Your browser does not fully support modern features. Please upgrade for a smoother experience.

Submitted Successfully!
Thank you for your contribution! You can also upload a video entry or images related to this topic.
For video creation, please contact our Academic Video Service.
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Alessandro Posa | -- | 970 | 2022-04-16 22:50:32 | | | |
| 2 | Rita Xu | Meta information modification | 970 | 2022-04-18 06:01:40 | | |
Video Upload Options
We provide professional Academic Video Service to translate complex research into visually appealing presentations. Would you like to try it?
Cite
If you have any further questions, please contact Encyclopedia Editorial Office.
Posa, A.; , .; Tanzilli, A.; Cilla, S.; Iezzi, R. Autoimmune Demyelinating Diseases. Encyclopedia. Available online: https://encyclopedia.pub/entry/21848 (accessed on 21 May 2026).
Posa A, , Tanzilli A, Cilla S, Iezzi R. Autoimmune Demyelinating Diseases. Encyclopedia. Available at: https://encyclopedia.pub/entry/21848. Accessed May 21, 2026.
Posa, Alessandro, , Alessandro Tanzilli, Savino Cilla, Roberto Iezzi. "Autoimmune Demyelinating Diseases" Encyclopedia, https://encyclopedia.pub/entry/21848 (accessed May 21, 2026).
Posa, A., , ., Tanzilli, A., Cilla, S., & Iezzi, R. (2022, April 16). Autoimmune Demyelinating Diseases. In Encyclopedia. https://encyclopedia.pub/entry/21848
Posa, Alessandro, et al. "Autoimmune Demyelinating Diseases." Encyclopedia. Web. 16 April, 2022.
Copy Citation
The magnetic resonance characteristics of autoimmune demyelinating diseases are complex and represent a challenge for the radiologist. Expected and unexpected findings of magnetic resonance imaging examination for autoimmune demyelinating diseases were reported in order to provide a valuable approach for diagnosis.
demyelinating diseases
spinal cord
neuromyelitis optica
disseminated encephalomyelitis
1. Introduction
Autoimmune demyelinating diseases (ADD) constitute a heterogeneous group of diseases with variable clinical and imaging manifestations[1]. Multiple sclerosis (MS), neuromyelitis optica spectrum disorder (NMOSD), acute disseminated encephalomyelitis (ADEM), myelin oligodendrocyte glycoprotein (MOG) encephalomyelitis, and idiopathic transverse myelitis (TM) represent the main diseases characterized by autoimmune demyelination [1][2]. ADD are chronic disorders that can manifest with severe and acute neurologic complications due to direct damage to brain tissue and spinal cord [3][4][5]. Among all ADD, the archetype is the MS, representing the most common variant.
Diagnosing ADD can be very challenging owing to their complex and variable radiological features. Magnetic resonance imaging (MRI) plays a pivotal role in the diagnostic workflow [6][7]. However, misdiagnosis of ADD remains a matter of concern [8][9]. A valuable diagnosis is therefore necessary to establish an appropriate therapy, as therapeutic strategies greatly differ between ADD pathologies, influencing the patient’s prognosis. In this scenario, the evaluation of MRI features represents a major challenge for the radiologist and, in some cases, it may be a source of confusion, as certain ADD can present overlapping radiological findings. Radiologist expertise in recognition of the typical signs of the various ADD phenotypes needs to be strengthened with the integration of clinical and laboratory findings.
2. Case Presentation 1
A 57-year-old male with a history of chronic alcoholism presented to the emergency department with severe muscle weakness, which started with a progressive impairment in walking, followed by a feverish state (38 Celsius degrees), worsening up to being unable to walk. The patient was alert, collaborating, oriented in space, disoriented in time. Due to this clinical presentation, the patient was transferred to the neurology department for appropriate treatment. Osteo-tendon reflexes were normal in the upper limbs but absent in the lower limbs. Autoimmune screening, blood chemistry tests, and neoplastic markers were within normal limits. The search for pathogenic microorganisms was negative. Cyanocobalamin serum levels were normal. Anti-Myelin Oligodendrocyte Glycoprotein (MOG) and anti-Aquaporin-4 (AQP4) antibodies were absent, and the search for oligoclonal bands turned negative. Cerebrospinal fluid (CSF) examination demonstrated a clear rock water, marked pleocytosis (132 cells/microLiter, 99% mononuclear), and high CSF proteins (128 mg/dL). MRI of the brain and spinal cord was acquired (Figure 1, Figure 2 and Figure 3).
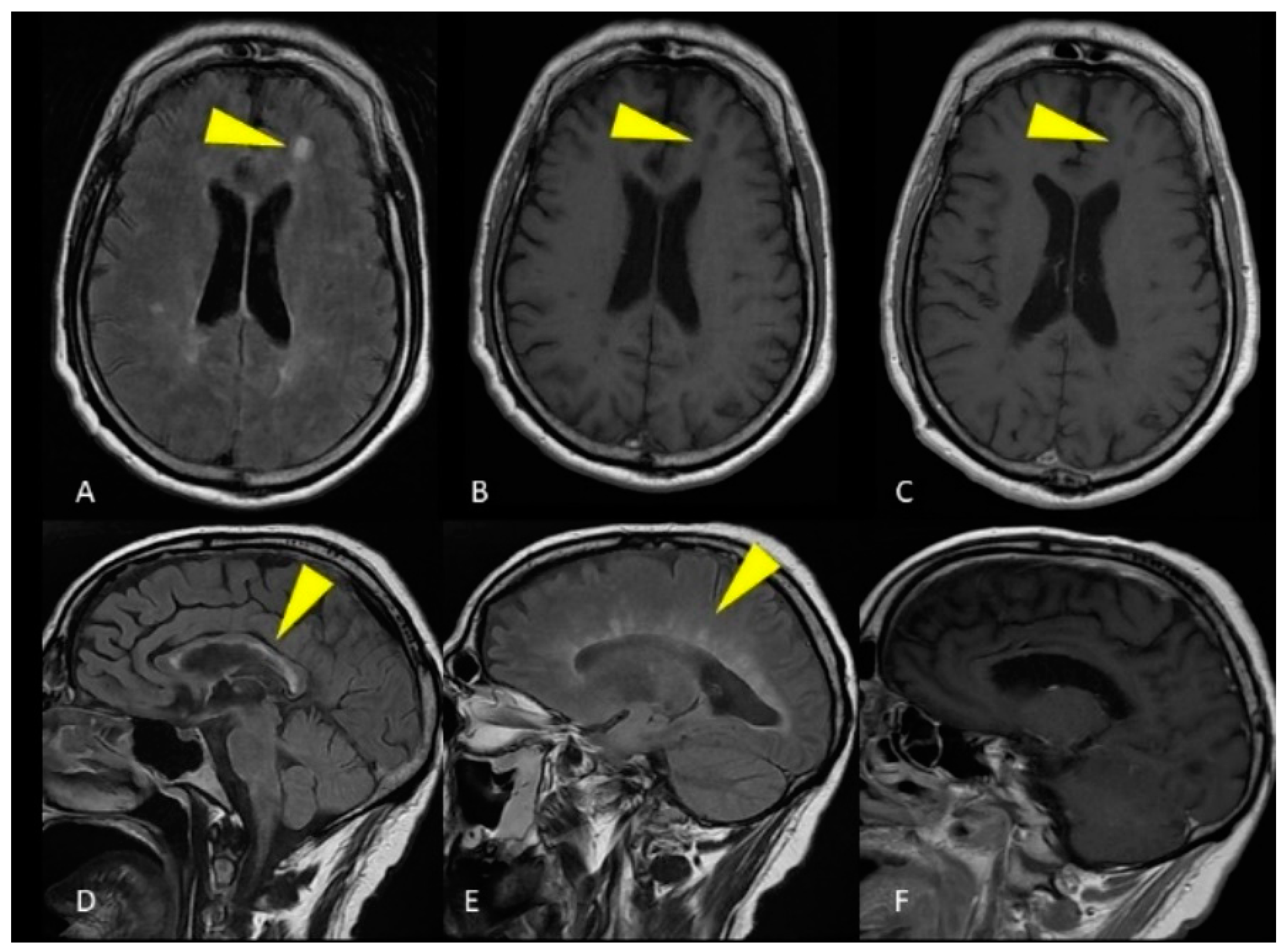
Figure 1. MRI of the brain. Axial FLAIR image (A) and unenhanced T1-weighted image (B) show a single ovoid-shaped lesion in the left frontal deep white matter, without enhancement on the T1-weighted post-gadolinium acquisition (C). Sagittal FLAIR image shows hyperintense lesions affecting the ependymal surface of the corpus callosum with a marble pattern (D). The so-called Dawson fingers are visible on the sagittal FLAIR image as hyperintense, ovoid lesions perpendicular to the body of the lateral ventricle (E). Corpus callosum and periventricular lesions don’t show enhancement on the T1-weighted post-gadolinium acquisition (F).
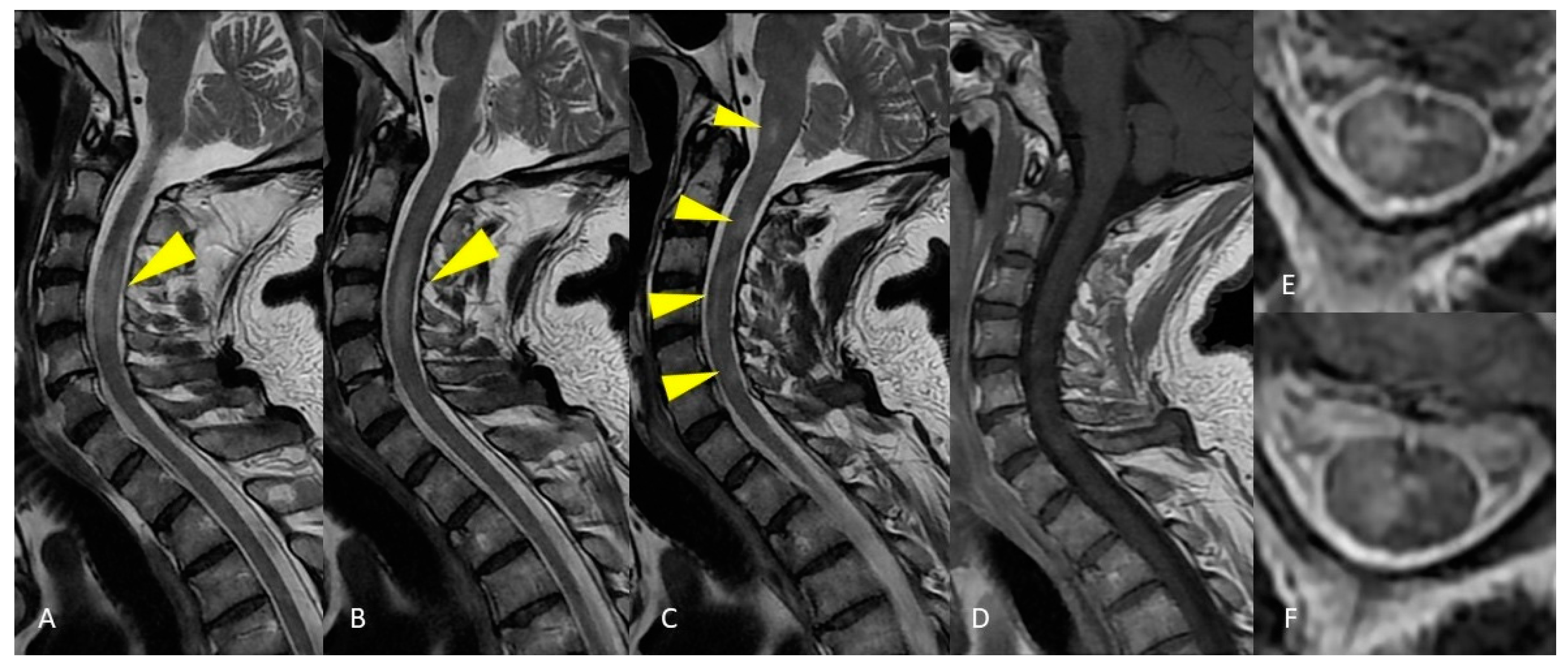
Figure 2. MRI of the cervical spinal cord. Sagittal T2-weighted images of the cervical spine show areas of patchy and long-segment (>1.5 vertebral body length) hyperintensity (A–C) without enhancement on the T1-weighted post-gadolinium acquisition (D). Axial T2-weighted image shows large hyperintensity involving half or more than half of the cross-sectional area of the spinal cord (E,F).
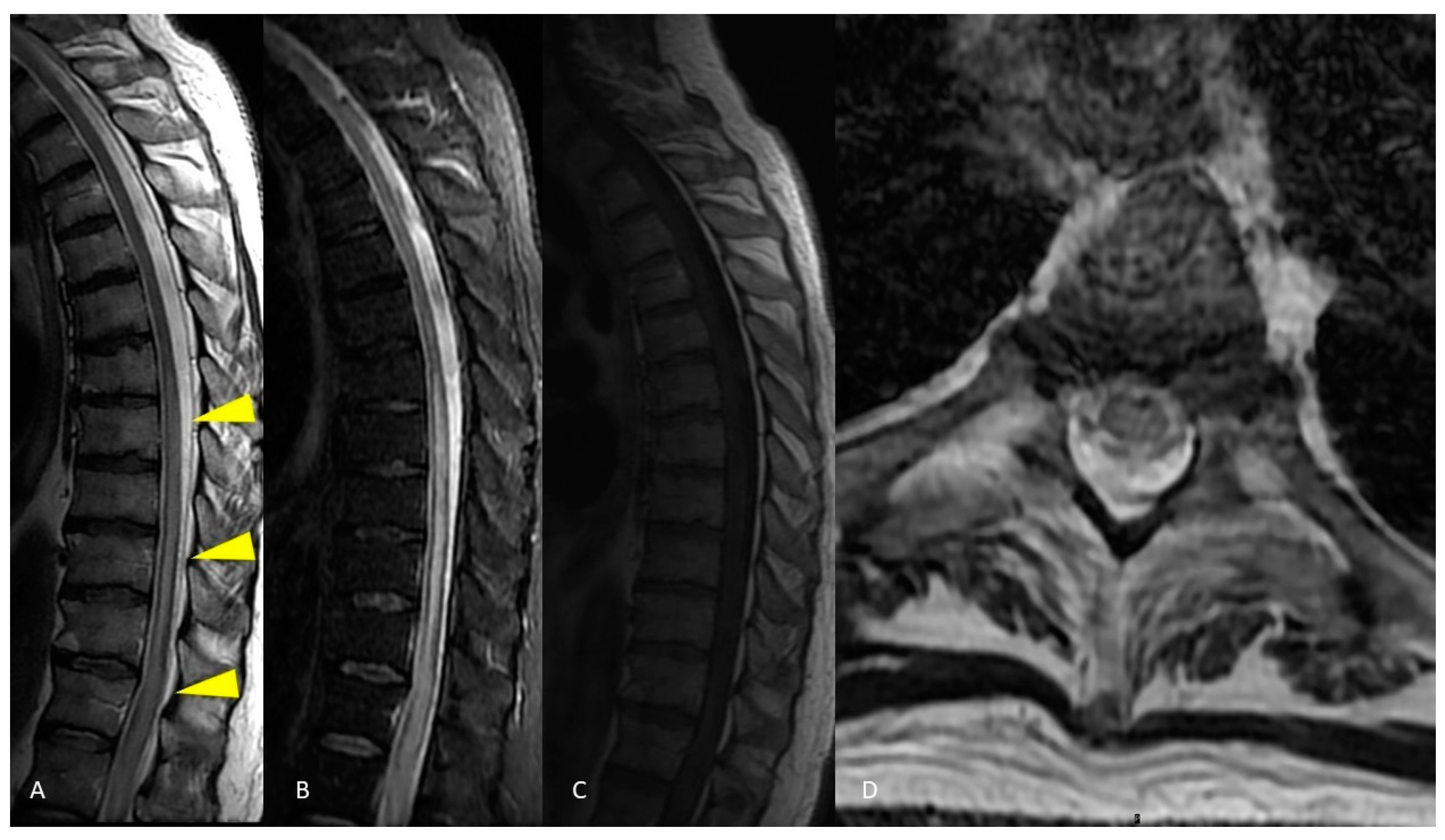
Figure 3. MRI of the thoracic spinal cord. Sagittal T2-weighted images demonstrate long segment hyperintensity of the thoracic cord without expansion or enhancement (A–C). Axial T2-weighted image shows large hyperintensity affecting all the cross-sectional area of the spinal cord (D).
The integration of clinical and imaging characteristics allowed ADEM diagnosis.
Follow-up imaging revealed that the ADEM lesions were resolved and that no new lesions had appeared. As a result, the disorder was monophasic.
3. Case Presentation 2
A 57-year-old female presented to the emergency department with paresthesia of the trunk, pelvis, and lower limbs with progressive difficulty of movement up to the inability to walk and collapse when standing. On the same day, double sphincter disorder was reported. The diagnosis of spinal cord syndrome was considered after neurological counseling. The patient was hospitalized for diagnostic and therapeutic evaluation. The patient was alert and cooperative, with visual acuity reduction on the left associated with retrobulbar pain. The patient presented weakness of the upper limbs and severe deficiency of the lower limbs. Osteo-tendon reflexes were normal in the upper limbs but absent in the lower limbs. Anti-MOG antibodies and oligoclonal bands were absent. Anti-AQP4 antibodies were present. Microbiological examination of CSF and peripheral blood were positive for previous and latent human beta-herpesvirus-7 (HHV 7) infection. Marker of active HHV 7 infection were excluded. MRI of brain and spinal cord was acquired (Figure 4 and Figure 5).
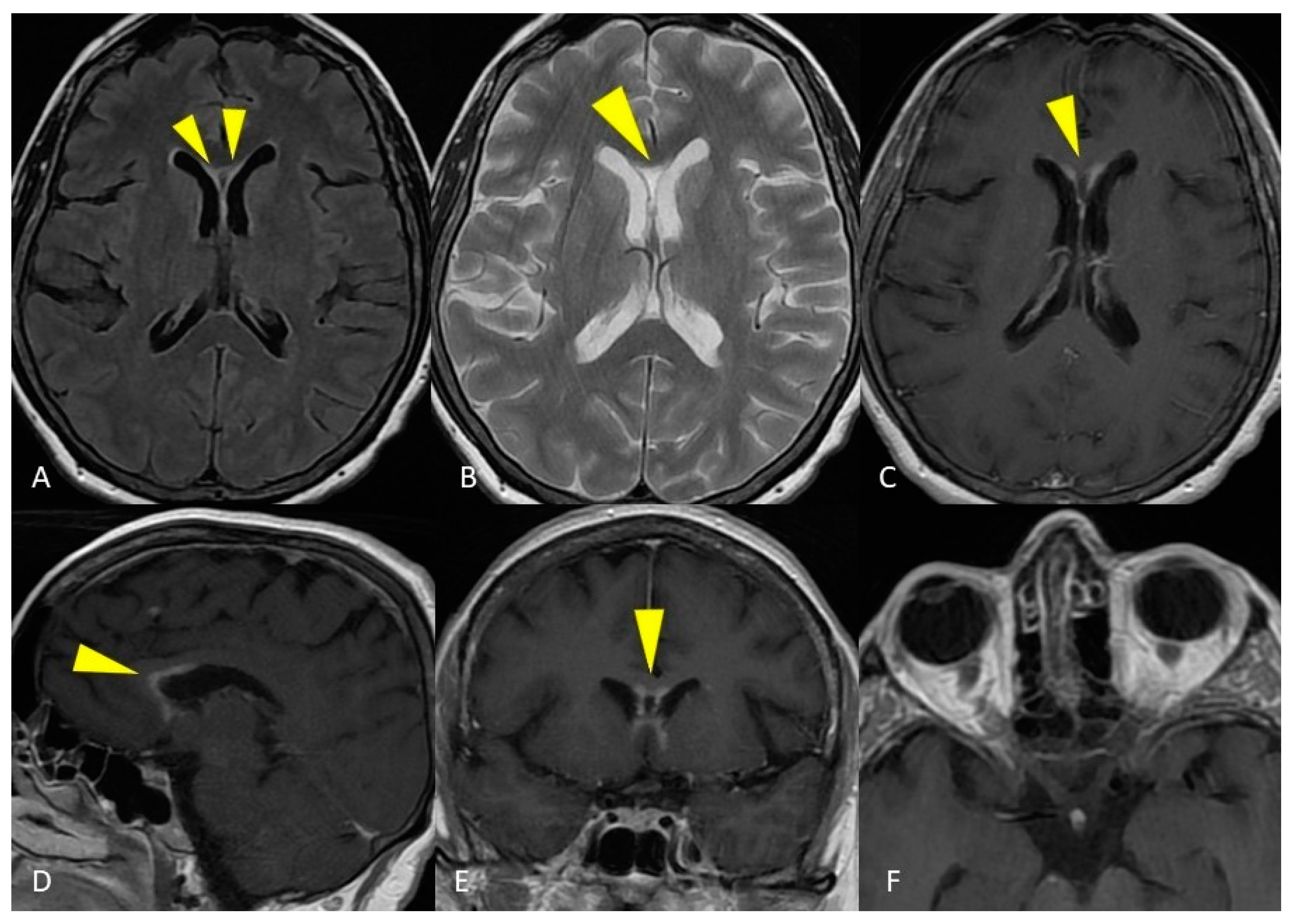
Figure 4. MRI of the brain. Axial T2-weighted and FLAIR images show curve hyperintense lesions affecting the ependymal surface of frontal horns of the lateral ventricles and corpus callosum just near the genu, with a symmetrical pattern (A,B). These sub-ependymal lesions show homogeneous enhancement on the T1-weighted post-gadolinium image (C–E). No optic nerve enhancement on T1-weighted post-gadolinium images was present (F).
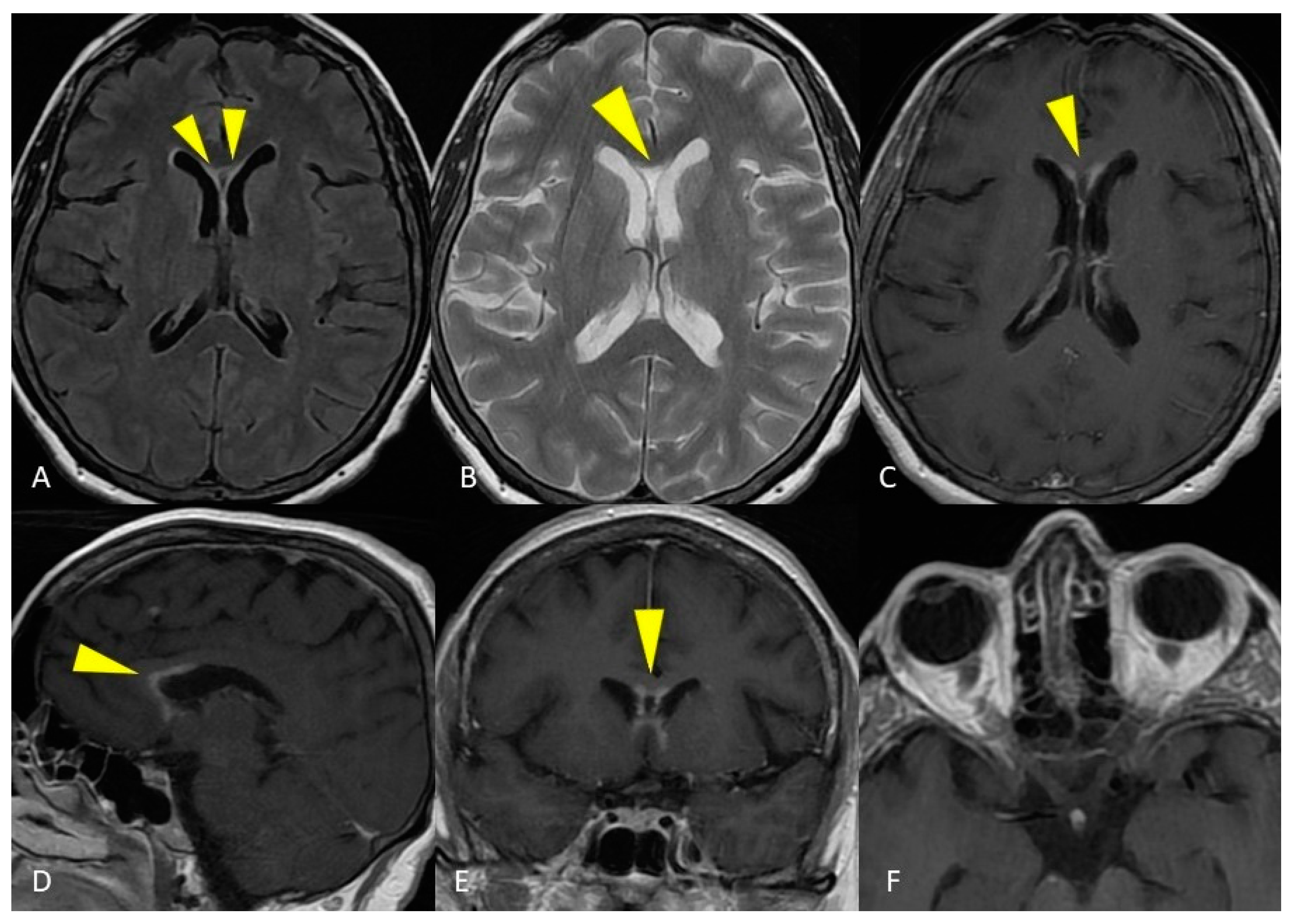
Figure 5. MRI of the spinal cord. Sagittal T2-weighted and STIR images show a long and continuous segment of abnormal hyperintensity affecting the cervico-thoracic spinal cord (A,B). Axial T2-weighted image shows diffuse hyperintensity involving all the cross-sectional area of the spinal cord at a more cranial level (D) and a predominant gray matter involvement at a more caudal level (E). Spinal cord swelling and enhancement were present at the segmental thoracic level, evident in both sagittal (C) and axial (F) images.
Despite the absence of alterations of optic nerves on MRI, which represents a major criteria for NMO diagnosis, the integration of clinical and imaging features allowed the aforementioned diagnosis.
References
- Michael J. Lee; Ryan Aronberg; Matthew S. Manganaro; Mohannad Ibrahim; Hemant A. Parmar; Diagnostic Approach to Intrinsic Abnormality of Spinal Cord Signal Intensity. RadioGraphics 2019, 39, 1824-1839, 10.1148/rg.2019190021.
- Yukio Miki; Magnetic resonance imaging diagnosis of demyelinating diseases: An update. Clinical and Experimental Neuroimmunology 2019, 10, 32-48, 10.1111/cen3.12501.
- Reem F. Bunyan; Junger Tang; Brian Weinshenker; Acute Demyelinating Disorders: Emergencies and Management. Neurologic Clinics 2012, 30, 285-307, 10.1016/j.ncl.2011.09.013.
- Chrysostomos Katsenos; Despoina Androulaki; Stavroula Lyra; Theodoros Tsoutsouras; Costas Mandragos; A 17 year-old girl with a demyelinating disease requiring mechanical ventilation: a case report. BMC Research Notes 2013, 6, 22-4, 10.1186/1756-0500-6-22.
- Andrea Rossi; Imaging of Acute Disseminated Encephalomyelitis. Neuroimaging Clinics of North America 2008, 18, 149-161, 10.1016/j.nic.2007.12.007.
- Stefan Weidauer; Peter Raab; Elke Hattingen; Diagnostic approach in multiple sclerosis with MRI: an update. Clinical Imaging 2021, 78, 276-285, 10.1016/j.clinimag.2021.05.025.
- Nicolae Sarbu; Valentina Lolli; James G. Smirniotopoulos; Magnetic resonance imaging in myelopathy: a pictorial review. Clinical Imaging 2019, 57, 56-68, 10.1016/j.clinimag.2019.05.002.
- Wallace J Brownlee; Todd A Hardy; Franz Fazekas; David H Miller; Diagnosis of multiple sclerosis: progress and challenges. The Lancet 2016, 389, 1336-1346, 10.1016/s0140-6736(16)30959-x.
- Andrew J. Solomon; John R. Corboy; The tension between early diagnosis and misdiagnosis of multiple sclerosis. Nature Reviews Neurology 2017, 13, 567-572, 10.1038/nrneurol.2017.106.
More
Information
Subjects:
Neuroimaging
Contributors
MDPI registered users' name will be linked to their SciProfiles pages. To register with us, please refer to https://encyclopedia.pub/register
:
View Times:
1.3K
Entry Collection:
Nuclear Magnetic Resonance
Revisions:
2 times
(View History)
Update Date:
18 Apr 2022
Notice
You are not a member of the advisory board for this topic. If you want to update advisory board member profile, please contact office@encyclopedia.pub.
OK
Confirm
Only members of the Encyclopedia advisory board for this topic are allowed to note entries. Would you like to become an advisory board member of the Encyclopedia?
Yes
No
${ textCharacter }/${ maxCharacter }
Submit
Cancel
Back
Comments
${ item }
|
More
No more~
There is no comment~
${ textCharacter }/${ maxCharacter }
Submit
Cancel
${ selectedItem.replyTextCharacter }/${ selectedItem.replyMaxCharacter }
Submit
Cancel
Confirm
Are you sure to Delete?
Yes
No





