetness, texture, and aroma characteristics of chocolate, or it could depend on the interaction between a person’s state and the post-gastronomic effects of chocolate. Surprisingly, there is little evidence of a relationship between chocolate addiction and chocolate liking [50]. However, chocolate consumption fails to activate the key structure for drug addiction, the nucleus accumbens [57][58][59].
The effect of chocolate on mood may be attributed to the affinity for adenosine and benzodiazepine (GABAa) receptors of polyphenolic compounds, which means that their ingestion may have a soothing effect [60]. Some polyphenolic compounds indeed have anxiolytic properties [61]. A small randomized controlled pilot study in humans with chronic fatigue reported a reduction in anxiety-related symptoms after eating polyphenol-rich chocolate, compared to polyphenol-poor chocolate [62].
Pase et al. [63] investigated the acute and subchronic effects of polyphenol supplementation on mood and cognitive performance in a randomized, placebo-controlled, double-blind study. Thirty days of treatment with a high dose of cocoa polyphenols reduced self-rated anxiety and contentment. No significant effect on cognitive performance was recorded with either the high or low dose in either the acute or chronic phase.
The optimal dosage of cocoa polyphenols needed to improve cognitive function and mood remains unclear.
4. Chocolate, Sleep and Circadian Rhythms
Our modern society functions at a hectic pace of activity 24/7, which leads individuals to sacrifice sleep hours and disregard daily sleep–wake rhythms. As experienced daily by shift workers, jet-lagged travelers, or those with so-called social jet-lag syndrome, disturbed sleep–wake rhythms create a conflict, a temporal mismatch between the circadian system and temporal signals derived from the cyclical environmental changes, such as the light–dark cycle, [64] or a desynchronization [65]. This condition, if prolonged over time, leads to chronic sleep disorders (CSD) that result in deficits to health and psychophysical well-being. The most frequent symptoms belong to the neurobehavioral sphere and are often associated with mood changes such as a tendency to depression and impairment of cognitive functions, especially executive functions, as well as cardio- and cerebrovascular disorders, stroke, hypertension, obesity, and diabetes [66]. CSD are a socio-economic and public health issue due to their high prevalence in the general and working population, their impact on health, and work output given the higher incidence of absenteeism, and increased rates of errors and accidents at work [67]. In addition, CSD often have a bidirectional relationship with stress [68].
Due to this complex interaction between external Zeitgeber and internal circadian rhythms, the greatest beneficial effects of entrainment on circadian function are seen when food intake coincides with the activity phase [69][70], whereas an inhibitory effect occurs when food is taken during the rest phase [71][72] (Figure 1).
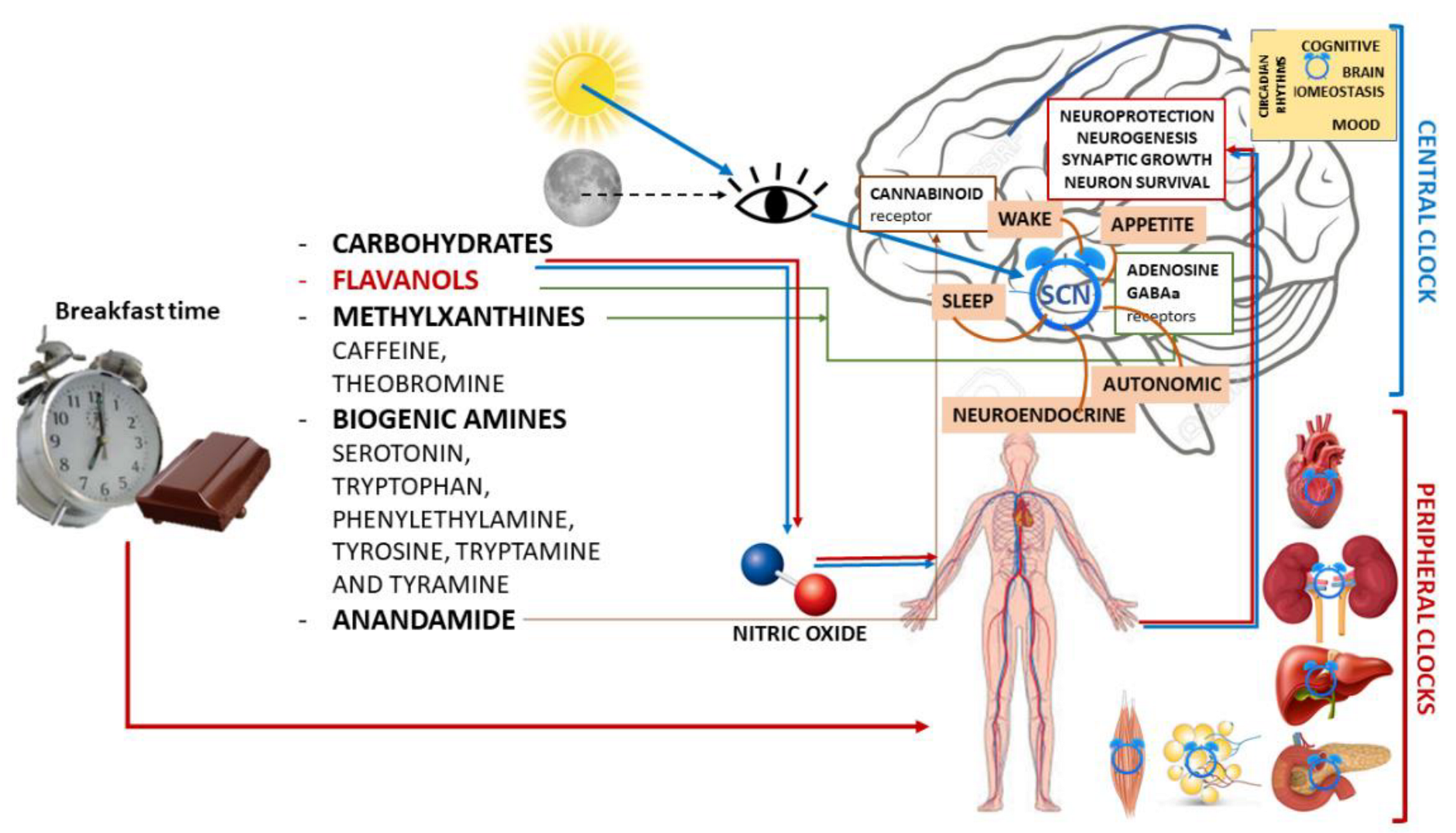
Figure 1. Schematic illustration of chrononutrition with chocolate for breakfast and components of chocolate with the main mechanisms of action in both the central nervous system and peripheral organs and their actions.
Furthermore, to maintain a coordinated and synchronized circadian function, food intake must be phased with the light–dark cycle. The main effect seems to be due to a direct synchronization action on brain oscillators and central and peripheral clocks [73][74].
Under normal light and dark (LD) conditions, programmed food intake does not shift the SCN phase [75][76]. Other studies indicate that SCN is inhibited during food anticipation and fasting as observed with c-Fos, a major early gene that is activated by external signals [77][78][79] or electrophysiological recordings, whereas the ventral SCN is activated both after re-feeding and with light [80].
Recent results indicate that the SCN may also respond to palatable food construed as hedonic information, via dopaminergic projections from the ventral tegmental area [81]. The rapid achievement of synchronization with limited daily chocolate intake may also be partly due to the increase in arousal induced by chocolate intake as a hedonic effect.
When planned for breakfast, an appetizing food, such as chocolate, can influence activation in the SCN, at the level of the dorsomedial region [82]. This rhythmic pattern in the dorsal SCN may promote faster re-entrainment [83] when bounded by a time window in which chocolate was administered during the active phase, whereas chocolate did not promote re-entrainment when administered during the resting phase.
Scheduled feeding has been shown to be a strong entrainment signal for circadian rhythm; especially when food intake is in phase with the period of activity. This exerts beneficial effects on the circadian system by favoring its synchronization and activation of the metabolism [84][85], as demonstrated in experimental studies on shift-worker models. Time-limited access to food accelerates resynchronization in a jet-lag model, prevents circadian desynchronization in a shift-work model, and induces positive effects in metabolism [86][87]. In contrast, food scheduled in the sleep–rest phase slows circadian synchronization and metabolism and alters behavior [86][87][88].
Recently, Oishi et al. [89] confirmed the positive action of cocoa on sleep disturbance induced by psychophysiological stress in mice using EEG. Cocoa intake attenuated the alteration of circadian sleep–wake rhythms. The EEG revealed that cocoa significantly improved both the increase in the level of alertness during the first half of the light period and the increase in NREM sleep during the first half of the dark period in mice with CSD. Under non-CSD conditions, cocoa does not appear to influence either the rhythms of run-rest activity or sleep–wake cycles. It is hypothesized that this positive action may be attributable to high concentrations of flavonoids in cocoa (epicatechin, catechin, and procyanidins), which improve blood flow and have antioxidant and neuroprotective properties. Indeed, in experimental animals, sleep deprivation and CSDs in general have been shown to increase oxidative stress levels in specific brain regions such as the hypothalamus, hippocampus, and thalamus [90].
Flavanols acting on endothelial function could also play a role in insomnia, as it is just endothelial dysfunction that appears to be responsible for some insomnia-related symptoms and the association between insomnia and cardiovascular disease [91]. Cocoa flavanols, by facilitating nitric oxide production, improve vascular endothelial function due to their vasodilatory effect [25]. CBF also appears to play an important role in sleep regulation [92] as well as cognitive and emotional processes, although it is not known how cerebral blood flow varies during alternating sleep–wake cycles [93]. Flavanol-rich cocoa significantly increases cerebral blood flow in humans [25][94] and attenuates the CSD-induced disturbance of circadian activity rhythm, sleep–wake cycles, cognitive functions by improving cerebral endothelial cell function, and blood flow, as demonstrated by Grassi et al. [23].
In addition, another pathway through which cocoa has a protective effect on the synchronous maintenance of the sleep–wake rhythm in subjects with CSD is the modulated neurotransmission of serotonin [95][96]. In fact, regular cocoa consumption has been shown to increase serotonin concentrations in the brain [97].
Acute administration of flavanol-rich chocolate can counteract the negative effects of total sleep deprivation both on working memory performance in healthy young people [23] and on endothelial and arterial function and, thus, on blood pressure (Figure 1). Natural cocoa seems to be an ideal nutrient for ameliorating stress-induced psychophysiological sleep disturbance without distorting behavioral or sleep regulation under normal conditions.
References
- Chemistry Behind the Sweet Lure of Chocolate. Available online: https://www.worldofchemicals.com/113/chemistry-articles/chemistry-behind-the-sweet-lure-of-chocolate.html (accessed on 22 December 2021).
- Scholey, A.; Owen, L. Effects of chocolate on cognitive function and mood: A systematic review. Nutr. Rev. 2013, 71, 665–681.
- Nehlig, A. The neuroprotective effects of cocoa flavanol and its influence on cognitive performance. Br. J. Clin. Pharmacol. 2013, 75, 716–727.
- Whiting, D.A. Natural phenolic compounds 1900–2000: A bird’s eye view of a century’s chemistry. Nat. Prod. Rep. 2001, 18, 583–606.
- Gu, L.; Kelm, M.A.; Hammerstone, J.F.; Beecher, G.; Holden, J.; Haytowitz, D.; Gebhardt, S.; Prior, R.L. Concentrations of Proanthocyanidins in Common Foods and Estimations of Normal Consumption. J. Nutr. 2004, 134, 613–617.
- Faria, A.; Pestana, D.; Teixeira, D.; Couraud, P.-O.; Romero, I.; Weksler, B.; de Freitas, V.; Mateus, N.; Calhau, C. Insights into the putative catechin and epicatechin transport across blood-brain barrier. Food Funct. 2010, 2, 39–44.
- Ghosh, D.; Scheepens, A. Vascular action of polyphenols. Mol. Nutr. Food Res. 2009, 53, 322–331.
- Datla, K.P.; Christidou, M.; Widmer, W.W.; Rooprai, H.K.; Dexter, D.T. Tissue distribution and neuroprotective effects of citrus flavonoid tangeretin in a rat model of Parkinson’s disease. NeuroReport 2001, 12, 3871–3875.
- Spencer, J.P.E. Food for thought: The role of dietary flavonoids in enhancing human memory, learning and neuro-cognitive performance. Proc. Nutr. Soc. 2008, 67, 238–252.
- Kelleher, R.J.; Govindarajan, A.; Jung, H.-Y.; Kang, H.; Tonegawa, S. Translational Control by MAPK Signaling in Long-Term Synaptic Plasticity and Memory. Cell 2004, 116, 467–479.
- Kim, D.H.; Jeon, S.J.; Son, K.H.; Jung, J.W.; Lee, S.; Yoon, B.H.; Choi, J.W.; Cheong, J.H.; Ko, K.H.; Ryu, J.H. Effect of the flavonoid, oroxylin A, on transient cerebral hypoperfusion-induced memory impairment in mice. Pharmacol. Biochem. Behav. 2006, 85, 658–668.
- Valente, T.; Hidalgo, J.; Bolea, I.; Ramirez, B.; Anglés, N.; Reguant, J.; Morelló, J.R.; Gutiérrez, C.; Boada, M.; Unzeta, M. A Diet Enriched in Polyphenols and Polyunsaturated Fatty Acids, LMN Diet, Induces Neurogenesis in the Subventricular Zone and Hippocampus of Adult Mouse Brain. J. Alzheimer’s Dis. 2009, 18, 849–865.
- González-Gallego, J.; Sánchez-Campos, S.; Tuñón, M.J. Anti-inflammatory properties of dietary flavonoids. Nutr. Hosp. 2007, 22, 287–293.
- Schroeter, H.; Heiss, C.; Balzer, J.; Kleinbongard, P.; Keen, C.L.; Hollenberg, N.K.; Sies, H.; Kwik-Uribe, C.; Schmitz, H.H.; Kelm, M. (-)-Epicatechin mediates beneficial effects of flavanol-rich cocoa on vascular function in humans. Proc. Natl. Acad. Sci. USA 2006, 103, 1024–1029.
- Flammer, A.J.; Hermann, F.; Sudano, I.; Spieker, L.; Hermann, M.; Cooper, K.A.; Serafini, M.; Lüscher, T.F.; Ruschitzka, F.; Noll, G.; et al. Dark Chocolate Improves Coronary Vasomotion and Reduces Platelet Reactivity. Circulation 2007, 116, 2376–2382.
- Grassi, D.; Necozione, S.; Lippi, C.; Croce, G.; Valeri, L.; Pasqualetti, P.; Desideri, G.; Blumberg, J.B.; Ferri, C. Cocoa Reduces Blood Pressure and Insulin Resistance and Improves Endothelium-Dependent Vasodilation in Hypertensives. Hypertension 2005, 46, 398–405.
- Grassi, D.; Desideri, G.; Necozione, S.; Lippi, C.; Casale, R.; Properzi, G.; Blumberg, J.B.; Ferri, C. Blood Pressure Is Reduced and Insulin Sensitivity Increased in Glucose-Intolerant, Hypertensive Subjects after 15 Days of Consuming High-Polyphenol Dark Chocolate. J. Nutr. 2008, 138, 1671–1676.
- Grassi, D.; Desideri, G.; Ferri, C. Protective effects of dark chocolate on endothelial function and diabetes. Curr. Opin. Clin. Nutr. Metab. Care 2013, 16, 662–668.
- Grassi, D.; Desideri, G.; Necozione, S.; di Giosia, P.; Barnabei, R.; Allegaert, L.; Bernaert, H.; Ferri, C. Cocoa consumption dose-dependently improves flow-mediated dilation and arterial stiffness decreasing blood pressure in healthy individuals. J. Hypertens. 2015, 33, 294–303.
- Shrime, M.G.; Bauer, S.R.; McDonald, A.C.; Chowdhury, N.H.; Coltart, C.E.M.; Ding, E.L. Flavonoid-Rich Cocoa Consumption Affects Multiple Cardiovascular Risk Factors in a Meta-Analysis of Short-Term Studies. J. Nutr. 2011, 141, 1982–1988.
- Hollenberg, N.K.; Fisher, N.D.; McCullough, M.L. Flavanols, the Kuna, cocoa consumption, and nitric oxide. J. Am. Soc. Hypertens. 2009, 3, 105–112.
- Kalt, W.; Hanneken, A.; Milbury, P.; Tremblay, F. Recent Research on Polyphenolics in Vision and Eye Health. J. Agric. Food Chem. 2010, 58, 4001–4007.
- Grassi, D.; Ferri, C.; Desideri, G. Brain Protection and Cognitive Function: Cocoa Flavonoids as Nutraceuticals. Curr. Pharm. Des. 2015, 22, 145–151.
- Gage, F.H. Mammalian Neural Stem Cells. Science 2000, 287, 1433–1438.
- Sokolov, A.N.; Pavlova, M.A.; Klosterhalfen, S.; Enck, P. Chocolate and the brain: Neurobiological impact of cocoa flavanols on cognition and behavior. Neurosci. Biobehav. Rev. 2013, 37 Pt 2, 2445–2453.
- Van Praag, H.; Lucero, M.J.; Yeo, E.; Stecker, K.; Heivand, N.; Zhao, C.; Yip, E.; Afanador, M.; Schroeter, H.; Hammerstone, J.; et al. Plant-Derived Flavanol (-)Epicatechin Enhances Angiogenesis and Retention of Spatial Memory in Mice. J. Neurosci. 2007, 27, 5869–5878, Erratum in J. Neurosci. 2007, 27.
- Spencer, J.P.E. The interactions of flavonoids within neuronal signalling pathways. Genes Nutr. 2007, 2, 257–273.
- Crichton, G.E.; Elias, M.F.; Alkerwi, A. Chocolate intake is associated with better cognitive function: The Maine-Syracuse Longitudinal Study. Appetite 2016, 100, 126–132.
- Commenges, D.; Scotet, V.; Renaud, S.; Jacqmin-Gadda, H.; Barberger-Gateau, P.; Dartigues, J.-F. Intake of flavonoids and risk of dementia. Eur. J. Epidemiol. 2000, 16, 357–363.
- Moreira, A.; Diógenes, M.J.; de Mendonça, A.; Lunet, N.; Barros, H. Chocolate Consumption is Associated with a Lower Risk of Cognitive Decline. J. Alzheimer’s Dis. 2016, 53, 85–93.
- Kuriyama, S.; Hozawa, A.; Ohmori, K.; Shimazu, T.; Matsui, T.; Ebihara, S.; Awata, S.; Nagatomi, R.; Arai, H.; Tsuji, I. Green tea consumption and cognitive function: A cross-sectional study from the Tsurugaya Project. Am. J. Clin. Nutr. 2006, 83, 355–361.
- Letenneur, L.; Proust-Lima, C.; Le Gouge, A.; Dartigues, J.F.; Barberger-Gateau, P. Flavonoid Intake and Cognitive Decline over a 10-Year Period. Am. J. Epidemiol. 2007, 165, 1364–1371.
- Nurk, E.; Refsum, H.; Drevon, C.A.; Tell, G.S.; Nygaard, H.A.; Engedal, K.; Smith, A.D. Intake of Flavonoid-Rich Wine, Tea, and Chocolate by Elderly Men and Women Is Associated with Better Cognitive Test Performance. J. Nutr. 2008, 139, 120–127.
- Smit, H.J.; Rogers, P.J. Effects of low doses of caffeine on cognitive performance, mood and thirst in low and higher caffeine consumers. Psychopharmacology 2000, 152, 167–173.
- Smit, H.J.; Gaffan, E.A.; Rogers, P.J. Methylxanthines are the psycho-pharmacologically active constituents of chocolate. Psychopharmacology 2004, 176, 412–419.
- Smit, H.J.; Blackburn, R.J. Reinforcing effects of caffeine and theobromine as found in chocolate. Psychopharmacology 2005, 181, 101–106.
- Nehlig, A. Is caffeine a cognitive enhancer? J. Alzheimer’s Dis. 2010, 20, 85–94.
- Parker, G.; Roy, K.; Mitchell, P.; Wilhelm, K.; Malhi, G.; Hadzi-Pavlovic, D. Atypical Depression: A Reappraisal. Am. J. Psychiatry 2002, 159, 1470–1479.
- Messaoudi, M.; Bisson, J.-F.; Nejdi, A.; Rozan, P.; Javelot, H. Antidepressant-like effects of a cocoa polyphenolic extract in Wistar–Unilever rats. Nutr. Neurosci. 2008, 11, 269–276.
- Benton, D.; Donohoe, R.T. The effects of nutrients on mood. Public Health Nutr. 1999, 2, 403–409.
- Reid, L.D. Endogenous opioid peptides and regulation of drinking and feeding. Am. J. Clin. Nutr. 1985, 42, 1099–1132.
- Giraudo, S.; Grace, M.; Welch, C.; Billington, C.; Levine, A. Naloxone’s anorectic effect is dependant upon the relative palatability of food. Pharmacol. Biochem. Behav. 1993, 46, 917–921.
- Young, S.N.; Smith, S.E.; Pihl, R.O.; Ervin, F.R. Tryptophan depletion causes a rapid lowering of mood in normal males. Psychopharmacology 1985, 87, 173–177.
- Si, E.; Bryant, H.; Yim, G. Opioid and non-opioid components of insulin-induced feeding. Pharmacol. Biochem. Behav. 1986, 24, 899–903.
- Ottley, C. Food and mood. Nurs. Stand. 2000, 15, 46–52, quiz 54–55.
- Parker, G.; Parker, I.; Brotchie, H. Mood state effects of chocolate. J. Affect. Disord. 2006, 92, 149–159.
- Fullerton, D.T.; Getto, C.J.; Swift, W.J.; Carlson, I.H. Sugar, opioids and binge eating. Brain Res. Bull. 1985, 14, 673–680.
- Willner, P.; Benton, D.; Brown, E.; Cheeta, S.; Roderique-Davies, G.; Morgan, J.; Morgan, M. “Depression” increases “craving” for sweet rewards in animal and human models of depression and craving. Psychopharmacology 1998, 136, 272–283.
- Hetherington, M.M.; MacDiarmid, J.I. “Chocolate Addiction”: A Preliminary Study of its Description and its Relationship to Problem Eating. Appetite 1993, 21, 233–246.
- Rozin, P.; Levine, E.; Stoess, C. Chocolate craving and liking. Appetite 1991, 17, 199–212.
- Smeets, P.A.M.; De Graaf, C.; Stafleu, A.; Van Osch, M.J.P.; Nievelstein, R.A.J.; Van Der Grond, J. Effect of satiety on brain activation during chocolate tasting in men and women. Am. J. Clin. Nutr. 2006, 83, 1297–1305.
- Michener, W.; Rozin, P. Pharmacological versus sensory factors in the satiation of chocolate craving. Physiol. Behav. 1994, 56, 419–422.
- Smits, M.; Peeters, R.R.; Van Hecke, P.; Sunaert, S. A 3 T event-related functional magnetic resonance imaging (fMRI) study of primary and secondary gustatory cortex localization using natural tastants. Neuroradiology 2006, 49, 61–71.
- Beaver, J.D.; Lawrence, A.D.; van Ditzhuijzen, J.; Davis, M.; Woods, A.; Calder, A.J. Individual Differences in Reward Drive Predict Neural Responses to Images of Food. J. Neurosci. 2006, 26, 5160–5166.
- Martin, G. Human electroencephalographic (EEG) response to olfactory stimulation: Two experiments using the aroma of food. Int. J. Psychophysiol. 1998, 30, 287–302.
- Rolls, E.T.; McCabe, C. Enhanced affective brain representations of chocolate in cravers vs. non-cravers. Eur. J. Neurosci. 2007, 26, 1067–1076.
- Nehlig, A. Are we dependent upon coffee and caffeine? A review on human and animal data. Neurosci. Biobehav. Rev. 1999, 23, 563–576.
- Di Chiara, G. Nucleus accumbens shell and core dopamine: Differential role in behavior and addiction. Behav. Brain Res. 2002, 137, 75–114.
- Schroeder, B.; Binzak, J.; Kelley, A. A common profile of prefrontal cortical activation following exposure to nicotine- or chocolate-associated contextual cues. Neuroscience 2001, 105, 535–545.
- Medina, J.H.; Viola, H.; Wolfman, C.; Marder, M.; Wasowski, C.; Calvo, D.; Paladini, A.C. Overview—Flavonoids: A New Family of Benzodiazepine Receptor Ligands. Neurochem. Res. 1997, 22, 419–425.
- Vignes, M.; Maurice, T.; Lanté, F.; Nedjar, M.; Thethi, K.; Guiramand, J.; Récasens, M. Anxiolytic properties of green tea polyphenol (−)-epigallocatechin gallate (EGCG). Brain Res. 2006, 1110, 102–115.
- Sathyapalan, T.; Beckett, S.; Rigby, A.S.; Mellor, D.D.; Atkin, S.L. High cocoa polyphenol rich chocolate may reduce the burden of the symptoms in chronic fatigue syndrome. Nutr. J. 2010, 9, 55.
- Pase, M.P.; Scholey, A.B.; Pipingas, A.; Kras, M.; Nolidin, K.; Gibbs, A.; Wesnes, K.; Stough, C. Cocoa polyphenols enhance positive mood states but not cognitive performance: A randomized, placebo-controlled trial. J. Psychopharmacol. 2013, 27, 451–458.
- Buijs, F.N.; Leon, L.; Guzmán-Ruiz, M.; Guerrero-Vargas, N.N.; Romo-Nava, F.; Buijs, R.M. The Circadian System: A Regulatory Feedback Network of Periphery and Brain. Physiology 2016, 31, 170–181.
- Bass, J.; Takahashi, J.S. Circadian Integration of Metabolism and Energetics. Science 2010, 330, 1349–1354.
- Knutson, K.L.; Spiegel, K.; Penev, P.; Van Cauter, E. The metabolic consequences of sleep deprivation. Sleep Med. Rev. 2007, 11, 163–178.
- Uehli, K.; Mehta, A.J.; Miedinger, D.; Hug, K.; Schindler, C.; Holsboer-Trachsler, E.; Leuppi, J.D.; Künzli, N. Sleep problems and work injuries: A systematic review and meta-analysis. Sleep Med. Rev. 2013, 18, 61–73.
- Kalmbach, D.A.; Anderson, J.R.; Drake, C.L. The impact of stress on sleep: Pathogenic sleep reactivity as a vulnerability to insomnia and circadian disorders. J. Sleep Res. 2018, 27, e12710.
- Salgado-Delgado, R.C.; Saderi, N.; Basualdo, M.D.C.; Guerrero-Vargas, N.N.; Escobar, C.; Buijs, R.M. Shift Work or Food Intake during the Rest Phase Promotes Metabolic Disruption and Desynchrony of Liver Genes in Male Rats. PLoS ONE 2013, 8, e60052.
- Schilperoort, M.; van den Berg, R.; Dollé, M.E.T.; Van Oostrom, C.T.M.; Wagner, K.; Tambyrajah, L.L.; Wackers, P.; DeBoer, T.; Hulsegge, G.; Proper, K.I.; et al. Time-restricted feeding improves adaptation to chronically alternating light-dark cycles. Sci. Rep. 2019, 9, 7874.
- Mukherji, A.; Kobiita, A.; Damara, M.; Misra, N.; Meziane, H.; Champy, M.-F.; Chambon, P. Shifting eating to the circadian rest phase misaligns the peripheral clocks with the master SCN clock and leads to a metabolic syndrome. Proc. Natl. Acad. Sci. USA 2015, 112, E6691–E6698.
- Yasumoto, Y.; Hashimoto, C.; Nakao, R.; Yamazaki, H.; Hiroyama, H.; Nemoto, T.; Yamamoto, S.; Sakurai, M.; Oike, H.; Wada, N.; et al. Short-term feeding at the wrong time is sufficient to desynchronize peripheral clocks and induce obesity with hyperphagia, physical inactivity and metabolic disorders in mice. Metabolism 2016, 65, 714–727.
- Wu, T.; Jin, Y.; Ni, Y.; Zhang, D.; Kato, H.; Fu, Z. Effects of light cues on re-entrainment of the food-dominated peripheral clocks in mammals. Gene 2008, 419, 27–34.
- Ekmekcioglu, C.; Touitou, Y. Chronobiological aspects of food intake and metabolism and their relevance on energy balance and weight regulation. Obes. Rev. 2010, 12, 14–25.
- Hara, R.; Wan, K.; Wakamatsu, H.; Aida, R.; Moriya, T.; Akiyama, M.; Shibata, S. Restricted feeding entrains liver clock without participation of the suprachiasmatic nucleus. Genes Cells 2001, 6, 269–278.
- Wakamatsu, H.; Yoshinobu, Y.; Aida, R.; Moriya, T.; Akiyama, M.; Shibata, S. Restricted-feeding-induced anticipatory activity rhythm is associated with a phase-shift of the expression of mPer1 and mPer2 mRNA in the cerebral cortex and hippocampus but not in the suprachiasmatic nucleus of mice. Eur. J. Neurosci. 2001, 13, 1190–1196.
- Herrera, D.; Robertson, H. Activation of c-fos in the brain. Prog. Neurobiol. 1996, 50, 83–107.
- Greco, C.; Sassone–Corsi, P. Circadian blueprint of metabolic pathways in the brain. Nat. Rev. Neurosci. 2018, 20, 71–82.
- Acosta-Galvan, G.; Yi, C.-X.; van der Vliet, J.; Jhamandas, J.H.; Panula, P.; Angeles-Castellanos, M.; Del Basualdo, M.C.; Escobar, C.; Buijs, R.M. Interaction between hypothalamic dorsomedial nucleus and the suprachiasmatic nucleus determines intensity of food anticipatory behavior. Proc. Natl. Acad. Sci. USA 2011, 108, 5813–5818.
- Saderi, N.; Cazarez-Márquez, F.; Buijs, F.; Salgado-Delgado, R.; Guzman-Ruiz, M.; Basualdo, M.D.C.; Escobar, C.; Buijs, R. The NPY intergeniculate leaflet projections to the suprachiasmatic nucleus transmit metabolic conditions. Neuroscience 2013, 246, 291–300.
- Grippo, R.M.; Purohit, A.M.; Zhang, Q.; Zweifel, L.S.; Güler, A.D. Direct Midbrain Dopamine Input to the Suprachiasmatic Nucleus Accelerates Circadian Entrainment. Curr. Biol. 2017, 27, 2465–2475.e3.
- Escobar, C.; Espitia-Bautista, E.; Guzmán-Ruiz, M.A.; Vargas, N.N.G.; Hernández-Navarrete, M.; Ángeles-Castellanos, M.; Morales-Pérez, B.; Buijs, R.M. Chocolate for breakfast prevents circadian desynchrony in experimental models of jet-lag and shift-work. Sci. Rep. 2020, 10, 6243.
- Ubaldo, L.; Buijs, R.; Escobar, C.; Angeles-Castellanos, M. Scheduled meal accelerates entrainment to a 6-h phase advance by shifting central and peripheral oscillations in rats. Eur. J. Neurosci. 2017, 46, 1875–1886.
- Ángeles-Castellanos, M.; Amaya, J.M.; Salgado-Delgado, R.; Buijs, R.M.; Escobar, C. Scheduled Food Hastens Re-Entrainment More Than Melatonin Does after a 6-h Phase Advance of the Light-Dark Cycle in Rats. J. Biol. Rhythm. 2011, 26, 324–334.
- Salgado-Delgado, R.; Angeles-Castellanos, M.; Saderi, N.; Buijs, R.M.; Escobar, C. Food Intake during the Normal Activity Phase Prevents Obesity and Circadian Desynchrony in a Rat Model of Night Work. Endocrinology 2010, 151, 1019–1029.
- Guerrero-Vargas, N.N.; Espitia-Bautista, E.; Buijs, R.M.; Escobar, C. Shift-work: Is time of eating determining metabolic health? Evidence from animal models. Proc. Nutr. Soc. 2018, 77, 199–215.
- Hutchison, A.T.; Regmi, P.; Manoogian, E.N.; Fleischer, J.G.; Wittert, G.A.; Panda, S.; Heilbronn, L.K. Time-restricted feeding improves glucose tolerance in men at risk for type 2 diabetes: A randomized crossover trial. Obesity 2019, 27, 724–732.
- Chaix, A.; Manoogian, E.N.; Melkani, G.C.; Panda, S. Time-Restricted Eating to Prevent and Manage Chronic Metabolic Diseases. Annu. Rev. Nutr. 2019, 39, 291–315.
- Oishi, K.; Okauchi, H.; Yamamoto, S.; Higo-Yamamoto, S. Dietary natural cocoa ameliorates disrupted circadian rhythms in locomotor activity and sleep-wake cycles in mice with chronic sleep disorders caused by psychophysiological stress. Nutrition 2020, 75–76, 110751.
- Villafuerte, G.; Miguel-Puga, A.; Rodríguez, E.M.; Machado, S.; Manjarrez, E.; Arias-Carrión, O. Sleep Deprivation and Oxidative Stress in Animal Models: A Systematic Review. Oxidative Med. Cell. Longev. 2015, 2015, 234952.
- Routledge, F.S.; Dunbar, S.B.; Higgins, M.; Rogers, A.E.; Feeley, C.; Ioachimescu, O.; Euwer, K.; Eapen, D.; Quyyumi, A. Insomnia Symptoms Are Associated With Abnormal Endothelial Function. J. Cardiovasc. Nurs. 2017, 32, 78–85.
- Braun, A.R.; Balkin, T.J.; Wesenten, N.J.; E Carson, R.; Varga, M.; Baldwin, P.; Selbie, S.; Belenky, G.; Herscovitch, P. Regional cerebral blood flow throughout the sleep-wake cycle. An H2(15)O PET study. Brain 1997, 120 Pt 7, 1173–1197.
- Elvsåshagen, T.; Mutsaerts, H.J.; Zak, N.; Norbom, L.; Quraishi, S.H.; Pedersen, P.Ø.; Malt, U.F.; Westlye, L.T.; van Someren, E.J.; Bjørnerud, A.; et al. Cerebral blood flow changes after a day of wake, sleep, and sleep deprivation. NeuroImage 2018, 186, 497–509.
- Socci, V.; Tempesta, D.; Desideri, G.; De Gennaro, L.; Ferrara, M. Enhancing Human Cognition with Cocoa Flavonoids. Front. Nutr. 2017, 4, 19.
- Pich, E.M.; Solfrini, V.; Biagini, G.; Fuxe, K.; Agnati, L.F. Effects of indole-pyruvic acid on sleep and food intake in the rat. Acta Physiol. Scand. 1990, 139, 583–589.
- Biagini, G.; Pich, E.M.; Carani, C.; Marrama, P.; Gustafsson, J.; Fuxe, K.; Agnati, L.F. Indole-pyruvic acid, a tryptophan ketoanalogue, antagonizes the endocrine but not the behavioral effects of repeated stress in a model of depression. Biol. Psychiatry 1993, 33, 712–719.
- Yamada, T.; Yamada, Y.; Okano, Y.; Terashima, T.; Yokogoshi, H. Anxiolytic effects of short- and long-term administration of cacao mass on rat elevated T-maze test. J. Nutr. Biochem. 2009, 20, 948–955.
More
Information
Subjects:
Neurosciences; Nutrition & Dietetics
Contributors
MDPI registered users' name will be linked to their SciProfiles pages. To register with us, please refer to https://encyclopedia.pub/register
:
View Times:
1.8K
Entry Collection:
Peptides for Health Benefits
Revisions:
2 times
(View History)
Update Date:
10 Aug 2022
Notice
You are not a member of the advisory board for this topic. If you want to update advisory board member profile, please contact office@encyclopedia.pub.
OK
Confirm
Only members of the Encyclopedia advisory board for this topic are allowed to note entries. Would you like to become an advisory board member of the Encyclopedia?
Yes
No
${ textCharacter }/${ maxCharacter }
Submit
Cancel
Back
Comments
${ item }
|
More
No more~
There is no comment~
${ textCharacter }/${ maxCharacter }
Submit
Cancel
${ selectedItem.replyTextCharacter }/${ selectedItem.replyMaxCharacter }
Submit
Cancel
Confirm
Are you sure to Delete?
Yes
No





