Your browser does not fully support modern features. Please upgrade for a smoother experience.

Submitted Successfully!
Thank you for your contribution! You can also upload a video entry or images related to this topic.
For video creation, please contact our Academic Video Service.
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Philippe Drabent | -- | 5626 | 2022-06-01 15:46:32 | | | |
| 2 | Vivi Li | -39 word(s) | 5587 | 2022-06-02 05:32:35 | | |
Video Upload Options
We provide professional Academic Video Service to translate complex research into visually appealing presentations. Would you like to try it?
Cite
If you have any further questions, please contact Encyclopedia Editorial Office.
Drabent, P.; Fraitag, S. Malignant Superficial Mesenchymal Tumors in Children. Encyclopedia. Available online: https://encyclopedia.pub/entry/23673 (accessed on 04 June 2026).
Drabent P, Fraitag S. Malignant Superficial Mesenchymal Tumors in Children. Encyclopedia. Available at: https://encyclopedia.pub/entry/23673. Accessed June 04, 2026.
Drabent, Philippe, Sylvie Fraitag. "Malignant Superficial Mesenchymal Tumors in Children" Encyclopedia, https://encyclopedia.pub/entry/23673 (accessed June 04, 2026).
Drabent, P., & Fraitag, S. (2022, June 01). Malignant Superficial Mesenchymal Tumors in Children. In Encyclopedia. https://encyclopedia.pub/entry/23673
Drabent, Philippe and Sylvie Fraitag. "Malignant Superficial Mesenchymal Tumors in Children." Encyclopedia. Web. 01 June, 2022.
Copy Citation
Malignant superficial mesenchymal tumors are a very diverse group of neoplasms with few clinical and radiological discriminatory factors. Hence, some of these cancers are rarely suspected based on clinical and radiological grounds, others may be easily misdiagnosed, and the histological analysis of a biopsy or resection is central in the diagnostic process. In children, the age at presentation is a major element of the differential diagnosis. Some tumors have a very distinct epidemiology, while others may be seen at any age. More recently, the advances in molecular biology have greatly improved the diagnosis of mesenchymal tumors and new entities are still being described.
children
skin
sarcoma
mesenchymal tumors
diagnosis
histology
genetics
1. Introduction
This entry focuses on primitive malignant mesenchymal tumors of the skin in children. Neuroectodermal tumors are included here as well, since they share some characteristics with soft tissue tumors of neural differentiation. Superficial leiomyosarcoma and epithelioid hemangioendothelioma are far too exceptional in children to have been included hereafter. Metastases are not discussed, or only briefly for differential diagnosis. Pseudotumors and benign tumors do not fall into the topic of this entry.
There are few studies that have focused on the epidemiology of malignant mesenchymal tumors of the skin specifically in children. It is important to bear in mind that, in children and adolescents, soft tissue tumors of intermediate malignant potential are by far more frequent than high grade sarcomas. In one American study, the most prevalent cancer was rhabdomyosarcoma (RMS), however, it was never primitively cutaneous and rather arose from the subcutis or deep soft tissues and involved the skin secondarily [1]. Although primitively cutaneous RMS is exceedingly rare, the fact that deeper seated RMS may involve the skin prompted researchers to discuss this cancer quite extensively. As expected, the second most prevalent cancer in the same study was dermatofibrosarcoma protuberans. However, this was followed by synovial sarcoma, primitive neuroectodermal tumor, and malignant peripheral nerve sheath tumor (MPNST), all tumors that, in researchers' experience, are very rare in the skin and arise more deeply. This underlines how there may be wide differences between centers, and how soft tissue sarcomas remain an important differential diagnosis. Even more significant for clinical practice, the age range is of the utmost importance when discussing mesenchymal tumors of the skin in children. This was highlighted by Liszewski et al., and also by the French network of cancer registries (FRANCIM) [2]. Soft tissue tumors that are predominate in the neonatal period (infantile fibrosarcoma, kaposiform hemangioendothelioma, melanotic neuroectodermal tumor of infancy, and others) are very different from those that are seen mainly in adolescents. Cutaneous soft tissue tumors of adolescents are close to that of adults, with the particularity of some genetic predispositions or medical contexts that may explain the early presentation of the cancer (e.g., MPNST in children with neurofibromatosis type 1 or with a history of radiation therapy). For practical purposes, the tumors discussed hereafter are classified according to their line of differentiation. In addition, the age ranges of these tumors, which may be useful in clinical practice, are outlined in Figure 1. Similarly, the different genetic anomalies that are currently recognized in these tumors are summarized in Table 1.
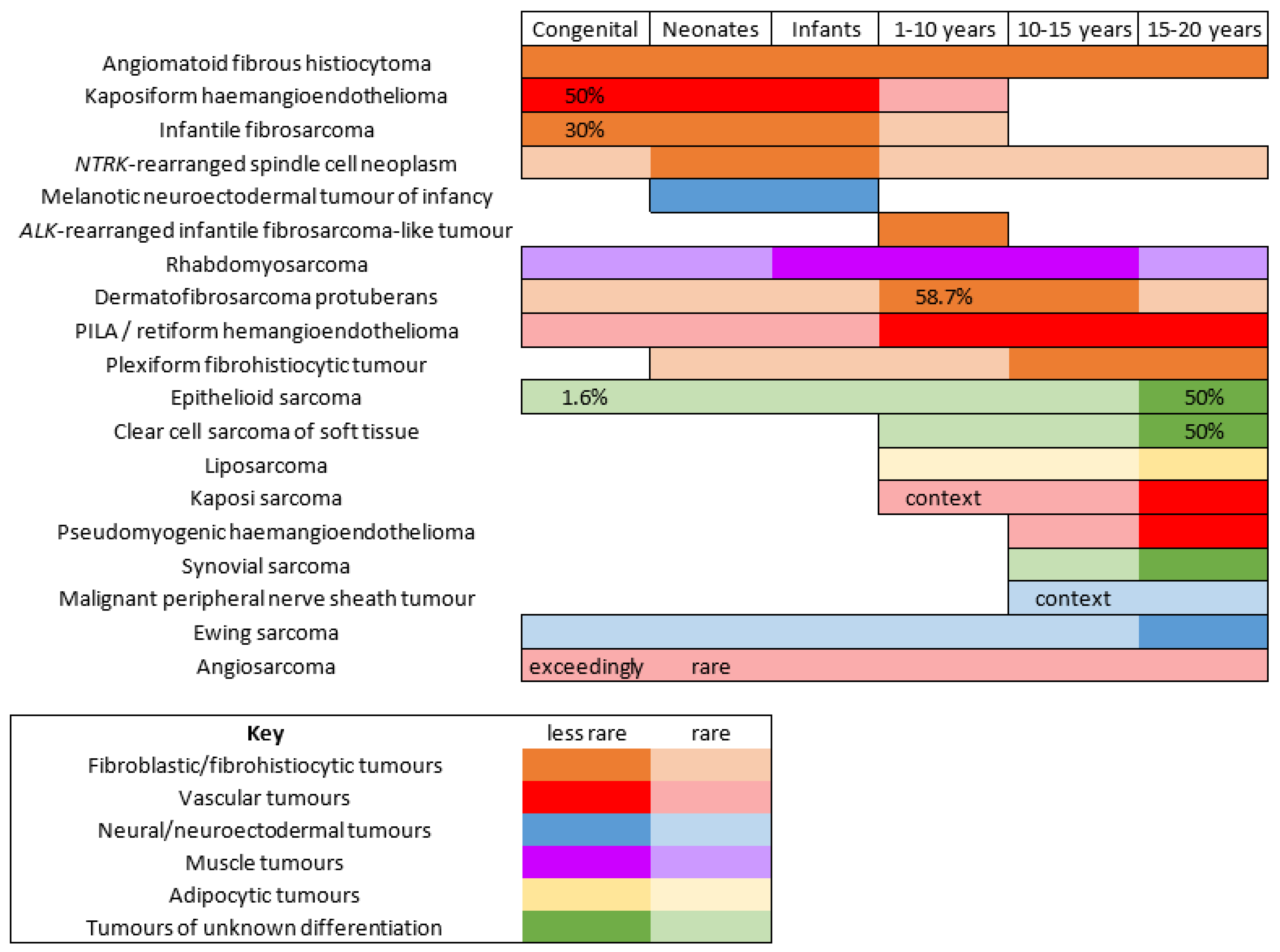
Figure 1. Malignant superficial mesenchymal tumors of the child by age range.
Table 1. Genetic anomalies in pediatric malignant superficial mesenchymal tumors (tumors are listed by order of appearance in the text).
| Tumor | Major Genetic Anomalies | Other Genetic Anomalies |
|---|---|---|
| Angiomatoid fibrous histiocytoma | EWSR1-CREB1 | EWSR1-ATF1, FUS-ATF1 |
| Infantile fibrosarcoma | ETV6-NTRK3 | EML4-NTRK3, RBPMS-NTRK3, SPECC1L-NTRK3 Other gene fusions involving NTRK1, NTRK2, MET, RET, RAF1 |
| NTRK-rearranged spindle cell neoplasms | LMNA-NTRK1 TPM3-NTRK1 TPR-NTRK1 |
SQSTM1-NTRK1 EML4-NTRK3 KHDRBS1-NTRK3 Other gene fusions involving NTRK1,2,3 |
| EWSR1-SMAD3-rearranged fibroblastic tumor | EWSR1-SMAD3 | |
| ALK-rearranged infantile fibrosarcoma-like tumor | ALK-AK5, ALK-ERC1 | Other gene fusions involving ALK? |
| Dermatofibrosarcoma protuberans/giant cell fibroblastoma | COL1A1-PDGFB | COL1A2-PDGFB, COL6A3-PDGFD, EMILIN2-PDGFD |
| Plexiform fibrohistiocytic tumor | Unknown | |
| Kaposiform hemangioendothelioma | - | Mutations in GNA14 |
| Papillary intralymphatic angioendothelioma (PILA)/retiform hemangioendothelioma | - | |
| Pseudomyogenic hemangioendothelioma | SERPINE1-FOSB | ACTB-FOSB, WWTR1-FOSB, CLTC-FOSB, EGFL7-FOSB |
| Angiosarcoma | Complex anomalies (high grade tumor) | |
| Alveolar rhabdomyosarcoma (ARMS) | PAX7-FOXO1, PAX3-FOXO1, or no FOXO1 fusion | |
| Embryonal rhabdomyosarcoma (ERMS) | Unknown | |
| Myxoid liposarcoma | FUS-DDIT3 | EWSR1-DDIT3 |
| Melanotic neuroectodermal tumor of infancy | unknown | BRAFV600E mutation (one case) |
| Ewing sarcoma | EWSR1-FLI1, EWSR1-ERG | EWSR1-ETV1, EWSR1-ETV4, EWSR1-FEV, FUS-ERG, FUS-FEV |
| Malignant peripheral nerve sheath tumor (MPNST) | Unknown | Loss of function mutations in SUZ12 or EED |
| Epithelioid sarcoma | SMARCB1 deletion, mutation or epigenetic silencing | SMARCA4, SMARCC1, SMARCC2 deletions |
| Clear cell sarcoma of soft tissue | EWSR1-ATF1 | EWSR1-CREB1 |
| Synovial sarcoma | SS18-SSX1, SS18-SSX2, SS18-SSX4 | SS18L1-SSX1 |
2. Fibroblastic and Fibrohistiocytic Tumors
2.1. Angiomatoid Fibrous Histiocytoma
Angiomatoid fibrous histiocytoma (AFH) can arise at any age in children and young adults. It affects either sex equally and is located in the limbs or trunk, exceptionally in the head and neck. AFH arises in the subcutaneous tissues, less commonly in deeper soft tissues, and very rarely in the dermis alone. Unusual visceral or deep locations have also been reported: lung, brain, mediastinum, retroperitoneum, ovary, or bone. The tumor presents as a slowly growing, usually non-tender nodule less than 2 cm in diameter, although some tenderness or pain may be present. Systemic symptoms have been described in some cases, for example, weight loss, fever, or anemia, but were not found in all series [3]. Imaging characteristics may be misleading. AFH is isointense to muscle on T1-weighted images, heterogeneously hyperintense on T2-weighted images, and shows variegated enhancement after gadolinium injection. It also often features a pseudo-capsule, cystic areas (sometimes with fluid-fluid levels), and peritumoral edema. The most frequent suggested diagnoses on MRI are those of hemangioma and vascular malformation [4][5].
Microscopically, AFH is well circumscribed, often with a partial fibrous pseudo-capsule. It is made of multiple nodules or lobules with characteristic blood-filled pseudo-angiomatous spaces, often more prominent at the periphery of the tumor (Figure 2A,B). Up to one third of cases may have completely solid morphology without pseudo-angiomatous spaces. A dense lymphocytic infiltrate is also frequent at the periphery of the lesion, often with lymphoid follicular hyperplasia and germinal center formation (Figure 2C). In the nodules, the tumor cells have a fibroblastic-like or histiocytoid morphology (Figure 2D). Mitoses are sparse but atypical forms may be found, with no clinical implication. No histopathologic feature can predict behavior. Immunophenotype is not specific but supportive findings are positivity for desmin or rarely other myoid differentiation markers (SMA, calponin); positivity for EMA; positivity for CD68; and cytoplasmic positivity for CD99 (Figure 2E,F). All vascular markers, cytokeratins, and S100 are negative [3][4].
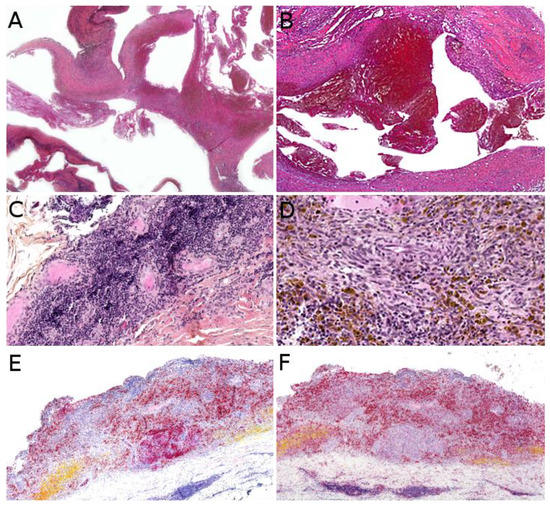
Figure 2. Angiomatoid fibrous histiocytoma: (A) Pseudo-angiomatous spaces (×25); (B) blood-filled pseudo-angiomatous space (×25); (C) dense lymphocytic infiltrate at the periphery of the tumor (×25); (D) histiocytoid tumor cells and hemosiderin deposits (×200); (E) positivity for desmin (×25) and (F) for CD68 (×25).
The most common chromosomal translocation in AFH is t(2;22)(q33;q12) resulting in the EWSR1-CREB1 fusion gene. Other translocations are t(12;22)(q13;q12) (EWSR1-ATF1) and t(12;16)(q13;p11) (FUS-ATF1). CREB1 and ATF1 are both members of the cAMP response element binding protein (CREB) family. EWSR1-CREB1 and EWSR1-ATF1 fusions are also seen in clear cell sarcoma and other non-cutaneous tumors [4].
Most AFH have a benign clinical course, with local recurrence in up to 15% of cases. Metastases to local lymph nodes occur in less than 5% of cases. Distant metastases to the lungs, liver, or brain are exceptional, with only very rare deaths occurring decades after initial presentation. Therefore, the recommended management of AFH is complete local excision, with adjuvant radiation therapy or chemotherapy being reserved only to metastatic or unresectable cases [4][5].
2.2. Infantile Fibrosarcoma
Infantile fibrosarcoma is the most frequent malignant soft tissue tumor of the skin and subcutis before the age of 1 [1]. Around a third of cases are congenital [6]. The median age is 3 to 4 months and 75% of cases occur in the first year of life [7].
About 90% of dermatologic cases are located in the subcutaneous tissue, the remaining cases resulting from the involvement of the subcutis by a more deeply located tumor [1]. There is a slight predilection for the lower extremities but all locations are seen [1][6]. The tumor is typically a painless, rapidly growing nodule or mass, ranging from 1 to >15 cm [8][9]. There is no specific imaging finding [9]. Ultrasound (US) typically shows a well-demarcated, homogeneous mass (54%), but heterogeneity is quite frequent (46%), consisting of hemorrhage foci or more rarely cystic components. On MRI, most tumors have isointense T1 and hyperintense T2 signal intensity with heterogeneous enhancement [9]. When dilated vessels and hemorrhage are prominent, the tumor may resemble a vascular tumor (Figure 3A). Cases with mild thrombocytopenia have been described, which should not be confused with Kasabach–Merritt phenomenon [8][10].
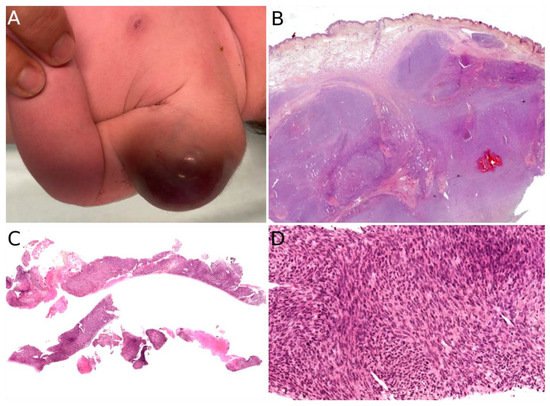
Figure 3. Infantile fibrosarcoma: (A) Large vascularized tumor on the arm of a newborn; (B) large surgical biopsy of a dermo-hypodermal multi-lobulated tumor with high cellularity (×25); (C) core-needle biopsy of another case (×25); (D) cellular, monomorphic spindle cell proliferation arranged in compact sheets or long fascicles (×100).
The diagnosis rests on the pathological analysis of a biopsy (including fine-needle biopsy), which should be as deep as possible. Indeed, a superficial sampling of the tumor may show misleading aspects, such as lipofibromatosis-like [11], hemangioma-like features [10][12] or fat necrosis. This can be avoided by favoring a deep surgical biopsy or fine needle biopsy (Figure 3B,C). More often than not, infantile fibrosarcoma demonstrates a cellular, monomorphic neoplasm made up of ovoid to spindle cells arranged in compact sheets or long fascicles, sometimes assuming the typical herringbone pattern (Figure 3D). Mitoses can be sparse or numerous from one tumor to another and in the same tumor, with no prognostic impact. Necrosis is possible and has no prognostic implication either. The tumor vessels may be numerous. In such cases, the tumor might even be misdiagnosed as a vascular anomaly. The immunophenotype is not specific. The tumor may show variable and focal expression of SMA, S100, CD34, and rarely desmin (or none) [13]. Expression of pan-TRK (neurotrophic tyrosine receptor kinase) is helpful for screening tumors with a NTRK gene rearrangement [14]. Indeed, most infantile fibrosarcomas exhibit an ETV6-NTRK3 gene fusion resulting from the t(12;15)(p13;q25) chromosomal translocation. Other NTRK3 fusion partners include EML4 [15], RBPMS [16], or SPECC1L [17]. Other tyrosine kinase genes are involved in fusions, including NTRK1, NTRK2, RET, MET, and RAF1, with a variety of partners [16][18][19][20]. BRAF fusions, complex deletions, and point mutations have also been evidenced, inducing constitutive activation of the RAF-MEK-ERK pathway [18][19]. Interestingly, other spindle cell tumors harboring RAF1, BRAF, and NTRK1/2 fusions and further characterized by S100 and CD34 co-expression (discussed hereafter, see Section 2.3), demonstrate a close proximity to some infantile fibrosarcomas by RNA unsupervised hierarchical clustering analysis [21]. This suggests a common pathogenesis between these tumors, or even a common spectrum of low-grade spindle cell tumors with activation of the RAF-MEK-ERK pathway.
Infantile fibrosarcoma has an overall favorable prognosis. It is locally aggressive and very rarely metastasizes. Metastases to the central nervous system, the lungs, and lymph nodes have been reported a few times [6][22][23][24][25][26][27]. There are rare reports of metastases to the heart, bones, liver, and adrenal glands [24][28][29]. The standard treatment includes complete surgical excision (as often as possible), associated with chemotherapy when necessary (incomplete or impossible excision). Targeted tyrosine kinase inhibitor therapies (larotrectinib, entrectinib, selitrectinib, and repotrectinib) seem to be promising alternatives [27][30][31][32].
2.3. NTRK-Rearranged Spindle Cell Neoplasms
This group of tumors is emerging as an entity based on molecular grounds. More than 55% of pediatric cases occur in the first year of life [16][20]. Some cases are congenital [33]. However, all ages can be affected [21][33][34].
Most NTRK-rearranged spindle cell neoplasms occur in the superficial soft tissues of the extremities, other locations being the trunk and scalp. They present as a subcutaneous nodule or mass ranging from 1 to 6 cm in greatest dimension [35]. Imaging characteristics of these tumors have never been studied; however, it is likely that they overlap with infantile fibrosarcoma, lipofibromatosis, and malignant peripheral nerve sheath tumor (MPNST), and thus, are not specific.
Indeed, NTRK-rearranged spindle cell neoplasms include a wide variety of morphologies with tumors described as infantile fibrosarcoma-like, inflammatory myofibroblastic tumor-like (IMT-like), lipofibromatosis-like neural tumor (LPF-NT), and MPNST-like [36]. Microscopically, the most consistent features are an infiltrative growth pattern within subcutaneous fat, dense spindle cells haphazardly arranged or in fascicles, elongated nuclei with mild atypia and hyperchromasia, inconspicuous nucleoli, and low mitotic activity (Figure 4A,B) [16][21][33][34][35][36]. A prominent inflammatory infiltrate has been described in IMT-like cases [16][21][34][37]. The clue to the diagnosis is the co-expression of CD34 and S100 by immunohistochemistry. The expression may be focal and vary among cases [34][35]. The retained expression of H3K27me3 can help in the differential diagnosis with a MPNST, a differential that may be considered only in adolescents. Screening with an anti-NTRK1 or panTRK antibody is also helpful (Figure 4C). The definitive diagnosis is made by molecular testing for NTRK1 gene fusions (in most cases, Figure 4D). The fusion partners include LMNA, TPM3, TPR, and SQSTM1 [16][34]. Other gene rearrangements have been identified in children, involving NTRK3 with EML4 or KHDRBS1 as partners [21][38][39]. Researchers have already discussed the possible relation between NTRK-rearranged spindle cell neoplasms and infantile fibrosarcoma (see Section 2.2).
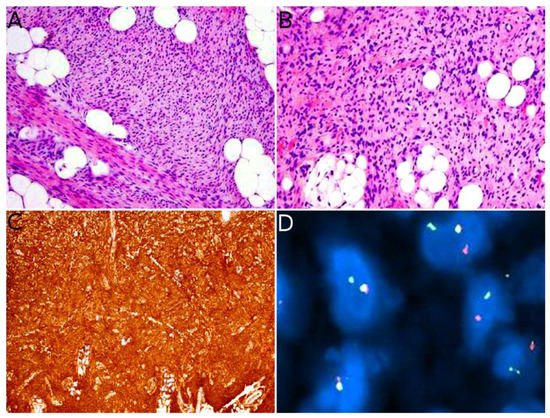
Figure 4. NTRK-rearranged spindle cell neoplasm: (A) Infiltrative growth pattern within the subcutaneous fat, with spindle cells haphazardly arranged or in fascicles (×100); (B) mild atypia and hyperchromasia (×100); (C) diffuse positivity for pan-TRK (×25); (D) fusion signals involving NTRK on FISH.
In children, for those tumors located in superficial soft tissues, the prognosis is favorable in most instances. The clinical course is marked by local recurrence, sometimes repeated [16][21]. Metastases are rare but have been recorded in one series in 3 children (out of 22) aged 0 to 5 years. The tumors were initially located in the foot, shoulder, and chest wall, respectively, with positive margins at initial resection in wo cases (third case unknown). Metastases were to the lung in all cases. Two children were alive after chemotherapy and one died of disease, in spite of the chemotherapy [16].
The treatment is, first and foremost, surgical. When excision cannot be complete, chemotherapy may be beneficial [16]. Similar to infantile fibrosarcoma, anti-TRK tyrosine kinase inhibitors (larotrectinib, entrectinib, and more recently selitrectinib and repotrectinib) will probably become a standard in the treatment of these tumors.
3. Vascular Tumors
3.1. Kaposiform Hemangioendothelioma
The incidence of kaposiform hemangioendothelioma (KHE) is probably under evaluated. It has been suggested that small asymptomatic or atypical KHE may be misdiagnosed as variants of other vascular tumors [40]. Although the exact incidence and prevalence of this vascular tumor are not known, it is clear that there is a peak within the first year of life. Around 50% of superficial cases are congenital [40]. It is admitted now that KHE and tufted angioma belong to the same spectrum and that they share the same biology and probably the same pathogenesis [41][42]. Actually, some specialists argue that KHE might only be a florid presentation of tufted angioma, and that the adverse clinical course of this tumor is purely dependent on the occurrence of Kasabach–Merritt phenomenon (KMP) or vital organ compression. For this reason, KHE may well be benign, the morbidity and mortality being linked to secondary complications. Researchers decided to include KHE in this entry anyway, but this decision is debatable and it will not be discussed extensively.
KHE usually involves the deep dermis and subcutis, but deeper lesions are possible, with no cutaneous signs in about 12% of cases [40][42]. There is a slightly higher prevalence in the limbs, but all sites are possible. Most of the time, the tumor is unique, and can adopt many clinical aspects: erythematous sometimes purple; papules, nodules or plaques or an indurated mass; with varying degrees of infiltration [42]. It is a big, rapidly growing tumor, 3 to 27 cm in greater dimension [43]. One of the most severe complications of KHE, also seen in tufted angioma, is the KMP, a life-threatening consumptive coagulopathy with severe thrombocytopenia. It occurs in 42 to 71% of cases [44][45][46]. There is a higher frequency of KMP in congenital, large KHE, especially above 8 cm [47]. In cases of KMP, the KHE lesions appear purpuric, congestive, and painful [48]. Spontaneous hemorrhage is rare but any invasive procedure (biopsy, excision), trauma, or ulceration may lead to significant bleeding. Other complications of KHE include musculoskeletal disorders due to tumor infiltration, lymphedema, and compression of vital structures (e.g., airway obstruction in a KHE of the neck) [42].
US reveals an ill-defined, heterogeneous lesion, most often hyperechoic [43]. On color Doppler US, the tumor is hypervascular. MRI reveals a heterogeneous, hyperintense appearance on T2-weighted images, with heterogeneous, generally intense enhancement [43]. Overall, the imaging findings lack specificity and are characterized by a wide range of aspects regarding the degree of infiltration, signal intensities, and enhancement patterns [43].
The gold standard for diagnosis is the biopsy. KHE is a deep dermal and subcutaneous, infiltrating proliferation of spindle endothelial cells arranged in nodules (Figure 5A). The tumor can adopt a “cannonball” pattern, especially in the dermis, similar to a tufted angioma, but the nodules are larger, less defined, and coalescent. The tumor is also more infiltrative. Mitoses are rare and not abnormal (Figure 5B) [42][44][49]. Most of the spindle cells are positive for lymphatic markers podoplanin, LYVE1, and PROX1 (Figure 5C) [44][50].
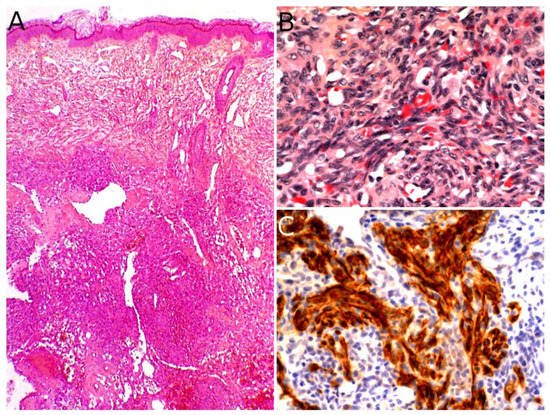
Figure 5. Kaposiform hemangioendothelioma: (A) Deep dermal and subcutaneous, infiltrating proliferation of spindle cells arranged in nodules (×25); (B) spindle endothelial cells with no mitoses (×200); (C) positivity for the lymphatic marker podoplanin in the spindle cells (×200).
Similar to other vascular anomalies, numerous mutations involving G proteins have been found in KHE and tufted angioma, especially in the GNA14 gene, a paralogue of GNAQ [51]. These genes encode the Gα subunit that, along with Gβ and Gγ in a heterotrimer, binds G protein-coupled receptors.
The prognosis depends on the size and location of the tumor. Large, deep KHE tumors are associated with higher morbidity and mortality due to tumor infiltration, compression of vital organs, and KMP [42]. Treatment with corticosteroids and/or vincristine has been recommended but is based on expert opinion and lack a well-designed clinical study [48]. Wide surgical excision can be curative but is rarely feasible. For a decade now, sirolimus has been emerging as a treatment option. The first report of a successful treatment in a child was published in 2010 [52]. Some of the concerns with sirolimus have been the occurrence of adverse events and the issue of the right dose in the pediatric population [53]. Recently, recommendations for the initial dose in children were proposed. Due to sirolimus being metabolized by cytochrome P450 3A (CYP3A), the initial dose depends both on the weight and the CYP3A5 genotype [54]. A recent prospective study of sirolimus for complicated vascular anomalies underlined the importance of closely monitoring possible adverse events, while confirming the effectiveness of the treatment [55]. In the study, the most frequent adverse events (>20% of cases) were mucositis, upper respiratory infections, and nausea/vomiting.
3.2. Papillary Intralymphatic Angioendothelioma (PILA)/Retiform Hemangioendothelioma
Papillary intralymphatic angioendothelioma (PILA, also known as Dabska tumor) and retiform hemangioendothelioma belong to the same spectrum. These two rare tumors are lymphatic in origin. Cases with overlapping features of PILA and retiform hemangioendothelioma have been described, which is the reason why some authors have suggested to include both tumors under the name hobnail hemangioendothelioma [56]. However, pure forms of retiform hemangioendothelioma are very rare in children; PILA or mixed tumors are much more frequent. PILA usually involves the dermis and subcutaneous tissues of the head and neck in children and adolescents, whereas retiform hemangioendothelioma more often involves the upper and lower limbs of young adults and adolescents, with some reported cases in the head or trunk [57]. Similar to Masson’s tumor, PILA has been reported to arise in a pre-existing vascular malformation, but also on chronic lymphedema and in intramuscular hemangiomas. There are a few reports of a retiform hemangioendothelioma arising on a pre-existing lymphatic malformation [58].
PILA and retiform hemangioendothelioma both present as a slowly growing, firm, solitary nodule or mass, more rarely plaque; all nonspecific findings, which explains the diagnosis not being raised by dermatologists or pediatricians. To the best of researchers' knowledge, there has been no study of the imaging characteristics of these tumors in the English literature.
Microscopically, PILA consists of vessels of various sizes, more or less dilated, containing papillary tufts protruding from the vessel wall and sometimes occluding the lumen. The papillae are centered by a hyalinized core and lined by hobnail endothelial cells with large hyperchromatic nuclei located towards the luminal pole of the cell (Figure 6A). Mitoses are sparse. Retiform hemangioendothelioma involves the whole dermis and often infiltrates the subcutaneous tissue. It is made of elongated, anastomosing vessels reminiscent of the architecture of the rete testis. In both PILA and retiform hemangioendothelioma, there is often a prominent lymphocytic infiltrate. The immunophenotype is similar in both tumors, with positivity for CD31 and CD34 in the endothelial cells. Prox1 and podoplanin (D2-40) are positive in most cases of PILA (Figure 6B) [59][60][61][62]. Of note, rare cases of composite hemangioendothelioma have been described in adolescents, consisting of areas of retiform hemangioendothelioma and solid areas with a vaguely neuroendocrine morphology. These cases showed no recurrence nor metastasis in children, however, both of these have been reported in adults, especially bone and lung metastases [60]. PILA has intermediate malignant potential with possible local recurrences and exceptional lymph node metastases. Lung metastases have been described in at least one case [63]. Retiform hemangioendothelioma shares a similar clinical course.
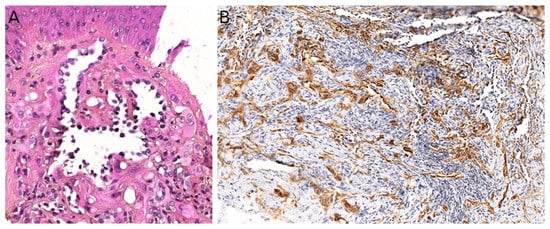
Figure 6. Papillary intralymphatic angioendothelioma: (A) Vascular proliferation with papillary tufts protruding from the vessel wall, lined by hobnail endothelial cells with large hyperchromatic nuclei (×200); (B) partial positivity for the lymphatic marker podoplanin (×100).
The treatment of choice of PILA and retiform hemangioendothelioma is complete surgical resection, in order to avoid recurrences. However, clear surgical margins may be hard to achieve in some cases of retiform hemangioendothelioma, due to its infiltrative growth pattern. In such cases, MMS may be useful, as reported in a 11-year-old girl with a tumor of the finger [64]. In unresectable cases, association of radiation therapy and chemotherapy with cisplatin might be an option, as reported in an adult [65].
3.3. Pseudomyogenic Haemangioendothelioma
Pseudomyogenic hemangioendothelioma (PMHE) is a rare vascular tumor of intermediate malignant potential. It affects adolescents and young adults, with a mean age at diagnosis close to 30 years and a strong male predominance (sex ratio about 4:1) [66][67]. The most frequently involved sites are the extremities, especially the lower limbs, followed by the trunk and upper limbs, but the tumor has been described in other locations. PMHE arises mainly in the soft tissues but may occur in the dermis, muscle, or bone [66]. A characteristic feature of this tumor is the frequent involvement of different tissue planes, resulting in a mass or deep-seated nodule, with pain in about half of cases. Multicentric presentation is also common [66][68][69]. On MRI, PMHE is hypointense on T1-weighted images and hyperintense on T2-weighted and stir-weighted images. It has been suggested that positron emission tomography scan (PET-scan) might be helpful to reveal possible occult deep lesions [8].
Grossly, PMHE is usually made of multifocal, ill-defined, 1 to 3 cm large, white- or brown-colored nodules [69]. Microscopically, PMHE has an infiltrative pattern of growth (Figure 7A). It is made of sheets and fascicles of epithelioid cells or plump spindle cells with a deeply eosinophilic cytoplasm which can be reminiscent of rhabdomyoblasts (Figure 7B). The stroma is loose, rarely myxoid, and contains numerous neutrophils in about half of cases (Figure 7C) [69]. Similar to dermatofibroma, epidermal hyperplasia may be present above the tumor [70]. Intravascular invasion has been described but does not seem to have any prognostic impact [71]. PMHE co-expresses cytokeratins and endothelial markers. It is diffusely positive for cytokeratin AE1/AE3 and ERG. CD31 is positive in about half of cases. SMA may be expressed focally in some cases. Interestingly, there is diffuse nuclear positivity for FOSB in 96% of cases, a very helpful finding since FOSB is expressed in only a couple of other vascular tumors: epithelioid hemangioma (54% of cases), which is not a histological differential diagnosis of PMHE, and some rare cases of epithelioid angiosarcoma (5%), spindle-cell angiosarcoma (10%), or epithelioid hemangioendothelioma (5%) [68]. One of the main differential diagnoses of PMHE is epithelioid sarcoma. However, INI1 is normally expressed in PMHE.
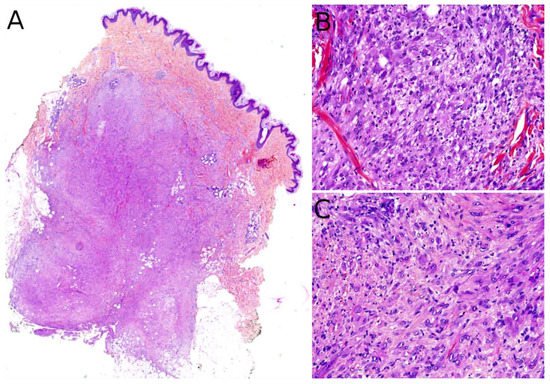
Figure 7. Pseudomyogenic hemangioendothelioma: (A) Dense cellular proliferation with an infiltrating pattern of growth (×25); (B) sheets of epithelioid cells or plump spindle cells with a deeply eosinophilic cytoplasm (×100); (C) rhabdomyoblast-like cells with moderate nuclear atypia and some neutrophils in the stroma (×200).
FOSB positivity is related to the presence of the t(7;19)(q22;q13) translocation responsible for a SERPINE1-FOSB gene fusion in the majority of PMHE [66][72]. Recently, other partners for FOSB have been found in some cases of PMHE: the beta-actin gene, responsible for the ACTB-FOSB gene fusion [73][74]; the WW domain-containing transcription regulator 1 gene, responsible for the WWTR1-FOSB gene fusion in an isolated case [75]; and more recently, the clathrin heavy chain gene, responsible for the CLTC-FOSB gene fusion [76]; and the epidermal growth factor-like 7 gene, responsible for the EGFL7-FOSB gene fusion [77]. It remains to be determined if these various gene fusions are associated with different clinicopathological features. To date, there is no targeted therapy for FOSB gene fusions.
The clinical course is characterized by local recurrences in about 60% of cases, often multiple and with possible additional nodules. Regional lymph node metastasis has been described in only one patient, and distant metastases in three patients; only two patients died of disease [71][78][79]. The treatment of choice is complete surgical excision. Some reports of clinical improvement following treatment with sirolimus or everolimus (anti-mTOR) are hopeful for non resectable cases [80][81].
3.4. Angiosarcoma
Angiosarcoma is extremely rare in children, therefore, it is impossible to draw reliable epidemiological or clinical data on angiosarcoma in children and in the skin. In children, it is mostly seen in association with genetic conditions such as xeroderma pigmentosum, or Aicardi syndrome, or after radiation therapy [49]. Therefore, angiosarcomas are almost always seen in older children and adolescents. Congenital or infantile cases are exceedingly rare. Angiosarcomas in children seem to predominate in girls and in the lower extremities, but these observations should be tempered since they are based on a very small series of 10 cases [82]. The tumor is often a rapidly enlarging nodule or mass, sometimes ulcerated (Figure 8A). It may be centered in the dermis, in the subcutaneous tissue, or in both. The tumor is made of a branching network of vessels admixed with more solid areas made of epithelioid cells (Figure 8B). Indeed, the epithelioid variant is particularly frequent in children (90%). Nuclear atypia is often prominent, at least in some areas. Mitotic activity is high in most cases (Figure 8G), but may be low. Necrosis is possible. Immunohistochemistry reveals a positivity for at least one endothelial marker, with a better sensitivity of CD31 and ERG as compared with CD34 (Figure 8C–E) [82]. Podoplanin is commonly positive. HHV8 is negative. Aberrant cytokeratin positivity is possible (Figure 8F). Wide surgical excision is the treatment of choice. Adjuvant chemotherapy may be useful but reported cases are too few to draw any recommendation and each case should be discussed collegially.
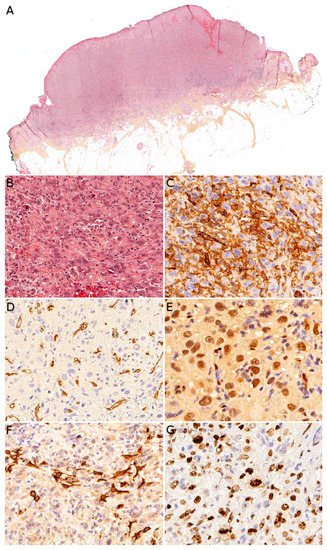
Figure 8. Angiosarcoma: (A) Ulcerated nodule (×25); (B) atypical epithelioid cells with numerous mitoses and a tripolar mitosis (center of the panel) (×200); (C) strong CD31 positivity (×200); (D) partial CD34 positivity on small dystrophic vascular structures (×200); (E) strong nuclear ERG positivity (×200); (F) partial positivity for the cytokeratin marker AE1/AE3 (×200); (G) high proliferation index and multiple mitoses (Ki67 ×200).
4. Muscular Tumors: Rhabdomyosarcomas
Primary cutaneous rhabdomyosarcoma (RMS) is a rare tumor, with about 55 cases reported in the literature [83][84]. It occurs mainly in the head and neck, and there seem to be no sex predominance [83]. The age range is wide with rare congenital/neonatal cases, but also cases in older children and adolescents. Importantly, the two main histological subtypes of RMS, embryonal RMS (ERMS) and alveolar RMS (ARMS), have distinct clinical and histological characteristics. Therefore, they are discussed separately hereafter. Contrary to non-cutaneous RMS, where ERMS account for nearly 75% of all RMS, primarily cutaneous RMS are more often of the alveolar subtype [83], even in congenital/neonatal cases. Indeed, in the skin and superficial soft tissues, after a short review of the literature, researchers found that congenital/neonatal ARMS were twice as often represented as congenital/neonatal ERMS (20 ARMS, 9 ERMS) [85][86][87][88][89][90]. In children, tumors with extensive pleomorphism should be considered anaplastic ERMS and not pleomorphic RMS [91]. To the best of researchers' knowledge, no pediatric primary cutaneous spindle-cell/sclerosing RMS has been reported in the literature. Still, it seems important to keep in mind this differential diagnosis when confronted with a spindle-cell tumor, since this RMS subtype is associated with a better prognosis and indolent clinical course. NCOA2 and VGLL2 rearrangements are the most common in spindle-cell/sclerosing RMS in children and should be sought for establishing this diagnosis [92].
4.1. Alveolar Rhabdomyosarcoma (ARMS)
Congenital/neonatal ARMS has a slight female predominance [88][93]. Although very rare in this age group, congenital/neonatal ARMS has other particularities which are worth mentioning: the tumor occurs mainly in the limbs, followed by the head and neck, and the trunk [88]; according to a recent review, cutaneous and subcutaneous locations are most often metastatic sites [88][94] however, at least four reported cases of congenital ARMS were primarily cutaneous [86][89][93][95]. All ages considered, ARMS is usually an erythematous mass or nodule, initially misdiagnosed as a hemangioma, but often rapidly growing, ranging from 4 to 12 cm in greater diameter [86][89][95]. When congenital/neonatal, it may be multiple, even in primarily cutaneous cases. In older children, the lesion is unique (Figure 9).
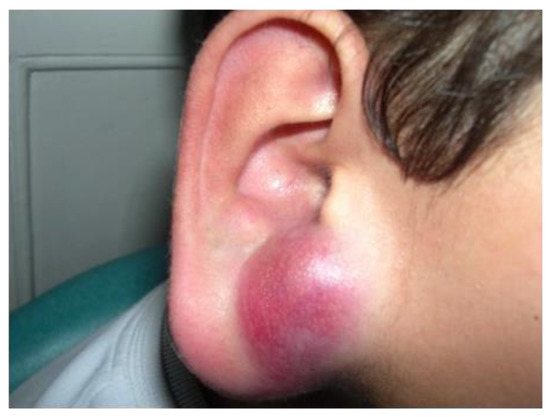
Figure 9. Rhabdomyosarcoma of the ear in a young boy: solitary erythematous nodule.
Microscopically, primarily cutaneous ARMS is similar to ARMS in other locations; it is composed of nests of undifferentiated small round cells with a high N/C ratio and a variable proportion of tumor cells with a moderate eosinophilic cytoplasm. True differentiated rhabdomyoblasts are rare. In the center of the nests, the cells show discohesion with a tendency to disaggregate, leading to the alveolar pattern. Multinucleated tumor cells are a rather common finding. Mitoses can be sparse or more prominent and necrosis is possible [85][86][90]. Tumor cells express skeletal muscle markers: desmin, myogenin (Myf4), and/or MyoD1.
The overall prognosis of congenital ARMS is poor, with about 75% of deaths in the review by Russo et al. However, about 75% of cases in this review also had metastases, with 41% of central nervous system metastases [88]. It is possible that primary cutaneous cases of congenital ARMS have a better prognosis, as reported [86][89][93][95]. Importantly, the prognosis of RMS, whatever the age of the child, has been linked to its genetic anomalies; patients with FOXO1 fusion positive RMS having a worse outcome than patients with FOXO1 fusion negative RMS, and the PAX7-FOXO1 fusion has a better prognosis than the PAX3-FOXO1 fusion [96]. Since 70% of ARMS show either one of the FOXO1 fusions, it makes sense that ARMS should have a poorer prognosis than ERMS. However, in congenital/neonatal ARMS cases, only three FOXO1 fusions have been reported, to researchers' knowledge, whereas the majority of cases seem to be negative for FOXO1 fusions [85][87][88][93]. This suggests a possible different pathogenesis for congenital/neonatal ARMS. For instance, some cases have been linked to Beckwith–Wiedemann syndrome and the tumors seem to arise from silencing of the CDKN1C gene through epigenetic mechanisms in this syndrome [87][93].
4.2. Embryonal Rhabdomyosarcoma (ERMS)
Congenital/neonatal ERMS mainly occurs in the head and neck, followed by the trunk. The male/female ratio is about two [88]. Only nine cases of congenital/neonatal ERMS of the skin or superficial soft tissues have been reported, to the best of researchers' knowledge, with fragmentary data, which makes it difficult to draw meaningful conclusions [85][97][98][99]. The locations include the cheek, scalp, neck, hand, foot, and lower back. All ages considered, the tumor is firm, ranges from 3 to 15 cm in greater dimension, may be ulcerated if large, with possible associated lymphadenopathy [85][97][99].
Microscopically, ERMS shows alternating loose and dense cellularity in a variably myxoid stroma. The tumor cells range from stellate to small round undifferentiated to spindle. Unlike ARMS, myogenin expression is more variable.
The prognosis is deemed to be better than that of ARMS, however, in congenital/neonatal cases at least three out of nine ERMS cases were fatal after only a few months: the tumor recurred in one case in spite of surgery, chemotherapy (specific RMS protocol), and radiation therapy; the tumor recurred in one other case after surgery; and one child did not receive any treatment [85][97][99]. The outcome was not known or favorable in other cases. Interestingly, one case exhibited a t(2;8)(q35;q13) translocation. When it was performed, cytogenetic techniques never evidenced any FOXO1 fusion. One case was associated with Apert syndrome (craniosynostosis, midface hypoplasia, and syndactyly of the hands and feet), a syndrome driven by mutations in FGFR2. In a recent study of germline predisposition variants in pediatric RMS, the authors found some cancer predisposition gene variants in ERMS occurring before 1 year of age, but not in ARMS in this age group. The exact age and location of the tumors are not discussed, but the most frequently involved genes have been NF1 (neurofibromatosis type 1), TP53 (Li–Fraumeni syndrome), HRAS (Costello syndrome), CBL (Noonan syndrome), and BRCA2 [100]. It is worth mentioning that some cases of congenital/neonatal ERMS may arise on congenital melanocytic nevi [99].
4.3. Rhabdomyosarcoma Risk Group Stratification and Treatment
The outcome and treatment of RMS are dependent on risk groups. Of note, the perineal location seems to have prognostic significance as well, with a poorer prognosis in those cases [84].
The stratification of patients into risk groups at diagnosis is essential. The definition of risk groups is different in Europe and in North America, with different treatment strategies. In Europe, four risk groups (low, standard, high, and very high) have been defined by the European Paediatric Soft Tissue Sarcoma Group (EpSSG), based on the Intergroup Rhabdomyosarcoma Study Group (IRS) post-surgical stage, the age, the tumor size, the histopathological subtype, the site of the primary tumor, and the nodal stage. According to the EpSSG, surgical resection follows induction chemotherapy when any residual mass can be completely excised (R0/R1) without causing a significant organ or functional impairment. For some standard risk cases, radiation therapy may be avoided if the resection is complete (R0). For more details on the management of RMS and the differences between North American and European approaches, the reader is referred to the review by Yechieli et al. [101].
References
- Liszewski, W.; Maguiness, S.; Greengard, E.; Boull, C. The Incidence of Pediatric Malignant Soft Tissue Tumors of the Skin and Subcutaneous Tissue. Pediatr. Dermatol. 2018, 35, e427–e429.
- Amadeo, B.; Penel, N.; Coindre, J.-M.; Ray-Coquard, I.; Ligier, K.; Delafosse, P.; Bouvier, A.-M.; Plouvier, S.; Gallet, J.; Lacourt, A.; et al. Incidence and Time Trends of Sarcoma (2000–2013): Results from the French Network of Cancer Registries (FRANCIM). BMC Cancer 2020, 20, 190.
- Bohelay, G.; Kluger, N.; Battistella, M.; Biaggi-Frassati, A.; Plantier, F.; Harraudeau, A.; Avril, M.-F.; Pedeutour, F.; Fraitag, S. Angiomatoid fibrous histiocytoma in children: 6 cases. Ann. Dermatol. Venereol. 2015, 142, 541–548.
- Thway, K.; Fisher, C. Angiomatoid Fibrous Histiocytoma: The Current Status of Pathology and Genetics. Arch. Pathol. Lab. Med. 2015, 139, 674–682.
- Saito, K.; Kobayashi, E.; Yoshida, A.; Araki, Y.; Kubota, D.; Tanzawa, Y.; Kawai, A.; Yanagawa, T.; Takagishi, K.; Chuman, H. Angiomatoid Fibrous Histiocytoma: A Series of Seven Cases Including Genetically Confirmed Aggressive Cases and a Literature Review. BMC Musculoskelet. Disord. 2017, 18, 31.
- Chung, E.B.; Enzinger, F.M. Infantile Fibrosarcoma. Cancer 1976, 38, 729–739.
- Orbach, D.; Rey, A.; Cecchetto, G.; Oberlin, O.; Casanova, M.; Thebaud, E.; Scopinaro, M.; Bisogno, G.; Carli, M.; Ferrari, A. Infantile Fibrosarcoma: Management Based on the European Experience. J. Clin. Oncol. Off. J. Am. Soc. Clin. Oncol. 2010, 28, 318–323.
- WHO Classification of Tumours Editorial Board. Soft Tissue and Bone Tumours; World Health Organization: Geneva, Switzerland, 2020; ISBN 978-92-832-4502-5.
- Ainsworth, K.E.; Chavhan, G.B.; Gupta, A.A.; Hopyan, S.; Taylor, G. Congenital Infantile Fibrosarcoma: Review of Imaging Features. Pediatr. Radiol. 2014, 44, 1124–1129.
- Yan, A.C.; Chamlin, S.L.; Liang, M.G.; Hoffman, B.; Attiyeh, E.F.; Chang, B.; Honig, P.J. Congenital Infantile Fibrosarcoma: A Masquerader of Ulcerated Hemangioma. Pediatr. Dermatol. 2006, 23, 330–334.
- Swiadkiewicz, R.; Galmiche, L.; Belhous, K.; Boccara, O.; Fraitag, S.; Pedeutour, F.; Dadone, B.; Buis, J.; Picard, A.; Orbach, D.; et al. Congenital Infantile Fibrosarcoma Associated with a Lipofibromatosis-Like Component: One Train May Be Hiding Another. Am. J. Dermatopathol. 2017, 39, 463–467.
- Enos, T.; Hosler, G.A.; Uddin, N.; Mir, A. Congenital Infantile Fibrosarcoma Mimicking a Cutaneous Vascular Lesion: A Case Report and Review of the Literature. J. Cutan. Pathol. 2017, 44, 193–200.
- Coffin, C.M.; Alaggio, R. Fibroblastic and Myofibroblastic Tumors in Children and Adolescents. Pediatr. Dev. Pathol. 2012, 15, 127–180.
- Hung, Y.P.; Fletcher, C.D.M.; Hornick, J.L. Evaluation of Pan-TRK Immunohistochemistry in Infantile Fibrosarcoma, Lipofibromatosis-like Neural Tumour and Histological Mimics. Histopathology 2018, 73, 634–644.
- Church, A.J.; Calicchio, M.L.; Nardi, V.; Skalova, A.; Pinto, A.; Dillon, D.A.; Gomez-Fernandez, C.R.; Manoj, N.; Haimes, J.D.; Stahl, J.A.; et al. Recurrent EML4-NTRK3 Fusions in Infantile Fibrosarcoma and Congenital Mesoblastic Nephroma Suggest a Revised Testing Strategy. Mod. Pathol. 2018, 31, 463–473.
- Davis, J.L.; Lockwood, C.M.; Stohr, B.; Boecking, C.; Al-Ibraheemi, A.; DuBois, S.G.; Vargas, S.O.; Black, J.O.; Cox, M.C.; Luquette, M.; et al. Expanding the Spectrum of Pediatric NTRK-Rearranged Mesenchymal Tumors. Am. J. Surg. Pathol. 2019, 43, 435–445.
- Zhao, X.; Kotch, C.; Fox, E.; Surrey, L.F.; Wertheim, G.B.; Baloch, Z.W.; Lin, F.; Pillai, V.; Luo, M.; Kreiger, P.A.; et al. NTRK Fusions Identified in Pediatric Tumors: The Frequency, Fusion Partners, and Clinical Outcome. JCO Precis. Oncol. 2021, 1, 204–214.
- Kao, Y.-C.; Fletcher, C.D.M.; Alaggio, R.; Wexler, L.; Zhang, L.; Sung, Y.-S.; Orhan, D.; Chang, W.-C.; Swanson, D.; Dickson, B.C.; et al. Recurrent BRAF Gene Fusions in a Subset of Pediatric Spindle Cell Sarcomas: Expanding the Genetic Spectrum of Tumors with Overlapping Features with Infantile Fibrosarcoma. Am. J. Surg. Pathol. 2018, 42, 28–38.
- Wegert, J.; Vokuhl, C.; Collord, G.; Velasco-Herrera, M.D.C.; Farndon, S.J.; Guzzo, C.; Jorgensen, M.; Anderson, J.; Slater, O.; Duncan, C.; et al. Recurrent Intragenic Rearrangements of EGFR and BRAF in Soft Tissue Tumors of Infants. Nat. Commun. 2018, 9, 2378.
- Davis, J.L.; Vargas, S.O.; Rudzinski, E.R.; Marti, J.M.L.; Janeway, K.; Forrest, S.; Winsnes, K.; Pinto, N.; Yang, S.E.; VanSandt, M.; et al. Recurrent RET Gene Fusions in Paediatric Spindle Mesenchymal Neoplasms. Histopathology 2020, 76, 1032–1041.
- Suurmeijer, A.J.H.; Dickson, B.C.; Swanson, D.; Zhang, L.; Sung, Y.-S.; Cotzia, P.; Fletcher, C.D.M.; Antonescu, C.R. A Novel Group of Spindle Cell Tumors Defined by S100 and CD34 Co-Expression Shows Recurrent Fusions Involving RAF1, BRAF, and NTRK1/2 Genes. Genes Chromosomes Cancer 2018, 57, 611–621.
- Shankwiler, R.A.; Athey, P.A.; Lamki, N. Aggressive Infantile Fibromatosis. Pulmonary Metastases Documented by Plain Film and Computed Tomography. Clin. Imaging 1989, 13, 127–129.
- Ramphal, R.; Manson, D.; Viero, S.; Zielenska, M.; Gerstle, T.; Pappo, A. Retroperitoneal Infantile Fibrosarcoma: Clinical, Molecular, and Therapeutic Aspects of an Unusual Tumor. Pediatr. Hematol. Oncol. 2003, 20, 635–642.
- Coulibaly, B.; Barel, E.; Soulier, M.; Bouvier, C.; Chaumoitre, K.; D’Ercole, C.; Liprandi, A. Prenatal Diagnosis of Infantile Fibrosarcoma with Diffuse Metastases. Prenat. Diagn. 2008, 28, 773–775.
- Parida, L.; Fernandez-Pineda, I.; Uffman, J.K.; Davidoff, A.M.; Krasin, M.J.; Pappo, A.; Rao, B.N. Clinical Management of Infantile Fibrosarcoma: A Retrospective Single-Institution Review. Pediatr. Surg. Int. 2013, 29, 703–708.
- Lo, C.-H.; Cheng, S.-N.; Lin, K.-T.; Jen, Y.-M. Successful Treatment of Infantile Fibrosarcoma Spinal Metastasis by Chemotherapy and Stereotactic Hypofractionated Radiotherapy. J. Korean Neurosurg. Soc. 2013, 54, 528–531.
- Orbach, D.; Sparber-Sauer, M.; Laetsch, T.W.; Minard-Colin, V.; Bielack, S.S.; Casanova, M.; Corradini, N.; Koscielniak, E.; Scheer, M.; Hettmer, S.; et al. Spotlight on the Treatment of Infantile Fibrosarcoma in the Era of Neurotrophic Tropomyosin Receptor Kinase Inhibitors: International Consensus and Remaining Controversies. Eur. J. Cancer 2020, 137, 183–192.
- Shah, N.N.; Price, M.R.; Loeb, D.M. Cardiac Metastasis and Hypertrophic Osteoarthropathy in Recurrent Infantile Fibrosarcoma. Pediatr. Blood Cancer 2012, 59, 179–181.
- Hiradfar, A.; Pourlak, T.; Badebarin, D. Primary Pulmonary Fibrosarcoma With Bone Metastasis: A Successful Treatment with Post-Operation Adjuvant Chemotherapy. Iran. J. Cancer Prev. 2015, 8, e2328.
- Laetsch, T.W.; DuBois, S.G.; Mascarenhas, L.; Turpin, B.; Federman, N.; Albert, C.M.; Nagasubramanian, R.; Davis, J.L.; Rudzinski, E.; Feraco, A.M.; et al. Larotrectinib for Paediatric Solid Tumours Harbouring NTRK Gene Fusions: Phase 1 Results from a Multicentre, Open-Label, Phase 1/2 Study. Lancet Oncol. 2018, 19, 705–714.
- Shulman, D.S.; DuBois, S.G. The Evolving Diagnostic and Treatment Landscape of NTRK-Fusion-Driven Pediatric Cancers. Pediatr. Drugs 2020, 22, 189–197.
- Caldwell, K.J.; De La Cuesta, E.; Morin, C.; Pappo, A.; Helmig, S. A Newborn with a Large NTRK Fusion Positive Infantile Fibrosarcoma Successfully Treated with Larotrectinib. Pediatr. Blood Cancer 2020, 67, e28330.
- Kao, Y.; Suurmeijer, A.J.H.; Argani, P.; Dickson, B.C.; Zhang, L.; Sung, Y.; Agaram, N.P.; Fletcher, C.D.M.; Antonescu, C.R. Soft Tissue Tumors Characterized by a Wide Spectrum of Kinase Fusions Share a Lipofibromatosis-like Neural Tumor Pattern. Genes Chromosomes Cancer 2020, 59, 575–583.
- Agaram, N.P.; Zhang, L.; Sung, Y.-S.; Chen, C.-L.; Chung, C.T.; Antonescu, C.R.; Fletcher, C.D. Recurrent NTRK1 Gene Fusions Define a Novel Subset of Locally Aggressive Lipofibromatosis-like Neural Tumors. Am. J. Surg. Pathol. 2016, 40, 1407–1416.
- Drabent, P.; Fraitag, S. Update on Superficial Spindle Cell Mesenchymal Tumors in Children. Dermatopathology 2021, 8, 285–300.
- Siozopoulou, V.; Smits, E.; De Winne, K.; Marcq, E.; Pauwels, P. NTRK Fusions in Sarcomas: Diagnostic Challenges and Clinical Aspects. Diagnostics 2021, 11, 478.
- Michal, M.; Hájková, V.; Skálová, A.; Michal, M. STRN-NTRK3-Rearranged Mesenchymal Tumor of the Uterus: Expanding the Morphologic Spectrum of Tumors with NTRK Fusions. Am. J. Surg. Pathol. 2019, 43, 1152–1154.
- Davis, J.L.; Lockwood, C.M.; Albert, C.M.; Tsuchiya, K.; Hawkins, D.S.; Rudzinski, E.R. Infantile NTRK-Associated Mesenchymal Tumors. Pediatr. Dev. Pathol. Off. J. Soc. Pediatr. Pathol. Paediatr. Pathol. Soc. 2018, 21, 68–78.
- Tallegas, M.; Fraitag, S.; Binet, A.; Orbach, D.; Jourdain, A.; Reynaud, S.; Pierron, G.; Machet, M.-C.; Maruani, A. Novel KHDRBS1-NTRK3 Rearrangement in a Congenital Pediatric CD34-Positive Skin Tumor: A Case Report. Virchows Arch. Int. J. Pathol. 2019, 474, 111–115.
- Ji, Y.; Yang, K.; Peng, S.; Chen, S.; Xiang, B.; Xu, Z.; Li, Y.; Wang, Q.; Wang, C.; Xia, C.; et al. Kaposiform Haemangioendothelioma: Clinical Features, Complications and Risk Factors for Kasabach-Merritt Phenomenon. Br. J. Dermatol. 2018, 179, 457–463.
- Ten Broek, R.W.; Koelsche, C.; Eijkelenboom, A.; Mentzel, T.; Creytens, D.; Vokuhl, C.; van Gorp, J.M.; Versleijen-Jonkers, Y.M.; van der Vleuten, C.J.; Kemmeren, P.; et al. Kaposiform Hemangioendothelioma and Tufted Angioma—(Epi)Genetic Analysis Including Genome-Wide Methylation Profiling. Ann. Diagn. Pathol. 2020, 44, 151434.
- Ji, Y.; Chen, S.; Yang, K.; Xia, C.; Li, L. Kaposiform Hemangioendothelioma: Current Knowledge and Future Perspectives. Orphanet J. Rare Dis. 2020, 15, 39.
- Ryu, Y.J.; Choi, Y.H.; Cheon, J.-E.; Kim, W.S.; Kim, I.-O.; Park, J.E.; Kim, Y.J. Imaging Findings of Kaposiform Hemangioendothelioma in Children. Eur. J. Radiol. 2017, 86, 198–205.
- Lyons, L.L.; North, P.E.; Mac-Moune Lai, F.; Stoler, M.H.; Folpe, A.L.; Weiss, S.W. Kaposiform Hemangioendothelioma: A Study of 33 Cases Emphasizing Its Pathologic, Immunophenotypic, and Biologic Uniqueness from Juvenile Hemangioma. Am. J. Surg. Pathol. 2004, 28, 559–568.
- Rodriguez, V.; Lee, A.; Witman, P.M.; Anderson, P.A. Kasabach-Merritt Phenomenon: Case Series and Retrospective Review of the Mayo Clinic Experience. J. Pediatr. Hematol. Oncol. 2009, 31, 522–526.
- Croteau, S.E.; Liang, M.G.; Kozakewich, H.P.; Alomari, A.I.; Fishman, S.J.; Mulliken, J.B.; Trenor, C.C. Kaposiform Hemangioendothelioma: Atypical Features and Risks of Kasabach-Merritt Phenomenon in 107 Referrals. J. Pediatr. 2013, 162, 142–147.
- Gruman, A.; Liang, M.G.; Mulliken, J.B.; Fishman, S.J.; Burrows, P.E.; Kozakewich, H.P.W.; Blei, F.; Frieden, I.J. Kaposiform Hemangioendothelioma without Kasabach-Merritt Phenomenon. J. Am. Acad. Dermatol. 2005, 52, 616–622.
- Drolet, B.A.; Trenor, C.C.; Brandão, L.R.; Chiu, Y.E.; Chun, R.H.; Dasgupta, R.; Garzon, M.C.; Hammill, A.M.; Johnson, C.M.; Tlougan, B.; et al. Consensus-Derived Practice Standards Plan for Complicated Kaposiform Hemangioendothelioma. J. Pediatr. 2013, 163, 285–291.
- Colmenero, I.; Hoeger, P.H. Vascular Tumours in Infants. Part II: Vascular Tumours of Intermediate Dignity and Malignant Tumours. Br. J. Dermatol. 2014, 171, 474–484.
- Le Huu, A.R.; Jokinen, C.H.; Rubin, B.P.; Ruben, B.P.; Mihm, M.C.; Weiss, S.W.; North, P.E.; Dadras, S.S. Expression of Prox1, Lymphatic Endothelial Nuclear Transcription Factor, in Kaposiform Hemangioendothelioma and Tufted Angioma. Am. J. Surg. Pathol. 2010, 34, 1563–1573.
- Lim, Y.H.; Bacchiocchi, A.; Qiu, J.; Straub, R.; Bruckner, A.; Bercovitch, L.; Narayan, D.; Yale Center for Mendelian Genomics; McNiff, J.; Ko, C.; et al. GNA14 Somatic Mutation Causes Congenital and Sporadic Vascular Tumors by MAPK Activation. Am. J. Hum. Genet. 2016, 99, 443–450.
- Blatt, J.; Stavas, J.; Moats-Staats, B.; Woosley, J.; Morrell, D.S. Treatment of Childhood Kaposiform Hemangioendothelioma with Sirolimus: Hemangioendothelioma and Sirolimus. Pediatr. Blood Cancer 2010, 55, 1396–1398.
- Rössler, J.; Baselga, E.; Davila, V.; Celis, V.; Diociaiuti, A.; El Hachem, M.; Mestre, S.; Haeberli, D.; Prokop, A.; Hanke, C.; et al. Severe Adverse Events during Sirolimus “Off-label” Therapy for Vascular Anomalies. Pediatr. Blood Cancer 2021, 68, e28936.
- Chen, X.; Wang, D.-D.; Xu, H.; Li, Z.-P. Initial Dose Recommendation for Sirolimus in Paediatric Kaposiform Haemangioendothelioma Patients Based on Population Pharmacokinetics and Pharmacogenomics. J. Int. Med. Res. 2020, 48, 030006052094762.
- Ji, Y.; Chen, S.; Yang, K.; Zhou, J.; Zhang, X.; Jiang, X.; Xu, X.; Lu, G.; Qiu, L.; Kong, F.; et al. A Prospective Multicenter Study of Sirolimus for Complicated Vascular Anomalies. J. Vasc. Surg. 2021, 74, 1673–1681.e3.
- Folpe, A.L.; Veikkola, T.; Valtola, R.; Weiss, S.W. Vascular Endothelial Growth Factor Receptor-3 (VEGFR-3): A Marker of Vascular Tumors with Presumed Lymphatic Differentiation, Including Kaposi’s Sarcoma, Kaposiform and Dabska-Type Hemangioendotheliomas, and a Subset of Angiosarcomas. Mod. Pathol. 2000, 13, 180–185.
- Requena, L.; Kutzner, H. Hemangioendothelioma. Semin. Diagn. Pathol. 2013, 30, 29–44.
- Albertini, A.-F.; Brousse, N.; Bodemer, C.; Calonje, E.; Fraitag, S. Retiform Hemangioendothelioma Developed on the Site of an Earlier Cystic Lymphangioma in a Six-Year-Old Girl. Am. J. Dermatopathol. 2011, 33, e84–e87.
- Miettinen, M.; Wang, Z.-F. Prox1 Transcription Factor as a Marker for Vascular Tumors—Evaluation of 314 Vascular Endothelial and 1086 Nonvascular Tumors. Am. J. Surg. Pathol. 2012, 36, 351–359.
- Perry, K.D.; Al-lbraheemi, A.; Rubin, B.P.; Jen, J.; Ren, H.; Jang, J.S.; Nair, A.; Davila, J.; Pambuccian, S.; Horvai, A.; et al. Composite Hemangioendothelioma with Neuroendocrine Marker Expression: An Aggressive Variant. Mod. Pathol. 2017, 30, 1589–1602.
- Fukunaga, M. Expression of D2-40 in Lymphatic Endothelium of Normal Tissues and in Vascular Tumours. Histopathology 2005, 46, 396–402.
- Parsons, A.; Sheehan, D.J.; Sangueza, O.P. Retiform Hemangioendotheliomas Usually Do Not Express D2-40 and VEGFR-3. Am. J. Dermatopathol. 2008, 30, 31–33.
- Schwartz, R.A.; Dabski, C.; Dabska, M. The Dabska Tumor: A Thirty-Year Retrospect. Dermatology 2000, 201, 1–5.
- Keiler, S.A.; Honda, K.; Bordeaux, J.S. Retiform Hemangioendothelioma Treated with Mohs Micrographic Surgery. J. Am. Acad. Dermatol. 2011, 65, 233–235.
- Hirsh, A.Z.; Yan, W.; Wei, L.; Wernicke, A.G.; Parashar, B. Unresectable Retiform Hemangioendothelioma Treated with External Beam Radiation Therapy and Chemotherapy: A Case Report and Review of the Literature. Sarcoma 2010, 2010, 756246.
- Walther, C.; Tayebwa, J.; Lilljebjörn, H.; Magnusson, L.; Nilsson, J.; von Steyern, F.V.; Øra, I.; Domanski, H.A.; Fioretos, T.; Nord, K.H.; et al. A Novel SERPINE1-FOSB Fusion Gene Results in Transcriptional up-Regulation of FOSB in Pseudomyogenic Haemangioendothelioma: SERPINE1-FOSB Fusion in Pseudomyogenic Haemangioendothelioma. J. Pathol. 2014, 232, 534–540.
- Raftopoulos, E.; Royer, M.; Warren, M.; Zhao, J.; Rush, W. Pseudomyogenic Hemangioendothelioma: Case Report and Review of the Literature. Am. J. Dermatopathol. 2018, 40, 597–601.
- Hung, Y.P.; Fletcher, C.D.M.; Hornick, J.L. FOSB Is a Useful Diagnostic Marker for Pseudomyogenic Hemangioendothelioma. Am. J. Surg. Pathol. 2017, 41, 596–606.
- Caballero, G.A.; Roitman, P.D. Pseudomyogenic Hemangioendothelioma (Epithelioid Sarcoma-Like Hemangioendothelioma). Arch. Pathol. Lab. Med. 2020, 144, 529–533.
- Requena, L.; Santonja, C.; Martinez-Amo, J.L.; Saus, C.; Kutzner, H. Cutaneous Epithelioid Sarcomalike (Pseudomyogenic) Hemangioendothelioma: A Little-Known Low-Grade Cutaneous Vascular Neoplasm. JAMA Dermatol. 2013, 149, 459–465.
- Hornick, J.L.; Fletcher, C.D.M. Pseudomyogenic Hemangioendothelioma: A Distinctive, Often Multicentric Tumor with Indolent Behavior. Am. J. Surg. Pathol. 2011, 35, 190–201.
- Trombetta, D.; Magnusson, L.; von Steyern, F.V.; Hornick, J.L.; Fletcher, C.D.M.; Mertens, F. Translocation t(7;19)(Q22;Q13)—A Recurrent Chromosome Aberration in Pseudomyogenic Hemangioendothelioma? Cancer Genet. 2011, 204, 211–215.
- Agaram, N.P.; Zhang, L.; Cotzia, P.; Antonescu, C.R. Expanding the Spectrum of Genetic Alterations in Pseudomyogenic Hemangioendothelioma with Recurrent Novel ACTB-FOSB Gene Fusions. Am. J. Surg. Pathol. 2018, 42, 1653–1661.
- Zhu, G.; Benayed, R.; Ho, C.; Mullaney, K.; Sukhadia, P.; Rios, K.; Berry, R.; Rubin, B.P.; Nafa, K.; Wang, L.; et al. Diagnosis of Known Sarcoma Fusions and Novel Fusion Partners by Targeted RNA Sequencing with Identification of a Recurrent ACTB-FOSB Fusion in Pseudomyogenic Hemangioendothelioma. Mod. Pathol. 2019, 32, 609–620.
- Panagopoulos, I.; Lobmaier, I.; Gorunova, L.; Heim, S. Fusion of the Genes WWTR1 and FOSB in Pseudomyogenic Hemangioendothelioma. Cancer Genom. Proteom. 2019, 16, 293–298.
- Bridge, J.A.; Sumegi, J.; Royce, T.; Baker, M.; Linos, K. A Novel CLTC-FOSB Gene Fusion in Pseudomyogenic Hemangioendothelioma of Bone. Genes Chromosomes Cancer 2021, 60, 38–42.
- Hakar, M.H.; White, K.; Hansford, B.G.; Swensen, J.; Davis, J.L. Novel EGFL7-FOSB Fusion in Pseudomyogenic Haemangioendothelioma with Widely Metastatic Disease. Histopathology 2021, 79, 888–891.
- Mirra, J.M.; Kessler, S.; Bhuta, S.; Eckardt, J. The Fibroma-like Variant of Epithelioid Sarcoma. A Fibrohistiocytic/Myoid Cell Lesion Often Confused with Benign and Malignant Spindle Cell Tumors. Cancer 1992, 69, 1382–1395.
- Billings, S.D.; Folpe, A.L.; Weiss, S.W. Epithelioid Sarcoma-like Hemangioendothelioma. Am. J. Surg. Pathol. 2003, 27, 48–57.
- Ozeki, M.; Nozawa, A.; Kanda, K.; Hori, T.; Nagano, A.; Shimada, A.; Miyazaki, T.; Fukao, T. Everolimus for Treatment of Pseudomyogenic Hemangioendothelioma. J. Pediatr. Hematol. Oncol. 2017, 39, e328–e331.
- Gabor, K.M.; Sapi, Z.; Tiszlavicz, L.G.; Fige, A.; Bereczki, C.; Bartyik, K. Sirolimus Therapy in the Treatment of Pseudomyogenic Hemangioendothelioma. Pediatr. Blood Cancer 2018, 65, e26781.
- Deyrup, A.T.; Miettinen, M.; Khoury, J.D.; Parham, D.M.; Shehata, B.M. Pediatric Cutaneous Angiosarcomas: A Clinicopathologic Study of 10 Cases. Am. J. Surg. Pathol. 2011, 35, 6.
- Kim, Y.S.; Lee, J.H.; Lee, J.Y.; Park, Y.M. Primary Cutaneous Rhabdomyosarcoma: Case Report and Review of Published Work. J. Dermatol. 2015, 42, 1014–1015.
- Gökdemir, G.; Ekmen, S.; Gungor, S.; Singer, R. Perianal Rhabdomyosarcoma: Report of a Case in an Infant and Review of the Literature: A Polypoid Anal Mass as Embryonal Rhabdomyosarcoma. Pediatr. Dermatol. 2013, 30, 97–99.
- Rekhi, B.; Qureshi, S.S.; Narula, G.; Gujral, S.; Kurkure, P. Rapidly Progressive Congenital Rhabdomyosarcoma Presenting with Multiple Cutaneous Lesions: An Uncommon Diagnosis and a Therapeutic Challenge. Pathol.-Res. Pract. 2014, 210, 328–333.
- Gavrilova, M.; Cabezas Macian, M.; Martin-Gorgojo, A.; Beteta, L.G.; Posadas, V.; Jorda-Cuevas, E. A Rapidly Enlarging Mass on the Right Leg. Pediatr. Dermatol. 2015, 32, 143–144.
- Piersigilli, F.; Auriti, C.; Mondì, V.; Francalanci, P.; Salvatori, G.; Danhaive, O. Decreased CDKN1C Expression in Congenital Alveolar Rhabdomyosarcoma Associated with Beckwith-Wiedemann Syndrome. Indian J. Pediatr. 2016, 83, 1476–1478.
- Russo, I.; Di Paolo, V.; Gurnari, C.; Mastronuzzi, A.; Del Bufalo, F.; Di Paolo, P.L.; Di Giannatale, A.; Boldrini, R.; Milano, G.M. Congenital Rhabdomyosarcoma: A Different Clinical Presentation in Two Cases. BMC Pediatr. 2018, 18, 166.
- Gong, Y.; Chao, J.; Bauer, B.; Sun, X.; Chou, P.M. Primary Cutaneous Alveolar Rhabdomyosarcoma of the Perineum. Arch. Pathol. Lab. Med. 2002, 126, 982–984.
- Grundy, R.; Anderson, J.; Gaze, M.; Gerrard, M.; Glaser, A.; Gordon, A.; Malone, M.; Pritchard-Jones, K.; Michalski, A. Congenital Alveolar Rhabdomyosarcomam: Clinical and Molecular Distinction from Alveolar Rhabdomyosarcoma in Older Children. Cancer 2001, 91, 606–612.
- Qualman, S.; Lynch, J.; Bridge, J.; Parham, D.; Teot, L.; Meyer, W.; Pappo, A. Prevalence and Clinical Impact of Anaplasia in Childhood Rhabdomyosarcoma: A Report from the Soft Tissue Sarcoma Committee of the Children’s Oncology Group. Cancer 2008, 113, 3242–3247.
- Montoya-Cerrillo, D.M.; Diaz-Perez, J.A.; Velez-Torres, J.M.; Montgomery, E.A.; Rosenberg, A.E. Novel Fusion Genes in Spindle Cell Rhabdomyosarcoma: The Spectrum Broadens. Genes Chromosomes Cancer 2021, 60, 687–694.
- Kuroiwa, M.; Sakamoto, J.; Shimada, A.; Suzuki, N.; Hirato, J.; Park, M.-J.; Sotomatsu, M.; Hayashi, Y. Manifestation of Alveolar Rhabdomyosarcoma as Primary Cutaneous Lesions in a Neonate with Beckwith-Wiedemann Syndrome. J. Pediatr. Surg. 2009, 44, e31–e35.
- Fraitag, S.; Boccara, O. What to Look Out for in a Newborn with Multiple Papulonodular Skin Lesions at Birth. Dermatopathology 2021, 8, 390–417.
- Brecher, A.R.; Reyes-Mugica, M.; Kamino, H.; Chang, M.W. Congenital Primary Cutaneous Rhabdomyosarcoma in a Neonate. Pediatr. Dermatol. 2003, 20, 335–338.
- Skapek, S.X.; Ferrari, A.; Gupta, A.A.; Lupo, P.J.; Butler, E.; Shipley, J.; Barr, F.G.; Hawkins, D.S. Rhabdomyosarcoma. Nat. Rev. Dis. Primer 2019, 5, 1.
- Marburger, T.B.; Gardner, J.M.; Prieto, V.G.; Billings, S.D. Primary Cutaneous Rhabdomyosarcoma: A Clinicopathologic Review of 11 Cases: Cutaneous Rhabdomyosarcoma. J. Cutan. Pathol. 2012, 39, 987–995.
- Esmaeili, H.; Azimpouran, M. Congenital Embryonal Rhabdomyosarcoma; Multiple Lesions. Int. J. Surg. Case Rep. 2017, 31, 47–50.
- Pitjadi, T.M.; Wadee, R.; Grayson, W. Rhabdomyosarcoma Arising in a Giant Congenital Melanocytic Naevus: Case Report and Literature Review. Dermatopathology 2019, 6, 91–98.
- Li, H.; Sisoudiya, S.D.; Martin-Giacalone, B.A.; Khayat, M.M.; Dugan-Perez, S.; Marquez-Do, D.A.; Scheurer, M.E.; Muzny, D.; Boerwinkle, E.; Gibbs, R.A.; et al. Germline Cancer Predisposition Variants in Pediatric Rhabdomyosarcoma: A Report from the Children’s Oncology Group. J. Natl. Cancer Inst. 2021, 113, 875–883.
- Yechieli, R.L.; Mandeville, H.C.; Hiniker, S.M.; Bernier-Chastagner, V.; McGovern, S.; Scarzello, G.; Wolden, S.; Cameron, A.; Breneman, J.; Fajardo, R.D.; et al. Rhabdomyosarcoma. Pediatr. Blood Cancer 2021, 68, e28254.
More
Information
Subjects:
Dermatology; Pediatrics
Contributors
MDPI registered users' name will be linked to their SciProfiles pages. To register with us, please refer to https://encyclopedia.pub/register
:
View Times:
1.7K
Revisions:
2 times
(View History)
Update Date:
02 Jun 2022
Notice
You are not a member of the advisory board for this topic. If you want to update advisory board member profile, please contact office@encyclopedia.pub.
OK
Confirm
Only members of the Encyclopedia advisory board for this topic are allowed to note entries. Would you like to become an advisory board member of the Encyclopedia?
Yes
No
${ textCharacter }/${ maxCharacter }
Submit
Cancel
Back
Comments
${ item }
|
More
No more~
There is no comment~
${ textCharacter }/${ maxCharacter }
Submit
Cancel
${ selectedItem.replyTextCharacter }/${ selectedItem.replyMaxCharacter }
Submit
Cancel
Confirm
Are you sure to Delete?
Yes
No





