
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Florence Pinet | + 674 word(s) | 674 | 2020-09-29 05:14:11 | | | |
| 2 | Florence Pinet | -13 word(s) | 661 | 2020-10-08 12:20:40 | | | | |
| 3 | Florence Pinet | -13 word(s) | 661 | 2020-10-08 12:21:26 | | | | |
| 4 | Conner Chen | Meta information modification | 661 | 2020-10-09 09:16:53 | | | | |
| 5 | Catherine Yang | Meta information modification | 661 | 2021-09-28 11:26:03 | | |
Video Upload Options
This entry aims to introduce the physiological roles and pathological implications of oxidative stress in cardiovascular tissues
Introduction
Cardiovascular diseases are multifactorial disorders that represent the leading causes of death worldwide according to the World Health Organization (WHO) [1]. The physiopathology of cardiovascular diseases, mainly caused by atherosclerosis, includes remodeling of blood vessels that can result in blood flow restrictions affecting the heart and the nervous system. Cardiovascular diseases comprise several disorders such as coronary artery diseases, stroke, hypertension, heart failure, congenital heart diseases, and vascular diseases. The main risk factors for cardiovascular diseases are obesity, diabetes, cigarette smoking, a sedentary and unhealthy lifestyle, and genetic predisposition [1]. Aging is also another risk factor, since it increases cardiovascular diseases prevalence mainly due to the accumulation of oxidative damage. Indeed, oxidative stress is an important factor involved in cardiovascular diseases progression. In this review, we will first detail the physiological role of reactive oxygen species (ROS) production in the heart and the vessels. Then, we will investigate the implication of oxidative stress in cardiovascular diseases and we will focus on ROS produced by NAPDH oxidase or during endothelial or mitochondrial dysfunction. In the last part of this review, we will detail the new therapeutic strategies potentially involved in cardiovascular protection and currently under study.
Physiological Roles of Oxidative Stress in Cardiovascular Tissues
Under physiological conditions, low levels of ROS production are equivalent to their detoxification and play a major role in cellular signaling and function [2]. This process is called redox signaling and is defined as the specific and reversible oxidation/reduction modification of cellular signaling components able to regulate gene expression, excitation–contraction coupling, or cell growth, migration, differentiation, and death [3][4] (Figure 1).
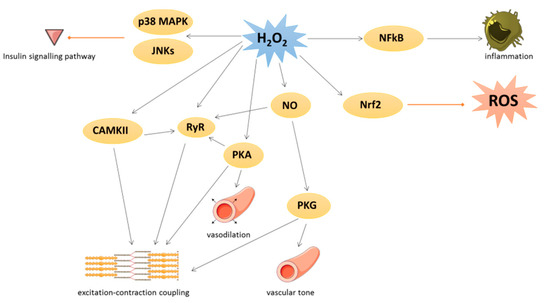
Figure 1. Physiological roles of oxidative stress in cardiovascular tissues. Black arrow represents activation and red arrow represents inhibition. p38 MAPK: p38 mitogen-activated protein kinase; JNK: c-Jun N-terminal kinase; H2O2: hydrogen peroxide; NFκB: nuclear factor-kappa beta; CAMKII: Ca/calmodulin-dependent kinase II; RyR: ryanodine receptor; PKA: cAMP-induced protein kinase A; NO: nitric oxide; PKG: protein kinase G; Nrf2: nuclear factor erythroid 2-related factor 2.
Several kinases are involved in redox signaling. For instance, H2O2 could activate Ca/calmodulin-dependent kinase II (CAMKII), leading to excitation–contraction coupling [4] or p38 mitogen-activated protein kinase (p38 MAPK) and c-Jun N-terminal kinase (JNKs) leading to inhibition of insulin signal transduction. cAMP-induced protein kinase A (PKA) is also activated by oxidation of its regulatory subunit R1α and translocated from cytosol to membrane, where PKA regulates cardiac excitation–contraction coupling in the heart and vasodilation in the vessels [4].
Many transcription factors are also regulated by redox signaling. As an example, the nuclear factor-kappa beta (NFκB) is activated when ROS have damaged its inhibitor (IkB) and regulates inflammation process [5]. The nuclear factor erythroid 2-related factor 2 (Nrf2) could be induced by lipid peroxidation to activate several key antioxidant enzymes containing an antioxidant/electrophile response element motif in their promoter, such as heme oxygenase 1, glutathione peroxidases, SOD, peroxiredoxins, thioredoxins, and thioredoxin reductases [6]. In detail, under unstressed conditions, Nrf2 is constitutively ubiquitinated by both Kelch-k-like ECH-associated protein 1 (Keap1) and Cullin-3 E3 ligase to be damaged [6][7]. The activation of Nrf2 is due to the oxidation of Keap1 that abrogated its negative control on Nrf2.
Under physiological conditions, NO is a cytoprotective molecule with a vasodilator action. NO inhibits the activation and adhesion of platelets and neutrophils and has protective effects against ischemia reperfusion and heart failure [8]. NO could exert its biological effects by binding to the soluble guanylate cyclase in order to produce cyclic guanosine monophosphate leading to protein kinase G (PKG) activation [9] or by S-nitrosylation. This latter modification could modulate several protein activities such as pro-caspase 3, myosin heavy chain, tropomyosin, peroxiredoxins, or ryanodine receptor (RyR). RyR, which mediates Ca2+ release from sarcoplasmic reticulum, is also activated by phosphorylation via PKA and CAMKII, themselves redox-regulated [4]. Moreover, PKG is activated by oxidation independently of NO and regulates vascular tone and cardiomyocyte contraction or hypertrophy [4].
References
- Benjamin, E.J.; Blaha, M.J.; Chiuve, S.E.; Cushman, M.; Das, S.R.; Deo, R.; de Ferranti, S.D.; Floyd, J.; Fornage, M.; Gillespie, C.; et al. Heart Disease and Stroke Statistics-2017 Update: A Report From the American Heart Association. Circulation 2017, 135, e146–e603.
- Tsutsui, H.; Kinugawa, S.; Matsushima, S. Mitochondrial oxidative stress and dysfunction in myocardial remodelling. Cardiovasc. Res. 2008, 81, 449–456.
- Sack, M.N.; Fyhrquist, F.Y.; Saijonmaa, O.J.; Fuster, V.; Kovacic, J.C. Basic Biology of Oxidative Stress and the Cardiovascular System: Part 1 of a 3-Part Series. J. Am. Coll. Cardiol. 2017, 70, 196–211.
- Burgoyne, J.R.; Mongue-Din, H.; Eaton, P.; Shah, A.M. Redox signaling in cardiac physiology and pathology. Circ. Res. 2012, 111, 1091–1106.
- Moris, D.; Spartalis, M.; Tzatzaki, E.; Spartalis, E.; Karachaliou, G.S.; Triantafyllis, A.S.; Karaolanis, G.I.; Tsilimigras, D.I.; Theocharis, S. The role of reactive oxygen species in myocardial redox signaling and regulation. Ann. Transl. Med. 2017, 5, 324.
- Lismont, C.; Revenco, I.; Fransen, M. Peroxisomal hydrogen peroxide metabolism and signaling in health and disease. Int. J. Mol. Sci. 2019, 20, 3673.
- Kasai, S.; Shimizu, S.; Tatara, Y.; Mimura, J.; Itoh, K. Regulation of Nrf2 by mitochondrial reactive oxygen species in physiology and pathology. Biomolecules 2020, 10, 320.
- Loyer, X.; Heymes, C.; Samuel, J.L. Constitutive nitric oxide synthases in the heart from hypertrophy to failure. Clin. Exp. Pharmacol. Physiol. 2008, 35, 483–488.
- Hammond, J.; Balligand, J.L. Nitric oxide synthase and cyclic GMP signaling in cardiac myocytes: From contractility to remodeling. J. Mol. Cell. Cardiol. 2012, 52, 330–340.





