
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Memoona Akhtar | -- | 7564 | 2022-07-05 11:15:27 | | | |
| 2 | Sirius Huang | -212 word(s) | 7352 | 2022-07-06 02:54:59 | | |
Video Upload Options
Studies have shown that metallic ions like Ag+, Sr2+, Mg2+, Mn2+, Cu2+, Ca2+, P+5, etc., have shown promising results in drug delivery systems and regenerative medicine. These metallic ions can be loaded in nanoparticles, mesoporous bioactive glass nanoparticles (MBGNs), hydroxyapatite (HA), calcium phosphates, polymeric coatings, and salt solutions. The metallic ions can exhibit different functions in the physiological environment such as antibacterial, antiviral, anticancer, bioactive, biocompatible, and angiogenic effects. Furthermore, the metals/metalloid ions can be loaded into scaffolds to improve osteoblast proliferation, differentiation, bone development, fibroblast growth, and improved wound healing efficacy. Moreover, different ions possess different therapeutic limits.
1. Introduction
2. Therapeutic Metallic Ions
2.1. Gallium
2.1.1. Gallium’s Physical, Chemical, and Biological Properties
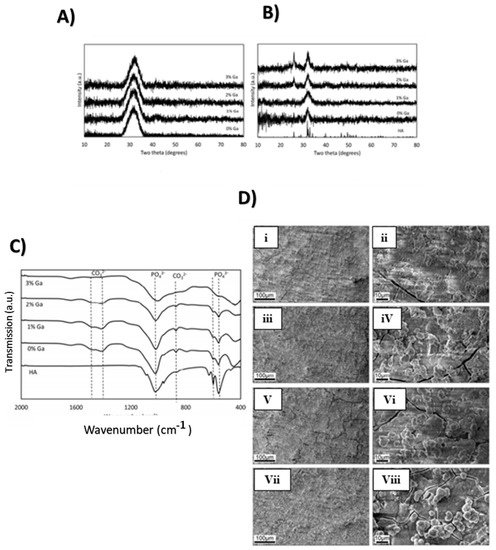
2.1.2. Properties and Applications
Anticancer Effects of Gallium
Antimicrobial Activity in Gallium Compounds
Gallium Compounds as Anti-Inflammatory and Immunosuppressive Agents
Effects of Gallium Associated with Hypercalcemia and Bone Metabolism
Antimalarial Agent
2.2. Bismuth
2.2.1. Properties and Applications
Antibacterial Action
Anti-Leishmaniasis Property
Inhibition of Enzyme Activity
Peptic Ulcer Treatment
2.3. Magnesium
2.3.1. Properties and Applications
Promotion of Osteoblast Cell Proliferation and Differentiation
Mg Ions as a Coagulant
2.4. Calcium
2.4.1. Properties and Applications
Cellular Proliferation and Differentiation
2.5. Germanium
2.5.1. Properties and Applications
Antitumor Activity/Malignant Pathology
Raynaud’s Disease
Antioxidant Effects
As a Protective Agent
As Biocompatible Coatings
2.6. Chromium
2.6.1. Properties and Applications
Diabetes Mellitus and Insulin Signaling
Anticarcinogenic Effect
2.7. Lithium
2.7.1. Properties and Applications
Manic Depression Treatment/Bipolar Disorder
Anti-Inflammatory Agent
Wound Healing/Anticoagulating Agent
Schizophrenic Disorders
Major Depressive Disorder
2.8. Potassium
2.8.1. Properties and Applications
Cellular Electrolyte Metabolism
Cell Signaling
Diuretic Agent
Nerve Functioning
Brushes Coated on Artificial Implants
2.9. Strontium
2.9.1. Properties and Applications
Treatment of Cancer
Osteoporosis Treatment
Osteogenic Response
References
- Bernstein, L.R. Mechanisms of therapeutic activity for gallium. Pharmacol. Rev. 1998, 50, 665–682.
- Mukherjee, A.; Sadler, P.J. Metals in Medicine: Therapeutic Agents. In Wiley Encyclopedia of Chemical Biology; John Wiley & Sons, Inc.: Hoboken, NJ, USA, 2009; pp. 1–47.
- Ayangbenro, A.S.; Babalola, O.O. A New Strategy for Heavy Metal Polluted Environments: A Review of Microbial Biosorbents. Int. J. Environ. Res. Public Health 2017, 14, 94.
- Brown, T. Design thinking. Harv. Bus. Rev. 2008, 86, 84–92.
- Bundschuh, M.; Filser, J.; Lüderwald, S.; McKee, M.S.; Metreveli, G.; Schaumann, G.E.; Schulz, R.; Wagner, S. Nanoparticles in the Environment: Where Do We Come From, Where Do We Go To? Environ. Sci. Eur. 2018, 30, 6.
- Eliaz, N. Corrosion of Metallic Biomaterials: A Review. Materials 2019, 12, 407.
- Zhao, J.; Zhai, Z.; Sun, D.; Yang, C.; Zhang, X.; Huang, N.; Jiang, X.; Yang, K. Antibacterial durability and biocompatibility of antibacterial-passivated 316L stainless steel in simulated physiological environment. Mater. Sci. Eng. C 2019, 100, 396–410.
- Ge, R.; Lin, M.; Li, X.; Liu, S.; Wang, W.; Li, S.; Zhang, X.; Liu, Y.; Liu, L.; Shi, F.; et al. Cu2+-Loaded Polydopamine Nanoparticles for Magnetic Resonance Imaging-Guided pH- and Near-Infrared-Light-Stimulated Thermochemotherapy. ACS Appl. Mater. Interfaces 2017, 9, 19706–19716.
- Kim, A.C.; Lim, S.; Kim, Y.K. Metal Ion Effects on Aβ and Tau Aggregation. Int. J. Mol. Sci. 2018, 19, 128.
- Liu, Y.; Nguyen, M.; Robert, A.; Meunier, B. Metal Ions in Alzheimer’s Disease: A Key Role or Not? Accounts Chem. Res. 2019, 52, 2026–2035.
- Liu, Y.; Zheng, Y.; Chen, X.-H.; Yang, J.-A.; Pan, H.; Chen, D.; Wang, L.; Zhang, J.; Zhu, D.; Wu, S.; et al. Fundamental Theory of Biodegradable Metals—Definition, Criteria, and Design. Adv. Funct. Mater. 2019, 29, 1805402.
- Mohammadinejad, R.; Shavandi, A.; Raie, D.S.; Sangeetha, J.; Soleimani, M.; Hajibehzad, S.S.; Thangadurai, D.; Hospet, R.; Popoola, J.O.; Arzani, A.; et al. Plant molecular farming: Production of metallic nanoparticles and therapeutic proteins using green factories. Green Chem. 2019, 21, 1845–1865.
- Törne, K.; Örnberg, A.; Weissenrieder, J. Influence of strain on the corrosion of magnesium alloys and zinc in physiological environments. Acta Biomater. 2017, 48, 541–550.
- Mohammed, L.; Gomaa, H.G.; Ragab, D.; Zhu, J. Magnetic nanoparticles for environmental and biomedical applications: A review. Particuology 2017, 30, 1–14.
- Zhang, D.; Zheng, A.; Li, J.; Wu, M.; Wu, L.; Wei, Z.; Liao, N.; Zhang, X.; Cai, Z.; Yang, H.; et al. Smart Cu(II)-aptamer complexes based gold nanoplatform for tumor micro-environment triggered programmable intracellular prodrug release, photodynamic treatment and aggregation induced photothermal therapy of hepatocellular carcinoma. Theranostics 2017, 7, 164–179.
- Mouriño, V.; Cattalini, J.P.; Boccaccini, A.R. Metallic ions as therapeutic agents in tissue engineering scaffolds: An overview of their biological applications and strategies for new developments. J. R. Soc. Interface 2012, 9, 401–419.
- Ren, Z.; Sun, S.; Sun, R.; Cui, G.; Hong, L.; Rao, B.; Li, A.; Yu, Z.; Kan, Q.; Mao, Z. A Metal–Polyphenol-Coordinated Nanomedicine for Synergistic Cascade Cancer Chemotherapy and Chemodynamic Therapy. Adv. Mater. 2020, 32, e1906024.
- Rogowska, J.; Olkowska, E.; Ratajczyk, W.; Wolska, L. Gadolinium as a new emerging contaminant of aquatic environments. Environ. Toxicol. Chem. 2018, 37, 1523–1534.
- Talha, M.; Ma, Y.; Kumar, P.; Lin, Y.; Singh, A. Role of protein adsorption in the bio corrosion of metallic implants—A review. Colloids Surf. B Biointerfaces 2019, 176, 494–506.
- Su, Y.; Yang, H.; Gao, J.; Qin, Y.; Zheng, Y.; Zhu, D. Interfacial Zinc Phosphate is the Key to Controlling Biocompatibility of Metallic Zinc Implants. Adv. Sci. 2019, 6, 1900112.
- Boros, E.; Dyson, P.J.; Gasser, G. Classification of Metal-Based Drugs according to Their Mechanisms of Action. Chem 2020, 6, 41–60.
- Boskey, A.; Ziecheck, W.; Guidon, P.; Doty, S. Gallium nitrate inhibits alkaline phosphatase activity in a differentiating mesenchymal cell culture. Bone Miner. 1993, 20, 179–192.
- Alzoman, H.; Diab, H.M. Effect of gallium aluminium arsenide diode laser therapy onPorphyromonas gingivalisin chronic periodontitis: A randomized controlled trial. Int. J. Dent. Hyg. 2016, 14, 261–266.
- Ehsan, Z. A Poly (Octanediol Citrate)/Gallium-Containing Bioglass Composite for Bone Tissue Regeneration/Ehsan Zeimaran. Ph.D. Thesis, University of Malaya, Kuala Lumpur, Malaysia, 2016.
- Rehman, J.C.I.; Knowles, W. Bonfield, Analysis ofin vitro reaction layers formed on Bioglass using thin-film X-ray diffraction and ATR-FTIR microspectroscopy. J. Biomed. Mater. Res. 1998, 41, 162–166.
- Bastos, R.W.; Rossato, L.; Valero, C.; Lagrou, K.; Colombo, A.L.; Goldman, G.H. Potential of Gallium as an Antifungal Agent. Front. Cell. Infect. Microbiol. 2019, 9, 414.
- Adamson, R.H.; Canellos, G.P.; Sieber, S.M. Sieber, Studies on the antitumor activity of gallium nitrate (NSC 15200) and other group IIIa metal salts. Cancer Chemother. Rep. 1975, 59, 599–610.
- Kaneko, Y.; Thoendel, M.; Olakanmi, O.; Britigan, B.E.; Singh, P.K. The transition metal gallium disrupts Pseudomonas aeruginosa iron metabolism and has antimicrobial and antibiofilm activity. J. Clin. Investig. 2007, 117, 877–888.
- Kratz, F.; Nuber, B.; Weis, J.; Keppler, B.K. Synthesis and Characterization of Potential Antitumor and Antiviral Gallium(III) Complexes of α-(N)-Heterocyclic Thiosemicarbazones. Synth. React. Inorg. Met.-Org. Chem. 1991, 21, 1601–1615.
- bin Othman, M.F.; Mitry, N.R.; Lewington, V.; Blower, P.; Terry, S.Y. Re-assessing gallium-67 as a therapeutic radionuclide. Nucl. Med. Biol. 2017, 46, 12–18.
- Thorson, J.A.; Smith, K.M.; Gomez, F.; Naumann, P.W.; Kemp, J.D. Role of iron in T cell activation: Th1 clones differ from TH2 clones in their sensitivity to inhibition of DNA synthesis caused by IGG mabs against the transferrin receptor and the iron chelator deferoxamine. Cell. Immunol. 1991, 134, 126–137.
- Chitambar, C.R. Medical Applications and Toxicities of Gallium Compounds. Int. J. Environ. Res. Public Health 2010, 7, 2337–2361.
- Goss, C.H.; Kaneko, Y.; Khuu, L.; Anderson, G.D.; Ravishankar, S.; Aitken, M.L.; Lechtzin, N.; Zhou, G.; Czyz, D.M.; McLean, K.; et al. Gallium disrupts bacterial iron metabolism and has therapeutic effects in mice and humans with lung infections. Sci. Transl. Med. 2018, 10, eaat7520.
- Gonçalves, R.; Mezencio, J.; Benevides, G.; Matta, S.; Neves, C.; Sarandy, M.; Vilela, E. Effect of gallium-arsenide laser, gallium-aluminum-arsenide laser and healing ointment on cutaneous wound healing in Wistar rats. Braz. J. Med. Biol. Res. 2010, 43, 350–355.
- Xu, Z.; Zhao, X.; Chen, X.; Chen, Z.; Xia, Z. Antimicrobial effect of gallium nitrate against bacteria encountered in burn wound infections. RSC Adv. 2017, 7, 52266–52273.
- Chitambar, C.R. The therapeutic potential of iron-targeting gallium compounds in human disease: From basic research to clinical application. Pharmacol. Res. 2017, 115, 56–64.
- Wang, Y.; Han, B.; Xie, Y.; Wang, H.; Wang, R.; Xia, W.; Li, H.; Sun, H. Combination of gallium(iii) with acetate for combating antibiotic resistant Pseudomonas aeruginosa. Chem. Sci. 2019, 10, 6099–6106.
- Dundar, U.; Evcik, D.; Samli, F.; Pusak, H.; Kavuncu, V. The effect of gallium arsenide aluminum laser therapy in the management of cervical myofascial pain syndrome: A double blind, placebo-controlled study. Clin. Rheumatol. 2006, 26, 930–934.
- Yang, Y.; Ouyang, R.; Xu, L.; Guo, N.; Li, W.; Feng, K.; Ouyang, L.; Yang, Z.; Zhou, S.; Miao, Y. Review: Bismuth complexes: Synthesis and applications in biomedicine. J. Coord. Chem. 2015, 68, 379–397.
- Rana, K.S.; Souza, L.; Isaacs, M.A.; Raja, F.N.S.; Morrell, A.P.; Martin, R.A. Development and Characterization of Gallium-Doped Bioactive Glasses for Potential Bone Cancer Applications. ACS Biomater. Sci. Eng. 2017, 3, 3425–3432.
- Barrioni, B.R.; Oliveira, A.C.; Leite, M.D.F.; Pereira, M. Sol–gel-derived manganese-releasing bioactive glass as a therapeutic approach for bone tissue engineering. J. Mater. Sci. 2017, 52, 8904–8927.
- Lambert, J.R. Pharmacology of Bismuth-Containing Compounds. Clin. Infect. Dis. 1991, 13, S691–S695.
- Cheng, Y.; Zhang, H. Novel Bismuth-Based Nanomaterials Used for Cancer Diagnosis and Therapy. Chem.—A Eur. J. 2018, 24, 17405–17418.
- Cacciotti, I. Bivalent cationic ions doped bioactive glasses: The influence of magnesium, zinc, strontium and copper on the physical and biological properties. J. Mater. Sci. 2017, 52, 8812–8831.
- Cheng, K.; Sano, M.; Jenkins, C.H.; Zhang, G.; Vernekohl, D.; Zhao, W.; Wei, C.; Zhang, Y.; Zhang, Z.; Liu, Y.; et al. Synergistically Enhancing the Therapeutic Effect of Radiation Therapy with Radiation Activatable and Reactive Oxygen Species-Releasing Nanostructures. ACS Nano 2018, 12, 4946–4958.
- Sisin, N.N.T.; Akasaka, H.; Sasaki, R.; Tominaga, T.; Miura, H.; Nishi, M.; Geso, M.; Mat, N.F.C.; Razak, K.A.; Rahman, W.N. Effects of Bismuth Oxide Nanoparticles, Cisplatin and Baicalein-rich Fraction on ROS Generation in Proton Beam irradiated Human Colon Carcinoma Cells. Pol. J. Med Phys. Eng. 2022, 28, 30–36.
- Klauder, J.V. Bismuth in the treatment of syphilis. Arch. Dermatol. Syphilol. 1923, 7, 721–744.
- Ayman, D. Bismuth subnitrate in the treatment of arteriolar (essential) hypertension. J. Am. Med. Assoc. 1932, 98, 545.
- Shavakhi, A.; Tabesh, E.; Yaghoutkar, A.; Hashemi, H.; Tabesh, F.; Khodadoostan, M.; Minakari, M.; Shavakhi, S.; Gholamrezaei, A. The Effects of Multistrain Probiotic Compound on Bismuth-Containing Quadruple Therapy for Helicobacter pylori Infection: A Randomized Placebo-Controlled Triple-Blind Study. Helicobacter 2013, 18, 280–284.
- Alkim, H.; Koksal, A.R.; Boga, S.; Sen, I.; Alkım, C. Role of Bismuth in the Eradication of Helicobacter pylori. Am. J. Ther. 2017, 24, e751–e757.
- Konturek, S.J.; Radecki, T.; Piastucki, I.; Drozdowicz, D. Advances in the Understanding of the Mechanism of Cytoprotective Action by Colloidal Bismuth Subcitrate. Scand. J. Gastroenterol. 1986, 21, 6–10.
- Whitehead, M.W.; Phillips, R.H.; Sieniawska, C.E.; Delves, H.T.; Seed, P.T.; Thompson, R.P.; Powell, J.J. Double-blind comparison of absorbable colloidal bismuth subcitrate and nonabsorbable bismuth subnitrate in the eradication of Helicobacter pylori and the relief of nonulcer dyspepsia. Helicobacter 2001, 5, 169–175.
- Hadrup, N.; Sharma, A.K.; Loeschner, K. Toxicity of silver ions, metallic silver, and silver nanoparticle materials after in vivo dermal and mucosal surface exposure: A review. Regul. Toxicol. Pharmacol. 2018, 98, 257–267.
- Wang, R.; Li, H.; Sun, H. Bismuth: Environmental Pollution and Health Effects. In Encyclopedia of Environmental Health, 2nd ed.; Elsevier: Amsterdam, The Netherlands, 2019; pp. 415–423.
- Li, H.; Wang, R.; Sun, H. Systems Approaches for Unveiling the Mechanism of Action of Bismuth Drugs: New Medicinal Applications beyond Helicobacter Pylori Infection. Accounts Chem. Res. 2018, 52, 216–227.
- Lambert, J.R.; Midolo, P. The actions of bismuth in the treatment of Helicobacter pylori infection. Aliment. Pharmacol. Ther. 1997, 11, 27–33.
- Armstrong, J.A.; Wee, S.H.; Goodwin, C.S.; Wilson, D.H. Response of Campylobacter pyloridis to antibiotics, bismuth and an acid-reducing agent in vitro--an ultrastructural study. J. Med. Microbiol. 1987, 24, 343–350.
- Dore, M.P.; Lu, H.; Graham, D.Y. Role of bismuth in improving Helicobacter pylori eradication with triple therapy. Gut 2016, 65, 870–878.
- Gorbach, S.L. Bismuth therapy in gastrointestinal diseases. Gastroenterology 1990, 99, 863–875.
- Rude, R.K.; Gruber, H.E. Magnesium deficiency and osteoporosis: Animal and human observations. J. Nutr. Biochem. 2004, 15, 710–716.
- De Baaij, J.H.F.; Hoenderop, J.G.J.; Bindels, R.J. Magnesium in Man: Implications for Health and Disease. Physiol. Rev. 2015, 95, 1–46.
- Leidi, M.; Dellera, F.; Mariotti, M.; Maier, J.A.M. High magnesium inhibits human osteoblast differentiation in vitro. Magnes. Res. 2011, 24, 1–6.
- Luo, J.; Stewart, R.; Berdeaux, R.; Hu, H. Tonic Inhibition of TRPV3 by Mg2+ in Mouse Epidermal Keratinocytes. J. Investig. Dermatol. 2012, 132, 2158–2165.
- Li, D.; Ma, W.; Wei, J.; Mao, Y.; Peng, Z.; Zhang, J.; Kong, X.; Han, Q.; Fan, W.; Yang, Y.; et al. Magnesium promotes root growth and increases aluminum tolerance via modulation of nitric oxide production in Arabidopsis. Plant Soil 2019, 457, 83–95.
- Rude, R.K.; Gruber, H.E.; Norton, H.J.; Wei, L.Y.; Frausto, A.; Mills, B.G. Bone Loss Induced by Dietary Magnesium Reduction to 10% of the Nutrient Requirement in Rats Is Associated with Increased Release of Substance P and Tumor Necrosis Factor-α. J. Nutr. 2004, 134, 79–85.
- Chen, X.; Liao, X.; Huang, Z.; You, P.; Chen, C.; Kang, Y.; Yin, G. Synthesis and characterization of novel multiphase bioactive glass-ceramics in the CaO-MgO-SiO2system. J. Biomed. Mater. Res. Part B Appl. Biomater. 2010, 9999B, 194–202.
- Saboori, A.; Rabiee, M.; Moztarzadeh, F.; Sheikhi, M.H.; Tahriri, M.; Karimi, M. Synthesis, characterization and in vitro bioactivity of sol-gel-derived SiO2–CaO–P2O5–MgO bioglass. Mater. Sci. Eng. C 2009, 29, 335–340.
- Sekiya, F.; Yoshida, M.; Yamashita, T.; Morita, T. Magnesium(II) Is a Crucial Constituent of the Blood Coagulation Cascade. J. Biol. Chem. 1996, 271, 8541–8544.
- Wong, H.M.; Zhao, Y.; Leung, F.K.L.; Xi, T.; Zhang, Z.; Zheng, Y.; Wu, S.; Luk, K.D.K.; Cheung, K.; Chu, P.K.; et al. Functionalized Polymeric Membrane with Enhanced Mechanical and Biological Properties to Control the Degradation of Magnesium Alloy. Adv. Healthc. Mater. 2017, 6, 1601269.
- Sgambato, A.; Wolf, F.I.; Faraglia, B.; Cittadini, A. Magnesium depletion causes growth inhibition, reduced expression of cyclin D1, and increased expression of p27(KIP1) in normal but not in transformed mammary epithelial cells. J. Cell. Physiol. 1999, 180, 245–254.
- Tommasino, F.; Scifoni, E.; Durante, M. New Ions for Therapy. Int. J. Part. Ther. 2015, 2, 428–438.
- Lankford, K.L.; Letourneau, P.C. Evidence that calcium may control neurite outgrowth by regulating the stability of actin filaments. J. Cell Biol. 1989, 109, 1229–1243.
- Maeno, S.; Niki, Y.; Matsumoto, H.; Morioka, H.; Yatabe, T.; Funayama, A.; Toyama, Y.; Taguchi, T.; Tanaka, J. The effect of calcium ion concentration on osteoblast viability, proliferation and differentiation in monolayer and 3D culture. Biomaterials 2005, 26, 4847–4855.
- Zhou, H.; Wei, J.; Wu, X.; Shi, J.; Liu, C.; Jia, J.; Dai, C.; Gan, Q. The bio-functional role of calcium in mesoporous silica xerogels on the responses of osteoblasts in vitro. J. Mater. Sci. Mater. Med. 2010, 21, 2175–2185.
- Gupta, S.P. Roles of metals in human health. MOJ Bioorganic Org. Chem. 2018, 2, 1.
- Moriyama, M.; Moriyama, H.; Uda, J.; Kubo, H.; Nakajima, Y.; Goto, A.; Akaki, J.; Yoshida, I.; Matsuoka, N.; Hayakawa, T. Beneficial Effects of the Genus Aloe on Wound Healing, Cell Proliferation, and Differentiation of Epidermal Keratinocytes. PLoS ONE 2016, 11, e0164799.
- Qiao, X.Y.; Nie, Y.; Ma, Y.X.; Chen, Y.; Cheng, R.; Yinrg, W.Y.; Hu, Y.; Xu, W.M.; Xu, L.Z. Irisin promotes osteoblast proliferation and differentiation via activating the MAP kinase signaling pathways. Sci. Rep. 2016, 6, 18732.
- Haimi, S.; Gorianc, G.; Moimas, L.; Lindroos, B.; Huhtala, H.; Räty, S.; Kuokkanen, H.; Sándor, G.K.; Schmid, C.; Miettinen, S. Characterization of zinc-releasing three-dimensional bioactive glass scaffolds and their effect on human adipose stem cell proliferation and osteogenic differentiation. Acta Biomater. 2009, 5, 3122–3131.
- Dimeloe, S.; Burgener, A.-V.; Grählert, J.; Hess, C. T-cell metabolism governing activation, proliferation and differentiation; a modular view. Immunology 2017, 150, 35–44.
- Bunting, S.; Di Silvio, L.; Deb, S.; Hall, S. Bioresorbable glass fibres facilitate peripheral nerve regeneration. J. Hand Surg. 2005, 30, 242–247.
- Dhivya, S.; Narayan, A.K.; Kumar, R.L.; Chandran, S.V.; Vairamani, M.; Selvamurugan, N. Proliferation and differentiation of mesenchymal stem cells on scaffolds containing chitosan, calcium polyphosphate and pigeonite for bone tissue engineering. Cell Prolif. 2018, 51, 1–13.
- Fan, J.; Wei, Q.; Liao, J.; Zou, Y.; Song, D.; Xiong, D.; Ma, C.; Hu, X.; Qu, X.; Chen, L.; et al. Noncanonical Wnt signaling plays an important role in modulating canonical Wnt-regulated stemness, proliferation and terminal differentiation of hepatic progenitors. Oncotarget 2017, 8, 27105–27119.
- Huang, Q.; Elkhooly, T.A.; Liu, X.; Zhang, R.; Yang, X.; Shen, Z.; Feng, Q. Effects of hierarchical micro/nano-topographies on the morphology, proliferation and differentiation of osteoblast-like cells. Colloids Surf. B Biointerfaces 2016, 145, 37–45.
- Li, L.; Peng, X.; Qin, Y.; Wang, R.; Tang, J.; Cui, X.; Wang, T.; Liu, W.; Pan, H.; Li, B. Acceleration of bone regeneration by activating Wnt/β-catenin signalling pathway via lithium released from lithium chloride/calcium phosphate cement in osteoporosis. Sci. Rep. 2017, 7, 45204.
- Nandi, S.K.; Mahato, A.; Kundu, B.; Mukherjee, P. Doped Bioactive Glass Materials in Bone Regeneration. In Advanced Techniques in Bone Regeneration; Zorzi, A.R., de Miranda, J.B., Eds.; IntechOpen: London, UK, 2016; pp. 275–328.
- Goodman, S. Therapeutic effects of organic Germanium. Med. Hypotheses 1988, 26, 207–215.
- Abrantes, A.M.; Pires, A.S.; Monteiro, L.; Teixo, R.; Neves, A.R.; Tavares, N.T.; Marques, I.A.; Botelho, M.F. Tumour functional imaging by PET. Biochim. Biophys. Acta (BBA) Mol. Basis Dis. 2020, 1866, 165717.
- Fukazawa, H.; Ohashi, Y.; Sekiyama, S.; Hoshi, H.; Abe, M.; Takahashi, M.; Sato, T. Multidisciplinary treatment of head and neck cancer using BCG, OK-432, and GE-132 as biologic response modifiers. Head Neck 1994, 16, 30–38.
- Jumina, J.; Harizal, H. Dermatologic Toxicities and Biological Activities of Chromium. In Trace Metals in the Environment-New Approaches and Recent Advances; IntechOpen: London, UK, 2021.
- Qi, S.S.; Zheng, H.X.; Jiang, H.; Yuan, L.P.; Dong, L.C. Protective Effects of Chromium Picolinate Against Diabetic-Induced Renal Dysfunction and Renal Fibrosis in Streptozotocin-Induced Diabetic Rats. Biomolecules 2020, 10, 398.
- Bozkurt, M.F.; Virgolini, I.; Balogova, S.; Beheshti, M.; Rubello, D.; Decristoforo, C.; Ambrosini, V.; Kjaer, A.; Delgado-Bolton, R.; Kunikowska, J.; et al. Guideline for PET/CT imaging of neuroendocrine neoplasms with 68Ga-DOTA-conjugated somatostatin receptor targeting peptides and 18F-DOPA. Eur. J. Nucl. Med. Mol. Imaging 2017, 44, 1588–1601, Erratum in 2017, 44, 2150–2151.
- Wang, C.; Chen, Z.; Pan, Y.; Gao, X.; Chen, H. Anti-diabetic effects of Inonotus obliquus polysaccharides-chromium (III) complex in type 2 diabetic mice and its sub-acute toxicity evaluation in normal mice. Food Chem. Toxicol. 2017, 108, 498–509.
- Ali, M.M.; Noaman, E.; Kamal, S.; Soliman, S.; Ismail, D.A. Role of germanium L-cysteine α-tocopherol complex as stimulator of some antioxidant defense systems in gamma-irradiated rats. Acta Pharm. 2007, 57, 1–12.
- Ngala, R.A.; Awe, M.A.; Nsiah, P. The effects of plasma chromium on lipid profile, glucose metabolism and cardiovascular risk in type 2 diabetes mellitus. A case—Control study. PLoS ONE 2018, 13, e0197977.
- Sala, A.; Anderson, D.J.; Brennan, P.M.; Butler, H.J.; Cameron, J.M.; Jenkinson, M.D.; Rinaldi, C.; Theakstone, A.G.; Baker, M.J. Biofluid diagnostics by FTIR spectroscopy: A platform technology for cancer detection. Cancer Lett. 2020, 477, 122–130.
- Prasad, A. Role of chromium compounds in diabetes. Indian J. Pharm. Pharmacol. 2016, 3, 17.
- Léonard, A.; Hantson, P.; Gerber, G. Mutagenicity, carcinogenicity and teratogenicity of lithium compounds. Mutat. Res. Genet. Toxicol. 1995, 339, 131–137.
- Anderson, R.A. Chromium and insulin resistance. Nutr. Res. Rev. 2003, 16, 267–275.
- Jain, S.K.; Kannan, K. Chromium Chloride Inhibits Oxidative Stress and TNF-α Secretion Caused by Exposure to High Glucose in Cultured U937 Monocytes. Biochem. Biophys. Res. Commun. 2001, 289, 687–691.
- Chen, G.; Liu, P.; Pattar, G.R.; Tackett, L.; Bhonagiri, P.; Strawbridge, A.B.; Elmendorf, J.S. Chromium Activates Glucose Transporter 4 Trafficking and Enhances Insulin-Stimulated Glucose Transport in 3T3-L1 Adipocytes via a Cholesterol-Dependent Mechanism. Mol. Endocrinol. 2006, 20, 857–870.
- Thompson, C.M.; Wolf, J.C.; McCoy, A.; Suh, M.; Proctor, D.M.; Kirman, C.R.; Haws, L.C.; Harris, M.A. Comparison of Toxicity and Recovery in the Duodenum of B6C3F1 Mice Following Treatment with Intestinal Carcinogens Captan, Folpet, and Hexavalent Chromium. Toxicol. Pathol. 2017, 45, 1091–1101.
- Deger, Y.; DeDe, S.; Belge, A.; Mert, N.; Kahraman, T.; Alkan, M. Effects of X-Ray Radiation on Lipid Peroxidation and Antioxidant Systems in Rabbits Treated with Antioxidant Compounds. Biol. Trace Elem. Res. 2003, 94, 149–156.
- Yao, H.; Guo, L.; Jiang, B.-H.; Luo, J.; Shi, X. Oxidative Stress and Chromium(VI) Carcinogenesis. J. Environ. Pathol. Toxicol. Oncol. 2008, 27, 77–88.
- Moradi, F.; Maleki, V.; Saleh-Ghadimi, S.; Kooshki, F.; Gargari, B.P. Potential roles of chromium on inflammatory biomarkers in diabetes: A Systematic. Clin. Exp. Pharmacol. Physiol. 2019, 46, 975–983.
- Zhitkovich, A.; Song, Y.; Quievryn, G.; Voitkun, V. Non-Oxidative Mechanisms Are Responsible for the Induction of Mutagenesis by Reduction of Cr(VI) with Cysteine: Role of Ternary DNA Adducts in Cr(III)-Dependent Mutagenesis. Biochemistry 2001, 40, 549–560.
- Jing, H.; Wang, F.; Gao, X. Lithium intoxication induced pyroptosis via ROS / NF-κB / NLRP3 inflammasome regulatory networks in kidney of mice. Environ. Toxicol. 2022, 37, 825–835.
- Sakrajda, K.; Szczepankiewicz, A. Inflammation-Related Changes in Mood Disorders and the Immunomodulatory Role of Lithium. Int. J. Mol. Sci. 2021, 22, 1532.
- Pessin, J.E.; Saltiel, A. Signaling pathways in insulin action: Molecular targets of insulin resistance. J. Clin. Investig. 2000, 106, 165–169.
- Undurraga, J.; Sim, K.; Tondo, L.; Gorodischer, A.; Azua, E.; Tay, K.H.; Tan, D.; Baldessarini, R.J. Lithium treatment for unipolar major depressive disorder: Systematic review. J. Psychopharmacol. 2019, 33, 167–176.
- Zhang, Y.; Peti-Peterdi, J.; Brandes, A.U.; Riquier-Brison, A.; Carlson, N.G.; Müller, C.E.; Ecelbarger, C.M.; Kishore, B.K. Prasugrel suppresses development of lithium-induced nephrogenic diabetes insipidus in mice. Purinergic Signal. 2017, 13, 239–248.
- Ge, W.; Jakobsson, E. Systems Biology Understanding of the Effects of Lithium on Affective and Neurodegenerative Disorders. Front. Neurosci. 2018, 12, 933.
- Zhang, K.; Alaohali, A.; Sawangboon, N.; Sharpe, P.T.; Brauer, D.S.; Gentleman, E. A comparison of lithium-substituted phosphate and borate bioactive glasses for mineralised tissue repair. Dent. Mater. 2019, 35, 919–927.
- Duvall, A.E.; Gallicchio, V.S. Lithium Treatment in Clinical Medicine: History, Current Status and Future Use. J. Cell Sci. Ther. 2017, 08, 1–9.
- Murphy, N.; Redahan, L.; Lally, J. Management of lithium intoxication. BJPsych Adv. 2022, 1–10.
- Youssef, F.S.; Menze, E.T.; Ashour, M.L. A Potent Lignan from Prunes Alleviates Inflammation and Oxidative Stress in Lithium/Pilocarpine-Induced Epileptic Seizures in Rats. Antioxidants 2020, 9, 575.
- Agrawal, S.; Gollapudi, S.; Gupta, S.; Agrawal, A. Dendritic cells from the elderly display an intrinsic defect in the production of IL-10 in response to Lithium Chloride. Exp. Gerontol. 2013, 48, 1285–1292.
- Gaddam, A.; Allu, A.R.; Ganisetti, S.; Fernandes, H.R.; Stan, G.E.; Negrila, C.C.; Jamale, A.P.; Mear, F.; Montagne, L.; Ferreira, J.M.F. Effect of Vanadium Oxide on the Structure and Li-Ion Conductivity of Lithium Silicate Glasses. J. Phys. Chem. C 2021, 125, 16843–16857.
- Ahmed, T.A.; Suso, H.-P.; Maqbool, A.; Hincke, M.T. Processed eggshell membrane powder: Bioinspiration for an innovative wound healing product. Mater. Sci. Eng. C 2019, 95, 192–203.
- Young, W. Review of Lithium Effects on Brain and Blood. Cell Transplant. 2009, 18, 951–975.
- Gitlin, M. Lithium side effects and toxicity: Prevalence and management strategies. Int. J. Bipolar Disord. 2016, 4, 1–10.
- Nassar, A.; Azab, A.N. Effects of Lithium on Inflammation. ACS Chem. Neurosci. 2014, 5, 451–458.
- Luo, D.-Z.; Chang, C.-Y.; Huang, T.-R.; Studer, V.; Wang, T.-W.; Lai, W.-S. Lithium for schizophrenia: Supporting evidence from a 12-year, nationwide health insurance database and from Akt1-deficient mouse and cellular models. Sci. Rep. 2020, 10, 1–13.
- Maçon, A.L.B.; Jacquemin, M.; Page, S.J.; Li, S.; Bertazzo, S.; Stevens, M.M.; Hanna, J.V.; Jones, J.R. Lithium-silicate sol–gel bioactive glass and the effect of lithium precursor on structure–property relationships. J. Sol-Gel Sci. Technol. 2016, 81, 84–94.
- Stone, M.S.; Martyn, L.; Weaver, C.M. Potassium Intake, Bioavailability, Hypertension, and Glucose Control. Nutrients 2016, 8, 444.
- Pérez-Granados, A.M.; Vaquero, M.P. Silicon, aluminium, arsenic and lithium: Essentiality and human health implications. J. Nutr. Health Aging 2002, 6, 154–162.
- Villette, J.; Cuéllar, T.; Verdeil, J.-L.; Delrot, S.; Gaillard, I. Grapevine Potassium Nutrition and Fruit Quality in the Context of Climate Change. Front. Plant Sci. 2020, 11, 123.
- Mierlo, L.A.J.V.; Arends, L.R.; Streppel, M.T.; A Zeegers, M.P.; Kok, F.J.; E Grobbee, D.; Geleijnse, J.M. Blood pressure response to calcium supplementation: A meta-analysis of randomized controlled trials. J. Hum. Hypertens. 2006, 20, 571–580.
- Marie, P.J. Strontium as therapy for osteoporosis. Curr. Opin. Pharmacol. 2005, 5, 633–636.
- Hackl, B.; Todt, H.; Kubista, H.; Hilber, K.; Koenig, X. Psilocybin Therapy of Psychiatric Disorders Is Not Hampered by hERG Potassium Channel–Mediated Cardiotoxicity. Int. J. Neuropsychopharmacol. 2022, 25, 280–282.
- Grynpas, M.; Hamilton, E.; Cheung, R.; Tsouderos, Y.; Deloffre, P.; Hott, M.; Marie, P. Strontium increases vertebral bone volume in rats at a low dose that does not induce detectable mineralization defect. Bone 1996, 18, 253–259.
- Cabrera, W.E.; Schrooten, I.; De Broe, M.E.; D’Haese, P.C. Strontium and Bone. J. Bone Miner. Res. 1999, 14, 661–668.
- Schrooten, I.; Cabrera, W.; Goodman, W.G.; Dauwe, S.; Lamberts, L.V.; Marynissen, R.; Dorriné, W.; De Broe, M.E.; D’Haese, P.C. Strontium causes osteomalacia in chronic renal failure rats. Kidney Int. 1998, 54, 448–456.
- Brandi, M.L. New treatment strategies: Ipriflavone, strontium, vitamin D metabolites and analogs. Am. J. Med. 1993, 95, S69–S74.
- Weaver, C. White Vegetables: A Forgotten Source of Nutrients Potassium and Health 1–3. Am. Soc. Nutr. 2013, 4, 3685–3775.
- Lanham-New, S.A.; Lambert, H.; Frassetto, L. Potassium. Adv. Nutr. Int. Rev. J. 2012, 3, 820–821.
- Mandakhbayar, N.; El-Fiqi, A.; Lee, J.-H.; Kim, H.-W. Evaluation of Strontium-Doped Nanobioactive Glass Cement for Dentin–Pulp Complex Regeneration Therapy. ACS Biomater. Sci. Eng. 2019, 5, 6117–6126.
- Naruphontjirakul, P.; Porter, A.E.; Jones, J.R. In vitro osteogenesis by intracellular uptake of strontium containing bioactive glass nanoparticles. Acta Biomater. 2018, 66, 67–80.
- Aqib, R.; Kiani, S.; Bano, S.; Wadood, A.; Rehman, M.A.U. Ag–Sr doped mesoporous bioactive glass nanoparticles loaded chitosan/gelatin coating for orthopedic implants. Int. J. Appl. Ceram. Technol. 2021, 18, 544–562.
- Saleem, O.; Wahaj, M.; Akhtar, M.A.; Rehman, M.A.U. Fabrication and Characterization of Ag–Sr-Substituted Hydroxyapatite/Chitosan Coatings Deposited via Electrophoretic Deposition: A Design of Experiment Study. ACS Omega 2020, 5, 22984–22992.
- Hoppe, A.; Sarker, B.; Detsch, R.; Hild, N.; Mohn, D.; Stark, W.; Boccaccini, A. In vitro reactivity of Sr-containing bioactive glass (type 1393) nanoparticles. J. Non-Crystalline Solids 2014, 387, 41–46.
- Bano, S.; Akhtar, M.; Yasir, M.; Maqbool, M.S.; Niaz, A.; Wadood, A.; Rehman, M.U. Synthesis and Characterization of Silver–Strontium (Ag-Sr)-Doped Mesoporous Bioactive Glass Nanoparticles. Gels 2021, 7, 34.
- Khaliq, H.; Juming, Z.; Ke-Mei, P. The Physiological Role of Boron on Health. Biol. Trace Elem. Res. 2018, 186, 31–51.
- Kargozar, S.; Montazerian, M.; Fiume, E.; Baino, F. Multiple and Promising Applications of Strontium (Sr)-Containing Bioactive Glasses in Bone Tissue Engineering. Front. Bioeng. Biotechnol. 2019, 7, 161.
- Kot, F.S. Boron sources, speciation and its potential impact on health. Rev. Environ. Sci. Bio/Technol. 2009, 8, 3–28.
- Growth, B.E. Variables, Studies of the Interaction Between Boron and Calcium, and its Modification by Magnesium and Potassium. Rats 1992, 35, 225–237.
- Naruphontjirakul, P.; Tsigkou, O.; Li, S.; Porter, A.E.; Jones, J.R. Human mesenchymal stem cells differentiate into an osteogenic lineage in presence of strontium containing bioactive glass nanoparticles. Acta Biomater. 2019, 90, 373–392.





