
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Lenaic Lartigue | + 1590 word(s) | 1590 | 2019-12-25 04:05:32 | | | |
| 2 | Catherine Yang | -12 word(s) | 1578 | 2020-01-08 02:23:39 | | | | |
| 3 | Catherine Yang | Meta information modification | 1578 | 2020-01-09 04:10:57 | | | | |
| 4 | Dean Liu | -44 word(s) | 1534 | 2021-10-13 08:11:09 | | |
Video Upload Options
The various imaging techniques used in clinic have advantages and disadvantages. The advent of nanotechnology offers the possibility to combine several imaging agents within the same nano-object. This will allow to perform multi-imaging and thus obtain additional information during the same clinical procedure. Here, the focus are made on the potentialities of biomodal agents labeled for MRI and optical imaging. The three main associations for obtaining magneto-fluorescent objects will be presented: (1) association by covalent bonding; (2) encapsulation in matrices; (3) dispersion in nanoassemblies.
1. Introduction
The coexistence of several imaging agents within the same nano-object suggests promising prospects in medical imaging field. The multifunctionality providing by these nano-tools allows to benefit from the complementarity of the various imaging techniques in terms of sensitivity, spatial resolution or depth penetration. Among the different possible combinations, growing interest has been shown in nanosystems comprising magnetic nanoparticles (MNPs) for MRI and luminescent entities for fluorescent imaging in recent years (Figure 1) [1]. Luminescence imaging, thanks to its multiple-label possibility, its high sensitivity and spatial resolution, is widely used to follow biological processes or in histopathology. However, extinction phenomena (diffusion and absorption of light by tissue) limit the depth penetration of this imaging technique. By contrast, MR present an unlimited depth penetration and greater soft-tissue contrast. Nevertherless, the low sensitivity of the MR technique makes it difficult to distinguish benign from malignant disease even with long acquisition time. So, combining these two safety techniques (using non-ionizing radiation) allows advantage to be taken of the high high sensitivity and spatial resolution of luminescence imaging, associated to the good spatial resolution and deep tissue penetration of MRI. This combination is particularly interesting for correlating in vitro monitoring and in vivo tracking [2]. We will focus, here, on structure integrating negative MRI constrast agent (i.e. superparamagnetic nanoparticles). Their efficiency will be linked to a very strong transverse relaxivity, r2, and a significant r2/r1 ratio.[3] The very good sensitivity of the luminophore will be proportional to its brightness. Its penetration into biological tissue will be all the greater if the luminescent entities emit into the biological window (700-950nm).[4]
 Figure 1. Fluorescence imaging vs. magnetic resonance imaging (MRI). Comparison between both imaging techniques showing their complementarity.
Figure 1. Fluorescence imaging vs. magnetic resonance imaging (MRI). Comparison between both imaging techniques showing their complementarity.
2. Association by Covalent Bonding (Nanoparticles)
Among the different association methods, grafting fluorescent entities on the surface of magnetic nanoparticles represents a simple approach (Figure 2) [5][6][7][8]. For example, fluorescent dyes (rhodamine B, λmaxem = 578 nm or fluorescein derivatives, λmaxem = 516 nm) are coupled to iron oxide surface [5]. These nanoparticles allow in cellulo motions of endosomes to be followed when exposed to a magnetic field gradient. Near-infrared (NIR) dyes have been also grafted as IR-820 cyanine derivative (λmaxem = 900 nm) [7] or dialkyl carbocyanine (λmaxem = 780 nm) [8] to obtain fluorescent systems emitting in the first biological window. Iron oxide multicore nanoparticles assembled by hydrophilic polymer have been envisaged to improve the MRI contrast agent properties [6][8]. Thus, the use of multicore Ferahme® iron oxide nanoparticles (dcore = 6–7 nm, dH = 17–31 nm) coupled to TO-PRO®-1 (λmaxem = 531 nm) gives bifunctional nanoparticles with a transverse relaxivity, r2 = 122 mM−1 s−1 (0.47 T, r2/r1 = 5) [6]. This transverse relaxivity is still improved using 8 nm iron oxide nanoparticles (r2 = 202 mM−1 s−1, r2/r1 = 3.8 at 0.47 T) embedded in polyacrylic acid matrix (dH = 90 nm) [8]. Moreover, these systems present NIR-emissive dye using a dialkylcarbocyanine as fluorophores (λmaxem in the region 751/780 nm and ε > 125,000 cm−1 M−1). The association of QDots, known to be brighter than small molecules, with magnetic nanoparticles is also envisaged [[9][10][11][12][13][14][15]]. The most common approach used is to prepare core–satellite systems. In this approach the core is composed of iron oxide nanoparticles surrounded by quantum dots (usually CdSe/ZnS) [9][10][11][15]. Pahari et al. have recently described an invert strategy where quantum dots (3.2 nm CdSe nanoparticles) are in the core and a shell of iron oxide is growned around (thickness of 1.3 nm) [14]. By this approach, a very good transverse relaxivity (r2 = 304 mM−1 s−1 at 9.4 T) is noted. Although these systems are easily synthetized, the close proximity between the luminophores and the metal core leads to a strong emission quenching. Indeed, electronic energy or electron transfers between both entities can take place while iron oxide nanoparticles significantly absorb at wavelengths less than 450 nm. The choice of fluorophores and their distance from the metallic core will therefore be essential. Moreover, direct exposure of fluorophores to the surrounding environment can modify the emissive properties of the system. Finally, the requirement of high colloidal stability of the final nanoassembly in aqueous solution excludes extensive grafting of fluorescent entities, especially if the latter are organic and hydrophobic. All together, these limitations produce low-emissive imaging agents.
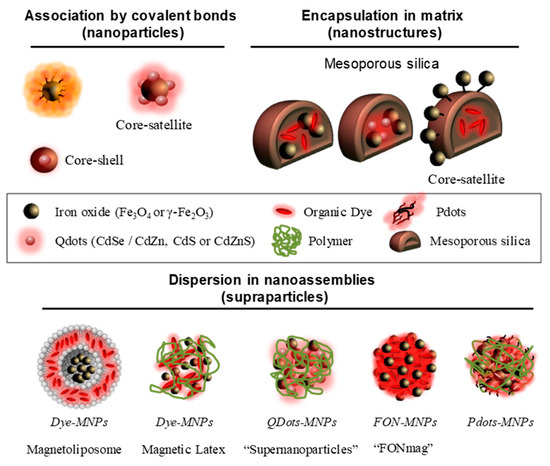
Figure 2. Schematic representation of magneto-fluorescent nanosystems.
3. Encapsulation in Silica Matrix (Nanostructure)
To protect the luminophores from quenching by the surrounding medium, the encapsulation of magnetic nanoparticles (γ-Fe2O3) and fluorescent units (small molecules, e.g., rhodamine or FITC derivatives; [[16][17][18][19][20]] or Qdots like CdSe/CdZn, CdS or CdZnS [[21][22][23][24][25]]) in mesoporous silica matrices has been envisaged (Figure 2). The encapsulation of magnetic nanoparticles and quantum dots leads to interactions between these two active units. This interaction induces (i) an increase of the magnetic anisotropy, (ii) a blue-shift of the fluorescence emission and (iii) a decrease of the quantum yield [21][24]. Silica-doped with organic dye core surrounded by magnetic nanoparticles (core-satellite assemblies) are also envisaged [16][17]. In these structures, the combination of several magnetic nanoparticles has the effect of drastically increasing the r2 value in comparison of magnetic nanoparticles alone. Lee et al. describe an r2 increase from 26.8 to 76.2 mM−1 s−1 (1.5 T) [16], and another study shows a rise from 116 to 397 mM−1 s−1 (9.4 T) [17]. These silica matrices show low cytotoxicity but provide only small amounts of encapsulated active units. In addition, although the fluorescent entities are protected from the external environment by encapsulation, they can diffuse freely outside the porous matrix as they are not covalently attached. To counter this phenomenon, hydrophobic fluorescent units amenable to self-assemble have been proposed to impart the magneto-fluorescent nanosystems with better structural stability and reduced dye leakage.
4. Dispersion in Nanoassemblies (Supraparticles)
The use of magneto-fluorescent nanoassemblies provides generally biodegradable systems that advantageously avoid bioaccumulation. In this context, several molecular matrices are envisaged composed of polymers [[26][27][28][29][30][31][32][33][34]], lipids [[35][36][37][38]], PDots [[39][40][41][42]] or organic molecule [[43][44][45][46][47]](Figure 2). In this type of organization we will distinguish the assemblies with inert matrices of those composed of active units. Inert matrices are mainly composed of lipids or polymers. For instance, magnetic nanoparticles can be encapsulated in liposomes, and subsequently functionalized by a fluorescent molecule, here rhodamine [35]. The magnetofluorescent liposome exhibits good T2-contrast agent properties with r2 = 268 mM−1 s−1 at 4.7 T (r2/r1 = 85). Another approach is based on the use of polymers to combine magnetic nanoparticles and fluorescent entities. In the work of R. K. Prud’homme et al., polyethylene glycol has also been used to assemble hydrophobic NIR fluorophores (λmaxem = 800 nm), tris-(porphyrinate) zinc (II), and magnetic nanoparticles [28]. The authors show an increase in r2 from 66 to 533 mM−1 s−1 as the wt% IO increases from 4 to 16% [28]. Bawendi et al. describe the association of quantum dots and densely packed magnetic nanoparticles into “supernanoparticles” thanks to poly(vinylpyrrolidone) (PVP) ethylene glycol (EG) [27]. These assemblies (dH = 120 nm) display a high r2 value of 402.7 mM−1 s−1 at 9.4 T [27]. In the three last cases, the effectiveness of these multimodal structures has been demonstrated in murine models. Correlative treatments of the MRI and fluorescence signals have proved the preservation of the in vivo integrity of the nanoassemblies and validate the design of multimodal nanostructures.
The second type of self-assembled systems implies functional units as molecular bricks (Figure 2), thus limiting the number of organic species administrated in vivo. Hyeon et al. have assembled magnetic nanoparticles with a polyethylene glycol block polymer . This polymer is functionalized with an imidazole derivative and fluorescent porphyrins (chlorin e6) whose emission is deactivated upon dye self-assembling [32]. The imidazole derivative is a pH-sensitive group which allows the disintegration of the nanostructure in the tumor medium (acidic pH) and leading to the reappearance of fluorescence. In this system (dH = 70 nm), the self-assembly of 3 nm iron oxide nanoparticles provides a transverse relaxivity r2 = 44 mM−1 s−1 at 1.5 T (r2/r1 = 13.3). Nanoassemblies incorporating Pdots and magnetic nanoparticles into phospholipid micelles improve relaxivity properties (r2 = 152mM−1 s−1 at 3.0 T) and enhance MRI contrast efficiency thereof [32]. Moreover, for these systems, important brightness and photostability under irradiation of the fluorophores have been demonstrated during in cellulo fluorescence microscopy . The use of small hydrophobic molecules with iron oxide nanoparticles chelating functions is another effective approach to obtain magnetofluorescent systems [43][44][45][46][47]. In these systems, the fluorescent core composed of 105 dyes is surrounded by magnetic nanoparticles shell [43]. This architecture deals with a very effective dual-mode contrast agent with a brightness around 107 L mol−1 cm−1 and transverse relaxivity r2 = 238 mM−1 s−1 (0.47 T) [43]. This contrast agent displays excellent properties in liver imaging on small rodents both as a cellular label and as in vivo follow-up [43]. In cellulo stability of these systems could be controlled by varying stabilizing ligands [45]. The use of polyacrylic acid allows a very cohesive architecture, when the stabilization by citrate ions allows a dissociation [45]. These systems have been functionalized with polyethylene glycol-based copolymers to increase their circulation time [47]. Moreover, it has been shown that the presence of a hydrophobic tail in the copolymer increases the r2/r1 two times compared to those which are without one [47].
References
- Nadja C. Bigall; Wolfgang J. Parak; Dirk Dorfs; Fluorescent, magnetic and plasmonic—Hybrid multifunctional colloidal nano objects. Nano Today 2012, 7, 282-296, 10.1016/j.nantod.2012.06.007.
- Serena A. Corr; Yury Petrovich Rakovich; Yurii K. Gun’Ko; Multifunctional Magnetic-fluorescent Nanocomposites for Biomedical Applications. Nanoscale Research Letters 2008, 3, 87-104, 10.1007/s11671-008-9122-8.
- Adrien Faucon; Arnaud Brosseau; Philippe Hulin; Steven Nedellec; Julie Hemez; Jérôme Fresnais; Eléna Ishow; Photoactive chelating organic nanospheres as central platforms of bimodal hybrid nanoparticles. Journal of Materials Chemistry C 2013, 1, 3879, 10.1039/c3tc30508b.
- Adrien Faucon; Thomas Maldiney; Olivier Clément; Philippe Hulin; Steven Nedellec; Myriam Robard; Nicolas Gautier; Evelien De Meulenaere; Koen Clays; Tomas Orlando; et al.Alessandro LascialfariCéline Fiorini-DebuisschertJérôme FresnaisEléna Ishow Highly cohesive dual nanoassemblies for complementary multiscale bioimaging. Journal of Materials Chemistry B 2014, 2, 7747-7755, 10.1039/c4tb01199f.
- Franck Bertorelle; Claire Wilhelm; Jacky Roger; Florence Gazeau; Christine Ménager; Valérie Cabuil; Fluorescence-Modified Superparamagnetic Nanoparticles: Intracellular Uptake and Use in Cellular Imaging. Langmuir 2006, 22, 5385-5391, 10.1021/la052710u.
- David Alcántara; Yanyan Guo; Hushan Yuan; Craig J. Goergen; Howard H. Chen; Hoonsung Cho; David E. Sosnovik; Lee Josephson; Fluorochrome-functionalized magnetic nanoparticles for high-sensitivity monitoring of the polymerase chain reaction by magnetic resonance.. Angewandte Chemie International Edition 2012, 51, 6904-6907, 10.1002/anie.201201661.
- Swee Kuan Yen; Dominik Jańczewski; Jeeva Lavanya Lakshmi; Surani Bin Dolmanan; Sudhiranjan Tripathy; Vincent H. B. Ho; Vimalan Vijayaragavan; Anushya Hariharan; Parasuraman Padmanabhan; Kishore K. Bhakoo; et al.Thankiah SudhaharanSohail AhmedYong ZhangSubramanian Tamil Selvan Design and Synthesis of Polymer-Functionalized NIR Fluorescent Dyes–Magnetic Nanoparticles for Bioimaging. ACS Nano 2013, 7, 6796-6805, 10.1021/nn401734t.
- Santimukul Santra; Charalambos Kaittanis; Jan Grimm; J. Manuel Perez; Drug/dye-loaded, multifunctional iron oxide nanoparticles for combined targeted cancer therapy and dual optical/magnetic resonance imaging.. Small 2009, 5, 1862-1868, 10.1002/smll.200900389.
- F. X. Redl; K.-S. Cho; C. B. Murray; Stephen Obrien; Three-dimensional binary superlattices of magnetic nanocrystals and semiconductor quantum dots. Nature 2003, 423, 968-971, 10.1038/nature01702.
- Minjung Cho; Elizabeth Q. Contreras; Seung Soo Lee; Christopher J. Jones; Wonhee Jang; Vicki L. Colvin; Characterization and Optimization of the Fluorescence of Nanoscale Iron Oxide/Quantum Dot Complexes. The Journal of Physical Chemistry C 2014, 118, 14606-14616, 10.1021/jp502194z.
- Edakkattuparambil S. Shibu; Kenji Ono; Sakiko Sugino; Ayami Nishioka; Akikazu Yasuda; Yasushi Shigeri; Shin-Ichi Wakida; Makoto Sawada; Vasudevanpillai Biju; Photouncaging Nanoparticles for MRI and Fluorescence Imaging in Vitro and in Vivo. ACS Nano 2013, 7, 9851-9859, 10.1021/nn4043699.
- Jinhao Gao; Wei Zhang; Pingbo Huang; Bei Zhang; Xixiang Zhang; Bing Xu; Intracellular Spatial Control of Fluorescent Magnetic Nanoparticles. Journal of the American Chemical Society 2008, 130, 3710-3711, 10.1021/ja7103125.
- Subramanian Tamil Selvan; Pranab K. Patra; Chung Yen Ang; Jackie Y. Ying; Synthesis of Silica-Coated Semiconductor and Magnetic Quantum Dots and Their Use in the Imaging of Live Cells. Angewandte Chemie International Edition 2007, 46, 2448-2452, 10.1002/anie.200604245.
- Sandip K. Pahari; Shunit Olszakier; Itamar Kahn; Lilac Amirav; Magneto-Fluorescent Yolk–Shell Nanoparticles. Chemistry of Materials 2018, 30, 775-780, 10.1021/acs.chemmater.7b04253.
- Desheng Wang; Jibao He; Nista Rosenzweig; Zeev Rosenzweig; Superparamagnetic Fe2O3Beads−CdSe/ZnS Quantum Dots Core−Shell Nanocomposite Particles for Cell Separation. Nano Letters 2004, 4, 409-413, 10.1021/nl035010n.
- Ji Eun Lee; Nohyun Lee; Hyoungsu Kim; Jaeyun Kim; Seung Hong Choi; Jeong Hyun Kim; Taeho Kim; In Chan Song; Seung Pyo Park; Woo Kyung Moon; Taeghwan Hyeon; Junyoung Lee; Uniform Mesoporous Dye-Doped Silica Nanoparticles Decorated with Multiple Magnetite Nanocrystals for Simultaneous Enhanced Magnetic Resonance Imaging, Fluorescence Imaging, and Drug Delivery. Journal of the American Chemical Society 2010, 132, 552-557, 10.1021/ja905793q.
- Jae-Hyun Lee; Young-Wook Jun; Soo-In Yeon; Jeon-Soo Shin; Jinwoo Cheon; Dual-Mode Nanoparticle Probes for High-Performance Magnetic Resonance and Fluorescence Imaging of Neuroblastoma. Angewandte Chemie 2006, 118, 8340-8342, 10.1002/ange.200603052.
- Jaemoon Yang; Jaewon Lee; Jinyoung Kang; Chan-Hwa Chung; Kwangyeol Lee; Jin-Suck Suh; Ho-Geun Yoon; Yong-Min Huh; Seungjoo Haam; Magnetic sensitivity enhanced novel fluorescent magnetic silica nanoparticles for biomedical applications. Nanotechnology 2008, 19, 075610, 10.1088/0957-4484/19/7/075610.
- Fang Wang; Xiaolan Chen; Zengxia Zhao; Shaoheng Tang; Xiaoqing Huang; Chenghong Lin; Congbo Cai; Nanfeng Zheng; Synthesis of magnetic, fluorescent and mesoporous core-shell-structured nanoparticles for imaging, targeting and photodynamic therapy. Journal of Materials Chemistry 2011, 21, 11244, 10.1039/c1jm10329f.
- Abu Zayed Md. Badruddoza; Taifur Rahman; Sudipa Ghosh; Zakir Hossain; Jizhong Shi; Kus Hidajat; Mohammad Shahab Uddin; β-Cyclodextrin conjugated magnetic, fluorescent silica core–shell nanoparticles for biomedical applications. Carbohydrate Polymers 2013, 95, 449-457, 10.1016/j.carbpol.2013.02.046.
- Ling Li; Eugene Shi Guang Choo; Zhaoyang Liu; Jun Ding; Junmin Xue; Double-layer silica core-shell nanospheres with superparamagnetic and fluorescent functionalities. Chemical Physics Letters 2008, 461, 114-117, 10.1016/j.cplett.2008.07.005.
- Numpon Insin; Joseph B. Tracy; Hakho Lee; John P. Zimmer; Robert M. Westervelt; Moungi G. Bawendi; Incorporation of Iron Oxide Nanoparticles and Quantum Dots into Silica Microspheres. ACS Nano 2008, 2, 197-202, 10.1021/nn700344x.
- Tushar R. Sathe; Amit Agrawal; Shuming Nie; Mesoporous Silica Beads Embedded with Semiconductor Quantum Dots and Iron Oxide Nanocrystals: Dual-Function Microcarriers for Optical Encoding and Magnetic Separation. Analytical Chemistry 2006, 78, 5627-5632, 10.1021/ac0610309.
- Dong Kee Yi; Subramanian Tamil Selvan; Su Seong Lee; Georgia C. Papaefthymiou; Darshan Kundaliya; Jackie Y. Ying; Silica-Coated Nanocomposites of Magnetic Nanoparticles and Quantum Dots. Journal of the American Chemical Society 2005, 127, 4990-4991, 10.1021/ja0428863.
- Jaeyun Kim; Ji Eun Lee; Jinwoo Lee; Jung Ho Yu; Byoung Chan Kim; Kwangjin An; Yosun Hwang; Chae-Ho Shin; Je-Geun Park; Jungbae Kim; Taeghwan Hyeon; Magnetic Fluorescent Delivery Vehicle Using Uniform Mesoporous Silica Spheres Embedded with Monodisperse Magnetic and Semiconductor Nanocrystals. Journal of the American Chemical Society 2006, 128, 688-689, 10.1021/ja0565875.
- Xiaohong He; Xue Shen; Ngming Li; Yiyao Liu; Kun Jia; Xiaobo Liu; Dual-Mode Fluorescence and Magnetic Resonance Imaging Nanoprobe Based on Aromatic Amphiphilic Copolymer Encapsulated CdSe@CdS and Fe3O4. ACS Applied Bio Materials 2018, 1, 520-528, 10.1021/acsabm.8b00240.
- Ou Chen; Lars Riedemann; Fred Etoc; Hendrik Herrmann; Mathieu Coppey; Mariya Barch; Christian T. Farrar; Jing Zhao; Oliver T. Bruns; He Wei; Peng Guo; Jian Cui; Russ Jensen; Yue Chen; Daniel K. Harris; Jose M. Cordero; Zhongwu Wang; Alan Jasanoff; Dai Fukumura; Rudolph Reimer; Maxime Dahan; Rakesh K. Jain; Moungi G. Bawendi; Magneto-fluorescent core-shell supernanoparticles.. Nature Communications 2014, 5, 5093-5093, 10.1038/ncomms6093.
- Nathalie M. Pinkerton; Marian E. Gindy; Victoria L. Calero-DdelC; Theodore Wolfson; Robert F. Pagels; Derek Adler; Dayuan Gao; Shike Li; Ruobing Wang; Margot Zevon; Nan Yao; Carlos Pacheco; Michael J. Therien; Carlos Rinaldi; Patrick J. Sinko; Robert K. Prud'homme; Single-Step Assembly of Multimodal Imaging Nanocarriers: MRI and Long-Wavelength Fluorescence Imaging.. Advanced Healthcare Materials 2015, 4, 1376-85, 10.1002/adhm.201400766.
- Manita Das; Archana Solanki; Apeksha Joshi; Ranjitsinh Devkar; Sriram Seshadri; Sonal Thakore; Solanki Archana; Joshi Apeksha; Devkar Ranjitsinh; Seshadri Sriram; Thakore Sonal; β-cyclodextrin based dual-responsive multifunctional nanotheranostics for cancer cell targeting and dual drug delivery. Carbohydrate Polymers 2019, 206, 694-705, 10.1016/j.carbpol.2018.11.049.
- Oliver Bixner; Noga Gal; Christoph Zaba; Andrea Scheberl; Erik Reimhult; Fluorescent Magnetopolymersomes: A Theranostic Platform to Track Intracellular Delivery. Materials 2017, 10, 1303, 10.3390/ma10111303.
- Riccardo Di Corato; Nadja C. Bigall; Andrea Ragusa; Dirk Dorfs; Alessandro Genovese; Roberto Marotta; Liberato Manna; Teresa Pellegrino; Multifunctional Nanobeads Based on Quantum Dots and Magnetic Nanoparticles: Synthesis and Cancer Cell Targeting and Sorting. ACS Nano 2011, 5, 1109-1121, 10.1021/nn102761t.
- Daishun Ling; Wooram Park; Sin-Jung Park; Yang Lu; Kyoung Sub Kim; Michael J. Hackett; Byung Hyo Kim; Hyeona Yim; Yong Sun Jeon; Kun Na; Taeghwan Hyeon; Multifunctional Tumor pH-Sensitive Self-Assembled Nanoparticles for Bimodal Imaging and Treatment of Resistant Heterogeneous Tumors. Journal of the American Chemical Society 2014, 136, 5647-5655, 10.1021/ja4108287.
- Demillo, V.G.; Zhu, X. Zwitterionic Amphiphile Coated Magnetofluorescent Nanoparticles - Synthesis, Characterization and Tumor Cell Targeting. J. Mater. Chem. B 2015, 3, 8328–8336.
- Artur Feld; Jan-Philip Merkl; Hauke Kloust; Sandra Flessau; Christian Schmidtke; Christopher Wolter; Johannes Ostermann; Michael Kampferbeck; Robin Eggers; Alf Mews; Theo Schotten; Horst Weller; A Universal Approach to Ultrasmall Magneto-Fluorescent Nanohybrids. Angewandte Chemie International Edition 2015, 54, 12468-12471, 10.1002/anie.201503017.
- Gaëlle Béalle; Riccardo Di Corato; Jelena Kolosnjaj-Tabi; Vincent Dupuis; Olivier Clément; Florence Gazeau; Claire Wilhelm; Christine Ménager; Ultra Magnetic Liposomes for MR Imaging, Targeting, and Hyperthermia. Langmuir 2012, 28, 11834-11842, 10.1021/la3024716.
- Grégory Beaune; Benoît Dubertret; Olivier Clément; Catherine Vayssettes; Valérie Cabuil; Christine Ménager; Giant Vesicles Containing Magnetic Nanoparticles and Quantum Dots: Feasibility and Tracking by Fiber Confocal Fluorescence Microscopy. Angewandte Chemie International Edition 2007, 46, 5421-5424, 10.1002/anie.200700581.
- Alexander Scheffold; Stefan Miltenyi; Andreas Radbruch; Magnetofluorescent liposomes for increased sensitivity of immunofluorescence. Immunotechnology 1995, 1, 127-137, 10.1016/1380-2933(95)00014-3.
- Grégory Beaune; Christine Ménager; Valérie Cabuil; Location of Magnetic and Fluorescent Nanoparticles Encapsulated inside Giant Liposomes. The Journal of Physical Chemistry B 2008, 112, 7424-7429, 10.1021/jp711811u.
- Guannan Wang; Xuanjun Zhang; Yaxu Liu; Zhangjun Hu; Xifan Mei; Kajsa Uvdal; Magneto-fluorescent nanoparticles with high-intensity NIR emission, T1- and T2-weighted MR for multimodal specific tumor imaging. Journal of Materials Chemistry B 2015, 3, 3072-3080, 10.1039/C5TB00155B.
- Kai Li; Dan Ding; Da Huo; Kan-Yi Pu; Ngo Nguyen Phuong Thao; Yong Hu; Zhi Li; Bin Liu; Conjugated Polymer Based Nanoparticles as Dual-Modal Probes for Targeted In Vivo Fluorescence and Magnetic Resonance Imaging. Advanced Functional Materials 2012, 22, 3107-3115, 10.1002/adfm.201102234.
- Vineeth M. Vijayan; Ansar Ereath Beeran; Sachin J Shenoy; Jayabalan Muthu; Vinoy Thomas; New Magneto-Fluorescent Hybrid Polymer Nanogel for Theranostic Applications. ACS Applied Bio Materials 2019, 2, 757-768, 10.1021/acsabm.8b00616.
- Philip Howes; Mark Green; Alex Bowers; David Parker; Gopal Varma; Mathew Kallumadil; Mary Hughes; Alice Warley; Anthony Brain; Rene Botnar; Magnetic Conjugated Polymer Nanoparticles as Bimodal Imaging Agents. Journal of the American Chemical Society 2010, 132, 9833-9842, 10.1021/ja1031634.
- Adrien Faucon; Thomas Maldiney; Olivier Clément; Philippe Hulin; Steven Nedellec; Myriam Robard; Nicolas Gautier; Evelien De Meulenaere; Koen Clays; Tomas Orlando; Alessandro Lascialfari; Céline Fiorini-Debuisschert; Jérôme Fresnais; Eléna Ishow; Highly cohesive dual nanoassemblies for complementary multiscale bioimaging. Journal of Materials Chemistry B 2014, 2, 7747-7755, 10.1039/c4tb01199f.
- Adrien Faucon; Arnaud Brosseau; Philippe Hulin; Steven Nedellec; Julie Hemez; Jérôme Fresnais; Eléna Ishow; Photoactive chelating organic nanospheres as central platforms of bimodal hybrid nanoparticles. Journal of Materials Chemistry C 2013, 1, 3879, 10.1039/c3tc30508b.
- Adrien Faucon; Houda Benhelli-Mokrani; Fabrice Fleury; Laurence Dubreil; Philippe Hulin; Steven Nedellec; Tristan Doussineau; Rodolphe Antoine; Tomas Orlando; Alessandro Lascialfari; Jérôme Fresnais; Lénaïc Lartigue; Eléna Ishow; Alessandro Lascialfar; Tuning the architectural integrity of high-performance magneto-fluorescent core-shell nanoassemblies in cancer cells. Journal of Colloid and Interface Science 2016, 479, 139-149, 10.1016/j.jcis.2016.06.064.
- Jã©Rã´me Fresnais; Elã©Na Ishow; Olivier Sandre; Jean-Franã§ois Berret; Electrostatic Co-assembly of Magnetic Nanoparticles and Fluorescent Nanospheres: A Versatile Approach Towards Bimodal Nanorods. Small 2009, 5, 2533-2536, 10.1002/smll.200900703.
- Linot, C.; Poly, J.; Boucard, J.; Pouliquen, D.; Nedellec, S.; Hulin, P.; Lecouvey, M.; Marec, N.; Arosio, P.; Lascialfari, A.; et al. PEGylated Anionic Magneto Fl Uorescent Nanoassemblies: Impact of Their Interface Structure on Magnetic Resonance Imaging Contrast and Cellular Uptake. ACS Appl. Mater. Interfaces 2017, 9, 14242–14257.





