Your browser does not fully support modern features. Please upgrade for a smoother experience.

Submitted Successfully!
Thank you for your contribution! You can also upload a video entry or images related to this topic.
For video creation, please contact our Academic Video Service.
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Igor Grivennikov | -- | 2454 | 2022-05-18 21:54:13 | | | |
| 2 | Camila Xu | Meta information modification | 2454 | 2022-05-19 03:28:37 | | |
Video Upload Options
We provide professional Academic Video Service to translate complex research into visually appealing presentations. Would you like to try it?
Cite
If you have any further questions, please contact Encyclopedia Editorial Office.
Grivennikov, I.; Dolotov, O.; Inozemtseva, L.; Myasoyedov, N. Astrocytes in Depression and Alzheimer’s Disease. Encyclopedia. Available online: https://encyclopedia.pub/entry/23090 (accessed on 26 May 2026).
Grivennikov I, Dolotov O, Inozemtseva L, Myasoyedov N. Astrocytes in Depression and Alzheimer’s Disease. Encyclopedia. Available at: https://encyclopedia.pub/entry/23090. Accessed May 26, 2026.
Grivennikov, Igor, Oleg Dolotov, Ludmila Inozemtseva, Nikolai Myasoyedov. "Astrocytes in Depression and Alzheimer’s Disease" Encyclopedia, https://encyclopedia.pub/entry/23090 (accessed May 26, 2026).
Grivennikov, I., Dolotov, O., Inozemtseva, L., & Myasoyedov, N. (2022, May 18). Astrocytes in Depression and Alzheimer’s Disease. In Encyclopedia. https://encyclopedia.pub/entry/23090
Grivennikov, Igor, et al. "Astrocytes in Depression and Alzheimer’s Disease." Encyclopedia. Web. 18 May, 2022.
Copy Citation
Astrocytes play a key role in the functioning of neurons in norm and pathology, being a target for stress and glucocorticoids, are a promising target for the treatment of stress-dependent depression and Alzheimer’s disease (AD). Depression, as a mental disorder, is characterized by two core symptoms, depressed mood and loss of interest or pleasure in nearly all activities, and may be accompanied by other symptoms such as cognitive impairments, sleep disturbance, psychomotor retardation or agitation, feelings of worthlessness or excessive or inappropriate guilt.
depression
neurodegeneration
Alzheimer’s disease
astrocytes
1. Introduction
According to Hans Selye, “stress is the nonspecific response of the body to any demand” [1]. Stress is also defined as a state of threatened (or perceived as threatened) internal dynamic balance (“homeostasis”) caused by external or internal stimuli (“stressors”) [2]. To achieve homeostasis, the highly conservative regulatory neuroendocrine system, the “stress system”, is activated through synchronized interaction between the hypothalamic–pituitary–adrenal axis (HPAA) and the autonomic nervous system [2]. In principle, stress is necessary to adapt to changing environmental or internal conditions and increase the chance of survival. It is known that moderate stress is able to activate mental and behavioral processes to find solutions to the challenges facing an individual. However, excessive and/or prolonged stressors and the consequent chronic deregulation of the stress system can lead to a wide range of chronic pathological conditions, including pathologies of the cardiovascular, endocrine, immune and nervous systems. One of the stress-related pathologies of the nervous system is depression (major depressive disorder). Currently, depression is a most widespread mental disorder worldwide, which, according to World Health Organization, affects approximately 280 million people in the world, or about 4.0% of the population, including at least 5% of adults [3]. The incidence of depression increases with age, so it is about 27% in the age group of 75–80 years, 33% in the age group of 81–85 years and reaches 46% in the age group of 91 years and older [4]. Considering that the frequency of neurodegenerative diseases (in particular, dementia) also increases with age and that depression often manifests itself in dementia, it is possible that pathological changes in the course of dementia are associated with the development of depression.
Depression, as a mental disorder, is characterized by two core symptoms, depressed mood and loss of interest or pleasure in nearly all activities, and may be accompanied by other symptoms such as cognitive impairments, sleep disturbance, psychomotor retardation or agitation, feelings of worthlessness or excessive or inappropriate guilt [5]. Depression significantly worsens the quality of life. A large percentage of suicides, especially among young people, is associated with depression.
Figure 1 schematically shows the general sequence of events leading to the development of chronic stress, depression and, ultimately, to the degeneration of nerve cells.
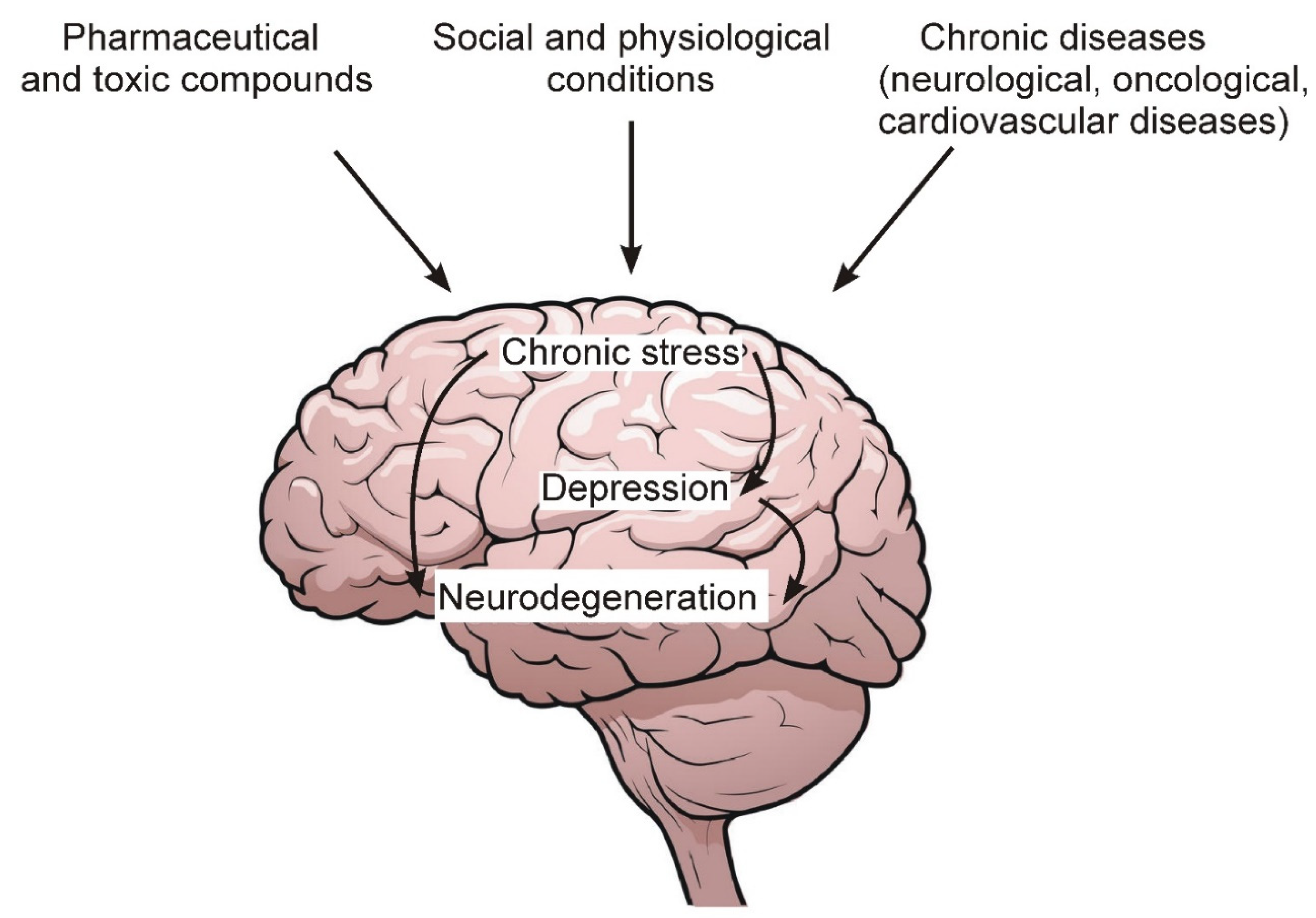 Figure 1. Some environmental factors, as well as chronic diseases that lead to the development of chronic stress and depression.
Figure 1. Some environmental factors, as well as chronic diseases that lead to the development of chronic stress and depression.Among the main factors leading to the development of chronic stress and depression, the following should be noted:
-
Chronic pathologies of the nervous and cardiovascular systems, as well as oncological diseases.
-
Social and psychological factors related to human living conditions and contacts with surrounding members of society and the external environment.
-
Excessive and prolonged intake of various pharmaceutical preparations, as well as toxic compounds from the external environment, the number of which increases with the deterioration of the overall environmental situation in the world.
Alzheimer’s disease (AD) is the most common cause of dementia, which is estimated to account for 60% to 80% of cases [6]. AD is considered one of the main causes of morbidity and mortality among the elderly [7]. The prevalence of AD in Europe is estimated at 5.0%, which is 3.3% in men and 7.1% in women [8]. AD is a slowly progressive brain pathology that begins many years before the onset of symptoms. Clinical symptoms in early stages of AD include difficulty remembering recent conversations, names, or events, which are often accompanied by apathy and depression. In later stages of AD, symptoms include impaired communication, disorientation, behavioral changes, and ultimately difficulty speaking, swallowing, and walking [6]. AD is characterized by the accumulation of beta-amyloid peptide (Aß) (amyloid plaques) in brain tissues and a destabilization of the cytoskeleton of neurons caused by hyperphosphorylation of microtubule-associated Tau-protein. However, the poor correlation between cognitive decline and amyloid plaques raises the question of whether Aß accumulation actually causes neurodegeneration in AD. The formation of neurofibrillary tangles of Tau correlates better with neurodegeneration and clinical symptoms, and although Aß can initiate a cascade of events leading to neurodegeneration, Tau hyperphosphorylation is assumed to be key in neurodegeneration in AD [9]. The vast majority of cases of AD belong to a sporadic form (usually, late onset of symptoms). The sporadic form of AD is associated with the interaction of genetic and environmental factors, and aging is the main risk factor. Two main genetic risk factors for sporadic AD have been identified. Firstly, the presence of the APOE4 allele encoding one of the three isoforms of Apolipoprotein E (apoE2, apoE3 and apoE4), the main transporter of cholesterol in the brain, which is synthesized and secreted by astrocytes [10], is the most significant risk factor for sporadic AD [11]. Other genetic risk factors for sporadic AD are polymorphisms and mutations in a number of genes expressed in microglia, in particular, polymorphism in the TREM2 gene encoding transmembrane glycoprotein, which acts as a receptor on the surface of microglia and perceives lipids that are exposed after cell damage [12]. The familial form of AD (early onset) accounts for about 5% of cases of AD and is associated with mutations in the genes encoding the precursor protein for Aß and presenilins 1 and 2, which leads to increased aggregation of Aß, but a small part of mutations in the gene encoding presenilin 1 is not familial and occurs de novo [13].
The development of depression is accompanied by changes in the metabolism of nerve and glial cells and an impairment of synaptic transmission between neurons. Prolonged action of harmful factors can eventually lead to the degeneration of nerve cells in the brain (Figure 1). Recent ideas about the functions of astrocytes assign them an extremely important role both in the normal functioning of the brain and in the development of brain pathologies [14][15]. In particular, astrocytes are a source of neurotrophic factors, regulate synaptic transmission and neurotransmitter levels in the synaptic cleft [15][16][17], and regulate neurogenesis in the adult hippocampus [17][18]. Thus, astrocytes are key actors in the processes, the deregulation of which is considered as an important component of the pathogenesis of depression. Depression is a frequent symptom of AD and may precede the manifestation of AD symptoms. Stress is an important risk factor for depression [19][20], while there are no direct data on whether stress is a risk factor for AD. The etiology and mechanisms of development of both pathologies are obviously complex and unclear, but the question arises whether there are common features in relation to astrocytes and stress response.
2. The Role of Astrocytes in the Functioning of Neurons
For a long time, it was believed that the human brain contains about 100 billion neurons and about one trillion glial cells (a ratio of 1:10). However, recent studies using more advanced cell counting methods have shown that the number of glial cells in the human brain is approximately equal to the number of neurons and ranges from 40 to 130 billion [21]. A characteristic feature of glial cells, both in the brain and on the periphery, is the lack of the ability to generate and conduct nerve impulses [22]. There are three main types of glial cells: astrocytes, microglia and myelin-producing cells (oligodendrocytes in the central nervous system (CNS) and Schwann cells in the peripheral nervous system). Astrocytes and oligodendrocytes are the most common type of glial cells in the CNS. For quite a long period of time, the main function of astrocytes was considered to be the passive support of neurons that transmit nerve impulses, process and store information in the brain. Currently, such views have been revised, and astrocytes are assigned an important role both in the normal functioning of the CNS and in the development of various pathologies [23][24][25].
In the brain, astrocytes are a structural component of the so-called tripartite synapse, which includes, in addition to astrocytes, pre- and postsynaptic endings of neurons [26][27][28]. The morphological complexity of these cells should be particularly noted. Recent data show that one mature rodent astrocyte covers from 20,000 to 80,000 μ3 of domain space in the brain, and at the same time can interact with 300–600 neuronal dendrites [29]. Moreover, morphological studies found that mature astrocytes are able to interact with many thousands of synapses and at the same time are able to unite with other astrocytes, occupying unique spatial regions in the brain [30]. Figure 2 illustrates some functions of astrocytes in the tripartite synapse.
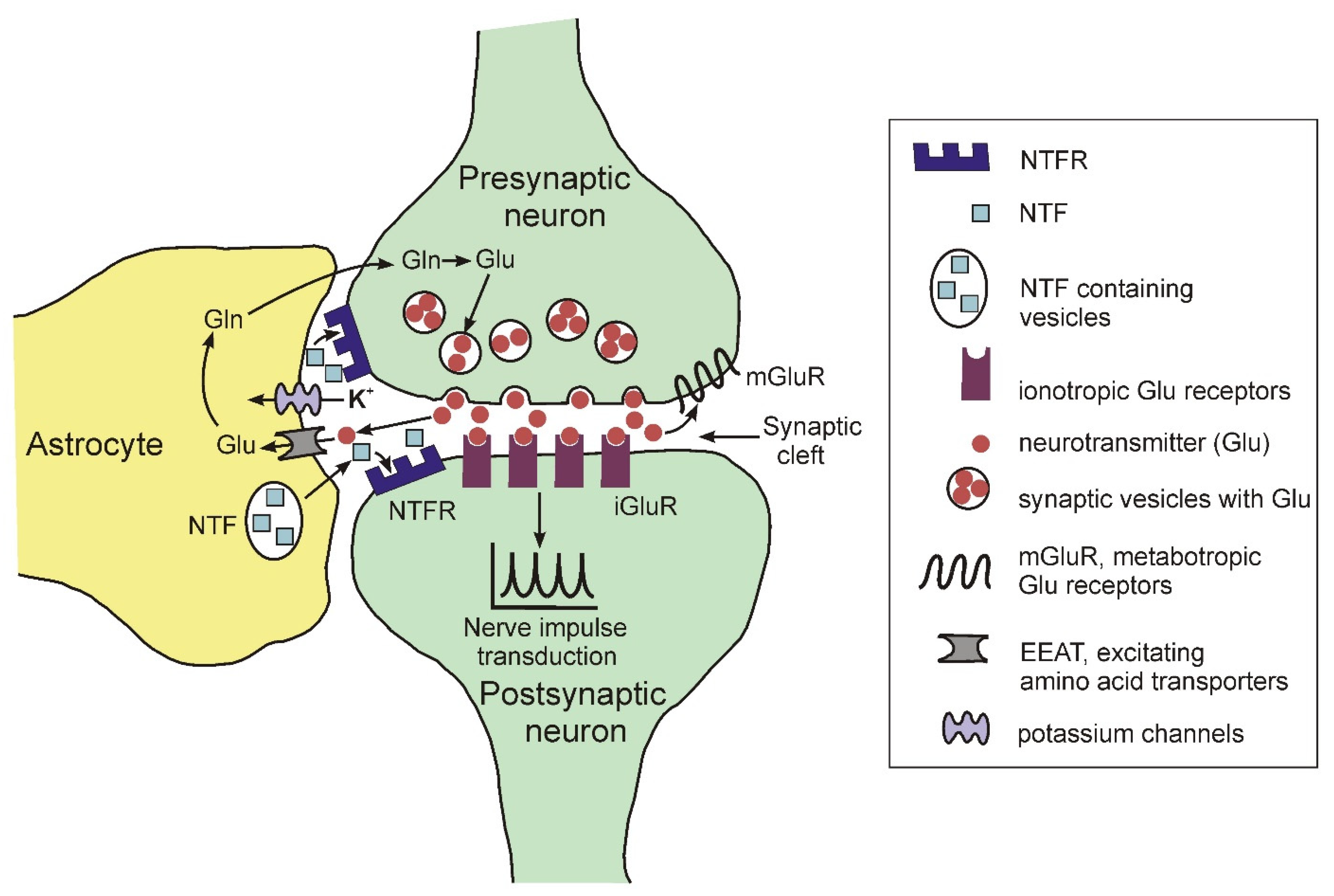 Figure 2. Schematic representation of the structure of the tripartite synapse.
Figure 2. Schematic representation of the structure of the tripartite synapse.The glutaminergic presynaptic neuron is shown as an example. The presynaptic terminal and postsynaptic neuron with astrocyte processes surrounding the synapse are shown. Glutamate is released into the synaptic cleft and activates iGluR on the postsynaptic membrane, facilitating further transmission of the nerve impulse. In addition, glutamate can also exert its effect through metabotropic receptors (mGluR) localized on the presynaptic membrane. Astrocytes remove excess glutamate from the synaptic cleft using the EEAT membrane transporter. Astrocytes express numerous neurotrophic factors (NTFS) that act through the corresponding receptors.
Among the most important functions of astrocytes, in addition to the structural support of neurons, the following should be noted:
-
The elimination of neurotransmitters such as glutamate, gamma-aminobutyric acid, dopamine, norepinephrine from the synaptic cleft during the transmission of a nerve impulse using specific transporters. For glutamate, EEAT serves as a transporter [31].
-
The regulation of the concentration of potassium ions in the synaptic cleft using specific inward K+ channels Kir 4.1. (for review see: [32]).
-
The expression and secretion of neurotrophic factors regulated the functioning and the viability of neurons, such as BDNF (brain-derived neurotrophic factor), FGF (fibroblast growth factor), NGF (nerve growth factor), GDNF (glial cell-derived neurotrophic factor), etc. (for review, see [33]).
In addition, astrocytes modulate the functioning of surrounding neurons by releasing gliotransmitters such as glutamate, ATP (adenosine triphosphate) and D-serine from cells. Calcium ions play an important role in this process [34][35]. It has now been established that astrocytes are also involved in the regulation of the neurogenesis in the adult hippocampus [17][18].
The mechanisms by which astrocytes modulate neural homeostasis, synaptic plasticity and memory are still poorly understood. It is known that astrocytes form intercellular networks by interaction of connexin 30 (Cx30) and connexin 43 (Cx43) proteins of gap junctions. In double-knockout mice with Cx30 and Cx43, sensorimotor disorders and a complete lack of spatial learning and memory were revealed, which shows that astrocytic connexins and an intact astroglial network in the brains of adult animals are important for maintaining neural homeostasis, plasticity and memory formation [36]. In general, astrocytes are key regulators of processes occurring in the nervous system, impairment of which can be considered as important components of various pathologies of the CNS, including AD and depression.
3. Astrocytes Atrophy in Depression and AD
Since astrocytes play a crucial role in the normal functioning of neurons, the question arises whether astrocytes undergo changes during depression and AD and the consequences of chronic stress. Astrocytes express receptors for all stress hormones and therefore are a target for them both in normal stress response and in conditions of impaired self-regulation of HPAA activity. Neuroimaging data show that in depression there are noticeable structural changes in a number of brain regions—first of all, a significant decrease in the volume of the closely interconnected medial PFC and hippocampus [14][16][37]. Apparently, such changes accompany a long-term and serious pathology, since a decrease in the volume of the hippocampus is observed only in cases of depression lasting longer than 2 years or in repeated episodes of the disease [38]. The interaction between the medial PFC and hippocampus integrates motivation, attention, memory and the results of past actions as the relevant circumstances change, which ensures adaptive behavior and mental health [39].
Atrophy of these brain regions is considered as a significant predictor of the development of clinical dementia [40][41][42]. Consequently, atrophy of the medial PFC and hippocampus may be associated with pre-existing depression and/or with early stages of dementia development. A decrease in the volume of these brain regions in patients with depression may be associated with both a decrease in astrocyte density [43][44], and neuronal atrophy [44][45] and a decrease in the number and functioning of synapses [37][46]. This brings depression closer to neurodegenerative diseases. Data on the presence of changes in the density of astrocytes and their sizes in various brain regions in depression are contradictory [47]. However, in particular, a decrease in astrocyte density in the hippocampus in patients with depression [43][48] or who received chronic treatment with glucocorticoids [48] has been shown. It should be noted that there is a significant problem in the identification of astrocytes, because astrocyte marker GFAP (glial fibrillary acidic protein) used in most studies can identify not all, and sometimes only a small part of astrocytes in tissues [49], which can lead to significant difficulties in assessing real differences in astrocyte density in brain tissues in various pathological conditions. In addition, a decrease in GFAP expression in astrocytes during depression can potentially lead to a decrease in the number of detected GFAP-positive cells [44][47].
A recent study shows that a decrease in hippocampal volume is associated with significant neurodegeneration in the CA1 region of the hippocampus at advanced stages of AD [50]. Additionally, there are indications of astrocyte atrophy in patients with advanced stages of AD [51]. Interestingly, astrocytes obtained from induced pluripotent stem cells (IPSC) of patients not only with familial, but also with a sporadic form of AD (the only patient studied), also showed features of atrophy in vitro in comparison with control astrocytes [52]. With regard to depression, a recent study using astrocytes differentiated from IPSC of healthy and depressed donors (treated with SSRIs) did not reveal morphological differences between cell lines, as well as significant differences in the expression of astrocytic markers and astrocytic glutamate transporter (EAAT2), and in the transcriptome profile [53]. There are still insufficient data on whether there is a correlation between the clinical manifestations of sporadic AD or depression and atrophy or other changes in astrocytes obtained from iPSC. However, it has already been shown that IPSC-derived human astrocytes carrying APOE ε4/ε4 genotype are less efficient than those with APOE ε3/ε3 in neuronal survival and synaptic integrity [54] and in the uptake and clearance of Aβ [55]. Research in this direction may shed light on how genetic risk factors for AD affect the functioning of astrocytes. Considering that the most significant genetic risk factor for sporadic AD is associated with the expression of isoforms of apolipoprotein E, the main source of which in the brain are astrocytes, this could help to identify the influence on the development of AD of the interaction of environmental factors, such as stress, with a genetic predisposition to AD.
References
- Selye, H. Forty years of stress research: Principal remaining problems and misconceptions. Can. Med. Assoc. J. 1976, 115, 53–56.
- Kazakou, P.; Nicolaides, N.C.; Chrousos, G.P. Basic Concepts and Hormonal Regulators of the Stress System. Horm. Res. Paediatr. 2022, 95.
- World Health Organization. Available online: http://www.who.int/news-room/fact-sheets/detail/depression (accessed on 12 April 2022).
- van’t Veer-Tazelaar, P.J.; van Marwijk, H.W.; Jansen, A.P.; Rijmen, F.; Kostense, P.J.; van Oppen, P.; van Hout, H.P.; Stalman, W.A.; Beekman, A.T. Depression in old age (75+), the PIKO study. J. Affect. Disord. 2008, 106, 295–299.
- Kennedy, S.H. Core symptoms of major depressive disorder: Relevance to diagnosis and treatment. Dialogues Clin. Neurosci. 2008, 10, 271–277.
- Alzheimer’s Association. 2021 Alzheimer’s disease facts and figures. Alzheimer’s Dement. J. Alzheimer’s Assoc. 2021, 17, 327–406.
- Tahami Monfared, A.A.; Byrnes, M.J.; White, L.A.; Zhang, Q. Alzheimer’s Disease: Epidemiology and Clinical Progression. Neurol. Ther. 2022, 11, 1–17.
- Niu, H.; Álvarez-Álvarez, I.; Guillén-Grima, F.; Aguinaga-Ontoso, I. Prevalence and incidence of Alzheimer’s disease in Europe: A meta-analysis. Neurologia 2017, 32, 523–532.
- Roda, A.R.; Serra-Mir, G.; Montoliu-Gaya, L.; Tiessler, L.; Villegas, S. Amyloid-beta peptide and tau protein crosstalk in Alzheimer’s disease. Neural. Regen Res. 2022, 17, 1666–1674.
- Pitas, R.E.; Boyles, J.K.; Lee, S.H.; Foss, D.; Mahley, R.W. Astrocytes synthesize apolipoprotein E and metabolize apolipoprotein E-containing lipoproteins. Biochim. Biophys. Acta 1987, 917, 148–161.
- Myers, R.H.; Schaefer, E.J.; Wilson, P.W.; D’Agostino, R.; Ordovas, J.M.; Espino, A.; Au, R.; White, R.F.; Knoefel, J.E.; Cobb, J.L.; et al. Apolipoprotein E epsilon4 association with dementia in a population-based study: The Framingham study. Neurology 1996, 46, 673–677.
- Efthymiou, A.G.; Goate, A.M. Late onset Alzheimer’s disease genetics implicates microglial pathways in disease risk. Mol. Neurodegener. 2017, 12, 43.
- Lanoiselée, H.M.; Nicolas, G.; Wallon, D.; Rovelet-Lecrux, A.; Lacour, M.; Rousseau, S.; Richard, A.C.; Pasquier, F.; Rollin-Sillaire, A.; Martinaud, O.; et al. APP, PSEN1, and PSEN2 mutations in early-onset Alzheimer disease: A genetic screening study of familial and sporadic cases. PLoS Med. 2017, 14, e1002270.
- Murphy-Royal, C.; Gordon, G.R.; Bains, J.S. Stress-induced structural and functional modifications of astrocytes-Further implicating glia in the central response to stress. Glia 2019, 67, 1806–1820.
- Zhou, X.; Xiao, Q.; Xie, L.; Yang, F.; Wang, L.; Tu, J. Astrocyte, a Promising Target for Mood Disorder Interventions. Front. Mol. Neurosci. 2019, 12, 136.
- Marathe, S.V.; D’Almeida, P.L.; Virmani, G.; Bathini, P.; Alberi, L. Effects of Monoamines and Antidepressants on Astrocyte Physiology: Implications for Monoamine Hypothesis of Depression. J. Exp. Neurosci. 2018, 12, 1179069518789149.
- Quesseveur, G.; Gardier, A.M.; Guiard, B.P. The monoaminergic tripartite synapse: A putative target for currently available antidepressant drugs. Curr. Drug Targets 2013, 14, 1277–1294.
- Cassé, F.; Richetin, K.; Toni, N. Astrocytes’ Contribution to Adult Neurogenesis in Physiology and Alzheimer’s Disease. Front. Cell. Neurosci. 2018, 12, 432.
- Nestler, E.J.; Barrot, M.; DiLeone, R.J.; Eisch, A.J.; Gold, S.J.; Monteggia, L.M. Neurobiology of depression. Neuron 2002, 34, 13–25.
- Hammen, C. Stress and Depression. Annu. Rev. Clin. Psychol. 2005, 1, 293–319.
- von Bartheld, C.S.; Bahney, J.; Herculano-Houzel, S. The search for true numbers of neurons and glial cells in the human brain: A review of 150 years of cell counting. J. Comp. Neurol. 2016, 524, 3865–3895.
- Fields, R.D.; Araque, A.; Johansen-Berg, H.; Lim, S.S.; Lynch, G.; Nave, K.A.; Nedergaard, M.; Perez, R.; Sejnowski, T.; Wake, H. Glial biology in learning and cognition. Neurosci. A Rev. J. BringingNeurobiol. Neurol. Psychiatry 2014, 20, 426–431.
- Barres, B.A. The mystery and magic of glia: A perspective on their roles in health and disease. Neuron 2008, 60, 430–440.
- Newman, E.A. New roles for astrocytes: Regulation of synaptic transmission. Trends Neurosci. 2003, 26, 536–542.
- Verkhratsky, A.; Parpura, V.; Vardjan, N.; Zorec, R. Physiology of Astroglia. Adv. Exp. Med. Biol. 2019, 1175, 45–91.
- Halassa, M.M.; Fellin, T.; Haydon, P.G. The tripartite synapse: Roles for gliotransmission in health and disease. Trends Mol. Med. 2007, 13, 54–63.
- Perea, G.; Navarrete, M.; Araque, A. Tripartite synapses: Astrocytes process and control synaptic information. Trends Neurosci. 2009, 32, 421–431.
- Santello, M.; Calì, C.; Bezzi, P. Gliotransmission and the tripartite synapse. Adv. Exp. Med. Biol. 2012, 970, 307–331.
- Halassa, M.M.; Fellin, T.; Takano, H.; Dong, J.-H.; Haydon, P.G. Synaptic islands defined by the territory of a single astrocyte. J. Neurosci. Off. J. Soc. Neurosci. 2007, 27, 6473–6477.
- Freeman, M.R. Specification and morphogenesis of astrocytes. Science 2010, 330, 774–778.
- Canul-Tec, J.C.; Kumar, A.; Dhenin, J.; Assal, R.; Legrand, P.; Rey, M.; Chamot-Rooke, J.; Reyes, N. The ion-coupling mechanism of human excitatory amino acid transporters. EMBO J. 2022, 41, e108341.
- Benarroch, E.E. Astrocyte signaling and synaptic homeostasis: I: Membrane channels, transporters, and receptors in astrocytes. Neurology 2016, 87, 324–330.
- Pöyhönen, S.; Er, S.; Domanskyi, A.; Airavaara, M. Effects of Neurotrophic Factors in Glial Cells in the Central Nervous System: Expression and Properties in Neurodegeneration and Injury. Front. Physiol. 2019, 10, 486.
- Bezzi, P.; Volterra, A. A neuron-glia signalling network in the active brain. Curr. Opin. Neurobiol. 2001, 11, 387–394.
- Araque, A.; Carmignoto, G.; Haydon, P.G.; Oliet, S.H.; Robitaille, R.; Volterra, A. Gliotransmitters travel in time and space. Neuron 2014, 81, 728–739.
- Hösli, L.; Binini, N.; Ferrari, K.D.; Thieren, L.; Looser, Z.J.; Zuend, M.; Zanker, H.S.; Berry, S.; Holub, M.; Möbius, W.; et al. Decoupling astrocytes in adult mice impairs synaptic plasticity and spatial learning. Cell Rep. 2022, 38.
- Duman, C.H.; Duman, R.S. Spine synapse remodeling in the pathophysiology and treatment of depression. Neurosci. Lett. 2015, 601, 20–29.
- McKinnon, M.C.; Yucel, K.; Nazarov, A.; MacQueen, G.M. A meta-analysis examining clinical predictors of hippocampal volume in patients with major depressive disorder. J. Psychiatry Neurosci. JPN 2009, 34, 41–54.
- Dolleman-van der Weel, M.J.; Griffin, A.L.; Ito, H.T.; Shapiro, M.L.; Witter, M.P.; Vertes, R.P.; Allen, T.A. The nucleus reuniens of the thalamus sits at the nexus of a hippocampus and medial prefrontal cortex circuit enabling memory and behavior. Learn Mem. 2019, 26, 191–205.
- den Heijer, T.; van der Lijn, F.; Koudstaal, P.J.; Hofman, A.; van der Lugt, A.; Krestin, G.P.; Niessen, W.J.; Breteler, M.M. A 10-year follow-up of hippocampal volume on magnetic resonance imaging in early dementia and cognitive decline. Brain A J. Neurol. 2010, 133, 1163–1172.
- Burgmans, S.; van Boxtel, M.P.; Smeets, F.; Vuurman, E.F.; Gronenschild, E.H.; Verhey, F.R.; Uylings, H.B.; Jolles, J. Prefrontal cortex atrophy predicts dementia over a six-year period. Neurobiol. Aging 2009, 30, 1413–1419.
- de Flores, R.; La Joie, R.; Chételat, G. Structural imaging of hippocampal subfields in healthy aging and Alzheimer’s disease. Neuroscience 2015, 309, 29–50.
- Cobb, J.A.; O’Neill, K.; Milner, J.; Mahajan, G.J.; Lawrence, T.J.; May, W.L.; Miguel-Hidalgo, J.; Rajkowska, G.; Stockmeier, C.A. Density of GFAP-immunoreactive astrocytes is decreased in left hippocampi in major depressive disorder. Neuroscience 2016, 316, 209–220.
- Rajkowska, G.; Stockmeier, C.A. Astrocyte pathology in major depressive disorder: Insights from human postmortem brain tissue. Curr. Drug Targets 2013, 14, 1225–1236.
- Stockmeier, C.A.; Mahajan, G.J.; Konick, L.C.; Overholser, J.C.; Jurjus, G.J.; Meltzer, H.Y.; Uylings, H.B.; Friedman, L.; Rajkowska, G. Cellular changes in the postmortem hippocampus in major depression. Biol. Psychiatry 2004, 56, 640–650.
- Price, R.B.; Duman, R. Neuroplasticity in cognitive and psychological mechanisms of depression: An integrative model. Mol. Psychiatry 2020, 25, 530–543.
- Dossi, E.; Vasile, F.; Rouach, N. Human astrocytes in the diseased brain. Brain Res. Bull. 2018, 136, 139–156.
- Müller, M.B.; Lucassen, P.J.; Yassouridis, A.; Hoogendijk, W.J.; Holsboer, F.; Swaab, D.F. Neither major depression nor glucocorticoid treatment affects the cellular integrity of the human hippocampus. Eur. J. Neurosci. 2001, 14, 1603–1612.
- Zhang, Z.; Ma, Z.; Zou, W.; Guo, H.; Liu, M.; Ma, Y.; Zhang, L. The Appropriate Marker for Astrocytes: Comparing the Distribution and Expression of Three Astrocytic Markers in Different Mouse Cerebral Regions. BioMed Res. Int. 2019, 2019, 9605265.
- Gonzalez-Rodriguez, M.; Villar-Conde, S.; Astillero-Lopez, V.; Villanueva-Anguita, P.; Ubeda-Banon, I.; Flores-Cuadrado, A.; Martinez-Marcos, A.; Saiz-Sanchez, D. Neurodegeneration and Astrogliosis in the Human CA1 Hippocampal Subfield Are Related to hsp90ab1 and bag3 in Alzheimer’s Disease. Int. J. Mol. Sci. 2021, 23, 165.
- Verkhratsky, A.; Rodrigues, J.J.; Pivoriunas, A.; Zorec, R.; Semyanov, A. Astroglial atrophy in Alzheimer’s disease. Pflug. Arch. Eur. J. Physiol. 2019, 471, 1247–1261.
- Jones, V.C.; Atkinson-Dell, R.; Verkhratsky, A.; Mohamet, L. Aberrant iPSC-derived human astrocytes in Alzheimer’s disease. Cell Death Dis. 2017, 8, e2696.
- Heard, K.J.; Shokhirev, M.N.; Becronis, C.; Fredlender, C.; Zahid, N.; Le, A.T.; Ji, Y.; Skime, M.; Nelson, T.; Hall-Flavin, D.; et al. Chronic cortisol differentially impacts stem cell-derived astrocytes from major depressive disorder patients. Transl Psychiatry 2021, 11, 608.
- Zhao, J.; Davis, M.D.; Martens, Y.A.; Shinohara, M.; Graff-Radford, N.R.; Younkin, S.G.; Wszolek, Z.K.; Kanekiyo, T.; Bu, G. APOE ε4/ε4 diminishes neurotrophic function of human iPSC-derived astrocytes. Hum. Mol. Genet. 2017, 26, 2690–2700.
- Lin, Y.T.; Seo, J.; Gao, F.; Feldman, H.M.; Wen, H.L.; Penney, J.; Cam, H.P.; Gjoneska, E.; Raja, W.K.; Cheng, J.; et al. APOE4 Causes Widespread Molecular and Cellular Alterations Associated with Alzheimer’s Disease Phenotypes in Human iPSC-Derived Brain Cell Types. Neuron 2018, 98, 1141–1154.
More
Information
Subjects:
Neurosciences
Contributors
MDPI registered users' name will be linked to their SciProfiles pages. To register with us, please refer to https://encyclopedia.pub/register
:
View Times:
1.4K
Entry Collection:
Neurodegeneration
Revisions:
2 times
(View History)
Update Date:
20 May 2022
Notice
You are not a member of the advisory board for this topic. If you want to update advisory board member profile, please contact office@encyclopedia.pub.
OK
Confirm
Only members of the Encyclopedia advisory board for this topic are allowed to note entries. Would you like to become an advisory board member of the Encyclopedia?
Yes
No
${ textCharacter }/${ maxCharacter }
Submit
Cancel
Back
Comments
${ item }
|
More
No more~
There is no comment~
${ textCharacter }/${ maxCharacter }
Submit
Cancel
${ selectedItem.replyTextCharacter }/${ selectedItem.replyMaxCharacter }
Submit
Cancel
Confirm
Are you sure to Delete?
Yes
No





