
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Dóra Hamar-Farkas | -- | 8669 | 2022-05-13 11:49:56 | | | |
| 2 | Jason Zhu | -91 word(s) | 8578 | 2022-05-16 04:54:56 | | | | |
| 3 | Jason Zhu | -617 word(s) | 7961 | 2022-05-16 08:46:35 | | | | |
| 4 | Dóra Hamar-Farkas | Meta information modification | 7961 | 2023-05-12 10:17:03 | | |
Video Upload Options
The biostimulant segment is becoming increasingly important worldwide. One of the reasons for this is that fewer plant protection products are placed on the market in the European Union, and environmental sustainability also plays an important role in their use. Biostimulants are often used in several horticultural sectors, including ornamentals, to strengthen plants, achieve commercial standards, produce quality goods, increase plant vitality, and aid harvesting.
1. Introduction
|
|
Filatov, 1951b |
Ikrina and Kolbin, 2004 |
Kauffman et al., 2007 |
Du Jardin, 2012 |
Calvo et al., 2014 |
Halpern et al., 2015 |
Du Jardin, 2015 |
Torre et al., 2016 |
|
1 |
Carboxylic fatty acids (oxalic acid and succiric acid) |
Microorganisms (bacteria, fungi) |
Humic substances |
Humic substances |
Microbial inoculants |
Humic substances |
Humic and fulvic acids |
Humic substances |
|
2 |
Carboxylic fatty hydroxyl acids (malic and tartaric acids) |
Plant materials (land, freshwater and marine) |
Hormone containing products (seaweed extracts) |
Complex organic materials |
Humic acids |
Protein hydrolysate and amino acid formulations |
Protein hydrolysates and other N-containing compounds |
Seaweed extracts |
|
3 |
Unsaturated fatty acids, aromatic and phenolic acids (cinnamic and hydroxycinnamic acids, coumarin) |
Sea shellfish, animals, bees |
Amino acid containing products |
Beneficial chemical elements |
Fulvic acids |
Seaweed extract |
Seaweed extracts and botanicals |
Hydrolyzed proteins and amino acids |
|
4 |
Phenolic aromatic acids containing several benzene rings linked via carbon atoms (humic acids) |
Humate- and humus-containing substances |
- |
Inorganic salts (such as phosphite) |
Protein hydrolysates and amino acids |
Plant-growth-promoting microorganism (including mycorrhizal fungi) |
Chitosan and other biopolymers |
Inorganic salts |
|
5 |
- |
Vegetable oils |
- |
Seaweed extracts |
Seaweed extracts |
- |
Inorganic compounds |
Microorganisms |
|
6 |
- |
Natural minerals |
- |
Chitin and chitosan derivatives |
- |
- |
Beneficial fungi |
- |
|
7 |
- |
Water (activated, degassed, thermal) |
- |
Free amino acids and other N-containing substances |
- |
- |
Beneficial bacteria |
- |
|
8 |
- |
Resins |
- |
|
- |
- |
- |
- |
|
9 |
- |
Other raw materials (oil and petroleum fraction, shale substance |
- |
- |
- |
- |
- |
- |
2. Groups of Biostimulants
2.1. Industrial By-Products: Protein Hydrolysates and Chitosans
2.2. Humic and Fulvic Acids
2.3. Algae Extracts
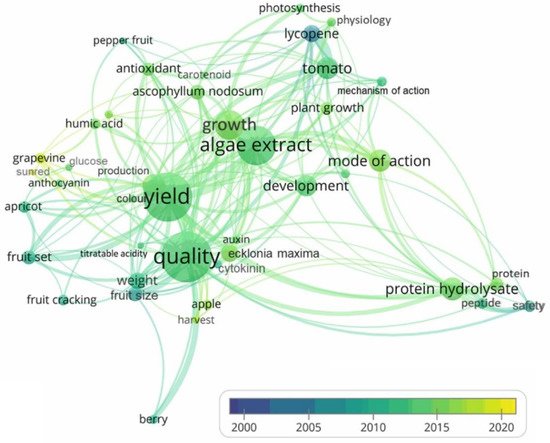
2.4. PGPR and PGPB
2.5. Fungal Inoculants
3. Abiotic and Biotic Stress and the Response of Ornamental Plants to Biostimulant Treatment
4. Effects and Application of Biostimulants in Ornamental Horticulture
4.1. The Role of Biostimulants in Ornamental Plant Production
4.2. The Role of Biostimulants in the Propagation of Ornamental Plants
4.3. Effect of Biostimulants on Plant Growth and Development
4.4. Post-Harvest Treatment of Ornamental Plants with the Use of Biostimulants
References
- Schmidt, R.E. Questions and Answers about Biostimulants; Hi Tech Ag Solutions: Davenport, WA, USA, 2003; p. 4.
- Zhang, X.; Schmidt, R.E. The impact of growth regulators on alpha-tocopherol status of water-stressed Poa pratensis L. Int. Turfgrass Soc. Res. J. 1997, 8, 1364–1373.
- Calvo, P.; Nelson, L.; Kloepper, J.W. Agricultural uses of plant biostimulants. Plant Soil 2014, 383, 3–41.
- Du Jardin, P. Plant biostimulants: Definition, concept, main categories and regulation. Sci. Hortic. 2015, 196, 3–14.
- Biostimulant Coalition. What Are Biostimulants? 2013. Available online: http://www.biostimulantcoalition.org/about/ (accessed on 9 February 2022).
- European Commission. Proposal for a Regulation Laying Down Rules on the Making Available on the Market of CE Marked Fertilizing Products and Amending Regulations (EC)1069/2009 and (EC)1107/2009.COM 2016; European Commission: Brussels, Belgium, 2016; p. 157.
- Council of the European Union. Proposal for a Regulation of the European Parliament and of the Council Laying Down Rules on the Making Available on the Market of CE Marked Fertilizing Products and Amending Regulations (EC) No 1069/2009 and (EC) No 1107/2009-Analysis of the Final Compromise Text with a View to Agreement. 2018. Available online: http://data.consilium.europa.eu/doc/document/ST-15103-2018-INIT/en/pdf (accessed on 20 December 2018).
- Rouphael, Y.; Colla, G. Biostimulants in agriculture. Front. Plant Sci. 2020, 11, 40.
- Zulfiqar, F.; Younis, A.; Finnegan, P.M.; Ferrante, A. Comparison of Soaking Corms with Moringa Leaf Extract Alone or in Combination with Synthetic Plant Growth Regulators on the Growth, Physiology and Vase Life of Sword Lily. Plants 2020, 9, 1590.
- Van Oosten, M.J.; Pepe, O.; De Pascale, S.; Silletti, S.; Maggio, A. The role of biostimulants and bioeffectors as alleviators of abiotic stress in crop plants. Chem. Biol. Technol. 2017, 4, 5.
- Toscano, S.; Romano, D.; Massa, D.; Bulgari, R.; Franzoni, G.; Ferrante, A. Biostimulant applications in low input horticultural cultivation systems. Italus Hortus 2018, 25, 27–36.
- Halpern, M.; Bar-Tal, A.; Ofek, M.; Minz, D.; Muller, T.; Yermiyahu, U. Chapter Two—The Use of Biostimulants for Enhancing Nutrient Uptake. In Advances in Agronomy; Sparks, D.L., Ed.; Academic Press: New York, NY, USA, 2015; pp. 141–174.
- Drobek, M.; Frąc, M.; Cybulska, J. Plant Biostimulants: Importance of the Quality and Yield of Horticultural Crops and the Improvement of Plant Tolerance to Abiotic Stress—A Review. Agronomy 2019, 9, 335.
- Kocira, A.; Świeca, M.; Kocira, S.; Złotek, U.; Jakubczyk, A. Enhancement of yield, nutritional and nutraceutical properties of two common bean cultivars following the application of seaweed extract (Ecklonia maxima). Saudi J. Biol. Sci. 2018, 25, 563–571.
- Abbott, L.K.; Macdonald, L.M.; Wong, M.T.F.; Webb, M.J.; Jenkins, S.N.; Farrell, M. Potential roles of biological amendments for profitable grain production—A review. Agric. Ecosyst. Environ. 2018, 256, 34–50.
- Yakhin, O.I.; Lubyanov, A.A.; Yakhin, I.A.; Brown, P.H. Biostimulants in Plant Science: A Global Perspective. Front. Plant Sci. 2017, 7, 2049.
- Caradonia, F.; Battaglia, V.; Righi, L.; Pascali, G.; La Torre, A. Plant Biostimulant Regulatory Framework: Prospects in Europe and Current Situation at International Level. J. Plant Growth Regul. 2019, 38, 438–448.
- Ertani, A.; Sambo, P.; Nicoletto, C.; Santagata, S.; Schiavon, M.; Nardi, S. The Use of Organic Biostimulants in Hot Pepper Plants to Help Low Input Sustainable Agriculture. Chem. Biol. Technol. Agric. 2015, 2, 11.
- Chen, S.K.; Subler, S.; Edwards, C.A. Effects of Agricultural Biostimulants on Soil Microbial Activity and Nitrogen Dynamics. Agric. Ecosyst. Environ. Appl. Soil Ecol. 2002, 19, 249–259.
- Lau, S.E.; Teo, W.F.A.; Teoh, E.Y.; Tan, B.C. Microbiome Engineering and Plant Biostimulants for Sustainable Crop Improvement and Mitigation of Biotic and Abiotic Stresses. Discover Food 2022, 2, 9.
- Dmytryk, A.; Chojnacka, K. Algae as fertilizers, biostimulants, and regulators of plant growth. In Algae Biomass: Characteristics and Applications; Springer: Cham, Switzerland, 2018; pp. 115–122.
- Wiszniewska, A.; Nowak, B.; Kołton, A.; Sitek, E.; Grabski, K.; Dziurka, M.; Długosz-Grochowska, O.; Dziurka, K.; Tukaj, Z. Rooting Response of Prunus domestica L. Microshoots in the Presence of Phytoactive Medium Supplements. Plant Cell Tissue Organ Cult. 2016, 125, 163–176.
- da Silva, J.A.T.; Pacholczak, A.; Ilczuk, A. Smoke Tree (Cotinus coggygria Scop.) Propagation and Biotechnology: A Mini-review. S. Afr. J. Bot. 2018, 114, 232–240.
- de Saeger, J.; Van Praet, S.; Vereecke, D.; Park, J.; Jacques, S.; Han, T.; Depuydt, S. Toward the Molecular Understanding of the Action Mechanism of Ascophyllum nodosum Extracts on Plants. J. Appl. Phycol. 2020, 32, 573–597.
- EUR-Lex. Available online: https://eur-lex.europa.eu/legal-content/EN/TXT/?uri=OJ:L:2019:170:TOC (accessed on 21 January 2021).
- Al-Juthery, H.W.A.; Abbas Drebee, H.; Al-Khafaji, B.M.K.; Hadi, R.F. Plant Biostimulants, Seaweeds Extract as a Model (Article Review). IOP Conf. Ser. Earth Environ. Sci. 2020, 553, 012015.
- Sible, C.N.; Seebauer, J.R.; Below, F.E. Plant Biostimulants: A Categorical Review, Their Implications for Row Crop Production, and Relation to Soil Health Indicators. Agronomy 2021, 11, 1297.
- Parađiković, N.; Teklić, T.; Zeljković, S.; Lisjak, M.; Špoljarević, M. Biostimulants research in some horticultural plant species—A review. Food Energy Secur. 2019, 8, e00162.
- Colla, G.; Rouphael, Y. Biostimulants in Horticulture. Sci. Hortic. 2015, 196, 1–134.
- Bulgari, R.; Franzoni, G.; Ferrante, A. Biostimulants application in horticultural crops under abiotic stress conditions. Agronomy 2019, 9, 306.
- Bhupenchandra, I.; Devi, S.H.; Basumatary, A.; Dutta, S.; Singh, L.K.; Kalita, P.; Bora, S.S.; Devi, S.R.; Saikia, A.; Sharma, P.; et al. Biostimulants: Potential and Prospects in Agriculture. Int. Res. J. Pure Appl. Chem. 2020, 21, 20–35.
- Nephali, L.; Piater, L.A.; Dubery, I.A.; Patterson, V.; Huyser, J.; Burgess, K.; Tugizimana, F. Biostimulants for Plant Growth and Mitigation of Abiotic Stresses: A Metabolomics Perspective. Metabolites 2020, 10, 505.
- Xu, L.; Geelen, D. Developing Biostimulants From Agro-Food and Industrial By-Products. Front Plant Sci. 2018, 9, 1567.
- El Boukhari, M.E.M.; Barakate, M.; Bouhia, Y.; Lyamlouli, K. Trends in Seaweed Extract Based Biostimulants: Manufacturing Process and Beneficial Effect on Soil-Plant Systems. Plants 2020, 9, 359.
- Critchley, A.T.; Critchley, J.S.C.; Norrie, J.; Gupta, S.; Van Staden, J. Chapter 13—Perspectives on the global biostimulant market: Applications, volumes, and values, 2016 data and projections to 2022. In Biostimulants for Crops from Seed Germination to Plant Development; Gupta, S., Van Staden, J., Eds.; Academic Press: New York, NY, USA, 2021; pp. 289–296.
- Marketsandmarkets.com. Biostimulants Market by Active Ingredient (Humic Substances, Seaweed, Microbials, Trace Minerals, Vitamins & Amino Acids), Crop Type (Row Crops, Fruits & Vegetables, Turf & Ornamentals), Formulation, Application Method, and Region—Global Forecast to 2022; MarketsandMarkets Inc.: Pune, India, 2017.
- Karagöz, F.P.; Dursun, A.; Tekiner, N.; Kul, R.; Kotan, R. Efficacy of Vermicompost and/or Plant Growth Promoting Bacteria on the Plant Growth and Development in Gladiolus. J. Ornam. Hortic. 2019, 25, 180–188.
- Allardice, R.P.; Kapp, C.; Botha, A.; és Valentine, A. A vermikomposzt koncentrációjának optimalizálása a hüvelyes lupinus angustifolius nitrogén táplálására és termelésére. Compos. Sci. Util. 2015, 23, 217–236.
- Ngoroyemoto, N.; Gupta, S.; Kulkarni, M.G.; Finnie, J.F.; Van Staden, J. Effect of organic biostimulants on the growth and biochemical composition of Amaranthus hybridus L. S. Afr. J. Bot. 2019, 124, 87–93.
- Moyo, M.; Aremu, A.O.; Amoo, S.O. Potential of seaweed extracts and humate-containing biostimulants in mitigating abiotic stress in plants. In Biostimulants for Crops from Seed Germination to Plant Development; Gupta, S., Van Staden, J., Eds.; Academic Press: New York, NY, USA, 2021; Chapter 14; pp. 297–332.
- Petropoulos, S.A. Practical applications of plant biostimulants in greenhouse vegetable crop production. Agronomy 2020, 10, 1569.
- Colla, G.; Hoagland, L.; Ruzzi, M.; Cardarelli, M.; Bonini, P.; Canaguier, R.; Rouphael, Y. Biostimulant Action of Protein Hydrolysates: Unraveling Their Effects on Plant Physiology and Microbiome. Front. Plant Sci. 2017, 8, 2202.
- Li, K.; Xing, R.; Liu, S.; Li, P. Chitin and Chitosan Fragments Responsible for Plant Elicitor and Growth Stimulator. J. Agric. Food Chem. 2020, 68, 12203–12211.
- Nguyen, H.T.; Boonyaritthongchai, P.; Buanong, M.; Supapvanich, S.; Wongs-Aree, C. Chitosan- and κ-carrageenan-based composite coating on dragon fruit (Hylocereus undatus) pretreated with plant growth regulators maintains bract chlorophyll and fruit edibility. Sci. Hortic. 2021, 281, 109916.
- Shahrajabian, M.H.; Chaski, C.; Polyzos, N.; Tzortzakis, N.; Petropoulos, S.A. Sustainable agriculture systems in vegetable production using chitin and chitosan as plant biostimulants. Biomolecules 2021, 11, 819.
- El Hadrami, A.; Adam, L.R.; El Hadrami, I.; Daayf, F. Chitosan in plant protection. Mar. Drugs 2010, 8, 968–987.
- Hidangmayum, A.; Dwivedi, P.; Katiyar, D.; Hemantaranjan, A. Application of chitosan on plant responses with special reference to abiotic stress. Physiol. Mol. Biol. Plants 2019, 25, 313–326.
- Zhang, X.; Li, K.; Xing, R.; Liu, S.; Chen, X.; Yang, H.; Li, P. miRNA and mRNA Expression Profiles Reveal Insight into Chitosan-Mediated Regulation of Plant Growth. J. Agric. Food Chem. 2018, 66, 3810–3822.
- Akter, J.; Jannat, R.; Hossain, M.M.; Ahmed, J.U.; Rubayet, M.T. Chitosan for plant growth promotion and disease suppression against anthracnose in Chilli. IJEAB 2018, 3, 806–817.
- Acemi, A.; Bayrak, B.; Çakır, M.; Demiryürek, E.; Gün, E.; El Gueddari, N.E.; Özen, F. Comparative analysis of the effects of chitosan and common plant growth regulators on in vitro propagation of Ipomoea purpurea (L.) roth from nodal explants. In Vitro Cell. Dev. Biol.-Plant 2018, 54, 537–544.
- Kumaraswamy, R.V.; Kumari, S.; Choudhary, R.C.; Pal, A.; Raliya, R.; Biswas, P.; Saharan, V. Engineered chitosan based nanomaterials: Bioactivities, mechanisms and perspectives in plant protection and growth. Int. J. Biol. Macromol. 2018, 113, 494–506.
- Kumar, S.M.; Zeng, X.; Wang, Y.; Su, S.; Soothar, P.; Bai, L.; Kumar, M.; Zhang, Y.; Mustafa, A.; Ye, N. The Short-Term Effects of Mineral- and Plant-Derived Fulvic Acids on Some Selected Soil Properties: Improvement in the Growth, Yield, and Mineral Nutritional Status of Wheat (Triticum aestivum L.) under Soils of Contrasting Textures. Plants 2020, 9, 205.
- Kumar, H.D.; Aloke, P. Role of biostimulant formulations in crop production: An overview. Int. J. Appl. Res. Vet. Med. 2020, 8, 38–46.
- Vujinović, T.; Zanin, L.; Venuti, S.; Contin, M.; Ceccon, P.; Tomasi, N.; Pinton, R.; Cesco, S.; De Nobili, M. Biostimulant action of dissolved humic substances from a conventionally and an organically managed soil on nitrate acquisition in maize plants. Front. Plant Sci. 2020, 10, 1652.
- Wang, Y.; Yang, R.; Zheng, J.; Shen, Z.; Xu, X. Exogenous foliar application of fulvic acid alleviate cadmium toxicity in lettuce (Lactuca sativa L.). Ecotoxic. Environ. Saf. 2019, 167, 10–19.
- Elansary, H.O.; Mahmoud, E.A.; El-Ansary, D.O.; Mattar, M.A. Effects of Water Stress and Modern Biostimulants on Growth and Quality Characteristics of Mint. Agronomy 2020, 10, 6.
- Ali, S.; Rizwan, M.; Waqas, A.; Hussain, M.B.; Hussain, A.; Liu, S.; Alqarawi, A.A.; Hashem, A.; Abd, A.E.F. Fulvic acid prevents chromium-induced morphological, photosynthetic, and oxidative alterations in wheat irrigated with tannery waste water. J. Plant Growth Regul. 2018, 37, 1357–1367.
- Yildirim, E.; Ekinci, M.; Turan, M.; Ağar, G.; Dursun, A.; Kul, R.; Alim, Z.; Argin, S. Humic + Fulvic acid mitigated Cd adverse effects on plant growth, physiology and biochemical properties of garden cress. Sci Rep 2021, 11, 8040.
- Stevenson, F.J. Humus Chemistry: Genesis, Composition, Reactions; John Wiley & Sons: New York, NY, USA, 1994.
- Chang, L.; Wu, Y.; Xu, W.; Nikbakht, A.; Xia, Y. Effects of Calcium and Humic Acid Treatment on the Growth and Nutrient Uptake of Oriental Lily. Afr. J. 2012, 11, 2218–2222.
- Conselvan, G.B.; Pizzeghello, D.; Francioso, O.; Di Foggia, M.; Nardi, S.; Carletti, P. Biostimulant activity of humic substances extracted from leonardites. Plant Soil 2017, 420, 119–134.
- Savy, D.; Brostaux, Y.; Cozzolino, V.; Delaplace, P.; du Jardin, P.; Piccolo, A. Quantitative structure-activity relationship of humic-like biostimulants derived from agro-industrial byproducts and energy crops. Front. Plant Sci. 2020, 11, 581.
- Vaughan, D.; Malcolm, R.E. Influence of humic substances on growth and physiological processes. In Soil Organic Matter and Biological Activity; Springer: Dordrecht, The Netherlands, 1985; pp. 37–75.
- Nardi, S.; Panuccio, M.R.; Abenavoli, M.R.; Muscolo, A. Auxin-like Effect of Humic Substances Extracted from Faeces of Allolobophora caliginosa and A. rosea. Soil Biol. Biochem. 1994, 26, 1341–1346.
- Elmongy, M.S.; Zhou, H.; Cao, Y.; Liu, B.; Xia, Y. The Effect of Humic Acid on Endogenous Hormone Levels and Antioxidant Enzyme Activity during in Vitro Rooting of Evergreen Azalea. Sci Hortic. 2018, 227, 234–243.
- del Buono, D. Can biostimulants be used to mitigate the effect of anthropogenic climate change on agriculture? It is time to respond. Sci. Total Environ. 2021, 751, 141763.
- Michalak, I.; Chojnacka, K. Algae as production systems of bioactive compounds. Eng. Life Sci. 2015, 15, 160–176.
- Hurtado, A.Q.; Critchley, A.T. A Review of Multiple Biostimulant and Bioeffector Benefits of AMPEP, an Extract of the Brown Alga Ascophyllum nodosum, as Applied to the Enhanced Cultivation and Micropropagation of the Commercially Important Red Algal Carrageenophyte Kappaphycus Alvarezii and its Selected Cultivars. J. Appl. Phycol. 2018, 30, 2859–2873.
- Rodrigues, M.; Baptistella, J.L.C.; Horz, D.C.; Bortolato, L.M.; Mazzafera, P. Organic Plant Biostimulants and Fruit Quality—A Review. Agronomy 2020, 10, 988.
- Gupta, V.; Kumar, M.; Brahmbhatt, H.; Reddy, C.R.K.; Seth, A.; Jha, B. Simultaneous determination of different endogenetic plant growth regulators in common green seaweeds using dispersive liquid–liquid microextraction method. Plant Physiol. Biochem. 2011, 49, 1259–1263.
- Hong, D.D.; Hien, H.M.; Son, P.N. Seaweeds from Vietnam Used for Functional Food, Medicine and Biofertilizer. J. Appl. Phycol. 2007, 19, 817–826.
- Khan, W.; Rayirath, U.P.; Subramanian, S.; Jithesh, M.N.; Rayorath, P.; Hodges, D.M.; Critchley, A.T.; Craigie, J.S.; Norrie, J.; Prithiviraj, B. Seaweed Extracts as Biostimulants of Plant Growth and Development. J. Plant Growth Regul. 2009, 28, 386–399.
- Behera, B.; Venkata, S.K.; Paramasivan, B. Integrated microalgal biorefinery for the production and application of biostimulants in circular bioeconomy. Bioresour. Technol. 2021, 339, 125588.
- Blunden, G.; El Barouni, M.M.; Gordon, S.M.; McLean, W.F.H.; Rogers, D.J. Extraction, Purification and Characterisation of Dragendorff-Positive Compounds from some British Marine Algae. Bot. Mar. 2013, 24, 451–456.
- Colapietra, M.; Alexander, A. Effect of Foliar Fertilization on Yield and Quality of Table Grapes. In Proceedings of the V International Symposium on Mineral Nutrition of Fruit Plants 721, Talca, Chile, 16–21 January 2005; pp. 213–218.
- Jayaraj, J.; Wan, A.; Rahman, M.; Punja, Z.K. Seaweed Extract Reduces Foliar Fungal Diseases on Carrot. Crop Prot. 2008, 27, 1360–1366.
- Bajpai, S.; Shukla, P.S.; Asiedu, S.; Pruski, K.; Prithiviraj, B. A Biostimulant Preparation of Brown Seaweed Ascophyllum nodosum Suppresses Powdery Mildew of Strawberry. Plant Pathol. J. 2019, 35, 406.
- Soppelsa, S.; Kelderer, M.; Casera, C.; Bassi, M.; Robatscher, P.; Andreotti, C. Use of biostimulants for organic apple production: Effects on tree growth, yield, and fruit quality at harvest and during storage. Front. Plant Sci. 2018, 9, 1324.
- Spann, T.M.; Little, H.A. Applications of a Commercial Extract of the Brown Seaweed Ascophyllum nodosum Increases Drought Tolerance in Container-Grown ‘Hamlin’Sweet Orange Nursery Trees. HortScience 2011, 46, 577–582.
- Shukla, P.S.; Borza, T.; Critchley, A.T.; Hiltz, D.; Norrie, J.; Prithiviraj, B. Ascophyllum nodosum Extract Mitigates Salinity Stress in Arabidopsis thaliana by Modulating the Expression of miRNA Involved in Stress Tolerance and Nutrient Acquisition. PLoS ONE 2018, 13, e0206221.
- Omidbakhshfard, M.A.; Sujeeth, N.; Gupta, S.; Omranian, N.; Guinan, K.J.; Brotman, Y.; Nikoloski, Z.; Fernie, A.R.; Mueller-Roeber, B.; Gechev, T.S. A Biostimulant Obtained from the Seaweed Ascophyllum nodosum Protects Arabidopsis thaliana from Severe Oxidative Stress. Int. J. Mol. Sci. 2020, 21, 474.
- Tandon, S.; Dubey, A. Effects of Biozyme (Ascophyllum nodosum) Biostimulant on Growth and Development of Soybean . Commun Soil Sci Plant Anal. 2015, 46, 845–858.
- Shukla, P.S.; Shotton, K.; Norman, E.; Neily, W.; Critchley, A.T.; Prithiviraj, B. Seaweed Extract Improve Drought Tolerance of Soybean by Regulating Stress-Response Genes. AoB Plants 2018, 10, plx051.
- Jayaraman, J.; Norrie, J.; Punja, Z.K. Commercial Extract from the Brown Seaweed Ascophyllum nodosum Reduces Fungal Diseases in Greenhouse Cucumber. J. Appl. Phycol. 2011, 23, 353–361.
- da Silva, C.P.; Laschi, D.; Ono, E.O.; Rodrigues, J.D.; Mogor, Á.F. Aplicação Foliar do Extrato de Alga Ascophyllum nodosum e do Ácido Glutâmico no Desenvolvimento Inicial de Crisântemos (Dendranthema morifolium (Ramat.) Kitam.) em Vasos. J. Ornam. Hortic. 2010, 16, 179–181.
- Wadas, W.; Dziugieł, T. Quality of new potatoes (Solanum tuberosum L.) in response to plant biostimulants application. Agriculture 2020, 10, 265.
- Prisa, D. Ascophyllum nodosum Extract on Growth Plants in Rebutia heliosa and Sulcorebutia canigueralli. GSC Biol. Pharm. Sci. 2020, 10, 039–045.
- Frioni, T.; Sabbatini, P.; Tombesi, S.; Norrie, J.; Poni, S.; Gatti, M.; Palliotti, A. Effects of a Biostimulant Derived from the Brown Seaweed Ascophyllum nodosum on Ripening Dynamics and Fruit Quality of Grapevines. Sci. Hortic. 2018, 232, 97–106.
- Kaviani, B.; Negahdar, N.; Hashemabadi, D. Improvement of Micropropagation and Proliferation of Robinia pseudoacasia L. Using Plant Growth Regulators and Extracts of Brown Seaweed Ascophyllum nodosum. J. Crop Prod. 2016, 6, 61–79.
- Ronga, D.; Biazzi, E.; Parati, K.; Carminati, D.; Carminati, E.; Tava, A. Microalgal Biostimulants and Biofertilisers in Crop Productions. Agronomy 2019, 9, 192.
- Kapoore, R.V.; Wood, E.E.; Llewellyn, C.A. Algae biostimulants: A critical look at microalgal biostimulants for sustainable agricultural practices. Biotechnol. Adv. 2021, 49, 107754.
- Sharma, H.S.; Fleming, C.; Selby, C.; Rao, J.R.; Martin, T. Plant biostimulants: A review on the processing of macroalgae and use of extracts for crop management to reduce abiotic and biotic stresses. J. Appl. Phycol. 2014, 26, 465–490.
- Rouphael, Y.; Giordano, M.; Cardarelli, M.; Cozzolino, E.; Mori, M.; Kyriacou, M.C.; Bonini, P.; Colla, G. Plant-and seaweed-based extracts increase yield but differentially modulate nutritional quality of greenhouse spinach through biostimulant action. Agronomy 2018, 8, 126.
- Murtic, S.; Oljaca, R.; Murtic, M.S.; Koleska, I.; Muhic, A. Enzymatic antioxidant responses to biostimulants in cherry tomato subjected to drought. JAPS 2019, 29, 1664–1672.
- Bashan, Y. Proposal for the Division of Plant Growth-Promoting Rhizobacteria into Two Classifications: Biocontrol-PGPB (Plant-Growth-Promoting Bacteria) and PGPB. Soil Biol. Biochem. 1998, 30, 1225–1228.
- Bashan, Y.; de-Bashan, L.E.; Prabhu, S.R.; Hernandez, J.P. Advances in plant growth-promoting bacterial inoculant technology: Formulations and practical perspectives (1998–2013). Plant Soil 2014, 378, 1–33.
- Bashan, Y.; Prabhu, S.R.; de-Bashan, L.E.; Kloepper, J.W. Disclosure of exact protocols of fermentation, identity of microorganisms within consortia, formation of advanced consortia with microbe-based products. Biol. Fertil. Soils 2000, 56, 443–445.
- Brock, A.K.; Berger, B.; Mewis, I.; Ruppel, S. Impact of the PGPB Enterobacter Radicincitans DSM 16656 on Growth, Glucosinolate Profile, and Immune Responses of Arabidopsis thaliana. Microb. Ecol. 2013, 65, 661–670.
- Ren, X.-M.; Guo, S.-J.; Tian, W.; Chen, Y.; Han, H.; Chen, E.; Li, B.-L.; Li, Y.-Y.; Chen, Z.-J. Effects of plant growth-promoting bacteria (pgpb) inoculation on the growth, antioxidant activity, cu uptake, and bacterial community structure of rape (Brassica napus l.) grown in cu-contaminated agricultural soil. Front. Microbiol. 2019, 10, 1455.
- Efthimiadou, A.; Katsenios, N.; Chanioti, S.; Giannoglou, M.; Djordjevic, N.; Katsaros, G. Effect of foliar and soil application of plant growth promoting bacteria on growth, physiology, yield and seed quality of maize under Mediterranean conditions. Sci. Rep. 2020, 10, 21060.
- Bhise, K.K.; Dandge, P.B. Mitigation of salinity stress in plants using plant growth promoting bacteria. Symbiosis 2019, 79, 191–204.
- Katsenios, N.; Andreou, V.; Sparangis, P.; Djordjevic, N.; Giannoglou, M.; Chanioti, S.; Stergiou, P.; Xanthou, M.-Z.; Kakabouki, I.; Vlachakis, D.; et al. Evaluation of Plant Growth Promoting Bacteria Strains on Growth, Yield and Quality of Industrial Tomato. Microorganisms 2021, 9, 2099.
- Yarte, M.E.; Gismondi, M.I.; Llorente, B.E.; Larraburu, E.E. Isolation of Endophytic Bacteria from the Medicinal, Forestal and Ornamental Tree Handroanthus impetiginosus. Environ. Technol. 2022, 43, 1129–1139.
- Manhães, N.E.; Jasmim, J.M.; Silva, L.A.A.; Castro, B.B.; Motta, N.L.; Pereira, V.R.; Erthal, A.P.R.C. Loofah Fiber and Sphagnum Moss in the Acclimatization of Cattleya guttata and Zygopetalum mackayi Inoculated with Plant Growth-Promoting Bacteria. Acta Hortic. 2015, 1076, 113–118.
- Hoda, E.E.; Mona, S. Effect of Bio and Chemical Fertilizers on Growth and Flowering of Petunia hybrida Plants. J. Plant Physiol. 2014, 9, 68–77.
- Toffoli, L.M.; Martínez-Zamora, M.G.; Medrano, N.N.; Fontana, C.A.; Lovaisa, N.C.; Delaporte-Quintana, P.; Elias, J.M.; Salazar, S.M.; Pedraza, R.O. Natural Occurrence of Azospirillum brasilense in Petunia with Capacity to Improve Plant Growth and Flowering. J. Basic Microbiol. 2021, 61, 662–673.
- Domenico, P. Optimised Fertilisation with Zeolitites Containing Plant Growth Promoting Rhizobacteria (PGPR) in Ranunculus asiaticus. SC Biol. Pharm. Sci. 2020, 10, 096–102.
- Ordookhani, K.; Sharafzadeh, S.; Zare, M. Influence of PGPR on Growth, Essential Oil and Nutrients Uptake of Sweet Basil. Adv. Environ. Biol. 2011, 5, 672–677.
- Khandan-Mirkohi, A.; Taheri, M.; Zafar-Farrokhi, F.; Rejali, F. Effects of Arbuscular Mycorrhizal Fungus and Plant Growth Promoting Rhizobacteria (PGPR) under Drought Stress on Growth of Ornamental Osteospermum (Osteospermum hybrida ‘Passion Mix’). Int. J. Hortic. Sci. Technol. 2016, 47, 177–191.
- Prisa, D.; Benati, A. Improving the Quality of Ornamental Bulbous with Plant Growth-Promoting Rhizobacteria (PGPR). EPRA Int. J. Multidiscip. Res. (IJMR) 2021, 7, 2455–3662.
- Park, H.G.; Lee, Y.S.; Kim, K.Y.; Park, Y.S.; Park, K.H.; Han, T.H.; Ahn, Y.S. Inoculation with Bacillus licheniformis MH48 Promotes Nutrient Uptake in Seedlings of the Ornamental Plant Camellia japonica Grown in Korean Reclaimed Coastal Lands. Hortic. Sci. Technol. 2017, 35, 11–20.
- Sezen, I.; Kaymak, H.Ç.; Aytatlı, B.; Dönmez, M.F.; Ercişli, S. Inoculations with Plant Growth Promoting Rhizobacteria (PGPR) Stimulate Adventitious Root Formation on Semi-Hardwood Stem Cuttings of Ficus benjamina L. Propag. Ornam. Plants 2014, 14, 152–157.
- Kumari, B.; Hora, A.; Mallick, M.A. Stimulatory effect of PGPR (Plant Growth Promoting Rhizospheric Bacteria) on Medicinal and Growth Properties of a Potential Medicinal Herb Chlorophytum borivilianum: A review. J. Plant Sci. Res. 2017, 33, 151–156.
- Parlakova Karagoz, F.; Dursun, A. Effects of PGPR Formulations, Chemical Fertilizers, and Their Combinations on Physiological Traits and Quality of Bracts of Poinsettia. J. Agric. 2020, 22, 775–787.
- Vinale, F.; Sivasithamparm, K. Beneficial effects of Trichoderma secondary metabolites on corps. Phyto. Res. 2020, 34, 2835–2842.
- Harman, G.E. Myths and Dogmas of Biocontrol Changes in Perceptions Derived from Research on Trichoderma harzinum T-22. Plant Dis. 2000, 84, 377–393.
- Blaszczyk, L.M.S.K.S.; Siwulski, M.; Sobieralski, K.; Lisiecka, J.; Jedryczka, M. Trichoderma spp.–application and prospects for use in organic farming and industry. J. Plant Prot. Res. 2014, 54, 309–317.
- López-Bucio, J.; Pelagio-Flores, R.; Herrera-Estrella, A. Trichoderma as Biostimulant: Exploiting the Multilevel Properties of a Plant Beneficial Fungus. Sci. Hortic. 2015, 196, 109–123.
- Fiorentino, N.; Ventorino, V.; Woo, S.L.; Pepe, O.; De Rosa, A.; Gioia, L.; Romano, I.; Lombardi, N.; Napolitano, M.; Colla, G.; et al. Trichoderma-Based Biostimulants Modulate Rhizosphere Microbial Populations and Improve N Uptake Efficiency, Yield, and Nutritional Quality of Leafy Vegetables. Front. Plant Sci. 2018, 9, 743.
- Şesan, T.E.; Oancea, A.O.; Ştefan, L.M.; Mănoiu, V.S.; Ghiurea, M.; Răut, I.; Constantinescu-Aruxandei, D.; Toma, Á.; Savin, S.; Bira, A.F.; et al. Effects of Foliar Treatment with a Trichoderma Plant Biostimulant Consortium on Passiflora caerulea L. Yield and Quality. Microorganisms 2020, 8, 123.
- Majkowska-Gadomska, J.; Francke, A.; Dobrowolski, A.; Mikulewicz, E. The Effect of Selected Biostimulants on Seed Germination of Four Plant Species. Acta Agrophys. 2017, 24, 591–599.
- Di Vaio, C.; Testa, A.; Cirillo, A.; Conti, S. Slow-Release Fertilization and Trichoderma harzianum-Based Biostimulant for the Nursery Production of Young Olive Trees (Olea europaea L.). Agronomy. 2021, 19, 3.
- Hasanuzzaman, M.; Bhuyan, M.H.M.B.; Zulfiqar, F.; Raza, A.; Mohsin, S.M.; Mahmud, J.A.; Fujita, M.; Fotopoulos, V. Reactive oxygen species and antioxidant defense in plants under abiotic stress: Revisiting the crucial role of a universal defense regulator. Antioxidants 2020, 9, 681.
- Teklić, T.; Parađiković, N.; Špoljarević, M.; Zeljković, S.; Lončarić, Z.; Lisjak, M. Linking abiotic stress, plant metabolites, biostimulants and functional food. Ann. Appl. Biol. 2021, 178, 169–191.
- Sangiorgio, D.; Cellini, A.; Donati, I.; Pastore, C.; Onofrietti, C.; Spinelli, F. Facing climate change: Application of microbial biostimulants to mitigate stress in horticultural crops. Agronomy 2020, 10, 794.
- de Pascali, M.; Vergine, M.; Sabella, E.; Aprile, A.; Nutricati, E.; Nicolì, F.; Buja, I.; Negro, C.; Miceli, A.; Rampino, P.; et al. Molecular effects of Xylella fastidiosa and drought combined stress in olive trees. Plants 2019, 8, 437.
- Ahanger, M.A.; Tyagi, S.R.; Wani, M.R.; Ahmad, P. Drought Tolerance: Role of Organic Osmolytes, Growth Regulators, and Mineral Nutrients. In Physiological Mechanisms and Adaptation Strategies in Plants Under Changing Environment: Volume 1; Ahmad, P., Wani, M.R., Eds.; Springer: Berlin, Germany, 2014; pp. 25–55.
- Zhang, H.; Zhu, J.; Gong, Z.; Zhu, J.-K. Abiotic Stress Responses in Plants. Nat. Rev. Genet. 2022, 23, 104–119.
- Gong, Z.; Xiong, L.; Shi, H.; Yang, S.; Herrera-Estrella, L.R.; Xu, G.; Chao, D.-Y.; Li, J.; Wang, P.-Y.; Qin, F.; et al. Plant abiotic stress response and nutrient use efficiency. Sci. China Life Sci. 2020, 63, 635–674.
- Kollist, H.; Zandalinas, S.I.; Sengupta, S.; Nuhkat, M.; Kangasjärvi, J.; Mittler, R. Rapid Responses to Abiotic Stress: Priming the Landscape for the Signal Transduction Network. Trends Plant Sci. 2019, 24, 25–37.
- Bechtold, U.; Field, B. Molecular mechanisms controlling plant growth during abiotic stress. J. Exp. Bot. 2018, 69, 2753–2758.
- Nemhauser, J.L.; Hong, F.; Chory, J. Different plant hormones regulate similar processes through largely nonoverlapping transcriptional responses. Cell 2006, 126, 467–475.
- Hepler, P. Calcium: A Central Regulator of Plant Growth and Development. Plant Cell 2005, 17, 2142–2155.
- Gupta, S.; Doležal, K.; Kulkarni, M.G.; Balázs, E.; Van Staden, J. Role of Non-Microbial Biostimulants in Regulation of Seed Germination and Seedling Establishment. Plant Growth Regul. 2022, 1–43.
- Kumar, J.S.P.; Rajendra Prasad, S.; Banerjee, R.; Thammineni, C. Seed Birth to Death: Dual Functions of Reactive Oxygen Species in Seed Physiology. Ann. Bot. 2015, 116, 663–668.
- Campobenedetto, C.; Grange, E.; Mannino, G.; Van Arkel, J.; Beekwilder, J.; Karlova, R.; Garabello, C.; Contartese, V.; Bertea, C.M. A Biostimulant Seed Treatment Improved Heat Stress Tolerance during Cucumber Seed Germination by Acting on the Antioxidant System and Glyoxylate Cycle. Front. Plant Sci. 2020, 11, 836.
- Pal, G.; Kumar, K.; Verma, A.; Verma, S.K. Application of Bacterial Biostimulants in Promoting Growth and Disease Prevention in Crop Plants. In Biostimulants for Crops from Seed Germination to Plant Development; Academic Press: New York, NY, USA, 2021; pp. 393–410. ISBN 9780128230480.
- Bayona-Morcillo, P.J.; Plaza, B.M.; Gómez-Serrano, C.; Rojas, E.; Jimenez-Becker, S. Effect of the Foliar Application of Cyanobacterial Hydrolysate (Arthrospira platensis) on the Growth of Petunia × hybrida Under Salinity Conditions. J. Appl. Phycol. 2020, 32, 4003–4011.
- Plaza, B.M.; Gómez-Serrano, C.; Acién-Fernández, F.G.; Jimenez-Becker, S. Effect of Microalgae Hydrolysate Foliar Application (Arthrospira platensis and Scenedesmus sp.) on Petunia × hybrida Growth. J. Appl. Phycol. 2018, 30, 2359–2365.
- Saini, I.; Aggarwal, A.; Kaushik, P. Influence of biostimulants on important traits of Zinnia elegans Jacq. under open field conditions. Int. J. Agron 2019, 2019, e3082967.
- Lin, Y.; Jones, M.L. Evaluating the growth-promoting effects of microbial biostimulants on greenhouse floriculture crops. HortScience 2022, 57, 97–109.
- Caser, M.; Lovisolo, C.; Scariot, V. The influence of water stress on growth, ecophysiology and ornamental quality of potted Primula vulgaris ‘Heidy’ Plants. New insights to increase water use efficiency in plant production. Plant Growth Regul. 2017, 83, 361–373.
- South, K.A.; Nordstedt, N.P.; Jones, M.L. Identification of Plant Growth Promoting Rhizobacteria That Improve the Performance of Greenhouse-Grown Petunias under Low Fertility Conditions. Plants 2021, 10, 1410.
- Nordstedt, N.P.; Chapin, L.J.; Taylor, C.G.; Jones, M.L. Identification of Pseudomonas spp. that Increase Ornamental Crop Quality During Abiotic Stress. Front. Plant Sci. 2020, 10, 1754.
- Van Loon, L.C. Plant Responses to Plant Growth-Promoting Rhizobacteria. In New Perspectives and Approaches in Plant Growth-Promoting Rhizobacteria Research; Springer: Dordrecht, The Netherlands, 2007; pp. 243–254.
- Shoresh, M.; Harman, G.E.; Mastouri, F. Induced systemic resistance and plant responses to fungal biocontrol agents. Ann. Rev. Phytopathol. 2010, 48, 21–43.
- Ertani, A.; Schiavon, M.; Muscolo, A.; Nardi, S. Alfalfa plant-derived biostimulant stimulate short-term growth of salt stressed Zea mays L. plants. Plant Soil 2013, 364, 145–158.
- Mutale-Joan, C.; Rachidi, F.; Mohamed, H.A.; Mernissi, N.E.; Aasfar, A.; Barakate, M.; Mohammad, D.; Sbabou, L.; Arroussi, H.E. Microalgae-cyanobacteria–based biostimulant effect on salinity tolerance mechanisms, nutrient uptake, and tomato plant growth under salt stress. J. Appl. Phycol. 2021, 33, 3779–3795.
- Rayirath, P.; Benkel, B.; Mark Hodges, D.; Allan-Wojtas, P.; MacKinnon, S.; Critchley, A.T.; Prithiviraj, B. Lipophilic components of the brown seaweed, Ascophyllum nodosum, enhance freezing tolerance in Arabidopsis thaliana. Planta 2009, 230, 135–147.
- Xu, C.; Leskovar, D.I. Effects of A. nodosum seaweed extracts on spinach growth, physiology and nutrition value under drought stress. Sci. Hortic. 2015, 183, 39–47.
- Möller, M.; Smith, M.L. The significance of the mineral component of seaweed suspensions on lettuce (Lactuca sativa L.) seedling growth. J. Plant Physiol. 1998, 153, 658–663.
- Khan, M.S.; Pandey, M.K.; Hemalatha, S. Comparative Studies on the Role of Organic Biostimulant in Resistant and Susceptible Cultivars of Rice Grown under Saline Stress—Organic Biostimulant Alleviate Saline Stress in Tolerant and Susceptible Cultivars of Rice. J. Crop Sci. Biotechnol. 2018, 21, 459–467.
- Hare, P.D.; Cress, W.A.; Van Staden, J. The involvement of cytokinins in plant responses to environmental stress. Plant Growth Regul. 1997, 23, 79–103.
- Toscano, S.; Ferrante, A.; Romano, D. Response of Mediterranean ornamental plants to drought stress. Horticulturae 2019, 5, 6.
- Battacharyya, D.; Babgohari, M.Z.; Rathor, P.; Prithiviraj, B. Seaweed extracts as biostimulants in horticulture. Sci. Hortic. 2015, 196, 39–48.
- Massa, D.; Lenzi, A.; Montoneri, E.; Ginepro, M.; Prisa, D.; Burchi, G. Plant response to biowaste soluble hydrolysates in hibiscus grown under limiting nutrient availability. J. Plant Nutr. 2018, 41, 396–409.
- Nordstedt, N.P.; Jones, M.L. Isolation of rhizosphere bacteria that improve quality and water stress tolerance in greenhouse ornamentals. Front. Plant Sci. 2020, 11, 826.
- Giordano, M.; Petropoulos, S.A.; Cirillo, C.; Rouphael, Y. Biochemical, physiological, and molecular aspects of ornamental plants adaptation to deficit irrigation. Horticulturae 2021, 7, 107.
- Chang, Y.-N.; Zhu, C.; Jiang, J.; Zhang, H.; Zhu, J.-K.; Duan, C.-G. Epigenetic regulation in plant abiotic stress responses. J. Integr. Plant Biol. 2020, 62, 563–580.
- Askari-Khorasgani, O.; Hatterman-Valenti, H.; Flores Pardo, F.B.; Pessarakli, M. Plant and symbiont metabolic regulation and biostimulants application improve symbiotic performance and cold acclimation. J. Plant Nutr. 2019, 42, 2151–2163.
- Hajizadeh, H.S.; Heidari, B.; Bertoldo, G.; Della Lucia, M.C.; Magro, F.; Broccanello, C.; Baglieri, A.; Puglisi, I.; Squartini, A.; Campagna, G.; et al. Expression Profiling of Candidate Genes in Sugar Beet Leaves Treated with Leonardite-Based Biostimulant. High-Throughput 2019, 8, 18.
- Sahana, B.N.; PrasannaKumar, M.K.; Mahesh, H.B.; Buela Parivallal, P.; Puneeth, M.E.; Gautam, C.; Girish, T.R.; Nori, S.; Suryanarayan, S. Biostimulants derived from red seaweed stimulate the plant defence mechanism in rice against Magnaporthe oryzae. J. Appl. Phycol. 2022, 34, 659–665.
- Campobenedetto, C.; Mannino, G.; Beekwilder, J.; Contartese, V.; Karlova, R.; Bertea, C.M. The application of a biostimulant based on tannins affects root architecture and improves tolerance to salinity in tomato plants. Sci. Rep. 2021, 11, 354.
- Casadesús, A.; Polo, J.; Munné-Bosch, S. Hormonal effects of an enzymatically hydrolyzed animal protein-based biostimulant (pepton) in water-stressed tomato plants. Front. Plant Sci. 2019, 10, 758.
- Vitale, E.; Velikova, V.; Tsonev, T.; Ferrandino, I.; Capriello, T.; Arena, C. The Interplay between Light Quality and Biostimulant Application Affects the Antioxidant Capacity and Photosynthetic Traits of Soybean (Glycine max L. Merrill). Plants 2021, 10, 861.
- Fan, H.M.; Wang, X.W.; Sun, X.; Li, Y.Y.; Sun, X.Z.; Zheng, C.S. Effects of Humic Acid Derived from Sediments on Growth, Photosynthesis and Chloroplast Ultrastructure in Chrysanthemum. Sci. Hortic. 2014, 177, 118–123.
- Krajnc, A.U.; Turinek, M.; Ivančič, A. Morphological and Physiological Changes during Adventitious Root Formation as Affected by Auxin Metabolism: Stimulatory Effect of Auxin Containing Seaweed Extract Treatment. Agricultura 2013, 10, 17–27.
- Tahiri, A.; Destain, J.; Thonart, P.; Druart, P. In vitro Model to Study the Biological Properties of Humic Fractions from Landfill Leachate and Leonardite during Root Elongation of Alnus glutinosa L. Gaertn and Betula pendula Roth. Plant Cell Tissue Organ Cult. 2015, 122, 739–749.
- Baldotto, L.E.; Baldotto, M.A. Adventitious Rooting on the Brazilian Red-Cloak and Sanchezia after Application of Indole-Butyric and Humic Acids. Hortic. Bras. 2014, 32, 434–439.
- Asrar, A.A.; Abdel-Fattah, G.M.; Elhindi, K.M. Improving growth, flower yield, and water relations of snapdragon (Antirhinum majus L.) plants grown under well-watered and water-stress conditions using arbuscular mycorrhizal fungi. Photosynthetica 2012, 50, 305–316.
- Monder, M.J.; Kozakiewicz, P.; Jankowska, A. The Role of Plant Origin Preparations and Phenological Stage in Anatomy Structure Changes in the Rhizogenesis of Rosa ‘Hurdal’. Front Plant Sci 2021, 12, 696998.
- Monder, M.J.; Kozakiewicz, P.; Jankowska, A. Anatomical Structure Changes in Stem Cuttings of Rambler Roses Induced with Plant Origin Preparations. Sci. Hortic. 2019, 255, 242–254.
- Megersa, H.G.; Lemma, D.T.; Banjawu, D.T. Effects of plant growth retardants and pot sizes on the height of potting ornamental plants: A short review. J. Hortic. 2018, 5, 1.
- Sriprapat, W.; Thiravetyan, P. Efficacy of Ornamental Plants for Benzene Removal from Contaminated Air and Water: Effect of Plant Associated Bacteria. Int. Biodeterior. Biodegrad. 2016, 113, 262–268.
- Lekawatana, S.; Suwannamek, B. Ornamental plants in Thailand. Acta Hortic. 2017, 11–16.
- Junqueira, A.H.; Peetz, M. Brazilian consumption of flowers and ornamental plants: Habits, practices and trends. OH 2017, 23, 178.
- Briercliffe, T. Growing the global market for ornamentals. Acta Hortic. 2017, 1–8.
- de Pascale, S.; Rouphael, Y.; Cirillo, C.; Colla, G. Plant Biostimulants in Greenhouse Horticulture: Recent Advances and Challenges Ahead. In Proceedings of the XXX International Horticultural Congress IHC2018: III International Symposium on Innovation and New Technologies in Protected 1271, Istanbul, Turkey, 12–18 August 2018; pp. 327–334.
- Gebashe, F.; Gupta, S.; Van Staden, J. Disease Management Using Biostimulants. In Biostimulants for Crops from Seed Germination to Plant Development; Academic Press: New York, NY, USA, 2021; pp. 411–425. ISBN 9780128230480.
- de Silva, T.S.; Silva, A.P.S.; de Almeida, S.A.; Ribeiro, K.G.; Souza, D.C.; Bueno, P.A.A.; Marques, M.M.M.; Almeida, P.M.; Peron, A.P. Cytotoxicity, Genotoxicity, and Toxicity of Plant Biostimulants Produced in Brazil: Subsidies for Determining Environmental Risk to Non-Target Species. Water Air Soil Pollut. 2020, 231, 233.
- Yuan, Y. Effects of Biostimulants on Ornamental Plants Grown in Solid Soil Less Cultural Systems. Ph.D. Thesis, Lincoln University, Lincoln, UK, 2021.
- Yücel, G.; Erken, K.; Doğan, Y.E. Organic Stimulant Uses in Natural Plant Production. EJOH 2020, 47, 119–128.
- Carmo, L.P.; Moura, C.W.N.; Lima-Brito, A. Red macroalgae extracts affect in vitro growth and bud formation in Comanthera mucugensis (Giul.) LR Parra & Giul., an endemic dry flower species from the Chapada Diamantina (Brazil). S. Afr. J. 2020, 135, 29–34.
- Florijančić, T.; Lužaić, R. Poljoprivredni Fakultet Sveučilišta Josipa Jurja Strossmayera u Osijeku. In Proceedings of the 44th Croatian and the 4th International Symposium of Agronomists, Opatija, Croatia, 16–20 February 2009.
- Parađiković, N.; Zeljković, S.; Tkalec, M.; Vinković, T.; Maksimović, I.; Haramija, J. Influence of Biostimulant Application on Growth, Nutrient Status and Proline Concentration of Begonia Transplants. Biol. Agric. 2017, 33, 89–96.
- Tavares, A.R.; dos Santos, P.L.F.; Zabotto, A.R.; do Nascimento, M.V.L.; Jordão, H.W.C.; Boas, R.L.V.; Broetto, F. Seaweed Extract to Enhance Marigold Seed Germination and Seedling Establishment. SN Appl. Sci. 2020, 2, 1–6.
- Zeljković, S.; Parađiković, N.; Vinković, T.; Tkalec, M. Biostimulant application in the production of seedlings of seasonal flowers. Agro-Knowl. J. 2011, 12, 175–181.
- Dudaš, S.; Šestan, I. Effect of Seedling Growing Technology and Bio-Algeen S-90 Application on Plantlets Quality of French Marigold (Tagetes patula L.) ‘Orange Boy’. Zb. Veleuč. Rij. 2014, 2, 333–342.
- Bolagam, R.; Natarajan, S. Economics of Cut Gladiolus (Gladiolus grandiflorus L.) Production with Application Biostimulants. J. Pharmacogn. Phytochem. 2019, 8, 1276–1279.
- Sankari, A.; Anand, M.; Arulmozhiyan, R. Effect of Biostimulants on Yield and Post Harvest Quality of Gladiolus cv. White Prosperity. J. Asian Hortic. 2015, 10, 86–94.
- Kumar, P.; Kumar, R.; Kumar, A. Effect of Organic Culture on Growth, Development and Post Harvest Life of Gladiolus (Gladiolus hybrida). J. Ornam. Hortic. 2008, 11, 127–130.
- Bhattacharyya, P.; Lalthafamkimi, L.; Van Staden, J. Insights into the Biostimulatory Effects of Chitosan in Propagation of Orchid Bioresources. In Biostimulants for Crops from Seed Germination to Plant Development; Academic Press: New York, NY, USA, 2021; pp. 197–210. ISBN 9780128230480.
- Abdalla, M.M. Boosting the growth of rocket plants in response to the application of Moringa olifera extracts as a biostimulant. Life Sci. 2014, 11, 1113–1121.
- Exposito-Rodriguez, M.; Laissue, P.P.; Yvon-Durocher, G.; Smirnoff, N.; Mullineaux, P.M. Photosynthesis-dependent H2O2 transfer from chloroplasts to nuclei provides a high-light signalling mechanism. Nat Commun 2017, 8, 49.
- Sharma, A.; Shahzad, B.; Rehman, A.; Bhardwaj, R.; Landi, M.; Zheng, B. Response of Phenylpropanoid Pathway and the Role of Polyphenols in Plants under Abiotic Stress. Molecules 2019, 24, 2452.
- Arjana, I.G.M.; Situmeang, Y.P.; Suaria, I.N.; Mudra, N.K.S. Effect of Plant Material and Variety for Production and Quality Chrysanthemum. Int. J. Adv. Sci. Eng. Inf. Technol. 2015, 5, 407–409.
- Pacholczak, A.; Jędrzejuk, A.; Sobczak, M. Shading and Natural Rooting Biostimulator Enhance Potential for Vegetative Propagation of Dogwood Plants (Cornus alba L.) Via Stem Cuttings. S. Afr. J 2017, 109, 34–41.
- Preece, J.E. A Century of Progress with Vegetative Plant Propagation. HortSci 2003, 38, 1015–1025.
- Wise, K.; Gill, H.; Selby-Pham, J. Willow Bark Extract and the Biostimulant Complex Root Nectar® Increase Propagation Efficiency in Chrysanthemum and Lavender Cuttings. Sci. Hort. 2020, 263, 109108.
- Abdel-Rahman, S.; Abdul-Hafeez, E.; Saleh, A.M. Improving Rooting and Growth of Conocarpus erectus Stem Cuttings Using Indole-3-Butyric Acid (IBA) and Some Biostimulants. SJFOP 2020, 7, 109–129.
- Ahkami, A.H.; Lischewski, S.; Haensch, K.T.; Porfirova, S.; Hofmann, J.; Rolletschek, H.; Melzer, M.; Franken, P.; Hause, B.; Druege, U.; et al. Molecular Physiology of Adventitious Root Formation in Petunia Hybrida Cuttings: Involvement of Wound Response and Primary Metabolism. New Phytol. 2009, 181, 613–625.
- Trofimuk, L.P.; Kirillov, P.S.; Egorov, A.A. Application of Biostimulants for Vegetative Propagation of Endangered Abies gracilis. J. For. Res. 2020, 31, 1195–1199.
- Stirk, W.A.; Van Staden, J. Comparison of Cytokinin-and Auxin-Like Activity in Some Commercially Used Seaweed Extracts. J. Appl. Phycol. 1996, 8, 503–508.
- Monder, M.J.; Woliński, K.; Niedzielski, M. The Propagation of Rosa gallica ‘Tuscany Superb’by Root Cuttings with the Use of IBA and Biostimulants. Not. Botan. Horti Agrobot. Cluj-Napoca 2019, 47, 691–698.
- Monder, M.J.; Niedzielski, M.; Woliński, K. The Pivotal Role of Phenological Stages Enhanced by Plant Origin Preparations in the Process of Rhizogenesis of Rosa ‘Hurdal’Stem Cuttings. Agriculture 2022, 12, 158.
- Norrie, J.; Critchley, A.T.; Gupta, S.; Van Staden, J. Biostimulants in modern agriculture: Fitting round biological effects into square regulatory holes. In Biostimulants for Crops from Seed Germination to Plant Development; Academic Press: New York, NY, USA, 2021; pp. 231–236. ISBN 9780128230480.
- dos Santos, P.L.F.; Zabotto, A.R.; Jordão, H.W.C.; Boas, R.L.V.; Broetto, F.; Tavares, A.R. Use of seaweed-based biostimulant (Ascophyllum nodosum) on ornamental sunflower seed germination and seedling growth. J. Ornam. Hortic. 2019, 25, 231–237.
- Makhaye, G.; Mofokeng, M.M.; Tesfay, S.; Aremu, A.O.; Van Staden, J.; Amoo, S.O. Influence of Plant Biostimulant Application on Seed Germination. In Biostimulants for Crops from Seed Germination to Plant Development; Academic Press: New York, NY, USA, 2021; pp. 109–135. ISBN 9780128230480.
- Szekely-Varga, Z.; Kentelky, E.; Cantor, M. Effect of Gibberellic Acid on the Seed Germination of Lavandula angustifolia Mill. RJH 2021, 2, 169–176.
- Zeljković, S.; Gidas, J.D.; Todorović, V.; Pašalić, M. Germination of floral species depending on the applied biostimulant. AgroReS 2019, 16, 77.
- Byczyńska, A. Chitosan improves growth and bulb yield of pineapple lily (Eucomis bicolor ‘Baker’) an ornamental and medicinal plant. WSN 2018, 110, 159–171.
- Aremu, A.O.; Masondo, N.A.; Rengasamy, K.R.; Amoo, S.O.; Gruz, J.; Bíba, O.; Šubrtová, M.; Pěnčík, A.; Novák, O.; Doležal, K.; et al. Physiological Role of Phenolic Biostimulants Isolated from Brown Seaweed Ecklonia maxima on plant growth and development. Planta 2015, 241, 1313–1324.
- Paris, L.; García-Caparrós, P.; Llanderal, A.; Silva, J.T.; Reca, J.; Lao, M. Plant Regeneration from Nodal Segments and Protocorm-Like Bodies (PLBs) Derived from Cattleya maxima J. Lindley in Response to Chitosan and Coconut Water. Propag. Ornam. Plants 2019, 19, 18–23.
- Gontijo, J.B.; Andrade, G.V.S.; Baldotto, M.A.; Baldotto, L.E.B. Bioprospecting and Selection of Growth-Promoting Bacteria for Cymbidium sp. orchids. Sci Agric 2018, 75, 368–374.
- Hasan, A.S.I.L. The Effect of Different Biostimulants Applications on Corm Characters of Saffron (Crocus sativus L.). In Academic Reseach in Life Sciences for Sustainibility; Artikel Akademi: Istanbul, Turkey, 2021; pp. 123–135.
- Monder, M.J. Rooting and Growth of Root Cuttings of Two Old Rose Cultivars “Harison’s Yellow” and “Poppius” Treated with IBA and Biostimulants. Acta Agrobot. 2019, 72.
- Zeljković, S.; Parađiković, N.; Vinković, T.; Oljača, R.; Tkalec, M. Contents of Mineral Elements in Nursery Stock of Marigold (Tagetes patula L.) Under Bio Stimulant Treatment. Agro-Knowl. J. 2020, 11, 127–134.
- Gomes, E.N.; Vieira, L.M.; Tomasi, J.D.C.; Tomazzoli, M.M.; Grunennvaldt, R.L.; Fagundes, C.D.M.; Machado, R.C.B. Brown Seaweed Extract Enhances Rooting and Roots Growth on Passiflora actinia Hook Stem Cuttings. Ornam. Hortic. 2018, 24, 269–276.
- Prisa, D. Possible Use of Spirulina and Klamath algae as Biostimulants in Portulaca grandiflora (Moss Rose). World J. Adv. Res. Rev. 2019, 3, 001–006.
- Bákonyi, N.; Kisvarga, S.; Barna, D.; Tóth, I.O.; El-Ramady, H.; Abdalla, N.; Kovács, S.; Rozbach, M.; Fehér, C.; Elhawat, N.; et al. Chemical traits of fermented alfalfa brown juice: Its implications on physiological, biochemical, anatomical, and growth parameters of celosia. Agronomy 2020, 10, 247.
- Kisvarga, S.; Barna, D.; Kovács, S.; Csatári, G.O.; Tóth, I.; Fári, M.G.; Makleit, P.; Veres, S.; Alshaal, T.; Bákonyi, N. Fermented Alfalfa Brown Juice Significantly Stimulates the Growth and Development of Sweet Basil (Ocimum basilicum L.) Plants. Agronomy 2020, 10, 657.
- Barna, D.; Kisvarga, S.; Kovács, S.; Csatári, G.; Tóth, I.O.; Fári, M.G.; Alshaal, T.; Bákonyi, N. Raw and fermented alfalfa brown juice induces changes in the germination and development of french marigold (Tagetes patula L.) plants. Plants 2021, 10, 1076.
- Jelačić, S.; Beatović, D.; Lakić, N. Effect of Natural Biostimulators and Slow-Disintegrating Fertilizers on the Quality of Sage Nursery Stock under Different Growing Conditions. In Proceedings of the XXIst Conference of Agronomist, Veterinarians and Technologists, Ministry of Science and Environmental Protection, Novi Sad, Serbia, 19–21 October 2007; pp. 145–156. Available online: https://agris.fao.org/agris-search/search.do?recordID=RS2010001902 (accessed on 20 January 2022).
- Sureshkumar, R.; Priya, G.S.; Rajkumar, M.; Sendhilnathan, R. Studies on the effect of organic manures, biostimulants and micronutrients on certain growth and flowering parameters of tuberose (Poianthes tuberosa L.) CV. Prajwal. Plant Arch. 2019, 19, 2436–2440.
- Hegde, P.P.; Patil, B.C.; Kulkarni, M.S.; Hegde, N.K.; Kukanoor, L.; Shiragur, M.; Harshavardhan, M. Efficacy of biostimulants on growth and flowering of Dendrobium orchid (Dendrobium nobile Lindl.) var. Sonia-17 under protected cultivation. J. Pharm. Innov. 2021, 10, 1189–1191.
- Ozbay, N.; Demirkiran, A.R. Enhancement of growth in ornamental pepper (Capsicum annuum L.) Plants with application of a commercial seaweed product, stimplex®. Appl. Ecol. Environ. Res. 2019, 17, 4361–4375.
- Marschner, H. (Ed.) Marschner’s Mineral Nutrition of Higher Plants; Academic Press: New York, NY, USA, 2011.
- Alhasan, A.S.; Aldahab, E.A.; Al-Ameri, D.T. Influence of Different Rates of Seaweed Extract on Chlorophyll Content, Vegetative Growth and Flowering Traits of Gerbera (Gerbera jamesonii L.) Grown under the Shade Net House Conditions. IOP Conf. Ser. Earth Environ. Sci. 2021, 923, 012019.
- Salachna, P. Effects of Depolymerized Gellan with Different Molecular Weights on the Growth of Four Bedding Plant Species. Agronomy 2020, 10, 169.
- Cristiano, G.; De Lucia, B. Petunia Performance under Application of Animal-Based Protein Hydrolysates: Effects on Visual Quality, Biomass, Nutrient Content, Root Morphology, and Gas Exchange. Front. Plant Sci. 2021, 12, 890.
- Cristiano, G.; Pallozzi, E.; Conversa, G.; Tufarelli, V.; De Lucia, B. Effects of an animal-derived biostimulant on the growth and physiological parameters of potted snapdragon (Antirrhinum majus L.). Front. Plant Sci. 2018, 9, 861.
- Kapczyńska, A.; Kowalska, I.; Prokopiuk, B.; Pawłowska, B. Rooting Media and Biostimulator Goteo Treatment effect the adventitious root formation of Pennisetum ‘Vertigo’cuttings and the Quality of the Final Product. Agriculture 2020, 10, 570.
- de Luca, V.; de Barreda, D.G.; Lidón, A.; Lull, C. Effect of Nitrogen-fixing Microorganisms and Amino Acid-based Biostimulants on Perennial Ryegrass. J. Am. Soc. Hortic. Sci. 2020, 30, 12.
- Godlewska, K.; Biesiada, A.; Michalak, I.; Pacyga, P. The effect of plant-derived biostimulants on white head cabbage seedlings grown under controlled conditions. Sustainability 2019, 11, 5317.





