
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Galya Bigman | -- | 3967 | 2022-05-06 20:05:58 | | | |
| 2 | Vivi Li | Meta information modification | 3967 | 2022-05-07 03:56:56 | | |
Video Upload Options
As human life expectancy is rising, the incidence of age-associated diseases will also increase. Scientific evidence has revealed that healthy diets, including good fats, vitamins, minerals, or polyphenolics, could have antioxidant and anti-inflammatory activities, with antiaging effects. Recent studies demonstrated that vitamin K is a vital cofactor in activating several proteins, which act against age-related syndromes. Thus, vitamin K can carboxylate osteocalcin (a protein capable of transporting and fixing calcium in bone), activate matrix Gla protein (an inhibitor of vascular calcification and cardiovascular events) and carboxylate Gas6 protein (involved in brain physiology and a cognitive decline and neurodegenerative disease inhibitor).
1. Introduction
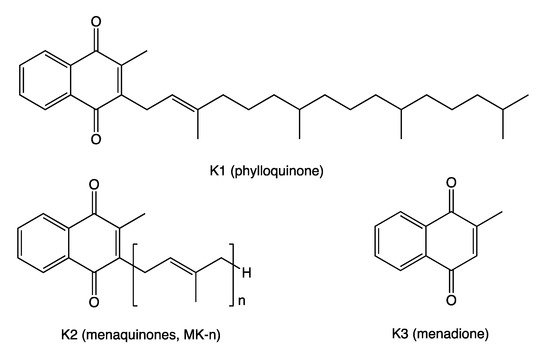
| Food Category | Food Source | VK2 * |
|---|---|---|
| Fermented foods | Natto Sauerkraut |
850–1000 (90% MK-7, 8% MK-8) 5.5 (31% MK-6, 23% MK-9, 17% MK-5 and -8) |
| Hard cheeses | 50–80 (15–67% MK-9, 6–22% MK-4, 6–22% MK-8) | |
| Soft cheeses | 30–60 (20–70% MK-9, 6–20% MK-4, 6–20% MK-8) | |
| Eggs | Yolk | 15–30 (MK-4) |
| Meats | Pork, beef, chicken | 1.4–10 (MK-4) |
2. Vitamin K in Bone Health
| Author, Year, Country [Ref.] | Subjects (W:M) Age (Mean ± SD) |
Design (Length) | Intervention Exposure |
Findings |
|---|---|---|---|---|
| Shiraki et al. 2000 Japan [40] | 241 PMO 67.2 y |
prospective 2 y |
45 mg/d MK-4 vs. control |
↓ ucOC (p < 0.0001) ↑ cOC (p = 0.0081) ↓ fracture risk (p = 0.0273) |
| Iwamoto et al. 2001 Japan [44] | 72 PMO 65.3 y |
prospective 2 y |
45 mg/d MK-4 + Ca vs. Ca |
↓ vertebral fractures (p < 0.0001) ↑ BMD (forearm) (p < 0.0001) |
| Purwosunu et al. 2006 Indonesia [45] | 63 PMO 60.8 y |
RCT 48 w |
45 mg/d MK-4 + Ca vs. Ca |
↓ ucOC (p ˂ 0.01) ↑ BMD (lumbar) (p < 0.05) |
| Bolton-Smith et al. 2007 UK [41] | 244 healthy W 68.2 y |
RCT 2 y |
200 μg/d VK1 + 10 μg/d vitD3 + Ca vs. placebo | ↓ ucOC (p < 0.001) ↑ BMD (ultradistal radius) (p < 0.01) |
| Knapen et al. 2007 Netherlands [46] | 325 PMW 66.0 y |
RCT 3 y |
45 mg/d MK-4 vs. placebo |
↑ BMC (p < 0.05) and bone strength (femoral neck) |
| Booth et al. 2008 USA [47] |
452 (267:185) 68.4 y |
RCT 3 y |
500 μg/d PK vs. control |
↓ ucOC (p ˂ 0.0001) |
| Cheung et al. 2008 Canada [48] |
400 PMOa 59.1 y |
RCT 2–4 y |
5 mg/d VK1 vs. placebo |
↓ fracture risk (p = 0.04) |
| Hirao et al. 2008 Japan [49] | 44 PMW 68.4 y |
prospective 1 y |
45 mg/d VK2 + 5 mg/d alendronate vs. 5 mg/d alendronate | ↓ ucOC (p = 0.014) ↓ ucOC:cOC (p = 0.007) ↑ BMD (femoral neck) (p = 0.03) |
| Tsugawa et al. 2008 Japan [50] | 379 W 63.0 y |
prospective 3 y |
high VK1 vs. low VK1 | ↓ vertebral fracture risk (p < 0.001) |
| Binkley et al. 2009 USA [42] | 381 PMW 62.5 y |
RCT 1 y |
1 mg/d VK1 or 45 mg/d MK-4 vs. placebo | ↓ ucOC (p < 0.001) for both VK1 and MK-4 groups |
| Yamauchi et al. 2010 Japan [51] | 221 healthy W 60.8 ± 9.5 y |
cross-sectional | 260±85 μg/d VK | ↓ ucOC (p < 0.0001) ↑ BMD (lumbar) (p = 0.015) |
| Je et al. 2011 Korea [52] |
78 PMW 67.8 y |
RCT 6 mo |
45 mg/d MK-4 + vitD + Ca vs. vitD + Ca | ↓ ucOC (p = 0.008) ↑ BMD (lumbar) (p = 0.049) |
| Kanellakis et al. 2012 Greece [53] | 173 PMW 62.0 y |
RCT 12 mo |
100 μg PK or MK-7 + vitD + Ca vs. control |
↓ ucOC (p = 0.001) * ↑ BMD (lumbar) (p < 0.05) * |
| Knapen et al. 2013 Netherlands [54] | 244 PMW 60.0 y |
RCT 3 y |
180 μg/d MK-7 vs. placebo |
↓ ucOC (p < 0.001) ↑ BMD (lumbar spine, femoral neck), bone strength (p < 0.05) |
| Jiang et al. 2014 China [55] | 213 PMW 64.4 y |
RCT 1 y |
45 mg/d MK-4 + Ca vs. Ca |
↓ ucOC (p < 0.001) ↑ BMD (lumbar) (p < 0.001) |
| Rønn et al. 2016 Denmark [43] | 148 PMOa 67.5 y |
RCT 1 y |
375 µg/d MK-7 vs. placebo |
↓ ucOC (p < 0.05) ↓ ucOC:cOC (p < 0.05) ↑ bone structure (tibia) (p < 0.05) |
| Bultynck et al. 2020 UK [56] | 62 (42:20) 80.0 ± 9.6 y |
Prospective | ↑ serum VK | ↓ hip fracture risk |
| Moore et al. 2020 UK [57] |
374 PMO 68.7 y |
cross-sectional | ↑ serum VK1 | ↓ fracture risk (p = 0.04) |
| Sim et al. 2020 Australia [58] |
30 (10:20) 61.8 ± 9.9 y |
RCT 12 w |
136.7 μg/d VK | ↓ ucOC and ucOC:tOC (p ≤ 0.01) |
3. Vitamin K in the Prevention and Therapy of Vascular Calcification and Cardiovascular Diseases
| Author, Year, Country (Ref.) | Subjects (W:M) Age (Mean ± SD) |
Design (Length) | Intervention Exposure |
Findings |
|---|---|---|---|---|
| Geleijnse et al. 2004 Netherlands [76] | 4807 (2971:1836) 67.5 y |
7 y | Q1 ˂ 21.6 μg/d VK2 Q2 21.6–32.7μg/d VK2 Q3 ˃ 32.7 μg/d VK2 |
↓ CHD mortality: RR = 0.43 (95% CI: 0.24–0.77, p = 0.005) Q3 vs. Q1 ↓ AC: OR = 0.48 (95% CI: 0.32–0.71, p ˂ 0.001) Q3 vs. Q1 |
| Gast et al. 2009 Netherlands [77] |
16,057 W 57.0 ± 6.0 y |
Longitudinal 8.1 y |
211.7μg/d VK1 29.1μg/d VK2 |
↓ CHD risk for 10 μg VK2: HR = 0.91 (95% CI: 0.85–1.00, p = 0.04) |
| Shea et al. 2009 USA [78] | 388 (235:153) 68 y |
RCT 3 y |
500 μg/d VK1 vs. control | ↓progression of CAC |
| Schurgers et al. 2010 France [79] | 107 (43:64) 67 ± 13 y |
18 mo | VK levels dp-ucMGP |
↓ VK levels ↑ dp-ucMGP levels with CKD stage |
| Ueland et al. 2010 Norway [80] |
147 (66:81) 74.0 ± 10 y |
20 mo | VK levels dp-ucMGP |
↓ VK levels ↑ dp-ucMGP in symptomatic AS |
| Schlieper et al. 2011 Serbia [81] |
188 (89:99) 58 ± 15 y |
Follow-up, 1104 days |
VK levels dp-ucMGP dp-cMGP |
↓ dp-cMGP ↑ CV: HR = 2.7 (95% CI: 1.2–6.2, p = 0.015) ↑ All-cause: HR = 2.16 (95% CI: 1.1–4.3, p = 0.027) |
| Ueland et al. 2011 Norway [82] |
179 (39:140) 56 y |
2.9 y | VK levels dp-ucMGP |
↓ VK levels; ↑ dp-ucMGP ↑ heart failure: HR=5.62 (95% CI: 2.05–15.46, p = 0.001) |
| Westenfeld et al. 2011 Germany [83] | 103 (48:55) ˃ 60.5 y |
RCT 6 w |
G1–45 µg/d MK-7 G2–135 µg/d MK-7 G3–360 µg/d MK-7 |
↓ dp-ucMGP by 77–93% G2 and G3 vs. control |
| Dalmeijer et al. 2012 Netherlands [84] |
60 (36:24) 59.5 y |
RCT 12 w |
G1–180 μg/d MK-7 G2–360 μg/d MK-7 |
↓ dp-ucMGP by 31% G1 and 46% G2 vs. placebo |
| van den Heuvel et al. 2013 Netherlands [85] | 577 (322:255) 59.9 ± 2.9 y |
Follow-up 5.6 y | VK levels dp-ucMGP |
↓ VK levels; ↑ dp-ucMGP ↑ CVD: HR=2.69 (95% CI: 1.09–6.62, p = 0.032) |
| Caluwé et al. 2014 Norway [86] |
165 (83:82) 70.8 y |
RCT 8 w |
360, 720 or 1080 μg MK-7 thrice weekly | ↓ dp-ucMGP by 17–33–46% |
| Liabeuf et al. 2014 France [87] |
198 (40:158) 64 ± 8 y |
Cross-sectional | VK levels dp-ucMGP |
↓ VK levels; ↑ dp-ucMGP ↑ PAC: OR = 1.88 (95% CI: 1.14–3.11, p = 0.014) |
| Cheung et al. 2015 USA [88] | 3401 (2245:1156) 61.9 y |
Follow-up 13.3 y |
↑ VK daily intake | ↓ CVD mortality: HR = 0.78 (95% CI: 0.64–0.95, p = 0.016) |
| Knapen et al. 2015 Norway [89] |
244 PMW 59.5 ± 3.3 y |
RCT 3 y |
180 µg/d MK-7 vs. placebo | ↓ Stiffness Index β: −0.67 ± 2.78 vs. +0.15 ± 2.51, p = 0.018 ↓ cfPWV: −0.36 ± 1.48 m/s vs. +0.021 ± 1.22 m/s, p = 0.040 |
| Kurnatowska et al. 2015 Poland [90] | 42 (20:22) 58 y |
RCT 270 days |
90 μg/d MK-7 + 10 μg/d vitD vs. control | ↑ CAC ↓dp-ucMGP |
| Asemi et al. 2016 Iran [91] |
66 (31:35) 65.5 y |
RCT 12 w |
180 µg/d MK-7 + 10 µg/d vitD + 1 g/d Ca vs. placebo | ↓ levels of left CIMT (p = 0.02) ↓ insulin (−0.9 vs. +2.6, p = 0.01) ↓ HOMA-IR (−0.4 vs. +0.7, p = 0.01) |
| Fulton et al. 2016 UK [92] |
80 (36:44) 77 ± 5 y |
RCT 6 mo |
100 µg MK-7 vs. placebo | ↓dp-ucMGP (p < 0.001) |
| Kurnatowska et al. 2016 Poland [93] | 38 (17:21) 58.6 y |
RCT 9 mo |
90 μg/d MK-7 + 10 μg/d vitD vs. control | ↓dp-ucMGP by 10.7% |
| Sardana et al. 2016 USA [94] |
66 (6:60) T2D 62 ± 2 y |
Cross-sectional | VK levels dp-ucMGP |
↓ VK levels; ↑ dp-ucMGP ↑ cfPWV (β = 0.40, p = 0.011) |
| Aoun et al. 2017 Lebanon [95] |
50 (20:30) 71.5 y |
RCT 4 w |
360 μg/d MK-7 | ↓ dp-ucMGP by 86% |
| Brandenburg et al. 2017 Germany [96] | 99 (18:81) 69.1 y |
RCT 1 y |
2 mg/d VK1 vs. placebo | ↓ progression of AVC (10.0% vs. 22.0%) |
| Shea et al. 2017 USA [97] |
1061 (615:446) 74 ± 5 y |
Follow-up 12.1 y |
VK1 levels dp-ucMGP |
↑ CVD risk in HBP patients (n = 489): HR = 2.94 (95% CI: 1.4–6.13, p ˂ 0.01) |
| Puzantian et al. 2018 USA [98] |
137 (8:129) 59.6 y |
VK levels dp-ucMGP |
↓ VK levels; ↑ dp-ucMGP ↑ cfPWV (β = 0.21; p = 0.019) |
|
| Dal Canto et al. 2020 Netherlands [99] |
601 (303:298) 70 ± 6 y |
Follow-up 7 and 17 y |
↓ VK levels ↓ vitD levels |
↑ LVMI: β = 5.9 g/m2.7 (95% CI: 1.8–10.0 g/2.7) ↑ All-cause mortality: HR = 1.64 (95% CI: 1.12–2.39, p = 0.011) |
| Roumeliotis et al. 2020 Greece [100] | 66 (31:35) diabetic CKD 68.5 ± 8.6 y |
Follow-up 7 y |
VK levels dp-ucMGP |
↓ VK levels; ↑ dp-ucMGP ↑ CVD mortality: HR = 2.82 (95% CI: 1.07–7.49, p = 0.037) |
| Shea et al. 2020 USA [101] |
3891 (2154:1737) 65 ± 11 y |
Follow-up 13 y |
↓ VK1 levels | ↑ CVD risk: HR = 1.12 (95% CI, 0.94–1.33) ↑ All-cause mortality |
| Wessinger et al. 2020 USA [102] | 60 (11:49) chronic stroke 61.7 ± 7.2 y |
Cross-sectional | VK dietary intake | Among stroke survivors, 82% reported consuming below the Dietary Reference Intake for VK |
References
- Franco, R.; Navarro, G.; Martínez-Pinilla, E. Hormetic and Mitochondria-Related Mechanisms of Antioxidant Action of Phytochemicals. Antioxidants 2019, 8, 373.
- Bjørklund, G.; Chirumbolo, S. Role of oxidative stress and antioxidants in daily nutrition and human health. Nutrition 2017, 33, 311–321.
- Maurya, P.K.; Kumar, P.; Chandra, P. Biomarkers of oxidative stress in erythrocytes as a function of human age. World J. Methodol. 2015, 5, 216–222.
- Rusu, M.E.; Gheldiu, A.-M.; Mocan, A.; Vlase, L.; Popa, D.-S. Anti-aging potential of tree nuts with a focus on phytochemical composition, molecular mechanisms and thermal stability of major bioactive compounds. Food Funct. 2018, 9, 2554–2575.
- Harshman, S.; Shea, M. The Role of Vitamin K in Chronic Aging Diseases: Inflammation, Cardiovascular Disease, and Osteoarthritis. Curr. Nutr. Rep. 2016, 5, 90–98.
- Braasch-Turi, M.; Crans, D.C. Synthesis of Naphthoquinone Derivatives: Menaquinones, Lipoquinones and Other Vitamin K Derivatives. Molecules 2020, 25, 4477.
- Schurgers, L.; Vermeer, C. Determination of phylloquinone and menaquinones in food. Effect of food matrix on circulating vitamin K concentrations. Haemostasis 2000, 30, 298–307.
- Turck, D.; Bresson, J.-L.; Burlingame, B.; Dean, T.; Fairweather-Tait, S.; Heinonen, M.; Hirsch-Ernst, K.I.; Mangelsdorf, I.; McArdle, H.; Naska, A.; et al. Dietary reference values for vitamin K. EFSA J. 2017, 15, e04780.
- Elder, S.J.; Haytowitz, D.B.; Howe, J.; Peterson, J.W.; Booth, S.L. Vitamin K Contents of Meat, Dairy, and Fast Food in the U.S. Diet. J. Agric. Food Chem. 2006, 54, 463–467.
- Melse-Boonstra, A. Bioavailability of Micronutrients from Nutrient-Dense Whole Foods: Zooming in on Dairy, Vegetables, and Fruits. Front. Nutr. 2020, 7, 101.
- Booth, S.L. Vitamin K: Food composition and dietary intakes. Food Nutr. Res. 2012, 56, 5505.
- Margier, M.; Antoine, T.; Siriaco, A.; Nowicki, M.; Halimi, C.; Maillot, M.; Georgé, S.; Reboul, E. The Presence of Pulses within a Meal can Alter Fat-Soluble Vitamin Bioavailability. Mol. Nutr. Food Res. 2019, 63, e1801323.
- Halder, M.; Petsophonsakul, P.; Akbulut, A.C.; Pavlic, A.; Bohan, F.; Anderson, E.; Maresz, K.; Kramann, R.; Schurgers, L. Vitamin K: Double Bonds beyond Coagulation Insights into Differences between Vitamin K1 and K2 in Health and Disease. Int. J. Mol. Sci. 2019, 20, 896.
- Wei, F.-F.; Trenson, S.; Verhamme, P.; Vermeer, C.; Staessen, J.A. Vitamin K-Dependent Matrix Gla Protein as Multifaceted Protector of Vascular and Tissue Integrity. Hypertension 2019, 73, 1160–1169.
- Gröber, U.; Reichrath, J.; Holick, M.F.; Kisters, K. Vitamin K: An old vitamin in a new perspective. Dermato Endocrinol. 2015, 6, e968490.
- Bender, D.; Vitamin, K. Nutritional Biochemistry of the Vitamins; Cambridge University Press: Cambridge, UK, 2003; pp. 131–147. ISBN 9780521803885.
- Simes, D.; Viegas, C.; Araújo, N.; Marreiros, C. Vitamin K as a Diet Supplement with Impact in Human Health: Current Evidence in Age-Related Diseases. Nutrients 2020, 12, 138.
- Li, J.; Lin, J.C.; Wang, H.; Peterson, J.W.; Furie, B.C.; Furie, B.; Booth, S.L.; Volpe, J.J.; Rosenberg, P.A. Novel Role ofVitamin K in Preventing Oxidative Injury to Developing Oligodendrocytes and Neurons. J. Neurosci. 2003, 23, 5816–5826.
- Sinbad, O.O.; Folorunsho, A.A.; Olabisi, O.L.; Ayoola, A.O.; Temitope, J. Vitamins as Antioxidants. J. Food Sci. Nutr. Res. 2019, 2, 214–235.
- Rusu, M.E.; Simedrea, R.; Gheldiu, A.-M.; Mocan, A.; Vlase, L.; Popa, D.-S.; Ferreira, I.C.F.R. Benefits of tree nut consumption on aging and age-related diseases: Mechanisms of actions. Trends Food Sci. Technol. 2019, 88, 104–120.
- Fusaro, M.; Gallieni, M.; Rizzo, M.A.; Stucchi, A.; Delanaye, P.; Cavalier, E.; Moysés, R.M.A.; Jorgetti, V.; Iervasi, G.; Giannini, S.; et al. Vitamin K plasma levels determination in human health. Clin. Chem. Lab. Med. 2017, 55, 789–799.
- DiNicolantonio, J.J.; Bhutani, J.; O’Keefe, J.H. The health benefits of vitamin K. Open Hear 2015, 2, e000300.
- Akbulut, A.; Pavlic, A.; Petsophonsakul, P.; Halder, M.; Maresz, K.; Kramann, R.; Schurgers, L. Vitamin K2 Needs an RDI Separate from Vitamin K1. Nutrients 2020, 12, 1852.
- Louka, M.; Fawzy, A.; Naiem, A.; Elseknedy, M.; Abdelhalim, A.; Abdelghany, M. Vitamin D and K signaling pathways in hepatocellular carcinoma. Gene 2017, 629, 108–116.
- Kim, Y.; Keogh, J.; Clifton, P. Benefits of nut consumption on insulin resistance and cardiovascular risk factors: Multiple potential mechanisms of actions. Nutrients 2017, 9, 1271.
- Paul, C.I.; Vitamin, K. Textbook of Natural Medicine, 5th ed.; Pizzorno, J.E., Murray, M.T., Eds.; Churchill Livingstone: St. Louis, MO, USA, 2020; pp. 919–947.e5. ISBN 978-0-323-52342-4.
- Vermeer, C.; Raes, J.; van’t Hoofd, C.; Knapen, M.H.J.; Xanthoulea, S. Menaquinone Content of Cheese. Nutrients 2018, 10, 446.
- Ferland, G. Vitamin K and brain function. Semin Thromb Hemost. 2013, 39, 849–855.
- Beulens, J.W.J.; Booth, S.L.; van den Heuvel, E.G.; Stoecklin, E.; Baka, A.; Vermeer, C. The role of menaquinones (vitamin K₂) in human health. Br. J. Nutr. 2013, 110, 1357–1368.
- Ferland, G.; Vitamin, K. An emerging nutrient in brain function. Biofactors 2012, 38, 151–157.
- Thijssen, H.; Drittij-Reijnders, M. Vitamin K status in human tissues: Tissue-specific accumulation of phylloquinone and menaquinone-4. Br. J. Nutr. 1996, 75, 121–127.
- Sato, T.; Inaba, N.; Yamashita, T. MK-7 and Its Effects on Bone Quality and Strength. Nutrients 2020, 12, 965.
- Kirk, B.; Feehan, J.; Lombardi, G.; Duque, G. Muscle, Bone, and Fat Crosstalk: The Biological Role of Myokines, Osteokines, and Adipokines. Curr. Osteoporos Rep. 2020, 18, 388–400.
- Hill, H.S.; Grams, J.; Walton, R.G.; Liu, J.; Moellering, D.R.; Garvey, W.T. Carboxylated and uncarboxylated forms of osteocalcin directly modulate the glucose transport system and inflammation in adipocytes. Horm. Metab. Res. 2014, 46, 341–347.
- Mohammad Rahimi, G.R.; Niyazi, A.; Alaee, S. The effect of exercise training on osteocalcin, adipocytokines, and insulin resistance: A systematic review and meta-analysis of randomized controlled trials. Osteoporos Int. 2021, 32, 213–224.
- Tsugawa, N.; Shiraki, M. Vitamin K Nutrition and Bone Health. Nutrients 2020, 12, 1909.
- Binkley, N.C.; Krueger, D.C.; Kawahara, T.N.; Engelke, J.A.; Chappell, R.J.; Suttie, J.W. A high phylloquinone intake is required to achieve maximal osteocalcin gamma-carboxylation. Am. J. Clin. Nutr. 2002, 76, 1055–1060.
- Lin, X.; Brennan-Speranza, T.C.; Levinger, I.; Yeap, B.B. Undercarboxylated Osteocalcin: Experimental and Human Evidence for a Role in Glucose Homeostasis and Muscle Regulation of Insulin Sensitivity. Nutrients 2018, 10, 847.
- Fusaro, M.; Cianciolo, G.; Brandi, M.L.; Ferrari, S.; Nickolas, T.L.; Tripepi, G.; Plebani, M.; Zaninotto, M.; Iervasi, G.; La Manna, G.; et al. Vitamin K and Osteoporosis. Nutrients 2020, 12, 3625.
- Shiraki, M.; Shiraki, Y.; Aoki, C.; Miura, M. Vitamin K2 (menatetrenone) effectively prevents fractuRes. and sustains lumbar bone mineral density in osteoporosis. J. Bone Min. Res. 2000, 15, 515–521.
- Bolton-Smith, C.; McMurdo, M.E.; Paterson, C.R.; Mole, P.A.; Harvey, J.M.; Fenton, S.T.; Prynne, C.J.; Mishra, G.D.; Shearer, M.J. Two-year randomized controlled trial of vitamin K1 (phylloquinone) and vitamin D3 plus calcium on the bone health of older women. J. Bone Min. Res. 2007, 22, 509–519.
- Binkley, N.; Harke, J.; Krueger, D.; Engelke, J.; Vallarta-Ast, N.; Gemar, D.; Checovich, M.; Chappell, R.; Suttie, J. Vitamin K Treatment Reduces Undercarboxylated Osteocalcin but Does Not Alter Bone Turnover, Density, or Geometry in Healthy Postmenopausal North American Women. J. Bone Min. Res. 2009, 24, 983–991.
- Rønn, S.; Harsløf, T.; Pedersen, S.; Langdahl, B. Vitamin K2 (menaquinone-7) prevents age-related deterioration of trabecular bone microarchitecture at the tibia in postmenopausal women. Eur. J. Endocrinol. 2016, 175, 541–549.
- Iwamoto, J.; Takeda, T.; Ichimura, S. Effect of menatetrenone on bone mineral density and incidence of vertebral fractuRes. in postmenopausal women with osteoporosis: A comparison with the effect of etidronate. J. Orthop. Sci. 2001, 6, 487–492.
- Purwosunu, Y.; Muharram; Rachman, I.A.; Reksoprodjo, S.; Sekizawa, A. Vitamin K 2 treatment for postmenopausal osteoporosis in Indonesia. J. Obs. Gynaecol. Res. 2006, 32, 230–234.
- Knapen, M.; Schurgers, L.; Vermeer, C. Vitamin K 2 supplementation improves hip bone geometry and bone strength indices in postmenopausal women. Osteoporos Int. 2007, 18, 963–972.
- Booth, S.L.; Dallal, G.; Shea, M.K.; Gundberg, C.; Peterson, J.W.; Dawson-Hughes, B. Effect of Vitamin K Supplementation on Bone Loss in Elderly Men and Women. J. Clin. Endocrinol. Metab. 2008, 93, 1217–1223.
- Cheung, A.M.; Tile, L.; Lee, Y.; Tomlinson, G.; Hawker, G.; Scher, J.; Hu, H.; Vieth, R.; Thompson, L.; Jamal, S.; et al. Vitamin K supplementation in postmenopausal women with osteopenia (ECKO trial): A randomized controlled trial. PLoS Med. 2008, 5, e196.
- Hirao, M.; Hashimoto, J.; Ando, W.; Ono, T.; Yoshikawa, H. Response of serum carboxylated and undercarboxylated osteocalcin to alendronate monotherapy and combined therapy with vitamin K2 in postmenopausal women. J. Bone Min. Metab. 2008, 26, 260–264.
- Tsugawa, N.; Shiraki, M.; Suhara, Y.; Kamao, M.; Ozaki, R.; Tanaka, K.; Okano, T. Low plasma phylloquinone concentration is associated with high incidence of vertebral fracture in Japanese women. J. Bone Min. Metab. 2008, 26, 79–85.
- Yamauchi, M.; Yamaguchi, T.; Nawata, K.; Takaoka, S.; Sugimoto, T. Relationships between undercarboxylated osteocalcin and vitamin K intakes, bone turnover, and bone mineral density in healthy women. Clin. Nutr. 2010, 29, 761–765.
- Je, S.H.; Joo, N.-S.; Choi, B.-H.; Kim, K.-M.; Kim, B.-T.; Park, S.-B.; Cho, D.-Y.; Kim, K.-N.; Lee, D.-J. Vitamin K Supplement Along with Vitamin D and Calcium Reduced Serum Concentration of Undercarboxylated Osteocalcin While Increasing Bone Mineral Density in Korean Postmenopausal Women over Sixty-Years-Old. J. Korean Med. Sci. 2011, 26, 1093–1098.
- Kanellakis, S.; Moschonis, G.; Tenta, R.; Schaafsma, A.; van den Heuvel, E.; Papaioannou, N.; Lyritis, G.; Manios, Y. Changes in parameters of bone metabolism in postmenopausal women following a 12-month intervention period using dairy products enriched with calcium, vitamin D, and phylloquinone (vitamin K(1)) or menaquinone-7 (vitamin K (2)): The Postmenopausal Health Study II. Calcif. Tissue Int. 2012, 90, 251–262.
- Knapen, M.; Drummen, N.; Smit, E.; Vermeer, C.; Theuwissen, E. Three-year low-dose menaquinone-7 supplementation helps decrease bone loss in healthy postmenopausal women. Osteoporos Int. 2013, 24, 2499–2507.
- Jiang, Y.; Zhang, Z.-L.; Zhang, Z.-L.; Zhu, H.-M.; Wu, Y.-Y.; Cheng, Q.; Wu, F.-L.; Xing, X.-P.; Liu, J.-L.; Yu, W.; et al. Menatetrenone versus alfacalcidol in the treatment of Chinese postmenopausal women with osteoporosis: A multicenter, randomized, double-blinded, double-dummy, positive drug-controlled clinical trial. Clin. Interv. Aging 2014, 9, 121–127.
- Bultynck, C.; Munim, N.; Harrington, D.; Judd, L.; Ataklte, F.; Shah, Z.; Dockery, F. Prevalence of vitamin K deficiency in older people with hip fracture. Acta Clin. Belg. 2020, 75, 136–140.
- Moore, A.E.; Kim, E.; Dulnoan, D.; Dolan, A.L.; Voong, K.; Ahmad, I.; Gorska, R.; Harrington, D.J.; Hampson, G. Serum vitamin K 1 (phylloquinone) is associated with fracture risk and hip strength in post-menopausal osteoporosis: A cross-sectional study. Bone 2020, 141, 115630.
- Sim, M.; Lewis, J.R.; Prince, R.L.; Levinger, I.; Brennan-Speranza, T.C.; Palmer, C.; Bondonno, C.P.; Bondonno, N.P.; Devine, A.; Ward, N.C.; et al. The effects of vitamin K-rich green leafy vegetables on bone metabolism: A 4-week randomised controlled trial in middle-aged and older individuals. Bone Rep. 2020, 12, 100274.
- Hooshmand, S.; Kern, M.; Metti, D.; Shamloufard, P.; Chai, S.C.; Johnson, S.A.; Payton, M.E.; Arjmandi, B.H. The effect of two doses of dried plum on bone density and bone biomarkers in osteopenic postmenopausal women: A randomized, controlled trial. Osteoporos Int. 2016, 27, 2271–2279.
- Higgs, J.; Derbyshire, E.; Styles, K. Nutrition and osteoporosis prevention for the orthopaedic surgeon: A wholefoods approach. EFORT Open Rev. 2017, 2, 300–308.
- Emaus, N.; Gjesdal, C.G.; Almås, B.; Christensen, M.; Grimsgaard, A.; Berntsen, G.; Salomonsen, L.; Fønnebø, V. Vitamin K2 supplementation does not influence bone loss in early menopausal women: A randomised double-blind placebo-controlled trial. Osteoporos Int. 2010, 21, 1731–1740.
- Feskanich, D.; Weber, P.; Willett, W.C.; Rockett, H.; Booth, S.L.; Colditz, G.A. Vitamin K intake and hip fractuRes. in women: A prospective study. Am. J. Clin. Nutr. 1999, 69, 74–79.
- Popa, D.-S.; Rusu, M.E. Isoflavones: Vegetable Sources, Biological Activity, and Analytical Methods for Their Assessment. In Superfood and Functional Food—The Development of Superfoods and Their Roles as Medicine; Shiomi, N., Waisundara, V., Eds.; InTech: London, UK, 2017; ISBN 978-953-51-2942-4.
- Lappe, J.; Kunz, I.; Bendik, I.; Prudence, K.; Weber, P.; Recker, R.; Heaney, R.P. Effect of a combination of genistein, polyunsaturated fatty acids and vitamins D3 and K1 on bone mineral density in postmenopausal women: A randomized, placebo-controlled, double-blind pilot study. Eur. J. Nutr. 2013, 52, 203–215.
- Capozzi, A.; Scambia, G.; Lello, S. Calcium, vitamin D, vitamin K2, and magnesium supplementation and skeletal health. Maturitas 2020, 140, 55–63.
- Goddek, S. Vitamin D3 and K2 and their potential contribution to reducing the COVID-19 mortality rate. Int. J. Infect. Dis. 2020, 99, 286–290.
- Mozos, I.; Stoian, D.; Luca, C.T. Crosstalk between Vitamins A, B12, D, K, C, and E Status and Arterial Stiffness. Dis. Markers. 2017, 2017, 8784971.
- Jaminon, A.M.G.; Dai, L.; Qureshi, A.R.; Evenepoel, P.; Ripsweden, J.; Söderberg, M.; Witasp, A.; Olauson, H.; Schurgers, L.J.; Stenvinkel, P. Matrix Gla protein is an independent predictor of both intimal and meDial. vascular calcification in chronic kidney disease. Sci. Rep. 2020, 10, 6586.
- Shioi, A.; Morioka, T.; Shoji, T.; Emoto, M. The Inhibitory Roles of Vitamin K in Progression of Vascular Calcification. Nutrients 2020, 12, 583.
- Dai, L.; Schurgers, L.J.; Shiels, P.G.; Stenvinkel, P. Early vascular ageing in chronic kidney disease: Impact of inflammation, vitamin K, senescence and genomic damage. Nephrol. Dial. Transplant. 2020, 35, ii31–ii37.
- Simes, D.C.; Viegas, C.S.B.; Araújo, N.; Marreiros, C. Vitamin K as a Powerful Micronutrient in Aging and Age-Related Diseases: Pros and Cons from Clinical Studies. Int. J. Mol. Sci. 2019, 20, 4150.
- Cozzolino, M.; Fusaro, M.; Ciceri, P.; Gasperoni, L.; Cianciolo, G. The Role of Vitamin K in Vascular Calcification. Adv. Chronic Kidney Dis. 2019, 26, 437–444.
- Dofferhoff, A.S.M.; Piscaer, I.; Schurgers, L.J.; Visser, M.P.J.; van den Ouweland, J.; de Jong, P.; Gosens, R.; Hackeng, T.; van Daal, H.; Lux, P.; et al. Reduced vitamin K status as a potentially modifiable risk factor of severe COVID-19. Clin. Infect. Dis. 2020, ciaa1258.
- Roumeliotis, S.; Dounousi, E.; Salmas, M.; Eleftheriadis, T.; Liakopoulos, V. Vascular Calcification in Chronic Kidney Disease: The Role of Vitamin K- Dependent Matrix Gla Protein. Front. Med. 2020, 7, 154.
- Shea, M.K.; Booth, S.L. Vitamin K, Vascular Calcification, and Chronic Kidney Disease: Current Evidence and Unanswered Questions. Curr. Dev. Nutr. 2019, 3, nzz077.
- Geleijnse, J.M.; Vermeer, C.; Grobbee, D.E.; Schurgers, L.J.; Knapen, M.H.J.; van der Meer, I.M.; Hofman, A.; Witteman, J.C.M. Dietary Intake of Menaquinone Is Associated with a Reduced Risk of Coronary Heart Disease: The Rotterd. Am. Study J. Nutr. 2004, 134, 3100–3105.
- Gast, G.C.M.; De Roos, N.M.; Sluijs, I.; Bots, M.L.; Beulens, J.W.J.; Geleijnse, J.M.; Witteman, J.C.; Grobbee, D.E.; Peeters, P.H.M.; Van Der Schouw, Y.T. A high menaquinone intake reduces the incidence of coronary heart disease. Nutr. Metab. Cardiovasc. Dis. 2009, 19, 504–510.
- Shea, M.K.; O’Donnell, C.J.; Hoffmann, U.; Dallal, G.E.; Dawson-Hughes, B.; Ordovas, J.; Price, P.A.; Williamson, M.K.; Booth, S.L. Vitamin K supplementation and progression of coronary artery calcium in older men and women. Am. J. Clin. Nutr. 2009, 89, 1799–1807.
- Schurgers, L.J.; Barreto, D.V.; Barreto, F.C.; Liabeuf, S.; Renard, C.; Magdeleyns, E.; Vermeer, C.; Choukroun, G.; Massy, Z. The circulating inactive form of matrix gla protein is a surrogate marker for vascular calcification in chronic kidney disease: A preliminary report. Clin. J. Am. Soc. Nephrol. 2010, 5, 568–575.
- Ueland, T.; Gullestad, L.; Dahl, C.P.; Aukrust, P.; Aakhus, S.; Solberg, O.G.; Vermeer, C.; Schurgers, L.J. Undercarboxylated matrix Gla protein is associated with indices of heart failure and mortality in symptomatic aortic stenosis. J. Int. Med. 2010, 268, 483–492.
- Schlieper, G.; Westenfeld, R.; Krüger, T.; Cranenburg, E.C.; Magdeleyns, E.J.; Brandenburg, V.M.; Djuric, Z.; Damjanovic, T.; Ketteler, M.; Vermeer, C.; et al. Circulating Nonphosphorylated Carboxylated Matrix Gla Protein Predicts Survival in ESRD. J. Am. Soc. Nephrol. 2011, 22, 387–395.
- Ueland, T.; Dahl, P.; Gullestad, L.; Aakhus, S.; Broch, K.; Skårdal, R.; Vermeer, C.; Aukrust, P.; Schurgers, L. Circulating levels of non-phosphorylated undercarboxylated matrix Gla protein are associated with disease severity in patients with chronic heart failure. Clin. Sci. (Lond.) 2011, 121, 119–127.
- Westenfeld, R.; Krueger, T.; Schlieper, G.; Cranenburg, E.C.M.; Magdeleyns, E.J.; Heidenreich, S.; Holzmann, S.; Vermeer, C.; Jahnen-Dechent, W.; Ketteler, M.; et al. Effect of vitamin K2 supplementation on functional vitamin K deficiency in hemodialysis patients: A randomized trial. Am. J. Kidney Dis. 2012, 59, 186–195.
- Dalmeijer, G.W.; van der Schouw, Y.T.; Magdeleyns, E.; Ahmed, N.; Vermeer, C.; Beulens, J.W.J. The effect of menaquinone-7 supplementation on circulating species of matrix Gla protein. Atherosclerosis 2012, 225, 397–402.
- Van Den Heuvel, E.G.H.M.; Van Schoor, N.M.; Lips, P.; Magdeleyns, E.J.P.; Deeg, D.J.H.; Vermeer, C.; Den Heijer, M. Circulating uncarboxylated matrix Gla protein, a marker of vitamin K status, as a risk factor of cardiovascular disease. Maturitas 2014, 77, 137–141.
- Caluwé, R.; Vandecasteele, S.; Van Vlem, B.; Vermeer, C.; De Vriese, A.S. Vitamin K2 supplementation in haemodialysis patients: A randomized dose-finding study. Nephrol. Dial. Transplant. 2014, 29, 1385–1390.
- Liabeuf, S.; Bourron, O.; Vemeer, C.; Theuwissen, E.; Magdeleyns, E.; Aubert, C.E.; Brazier, M.; Mentaverri, R.; Hartemann, A.; Massy, Z.A. Vascular calcification in patients with type 2 diabetes: The involvement of matrix Gla protein. Cardiovasc. Diabetol. 2014, 13, 85.
- Cheung, C.-L.; Sahni, S.; Cheung, B.M.Y.; Sing, C.-W.; Wong, I.C.K. Vitamin K intake and mortality in people with chronic kidney disease from NHANES III. Clin. Nutr. 2015, 34, 235–240.
- Knapen, M.H.J.; Braam, L.A.J.L.M.; Drummen, N.E.; Bekers, O.; Hoeks, A.P.G.; Vermeer, C. Menaquinone-7 supplementation improves arterial stiffness in healthy postmenopausal women. A double-blind randomised clinical trial. Thromb. Haemost. 2015, 113, 1135–1144.
- Kurnatowska, I.; Grzelak, P.; Masajtis-Zagajewska, A.; Kaczmarska, M.; Stefańczyk, L.; Vermeer, C.; Maresz, K.; Nowicki, M. Effect of vitamin K2 on progression of atherosclerosis and vascular calcification in nondialyzed patients with chronic kidney disease stages 3-5. Pol. Arch. Med. Wewn. 2015, 125, 631–640.
- Asemi, Z.; Raygan, F.; Bahmani, F.; Rezavandi, Z.; Talari, H.R.; Rafiee, M.; Poladchang, S.; Mofrad, M.D.; Taheri, S.; Mohammadi, A.A.; et al. The effects of vitamin D, K and calcium co-supplementation on carotid intima-media thickness and metabolic status in overweight type 2 diabetic patients with CHD. Br. J. Nutr. 2016, 116, 286–293.
- Fulton, R.L.; McMurdo, M.E.T.; Hill, A.; Abboud, R.J.; Arnold, G.P.; Struthers, A.D.; Khan, F.; Vermeer, C.; Knappen, M.H.J.; Drummen, N.E.A.; et al. Effect of Vitamin K on Vascular Health and Physical Function in Older People with Vascular DiseaseA Randomised Controlled Trial. J. Nutr. Heal Aging 2016, 20, 325–333.
- Kurnatowska, I.; Grzelak, P.; Masajtis-Zagajewska, A.; Kaczmarska, M.; Stefańczyk, L.; Vermeer, C.; Maresz, K.; Nowicki, M. Plasma Desphospho-Uncarboxylated Matrix Gla Protein as a Marker of Kidney Damage and Cardiovascular Risk in Advanced Stage of Chronic Kidney Disease. Kidney Blood Press Res. 2016, 41, 231–239.
- Sardana, M.; Vasim, I.; Varakantam, S.; Kewan, U.; Tariq, A.; Koppula, M.R.; Syed, A.A.; Beraun, M.; Drummen, N.E.A.; Vermeer, C.; et al. Inactive Matrix Gla-Protein and Arterial Stiffness in Type 2 Diabetes Mellitus. Am. J. Hypertens. 2016, 30, 196–201.
- Aoun, M.; Makki, M.; Azar, H.; Matta, H.; Chelala, D.N. High Dephosphorylated-Uncarboxylated MGP in Hemodialysis patients: Risk factors and response to vitamin K2, A pre-post intervention clinical trial. BMC Nephrol. 2017, 18, 191.
- Brandenburg, V.; Reinartz, S.; Kaesler, N.; Krüger, T.; Dirrichs, T.; Kramann, R.; Peeters, F.; Floege, J.; Keszei, A.; Marx, N.; et al. Slower Progress of Aortic Valve Calcification With Vitamin K Supplementation: Results From a Prospective Interventional Proof-of-Concept Study. Circulation 2017, 135, 2081–2084.
- Shea, M.K.; Booth, S.L.; Weiner, D.E.; Brinkley, T.E.; Kanaya, A.M.; Murphy, R.A.; Simonsick, E.M.; Wassel, C.L.; Vermeer, C.; Kritchevsky, S.B. Circulating Vitamin K Is Inversely Associated with Incident Cardiovascular Disease Risk among Those Treated for Hypertension in the Health, Aging, and Body Composition Study (Health ABC). J. Nutr. 2017, 147, 888–895.
- Puzantian, H.; Akers, S.R.; Oldland, G.; Javaid, K.; Miller, R.; Ge, Y.; Ansari, B.; Lee, J.; Suri, A.; Hasmath, Z.; et al. Circulating Dephospho-Uncarboxylated Matrix Gla-Protein Is Associated With Kidney Dysfunction and Arterial Stiffness. Am. J. Hypertens. 2018, 31, 988–994.
- Dal Canto, E.; Beulens, J.W.J.; Elders, P.; Rutters, F.; Stehouwer, C.D.A.; Van Der Heijden, A.A.; Van Ballegooijen, A.J. The Association of Vitamin D and Vitamin K Status with Subclinical MeasuRes. of Cardiovascular Health and All-Cause Mortality in Older Adults: The Hoorn Study. J. Nutr. 2020, 150, 3171–3179.
- Roumeliotis, S.; Roumeliotis, A.; Stamou, A.; Leivaditis, K.; Kantartzi, K.; Panagoutsos, S.; Liakopoulos, V. The Association of dp-ucMGP with Cardiovascular Morbidity and Decreased Renal Function in Diabetic Chronic Kidney Disease. Int. J. Mol. Sci. 2020, 21, 6035.
- Shea, M.K.; Barger, K.; Booth, S.L.; Matuszek, G.; Cushman, M.; Benjamin, E.J.; Kritchevsky, S.B.; Weiner, D.E. Vitamin K status, cardiovascular disease, and all-cause mortality: A participant-level meta-analysis of 3 US cohorts. Am. J. Clin. Nutr. 2020, 111, 1170–1177.
- Wessinger, C.; Hafer-Macko, C.; Ryan, A.S. Vitamin K Intake in Chronic Stroke: Implications for Dietary Recommendations. Nutrients 2020, 12, 3059.
- Haugsgjerd, T.R.; Egeland, G.M.; Nygård, O.K.; Vinknes, K.J.; Sulo, G.; Lysne, V.; Igland, J.; Tell, G.S. Association of dietary vitamin K and risk of coronary heart disease in middle-age adults: The Hordaland Health Study Cohort. BMJ Open 2020, 10, e035953.
- Caluwé, R.; Verbeke, F.; De Vriese, A.S. Evaluation of vitamin K status and rationale for vitamin K supplementation in dialysis patients. Nephrol. Dial. Transplant. 2020, 35, 23–33.
- Liu, Y.-P.; Gu, Y.-M.; Thijs, L.; Knapen, M.H.J.; Salvi, E.; Citterio, L.; Petit, T.; Carpini, S.D.; Zhang, Z.; Jacobs, L.; et al. Inactive Matrix Gla Protein Is Causally Related to Adverse Health Outcomes: A Mendelian Randomization Study in a Flemish Population. Hypertension 2015, 65, 463–470.





