
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Abolanle A. A Kayode | -- | 1138 | 2022-05-20 19:32:40 | | | |
| 2 | Catherine Yang | Meta information modification | 1138 | 2022-05-23 03:39:44 | | |
Video Upload Options
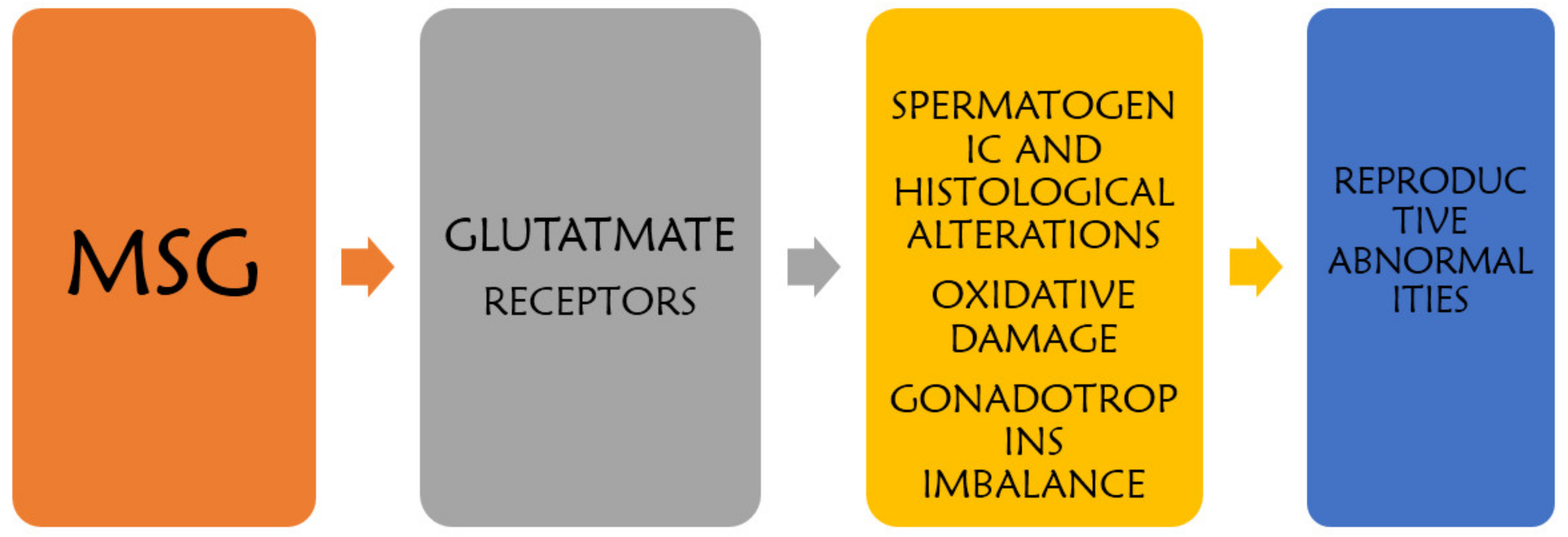
Reproductive dysfunction is often characterized by malfunction of the reproductive tissues, which may lead to disruption of the synergistic rhythm that should bring about a progression of sexual events and the conception of new life. This may therefore result in the sexual dysfunction and infertility that can be seen in couples having prolonged biological difficulty in reproducing their offspring after having unrestricted sexual intercourse for at least twelve months. Several factors have been implicated in the cause and progression of reproductive dysfunction, including poor nutrition, drug side effects, disease states, and toxicant ingestion. A well-known food additive that has been found to be potent at initiating reproductive anomalies in males is monosodium glutamate (MSG).
1. Introduction
2. Mechanism of MSG-Induced Testicular Alteration
2.1. Oxidative Stress
2.2. Neurotoxicity
2.3. Histomorphological Alterations
2.4. Glutamate Receptor Dysfunction
2.5. Brief Clinically Observed Adverse Effect of MSG
References
- Guyton, A.C.; Hall, J.E. Textbook of Medical Physiology, 10th ed.; Harcourt International Edition; W.B. Saunder Company: Philadelphia, PA, USA, 2000; pp. 279–281.
- Ganong, W.F. Review of Medical Physiology, 20th ed.; Lange Medical Books/McGraw-Hill Medical Publishing Division: London, UK, 2001; p. 543.
- Kandeel, F.R.; Koussa, V.K.; Swerdloff, R.S. Male sexual function and its disorders: physiology, pathophysiology, clinical investigation, and treatment. Endocr. Rev. 2001, 22, 342.
- Kayode, O.T.; Yakubu, M.T. Parquetina nigrescens leaves: Chemical profile and effects of its Aqueous Extract on the Physical and Biochemical Parameters of Sexual Behaviour of Male Rats. J. Integr. Med. 2017, 15, 64–76.
- Harchegani, A.B.; Irandoost, A.; Mirnamniha, M.; Rahmani, H.; Tahmasbpour, E.; Shahriary, A. Possible Mechanisms for The Effects of Calcium Deficiency on Male Infertility. Int. J. Fertil. Steril. 2019, 12, 267–272.
- Bera, T.K.; Kar, S.K.; Yadav, P.K.; Mukherjee, P.; Yadav, S.; Joshi, B. Effects of monosodium glutamate on human health: A systematic review. World J. Pharm. Sci. 2017, 5, 139–144.
- Hamza, R.Z.; AL-Harbi, M.S. Monosodium glutamate induced testicular toxicity and the possible ameliorative role of vitamin E or selenium in male rats. Toxicol. Report 2014, 1, 1037–1045.
- Kazmi, Z.; Fatima, I.; Perveen, S.; Malik, S.S. Monosodium glutamate: Review on clinical reports. Int. J. Food Prop. 2017, 20 (Suppl. 2), 1807–1815.
- Hanipah, E.N.A.; Yahya, N.J.; Ajik, E.M.; Yusoff, N.A.; Taib, I.S. Monosodium Glutamate Induced Oxidative Stress in Accessory Reproductive Organs of Male Sprague-Dawley Rats. J. Sains Kesihat. Malays. 2018, 16, 67–73.
- El-Sawy, H.B.I.; Soliman, M.M.; El-Shazly, S.A.; Ali, H.A. Protective effects of camel milk and vitamin E against monosodium glutamate induced biochemical and testicular dysfunctions. Prog. Nutr. 2018, 20, 76–85.
- Khaled, F.A.; Yousef, M.I.; Kamel, K.I. The protective role of propolis against the reproductive toxicity of mono-sodium glutamine in male rabbits. Int. J. Chem. Stud. 2016, 4, 4–9.
- Kianifard, D.; Gholamreza, S.V.; Farhad, R. Study of the protective effects of quince (Cydonia oblonga) leaf extract on fertility alterations and gonadal dysfunction induced by Monosodium glutamate in adult male wistar rats. Rom. J. Diabetes Nutr. Metab. Dis. 2015, 22, 375–384.
- Abd-Elaziz, A.M.S.; Ashoush, I.S. Effect of monosodium glutamate administration on the reproductive performance in male male albino rats. Egypy J. Basic Appl. Physiol. 2007, 6, 101–110.
- Das, R.S.; Ghosh, S.K. Long term effects of monosodium glutamate on spermatogenesis following neonatal exposure in albino mice—A histological study. Nepal Med. Coll. J. 2010, 12, 149–153.
- Igwebuike, U.M. The effects of oral administration of monosodium glutamaste (msg) on the testicular morphology and cauda epididymal sperm reserves of young and adult male rats. Vet. Arh. 2011, 81, 525–534.
- Fernandes, G.S.A.; Arena, A.C.; Campos, K.E.; Volpato, G.T.; Anselmo-Franci, J.A.; Damasceno, D.C.; Kempinas, W.G. Glutamate-induced obesity leads to decreased sperm reserves and acceleration of transit time in the epididymis of adult male rats. Reprod. Biol. Endocrinol. 2012, 10, 105.
- Iamsaard, S.; Sukhorum, W.; Samrid, R.; Yimdee, J.; Kanla, P.; Chaisiwamongkol, K.; Hipkaeo, W.; Fongmoon, D.; Kondo, H. The sensitivity of male rat reproductive organs to monosodium glutamate. Acta Med. Acad. 2014, 43, 3–9.
- Nosseir, N.S.; Ali, M.H.N.; Ebaid, H.M. A Histological and Morphometric Study of Monosodium Glutamate Toxic Effect on Testicular Structure and Potentiality of Recovery in Adult Albino Rats. Res. J. Biol. 2012, 2, 66–78.
- Sakr, S.A.; Bada, G.M. Protective Effect of Curcumin on Monosodium Glutamate-Induced Reproductive Toxicity in Male Albino Rats. Glob. J. Pharmacol. 2013, 7, 416–422.
- Abd-Ella, E.M.M.; Mohammed, A.M. Attenuation of Monosodium Glutamate-Induced Hepatic and Testicular Toxicity in Albino Rats by Annona Muricata Linn. (Annonaceae) Leaf Extract. IOSR J. Pharm. Biol. Sci. 2016, 11, 61–69.
- Ekaluo, U.B.; Ikpeme, E.V.; Ibiang, Y.B.; Amaechina, O.S. Attenuating role of vitamin C on sperm toxicity induced by monosodium glutamate in albino rats. J. Biol. Sci. 2013, 13, 298–301.
- Yeomans, M.R.; Gould, N.J.; Mobini, S.; Prescott, J. Acquired Flavor Acceptance and Intake Facilitated by Monosodium Glutamate in Humans. Physiol. Behav. 2008, 93, 958–966.
- Bellisle, F.; Monneuse, M.O.; Chabert, M.; Larue-Achagiotis, C.; Lanteaume, M.T.; Louis-Sylvestre, J. Monosodium Glutamate as a Palatability Enhancer in the European Diet. Physiol. Behav. 1991, 49, 869–873.
- He, K.; Zhao, L.; Daviglus, M.L.; Dyer, A.R.; Horn, L.; Garside, D.; Stamler, J. Association of Monosodium Glutamate Intake with Overweight in Chinese Adults: The INTERMAP Study. Obesity 2008, 16, 1875–1880.
- Shi, Z.; Luscombe-Marsh, N.D.; Wittert, G.A.; Yuan, B.; Dai, Y.; Pan, X.; Taylor, A.W. Monosodium Glutamate Is Not Associated with Obesity or a Greater Prevalence of Weight Gain over 5 Years: Findings from the Jiangsu Nutrition Study of Chinese Adults. Br. J. Nutar. 2010, 104, 457–463.
- Insawang, T.; Selmi, C.; Cha’on, U.; Pethlert, S.; Yongvanit, P.; Areejitranusorn, P.; Prasongwattana, V. Monosodium Glutamate (MSG) Intake Is Associated with the Prevalence of Metabolic Syndrome in a Rural Thai Population. Nutr. Metab. 2012, 9, 1.





