
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Yi-Qun Wang | -- | 2113 | 2022-06-22 13:45:09 | | | |
| 2 | Lindsay Dong | -17 word(s) | 2096 | 2022-06-23 10:24:35 | | |
Video Upload Options
Sleep and wakefulness are basic behavioral states that require coordination between several brain regions, and they involve multiple neurochemical systems, including neuropeptides. Neuropeptides are a group of peptides produced by neurons and neuroendocrine cells of the central nervous system. Like traditional neurotransmitters, neuropeptides can bind to specific surface receptors and subsequently regulate neuronal activities. For example, orexin is a crucial component for the maintenance of wakefulness and the suppression of rapid eye movement (REM) sleep. In addition to orexin, melanin-concentrating hormone, and galanin may promote REM sleep. These results suggest that neuropeptides play an important role in sleep–wake regulation. These neuropeptides can be divided into three categories according to their effects on sleep–wake behaviors in rodents and humans. (i) Galanin, melanin-concentrating hormone, and vasoactive intestinal polypeptide are sleep-promoting peptides. It is also noticeable that vasoactive intestinal polypeptide particularly increases REM sleep. (ii) Orexin and neuropeptide S have been shown to induce wakefulness. (iii) Neuropeptide Y and substance P may have a bidirectional function as they can produce both arousal and sleep-inducing effects.
1. Introduction
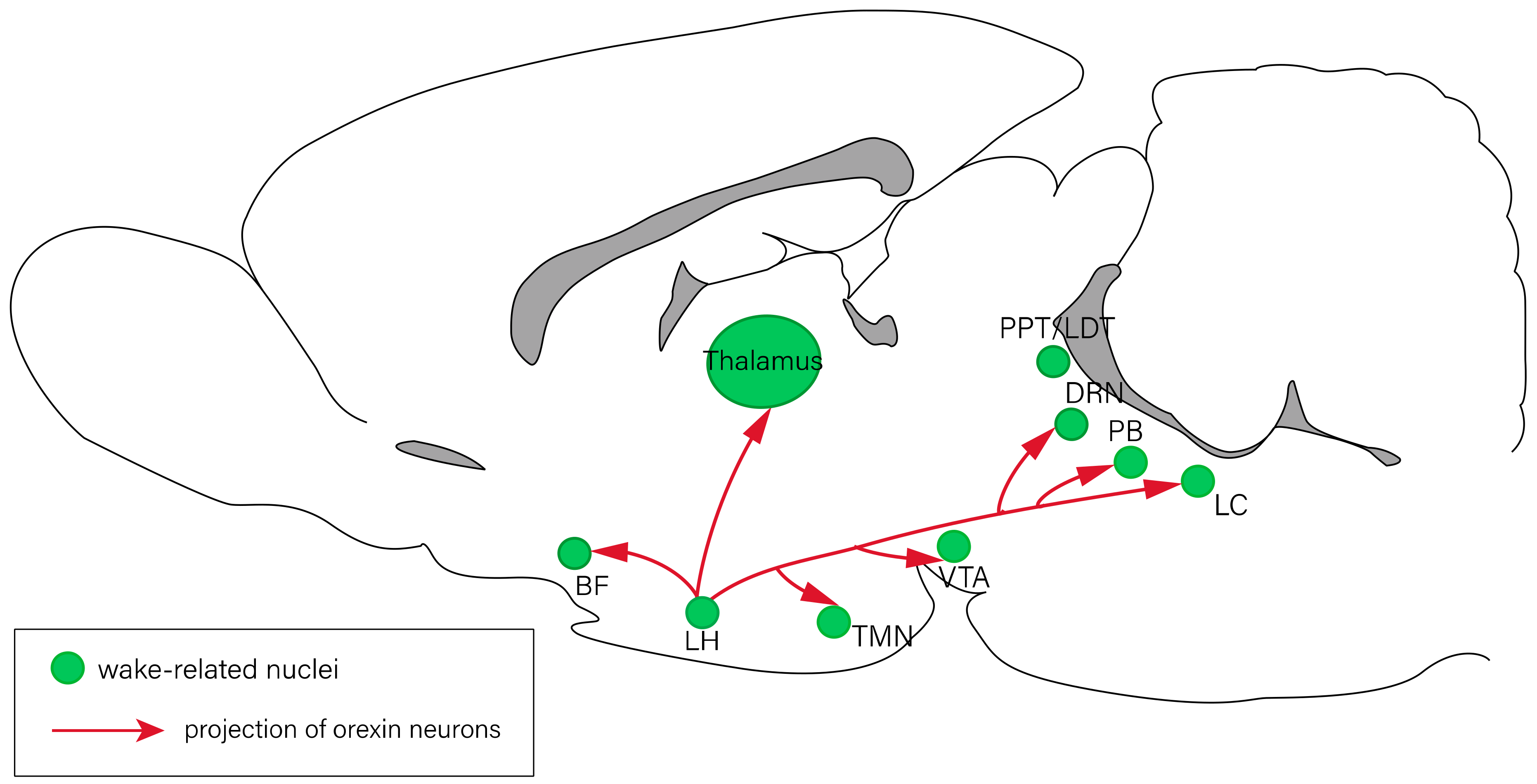
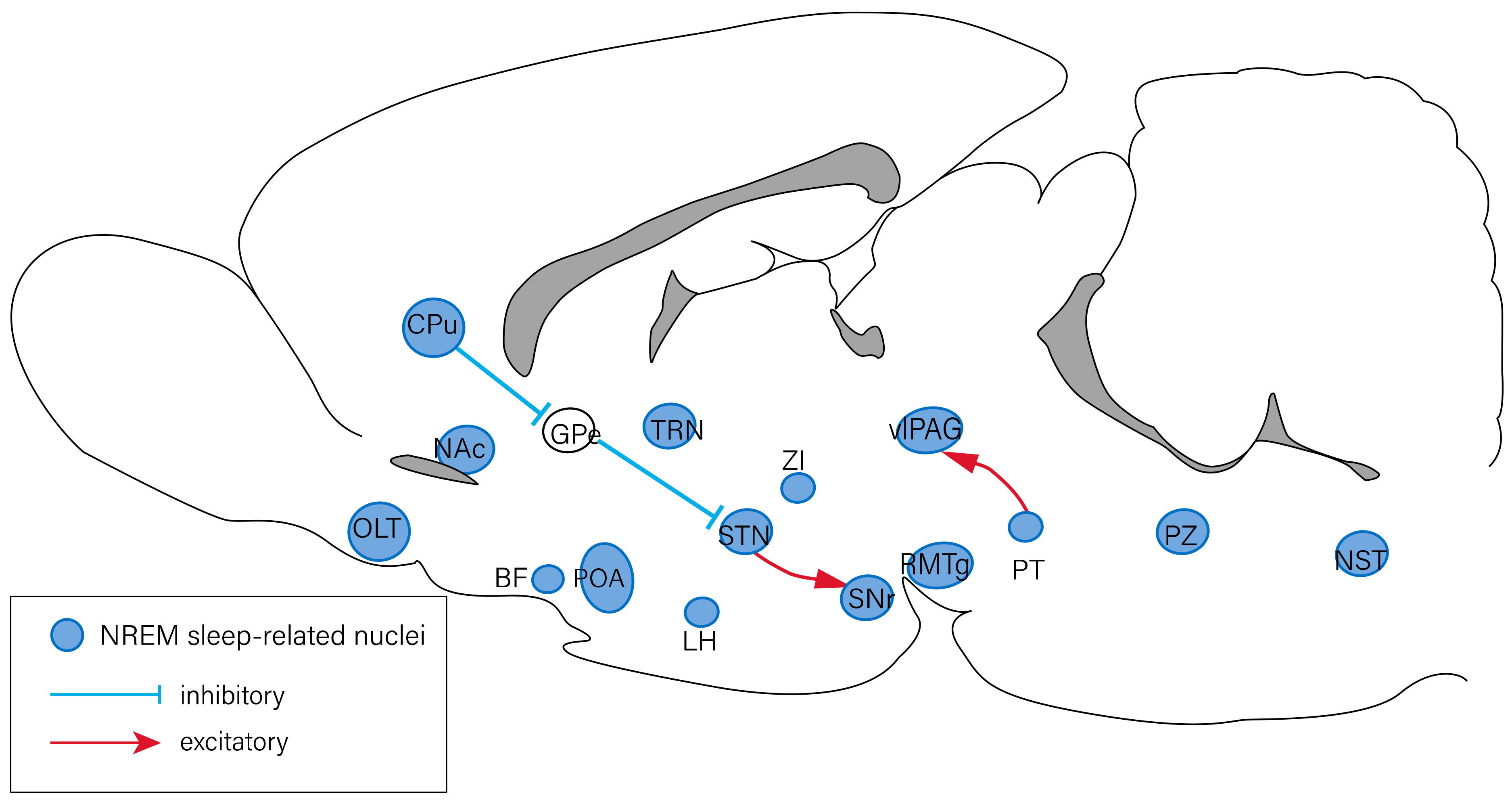
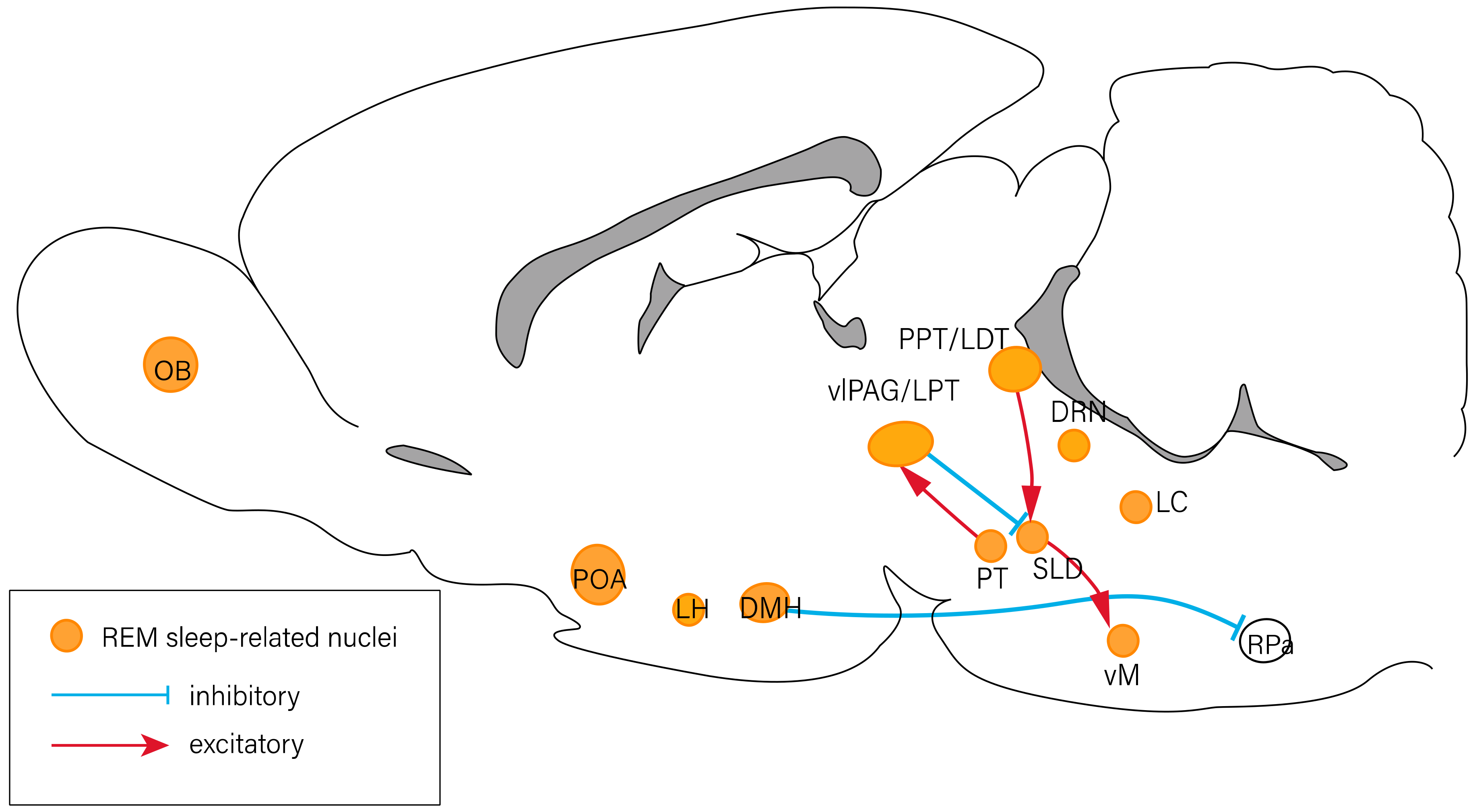
2. The Neuropeptides Involved in Sleep–Wake Regulation
2.1. Galanin Promotes Sleep
2.2. Orexin Consolidates Wakefulness and Inhibits REM Sleep
2.3. Melanin-Concentrating Hormone Has Positive Influence on Sleep
2.4. Neuropeptide S Is Associated with Arousal Induction
2.5. Neuropeptide Y Has a Dual Impact on Sleep–Wake Behaviors
2.6. Substance P Induces Either Sleep or Arousal
2.7. Vasoactive Intestinal Peptide Promotes REM Sleep
2.8. Other Neuropeptides Associated with Sleep and Wake Regulation
References
- Wang, Y.Q.; Li, R.; Zhang, M.Q.; Zhang, Z.; Qu, W.M.; Huang, Z.L. The neurobiological mechanisms and treatments of rem sleep disturbances in depression. Curr. Neuropharmacol. 2015, 13, 543–553.
- Chen, L.; Yin, D.; Wang, T.-X.; Guo, W.; Dong, H.; Xu, Q.; Luo, Y.-J.; Cherasse, Y.; Lazarus, M.; Qiu, Z.-l.; et al. Basal forebrain cholinergic neurons primarily contribute to inhibition of electroencephalogram delta activity; rather than inducing behavioral wakefulness in mice. Neuropsychopharmacology 2016, 41, 2133–2146.
- Wang, R.-F.; Guo, H.; Jiang, S.-Y.; Liu, Z.-L.; Qu, W.-M.; Huang, Z.-L.; Wang, L. Control of wakefulness by lateral hypothalamic glutamatergic neurons in male mice. J. Neurosci. Res. 2021, 99, 1689–1703.
- Yin, D.; Dong, H.; Wang, T.-X.; Hu, Z.-Z.; Chang, N.-N.; Qu, W.-M.; Huang, Z.-L. Glutamate activates the histaminergic tuberomammillary nucleus and increases wakefulness in rats. Neuroscience 2019, 413, 86–98.
- Yu, X.; Li, W.; Ma, Y.; Tossell, K.; Harris, J.J.; Harding, E.C.; Ba, W.; Miracca, G.; Wang, D.; Li, L.; et al. Gaba and glutamate neurons in the vta regulate sleep and wakefulness. Nat. Neurosci. 2019, 22, 106–119.
- Li, Y.-D.; Luo, Y.-J.; Xu, W.; Ge, J.; Cherasse, Y.; Wang, Y.-Q.; Lazarus, M.; Qu, W.-M.; Huang, Z.-L. Ventral pallidal gabaergic neurons control wakefulness associated with motivation through the ventral tegmental pathway. Mol. Psychiatry 2021, 26, 2912–2928.
- Kroeger, D.; Ferrari, L.L.; Petit, G.; Mahoney, C.E.; Fuller, P.M.; Arrigoni, E.; Scammell, T.E. Cholinergic, glutamatergic, and gabaergic neurons of the pedunculopontine tegmental nucleus have distinct effects on sleep/wake behavior in mice. J. Neurosci. 2017, 37, 1352–1366.
- Cui, S.-Y.; Li, S.-J.; Cui, X.-Y.; Zhang, X.-Q.; Yu, B.; Huang, Y.-L.; Cao, Q.; Xu, Y.-P.; Yang, G.; Ding, H.; et al. Ca2+ in the dorsal raphe nucleus promotes wakefulness via endogenous sleep-wake regulating pathway in the rats. Mol. Brain 2016, 9, 71.
- Xu, Q.; Wang, D.-R.; Dong, H.; Chen, L.; Lu, J.; Lazarus, M.; Cherasse, Y.; Chen, G.-H.; Qu, W.-M.; Huang, Z.-L. Medial parabrachial nucleus is essential in controlling wakefulness in rats. Front. Neurosci. 2021, 15, 645877.
- Liang, Y.; Shi, W.; Xiang, A.; Hu, D.; Wang, L.; Zhang, L. The naergic locus coeruleus- ventrolateral preoptic area neural circuit mediates rapid arousal from sleep. Curr. Biol. 2021, 31, 3729–3742.
- Li, R.; Wang, Y.-Q.; Liu, W.-Y.; Zhang, M.-Q.; Li, L.; Cherasse, Y.; Schiffmann, S.N.; d’Exaerde, A.d.K.; Lazarus, M.; Qu, W.-M.; et al. Activation of adenosine a(2a) receptors in the olfactory tubercle promotes sleep in rodents. Neuropharmacology 2020, 168, 107923.
- Weber, F.; Do, J.P.H.; Chung, S.; Beier, K.T.; Bikov, M.; Doost, M.S.; Dan, Y. Regulation of rem and non-rem sleep by periaqueductal gabaergic neurons. Nat. Commun. 2018, 9, 354.
- Benedetto, L.; Chase, M.H.; Torterolo, P. Gabaergic processes within the median preoptic nucleus promote nrem sleep. Behav. Brain Res. 2012, 232, 60–65.
- Yang, S.-R.; Hu, Z.-Z.; Luo, Y.-J.; Zhao, Y.-N.; Sun, H.-X.; Yin, D.; Wang, C.-Y.; Yan, Y.-D.; Wang, D.-R.; Yuan, X.-S.; et al. The rostromedial tegmental nucleus is essential for non-rapid eye movement sleep. PLoS Biol. 2018, 16, e2002909.
- Ni, K.-M.; Hou, X.-J.; Yang, C.-H.; Dong, P.; Li, Y.; Zhang, Y.; Jiang, P.; Berg, D.K.; Duan, S.; Li, X.-M. Selectively driving cholinergic fibers optically in the thalamic reticular nucleus promotes sleep. eLife 2016, 5, e10382.
- Wang, Y.-Q.; Li, R.; Wang, D.-R.; Cherasse, Y.; Zhang, Z.; Zhang, M.-Q.; Lavielle, O.; McEown, K.; Schiffmann, S.N.; d’Exaerde, A.d.K.; et al. Adenosine a(2a) receptors in the olfactory bulb suppress rapid eye movement sleep in rodents. Brain Struct. Funct. 2017, 222, 1351–1366.
- Gvilia, I.; Turner, A.; McGinty, D.; Szymusiak, R. Preoptic area neurons and the homeostatic regulation of rapid eye movement sleep. J. Neurosci. 2006, 26, 3037–3044.
- Monti, J.M.; Monti, D. Role of dorsal raphe nucleus serotonin 5-ht1a receptor in the regulation of rem sleep. Life Sci. 2000, 66, 1999–2012.
- Schwartz, M.D.; Nguyen, A.T.; Warrier, D.R.; Palmerston, J.B.; Thomas, A.M.; Morairty, S.R.; Neylan, T.C.; Kilduff, T.S. Locus coeruleus and tuberomammillary nuclei ablations attenuate hypocretin/orexin antagonist-mediated rem sleep. eNeuro 2016, 3.
- Liu, D.; Dan, Y. A motor theory of sleep-wake control: Arousal-action circuit. Annu. Rev. Neurosci. 2019, 42, 27–46.
- Wang, Y.-Q.; Liu, W.-Y.; Li, L.; Qu, W.-M.; Huang, Z.-L. Neural circuitry underlying rem sleep: A review of the literature and current concepts. Prog. Neurobiol. 2021, 204, 102106.
- Burbach, J.P. What are neuropeptides? Methods Mol. Biol. 2011, 789, 1–36.
- Salio, C.; Lossi, L.; Ferrini, F.; Merighi, A. Neuropeptides as synaptic transmitters. Cell Tissue Res. 2006, 326, 583–598.
- Hook, V.; Funkelstein, L.; Lu, D.; Bark, S.; Wegrzyn, J.; Hwang, S.-R. Proteases for processing proneuropeptides into peptide neurotransmitters and hormones. Annu. Rev. Pharmacol. Toxicol. 2008, 48, 393–423.
- Ludwig, M.; Leng, G. Dendritic peptide release and peptide-dependent behaviours. Nat. Rev. Neurosci. 2006, 7, 126–136.
- Hartman, K.; Mielczarek, P.; Smoluch, M.; Silberring, J. Inhibitors of neuropeptide peptidases engaged in pain and drug dependence. Neuropharmacology 2020, 175, 108137.
- Lang, R.; Gundlach, A.L.; Kofler, B. The galanin peptide family: Receptor pharmacology, pleiotropic biological actions, and implications in health and disease. Pharmacol. Ther. 2007, 115, 177–207.
- Branchek, T.A.; Smith, K.E.; Gerald, C.; Walker, M.W. Galanin receptor subtypes. Trends Pharmacol. Sci. 2000, 21, 109–116.
- Mitsukawa, K.; Lu, X.; Bartfai, T. Galanin, galanin receptors and drug targets. Cell Mol. Life Sci. 2008, 65, 1796–1805.
- Qualls-Creekmore, E.; Yu, S.; Francois, M.; Hoang, J.; Huesing, C.; Bruce-Keller, A.; Burk, D.; Berthoud, H.R.; Morrison, C.D.; Munzberg, H. Galanin-expressing gaba neurons in the lateral hypothalamus modulate food reward and noncompulsive locomotion. J. Neurosci. 2017, 37, 6053–6065.
- Idelevich, A.; Sato, K.; Nagano, K.; Rowe, G.; Gori, F.; Baron, R. Deltafosb requires galanin, but not leptin, to increase bone mass via the hypothalamus, but both are needed to increase energy expenditure. J. Bone Miner. Res. 2019, 34, 1707–1720.
- Koenig, J.I.; Hooi, S.; Gabriel, S.M.; Martin, J.B. Potential involvement of galanin in the regulation of fluid homeostasis in the rat. Regul. Pept. 1989, 24, 81–86.
- Scheller, K.J.; Williams, S.J.; Lawrence, A.J.; Djouma, E. The galanin-3 receptor antagonist, snap 37889, suppresses alcohol drinking and morphine self-administration in mice. Neuropharmacology 2017, 118, 1–12.
- Yang, Y.; Zhang, Y.; Li, X.H.; Li, Y.; Qian, R.; Li, J.; Xu, S.L. Involvements of galanin and its receptors in antinociception in nucleus accumbens of rats with inflammatory pain. Neurosci. Res. 2015, 97, 20–25.
- Gaus, S.E.; Strecker, R.E.; Tate, B.A.; Parker, R.A.; Saper, C.B. Ventrolateral preoptic nucleus contains sleep-active, galaninergic neurons in multiple mammalian species. Neuroscience 2002, 115, 285–294.
- Steininger, T.L.; Gong, H.; McGinty, D.; Szymusiak, R. Subregional organization of preoptic area/anterior hypothalamic projections to arousal-related monoaminergic cell groups. J. Comp. Neurol. 2001, 429, 638–653.
- Saito, Y.C.; Tsujino, N.; Abe, M.; Yamazaki, M.; Sakimura, K.; Sakurai, T. Serotonergic input to orexin neurons plays a role in maintaining wakefulness and rem sleep architecture. Front. Neurosci. 2018, 12, 892.
- Thomas, S.; Kilduff, C.P. The hypocretin/orexin ligand–receptor system: Implications for sleep and sleep disorders. Trends Neurosci. 2000, 23, 359–365.
- Peyron, C.; Tighe, D.K.; van den Pol, A.N.; de Lecea, L.; Heller, H.C.; Sutcliffe, J.G.; Kilduff, T.S. Neurons containing hypocretin (orexin) project to multiple neuronal systems. J. Neurosci. 1998, 18, 9996–10015.
- Arima, Y.; Yokota, S.; Fujitani, M. Lateral parabrachial neurons innervate orexin neurons projecting to brainstem arousal areas in the rat. Sci. Rep. 2019, 9, 2830.
- Soya, S.; Takahashi, T.M.; McHugh, T.J.; Maejima, T.; Herlitze, S.; Abe, M.; Sakimura, K.; Sakurai, T. Orexin modulates behavioral fear expression through the locus coeruleus. Nat. Commun. 2017, 8, 1606.
- Yang, C.; Zhang, L.; Hao, H.; Ran, M.; Li, J.; Dong, H. Serotonergic neurons in the dorsal raphe nucleus mediate the arousal-promoting effect of orexin during isoflurane anesthesia in male rats. Neuropeptides 2019, 75, 25–33.
- Bayer, L.; Eggermann, E.; Serafin, M.; Saint-Mleux, B.; Machard, D.; Jones, B.; Muhlethaler, M. Orexins (hypocretins) directly excite tuberomammillary neurons. Eur. J. Neurosci. 2001, 14, 1571–1575.
- Ishibashi, M.; Gumenchuk, I.; Kang, B.; Steger, C.; Lynn, E.; Molina, N.E.; Eisenberg, L.M.; Leonard, C.S. Orexin receptor activation generates gamma band input to cholinergic and serotonergic arousal system neurons and drives an intrinsic ca(2+)-dependent resonance in ldt and ppt cholinergic neurons. Front. Neurol. 2015, 6, 120.
- Zhang, L.N.; Yang, C.; Ouyang, P.R.; Zhang, Z.C.; Ran, M.Z.; Tong, L.; Dong, H.L.; Liu, Y. Orexin-a facilitates emergence of the rat from isoflurane anesthesia via mediation of the basal forebrain. Neuropeptides 2016, 58, 7–14.
- Torterolo, P.; Lagos, P.; Monti, J.M. Melanin-concentrating hormone: A new sleep factor? Front. Neurol. 2011, 2, 14.
- Sita, L.V.; Elias, C.F.; Bittencourt, J.C. Dopamine and melanin-concentrating hormone neurons are distinct populations in the rat rostromedial zona incerta. Brain Res. 2003, 970, 232–237.
- Pissios, P. Animals models of mch function and what they can tell us about its role in energy balance. Peptides 2009, 30, 2040–2044.
- Bittencourt, J.C.; Presse, F.; Arias, C.; Peto, C.; Vaughan, J.; Nahon, J.L.; Vale, W.; Sawchenko, P.E. The melanin-concentrating hormone system of the rat brain: An immuno- and hybridization histochemical characterization. J. Comp. Neurol. 1992, 319, 218–245.
- Sita, L.V.; Elias, C.F.; Bittencourt, J.C. Connectivity pattern suggests that incerto-hypothalamic area belongs to the medial hypothalamic system. Neuroscience 2007, 148, 949–969.
- Monti, J.M.; Torterolo, P.; Lagos, P. Melanin-concentrating hormone control of sleep-wake behavior. Sleep Med. Rev. 2013, 17, 293–298.
- Reinscheid, R.K.; Xu, Y.L. Neuropeptide s as a novel arousal promoting peptide transmitter. FEBS J. 2005, 272, 5689–5693.
- Koob, G.F.; Greenwell, T.N. Neuropeptide s: A novel activating anxiolytic? Neuron 2004, 43, 441–442.
- Okamura, N.; Reinscheid, R.K. Neuropeptide s: A novel modulator of stress and arousal. Stress 2007, 10, 221–226.
- Xu, Y.L.; Reinscheid, R.K.; Huitron-Resendiz, S.; Clark, S.D.; Wang, Z.; Lin, S.H.; Brucher, F.A.; Zeng, J.; Ly, N.K.; Henriksen, S.J.; et al. Neuropeptide s: A neuropeptide promoting arousal and anxiolytic-like effects. Neuron 2004, 43, 487–497.
- Castro, A.A.; Moretti, M.; Casagrande, T.S.; Martinello, C.; Petronilho, F.; Steckert, A.V.; Guerrini, R.; Calo, G.; Dal Pizzol, F.; Quevedo, J.; et al. Neuropeptide s produces hyperlocomotion and prevents oxidative stress damage in the mouse brain: A comparative study with amphetamine and diazepam. Pharmacol. Biochem. Behav. 2009, 91, 636–642.
- Beck, B.; Fernette, B.; Stricker-Krongrad, A. Peptide s is a novel potent inhibitor of voluntary and fast-induced food intake in rats. Biochem. Biophys. Res. Commun. 2005, 332, 859–865.
- Niimi, M. Centrally administered neuropeptide s activates orexin-containing neurons in the hypothalamus and stimulates feeding in rats. Endocrine 2006, 30, 75–79.
- Peng, Y.L.; Han, R.W.; Chang, M.; Zhang, L.; Zhang, R.S.; Li, W.; Han, Y.F.; Wang, R. Central neuropeptide s inhibits food intake in mice through activation of neuropeptide s receptor. Peptides 2010, 31, 2259–2263.
- Blomqvist, A.G.; Herzog, H. Y-receptor subtypes—How many more? Trends Neurosci. 1997, 20, 294–298.
- Diaz-delCastillo, M.; Woldbye, D.P.D.; Heegaard, A.M. Neuropeptide y and its involvement in chronic pain. Neuroscience 2018, 387, 162–169.
- Kautz, M.; Charney, D.S.; Murrough, J.W. Neuropeptide y, resilience, and ptsd therapeutics. Neurosci. Lett. 2017, 649, 164–169.
- Stanley, B.G.; Leibowitz, S.F. Neuropeptide y: Stimulation of feeding and drinking by injection into the paraventricular nucleus. Life Sci. 1984, 35, 2635–2642.
- Kornhuber, J.; Zoicas, I. Neuropeptide y reduces expression of social fear via simultaneous activation of y1 and y2 receptors. J. Psychopharmacol. 2019, 33, 1533–1539.
- Erickson, J.C.; Hollopeter, G.; Palmiter, R.D. Attenuation of the obesity syndrome of ob/ob mice by the loss of neuropeptide y. Science 1996, 274, 1704–1707.
- Harrington, M.; Molyneux, P.; Soscia, S.; Prabakar, C.; McKinley-Brewer, J.; Lall, G. Behavioral and neurochemical sources of variability of circadian period and phase: Studies of circadian rhythms of npy-/- mice. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2007, 292, R1306–R1314.
- Chang, R.S.; Lotti, V.J.; Chen, T.B.; Cerino, D.J.; Kling, P.J. Neuropeptide y (npy) binding sites in rat brain labeled with 125i-bolton-hunter npy: Comparative potencies of various polypeptides on brain npy binding and biological responses in the rat vas deferens. Life Sci. 1985, 37, 2111–2122.
- De Quidt, M.E.; Emson, P.C. Distribution of neuropeptide y-like immunoreactivity in the rat central nervous system—II. Immunohistochemical analysis. Neuroscience 1986, 18, 545–618.
- Lynch, D.R.; Walker, M.W.; Miller, R.J.; Snyder, S.H. Neuropeptide y receptor binding sites in rat brain: Differential autoradiographic localizations with 125i-peptide yy and 125i-neuropeptide y imply receptor heterogeneity. J. Neurosci. 1989, 9, 2607–2619.
- Dyzma, M.; Boudjeltia, K.Z.; Faraut, B.; Kerkhofs, M. Neuropeptide y and sleep. Sleep Med. Rev. 2010, 14, 161–165.
- Ribeiro-da-Silva, A.; Hokfelt, T. Neuroanatomical localisation of substance p in the cns and sensory neurons. Neuropeptides 2000, 34, 256–271.
- Yip, J.; Chahl, L.A. Localization of tachykinin receptors and fos-like immunoreactivity induced by substance p in guinea-pig brain. Clin. Exp. Pharmacol. Physiol. 2000, 27, 943–946.
- Kramer, M.S.; Cutler, N.; Feighner, J.; Shrivastava, R.; Carman, J.; Sramek, J.J.; Reines, S.A.; Liu, G.; Snavely, D.; Wyatt-Knowles, E.; et al. Distinct mechanism for antidepressant activity by blockade of central substance p receptors. Science 1998, 281, 1640–1645.
- Lisowska, B.; Siewruk, K.; Lisowski, A. Substance p and acute pain in patients undergoing orthopedic surgery. PLoS ONE 2016, 11, e0146400.
- Dam, T.V.; Escher, E.; Quirion, R. Evidence for the existence of three classes of neurokinin receptors in brain. Differential ontogeny of neurokinin-1, neurokinin-2 and neurokinin-3 binding sites in rat cerebral cortex. Brain Res. 1988, 453, 372–376.
- Brown, R.E.; Basheer, R.; McKenna, J.T.; Strecker, R.E.; McCarley, R.W. Control of sleep and wakefulness. Physiol. Rev. 2012, 92, 1087–1187.
- Zhang, G.; Wang, L.; Liu, H.; Zhang, J. Substance p promotes sleep in the ventrolateral preoptic area of rats. Brain Res. 2004, 1028, 225–232.
- Zielinski, M.R.; Karpova, S.A.; Yang, X.; Gerashchenko, D. Substance p and the neurokinin-1 receptor regulate electroencephalogram non-rapid eye movement sleep slow-wave activity locally. Neuroscience 2015, 284, 260–272.
- Zielinski, M.R.; Gerashchenko, D. Sleep-inducing effect of substance p-cholera toxin a subunit in mice. Neurosci. Lett. 2017, 659, 44–47.
- Andersen, M.L.; Nascimento, D.C.; Machado, R.B.; Roizenblatt, S.; Moldofsky, H.; Tufik, S. Sleep disturbance induced by substance p in mice. Behav. Brain Res. 2006, 167, 212–218.
- Lieb, K.; Ahlvers, K.; Dancker, K.; Strohbusch, S.; Reincke, M.; Feige, B.; Berger, M.; Riemann, D.; Voderholzer, U. Effects of the neuropeptide substance p on sleep, mood, and neuroendocrine measures in healthy young men. Neuropsychopharmacology 2002, 27, 1041–1049.
- Sergeeva, O.A.; Mazur, K.; Kernder, A.; Haas, H.L.; De Luca, R. Tachykinins amplify the action of capsaicin on central histaminergic neurons. Peptides 2021, 150, 170729.
- Fahrenkrug, J.; Emson, P.C. Vasoactive intestinal polypeptide: Functional aspects. Br. Med. Bull. 1982, 38, 265–270.
- Milena Pavlova, M. Circadian rhythm sleep-wake disorders. Continuum 2017, 23, 1051–1063.
- Welsh, D.K.; Logothetis, D.E.; Meister, M.; Reppert, S.M. Individual neurons dissociated from rat suprachiasmatic nucleus express independently phased circadian firing rhythms. Neuron 1995, 14, 697–706.
- Vosko, A.M.; Schroeder, A.; Loh, D.H.; Colwell, C.S. Vasoactive intestinal peptide and the mammalian circadian system. Gen. Comp. Endocrinol. 2007, 152, 165–175.
- Abrahamson, E.E.; Moore, R.Y. Suprachiasmatic nucleus in the mouse: Retinal innervation, intrinsic organization and efferent projections. Brain Res. 2001, 916, 172–191.
- Hannibal, J.; Fahrenkrug, J. Circadian rhythm regulation: A central role for the neuropeptide vasoactive intestinal polypeptide. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2003, 285, R935–R936.
- Chaki, S.; Kawashima, N.; Suzuki, Y.; Shimazaki, T.; Okuyama, S. Cocaine- and amphetamine-regulated transcript peptide produces anxiety-like behavior in rodents. Eur. J. Pharmacol. 2003, 464, 49–54.
- Esposito, M.; Pellinen, J.; Kapas, L.; Szentirmai, E. Impaired wake-promoting mechanisms in ghrelin receptor-deficient mice. Eur. J. Neurosci. 2012, 35, 233–243.
- Furutani, N.; Hondo, M.; Kageyama, H.; Tsujino, N.; Mieda, M.; Yanagisawa, M.; Shioda, S.; Sakurai, T. Neurotensin co-expressed in orexin-producing neurons in the lateral hypothalamus plays an important role in regulation of sleep/wakefulness states. PLoS ONE 2013, 8, e62391.
- Martel, G.; Dutar, P.; Epelbaum, J.; Viollet, C. Somatostatinergic systems: An update on brain functions in normal and pathological aging. Front. Endocrinol. 2012, 3, 154.
- Ahnaou, A.; Basille, M.; Gonzalez, B.; Vaudry, H.; Hamon, M.; Adrien, J.; Bourgin, P. Long-term enhancement of rem sleep by the pituitary adenylyl cyclase-activating polypeptide (pacap) in the pontine reticular formation of the rat. Eur. J. Neurosci. 1999, 11, 4051–4058.
- Chastrette, N.; Cespuglio, R.; Jouvet, M. Proopiomelanocortin (pomc)-derived peptides and sleep in the rat. Part 1—Hypnogenic properties of acth derivatives. Neuropeptides 1990, 15, 61–74.
- Fabio García-García, E.J.-A.; Santiago-García, J.; Cardinali, D.P. Ghrelin and its interactions with growth hormone, leptin and orexins: Implications for the sleepewake cycle and metabolism. Sleep Med. Rev. 2013, 18, 89–97.
- Hirashima, N.; Ichiki, K.; Tanaka, H.; Kilduff, T.S.; Yamanaka, A. Neuropeptide b induces slow wave sleep in mice. Sleep 2010, 34, 31–37.
- Obal, F.; Alt, J.; Taishi, P.; Gardi, J.; Krueger, J.M. Sleep in mice with nonfunctional growth hormone-releasing hormone receptors. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2002, 284, R131–R139.
- Rios, M.; Fan, G.; Fekete, C.; Kelly, J.; Bates, B.; Kuehn, R.; Lechan, R.M.; Jaenisch, R. Conditional deletion of brain-derived neurotrophic factor in the postnatal brain leads to obesity and hyperactivity. Mol. Endocrin. 2001, 15, 1748–1757.
- Sinton, C.M.; Fitch, T.E.; Gershenfeld, H.K. The effects of leptin on rem sleep and slow wave delta in rats are reversed by food deprivation. J. Sleep Res. 1999, 8, 197–203.
- Jewett, K.A.; Krueger, J.M. Humoral sleep regulation; interleukin-1 and tumor necrosis factor. Vitam. Horm. 2012, 89, 241–257.
- Wang, D.; Teichtahl, H. Opioids, steep architecture and steep-disordered breathing. Sleep Med. Rev. 2007, 11, 35–46.
- Kramer, A.; Yang, F.C.; Snodgrass, P.; Li, X.; Scammell, T.E.; Davis, F.C.; Weitz, C.J. Regulation of daily locomotor activity and sleep by hypothalamic egf receptor signaling. Science 2001, 294, 2511–2515.





