Your browser does not fully support modern features. Please upgrade for a smoother experience.

Submitted Successfully!
Thank you for your contribution! You can also upload a video entry or images related to this topic.
For video creation, please contact our Academic Video Service.
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Sayan Basu | -- | 2992 | 2022-04-07 09:29:08 | | | |
| 2 | Conner Chen | -1568 word(s) | 1424 | 2022-04-07 09:54:44 | | | | |
| 3 | Conner Chen | Meta information modification | 1424 | 2022-04-07 09:57:19 | | | | |
| 4 | Conner Chen | Meta information modification | 1424 | 2022-04-08 05:05:39 | | | | |
| 5 | Conner Chen | Meta information modification | 1424 | 2022-05-19 08:21:45 | | |
Video Upload Options
We provide professional Academic Video Service to translate complex research into visually appealing presentations. Would you like to try it?
Cite
If you have any further questions, please contact Encyclopedia Editorial Office.
Basu, S.; , . Principle of Optical Coherence Tomography Angiography. Encyclopedia. Available online: https://encyclopedia.pub/entry/21445 (accessed on 10 May 2026).
Basu S, . Principle of Optical Coherence Tomography Angiography. Encyclopedia. Available at: https://encyclopedia.pub/entry/21445. Accessed May 10, 2026.
Basu, Sayan, . "Principle of Optical Coherence Tomography Angiography" Encyclopedia, https://encyclopedia.pub/entry/21445 (accessed May 10, 2026).
Basu, S., & , . (2022, April 07). Principle of Optical Coherence Tomography Angiography. In Encyclopedia. https://encyclopedia.pub/entry/21445
Basu, Sayan and . "Principle of Optical Coherence Tomography Angiography." Encyclopedia. Web. 07 April, 2022.
Copy Citation
Diagnosis and quantification of ischemia in ocular burns is essential to determine the need for interventions in the acute phase and the final prognosis. Clinical signs in isolation are rarely sufficient to accurately assess limbal ischemia. With anterior segment optical coherence tomography angiography (AS-OCTA), the presence and extent of ischemia can be objectively confirmed and quantified.
optical coherence tomography
angiography
anterior segment OCT
Corneal angiography
Ocular burns
limbal ischemia
1. Introduction
Ocular burns can present with a myriad of clinical manifestations in the acute phase. The grade of injury and the extent of ocular structures involved often determines the type and severity of the chronic sequelae that ensue. Limbal stem cell deficiency (LSCD) is one such chronic feature that can cause significant visual morbidity due to the corneal scarring and vascularization that occurs in its end stages [1]. The development of this pathology is usually heralded by the presence of limbal ischemia in the acute phase of the disease [2]. The detection and grading of this finding can help identify those individuals who are at a higher risk of developing LSCD. This cohort of patients can then be subjected to regular monitoring which will facilitate early identification and treatment of LSCD to prevent its complications and restore visual function.
However, the diagnosis of ischemia in the acute phase is based on subjective clinical findings which are often difficult to discern in an inflamed eye [2]. Thus, the adoption of diagnostic tools can provide an objective method of confirming ischemia of the limbal vasculature and assessing its progression. An anterior segment optical coherence tomography (AS-OCT) is usually available in most anterior segment practices and provides rapid, high-definition images of sections of the cornea with relative ease [3]. The utility of its angiography feature (AS-OCTA) has recently been explored to detect ischemia in acute burns as it can provide a real time in-vivo image of the limbal and perilimbal vasculature [4][5]. The use of image processing software has also aided in objectively quantifying the degree of vascular changes observed [6].
2. Principle and Technology of Optical Coherence Tomography Angiography (OCTA)
2.1. Principle
Optical coherence tomography (OCT) measures the signal of backscattered low coherence light incident on the area of interest and uses it to construct sectional and en-face images of different structures of the eye [7][8]. The angiography images are created by obtaining multiple b-scans of the same section over a period of time and assessing the difference in the signal of each scan [7]. This difference is attributed to the movement of erythrocytes within the blood vessels, and by removing the signal from the static surrounding structures, an angiography image is produced by isolating the temporal changes in these dynamic signals [9][10]. Several algorithms are employed to facilitate the same which include the full spectrum or split spectrum amplitude decorrelation angiography (FS or SSADA), optical microangiography (OMAG), intensity ratio analysis (OCTA-RA), etc. They assess the change in the phase of the signal, its amplitude, or both [10].
In SSADA, smaller spectral bands of light are created from the full spectrum and individual analysis is carried out in each band and then averaged [11]. This contrasts with FSADA where the full spectrum is analyzed as a whole. Thus, the SSADA produces images that have a better ratio of signal to noise. However, this is at the expense of axial resolution and this disadvantage is overcome by the OMAG and OCTA-RA algorithms. The former uses both phase and amplitude differences for its images while the latter is independent of the decorrelation principle and assesses the maximum and minimum signal intensity measured between two frames [12][13][14].
2.2. Technology
Broadly, the OCT machines can be classified as time-domain and Fourier-domain machines [4]. The current OCTA machines are all Fourier-domain based and consist of the spectral domain {AngioVue (Optovue, Inc., Fremont, CA, USA), Angioscan (Nidek Co., Ltd., Gamagori, Aichi, Japan)} and swept-source OCTA systems {Triton DRI-OCT (Topcon Corporation, Tokyo, Japan), PLEX Elite Prototype 9000 (Carl Zeiss Meditec, Dublin, CA, USA)}. The swept-source OCTAs have a longer wavelength (1050 nm) which improves the depth of structures imaged [15]. This, in combination with a faster speed of scan acquisition (200,000 scans/s), helps improve the resolution and provides a wider field of view [4]. Additionally, the eye tracking software of PLEX Elite is unique to anterior segment devices and can help decrease the artifacts produced [4]. However, the images procured by the AngioVue system have the highest axial and lateral resolution of 5 and 15 μm, respectively [9]. Its speed of acquisition is also the highest at 3–4 s per scan because of which the motion artifacts generated with this device are the least. In comparison with dye-based angiography, both AngioVue and Angioscan machines were found to be comparable, yielding quantifying parameters that correlated well with each other [16][17][18][19]. Although the inter-device values correlate well with each other, the absolute values were found to be greater with AngioVue, implying that these inter-system values cannot be compared with each other [20]. AS-OCTA can not only confirm the presence or absence of vasculature but can also reliably and accurately detect changes in vessel caliber [6].
3. Comparison with Fluorescein Angiography (FA) and Indocyanine Green Angiography (ICG)
The most important factor that sets OCTA apart from its counterpart vessel-imaging modalities is its non-invasive nature as it is not dependent on the use of a dye [21][22]. This obviates the injection-associated trauma and dye-associated risks such as nausea, vomiting, and, rarely, anaphylaxis [23][24]. By avoiding the use of an external dye, the time taken for the angiography is significantly decreased with the use of OCTA which is especially desirable in patients with painful disorders such as acute burns. The test can be repeated multiple times without any need for recurrent dye injections. Additionally, the ischemic zones may be obscured by leakage of the dye from the inflamed vessels in these eyes and this disadvantage is circumvented with OCTA [25].
The use of light in the infrared wavelength by the OCTA provides an added advantage as the device can image vessels beneath hemorrhages from superficial conjunctival vessels [6][26]. This is of particular significance in eyes with acute burns as these traumatic sub-conjunctival hemorrhages (SCH) are common and may mask areas of ischemia when viewed via conventional angiographic techniques. However, shadow artifacts can also be seen in AS-OCTA images due to these hemorrhages. This contrast has been depicted in Figure 1 where in the inferior area the vasculature is obscured by the SCH. At the same time in the region abutting the inferior limbus, the vasculature is clear despite the presence of the SCH. One possible explanation for this discrepancy can be the relative position of the SCH when compared to the vessels. Very superficial and large hemorrhages may block the view of all the underlying vessels as opposed to deeper SCH which may allow the imaging of vessels overlying the hemorrhage.
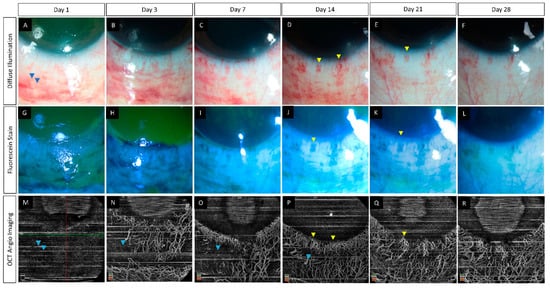
Figure 1. (A–R) Serial monitoring with slit lamp photographs (without and with fluorescein staining) along with anterior segment optical coherence tomography angiography (AS-OCTA) imaging in an eye with acute chemical burns. (A–L) These images show the progressive healing of the corneal and conjunctival epithelial defects in the inferior part of the eye which is complete by day 7. (M–R) AS-OCTA images show a blocked signal in the initial three visits due to the subconjunctival hemorrhages inferiorly (blue arrow heads) with clear delineation of the vasculature following the resolution of the hemorrhages. The hemorrhages abutting the limbus, however, have not affected the vascular signal (yellow arrow heads).
The infrared wavelength of light also allows for better patient comfort and has a good safety profile as well [27]. Localization of the exact depth of a vascular lesion is now feasible because OCTA allows segmentation of different layers of the imaged vessels [16][28][29]. This is in contrast to FA and ICG which generate a composite two-dimensional image. The major drawback of OCTA when compared to the traditional angiography tools lies in its inability to identify the flow characteristics and thus, patterns of leakage, the difference between afferent and efferent systems, etc. cannot be captured with OCTA [30][31]. In the context of ocular burns, this is relevant as images from OCTA may not distinguish vasospasm from true ischemia and thus caution must be exercised during interpretation of OCTA scans. Furthermore, the field of view is larger in FA and ICG and multiple scans would be required by OCTA to cover the same surface area [3][24]. As OCTA images are derived from the motion of blood cells, this process can be confounded by motion originating from other sources, such as movements of the patient’s eyes or head. Although the eye tracking feature of retinal OCTA can counteract these artifacts, its incorporation in anterior segment images has not been completely established.
References
- Haagdorens, M.; Van Acker, S.I.; Van Gerwen, V.; Ní Dhubhghaill, S.; Koppen, C.; Tassignon, M.-J.; Zakaria, N. Limbal Stem Cell Deficiency: Current Treatment Options and Emerging Therapies. Stem Cells Int. 2016, 2016, 9798374.
- Kam, K.W.; Patel, C.N.; Nikpoor, N.; Yu, M.; Basu, S. Limbal Ischemia: Reliability of Clinical Assessment and Implications in the Management of Ocular Burns. Indian J. Ophthalmol. 2019, 67, 32–36.
- Ayres, M.; Smallwood, R.; Brooks, A.M.; Chan, E.; Fagan, X. Anterior Segment Optical Coherence Tomography Angiography. J. Vis. Commun. Med. 2019, 42, 153–157.
- Lee, W.D.; Devarajan, K.; Chua, J.; Schmetterer, L.; Mehta, J.S.; Ang, M. Optical Coherence Tomography Angiography for the Anterior Segment. Eye Vis. 2019, 6, 4.
- Cozzi, M.; Staurenghi, G.; Invernizzi, A. Anterior Segment and Ocular Adnexa OCT Angiography. Ophthalmology 2020, 127, 220.
- Patel, C.N.; Antony, A.K.; Kommula, H.; Shah, S.; Singh, V.; Basu, S. Optical Coherence Tomography Angiography of Perilimbal Vasculature: Validation of a Standardised Imaging Algorithm. Br. J. Ophthalmol. 2020, 104, 404–409.
- Fujimoto, J.G.; Pitris, C.; Boppart, S.A.; Brezinski, M.E. Optical Coherence Tomography: An Emerging Technology for Biomedical Imaging and Optical Biopsy. Neoplasia 2000, 2, 9–25.
- Borrelli, E.; Parravano, M.; Sacconi, R.; Costanzo, E.; Querques, L.; Vella, G.; Bandello, F.; Querques, G. Guidelines on Optical Coherence Tomography Angiography Imaging: 2020 Focused Update. Ophthalmol. Ther. 2020, 9, 697–707.
- Munk, M.R.; Giannakaki-Zimmermann, H.; Berger, L.; Huf, W.; Ebneter, A.; Wolf, S.; Zinkernagel, M.S. OCT-Angiography: A Qualitative and Quantitative Comparison of 4 OCT-A Devices. PLoS ONE 2017, 12, e0177059.
- Onishi, A.C.; Fawzi, A.A. An Overview of Optical Coherence Tomography Angiography and the Posterior Pole. Ther. Adv. Ophthalmol. 2019, 11, 2515841419840249.
- Jia, Y.; Tan, O.; Tokayer, J.; Potsaid, B.; Wang, Y.; Liu, J.J.; Kraus, M.F.; Subhash, H.; Fujimoto, J.G.; Hornegger, J.; et al. Split-Spectrum Amplitude-Decorrelation Angiography with Optical Coherence Tomography. Opt. Express 2012, 20, 4710–4725.
- An, L.; Wang, R.K. In Vivo Volumetric Imaging of Vascular Perfusion within Human Retina and Choroids with Optical Micro-Angiography. Opt. Express 2008, 16, 11438–11452.
- Wang, R.K.; An, L.; Francis, P.; Wilson, D.J. Depth-Resolved Imaging of Capillary Networks in Retina and Choroid Using Ultrahigh Sensitive Optical Microangiography. Opt. Lett 2010, 35, 1467–1469.
- Stanga, P.E.; Tsamis, E.; Papayannis, A.; Stringa, F.; Cole, T.; Jalil, A. Swept-Source Optical Coherence Tomography AngioTM (Topcon Corp, Japan): Technology Review. Dev. Ophthalmol. 2016, 56, 13–17.
- Ang, M.; Cai, Y.; Tan, A.C.S. Swept Source Optical Coherence Tomography Angiography for Contact Lens-Related Corneal Vascularization. J. Ophthalmol. 2016, 2016, 9685297.
- Devarajan, K.; Di Lee, W.; Ong, H.S.; Lwin, N.C.; Chua, J.; Schmetterer, L.; Mehta, J.S.; Ang, M. Vessel Density and En-Face Segmentation of Optical Coherence Tomography Angiography to Analyse Corneal Vascularisation in an Animal Model. Eye Vis. 2019, 6, 2.
- Ang, M.; Cai, Y.; MacPhee, B.; Sim, D.A.; Keane, P.A.; Sng, C.C.A.; Egan, C.A.; Tufail, A.; Larkin, D.F.; Wilkins, M.R. Optical Coherence Tomography Angiography and Indocyanine Green Angiography for Corneal Vascularisation. Br. J. Ophthalmol. 2016, 100, 1557–1563.
- Hong, J.; Zhu, W.; Zhuang, H.; Xu, J.; Sun, X.; Le, Q.; Li, G.; Wang, Y. In Vivo Confocal Microscopy of Conjunctival Goblet Cells in Patients with Sjogren’s Syndrome Dry Eye. Br. J. Ophthalmol. 2010, 94, 1454–1458.
- Stanzel, T.P.; Devarajan, K.; Lwin, N.C.; Yam, G.H.; Schmetterer, L.; Mehta, J.S.; Ang, M. Comparison of Optical Coherence Tomography Angiography to Indocyanine Green Angiography and Slit Lamp Photography for Corneal Vascularization in an Animal Model. Sci. Rep. 2018, 8, 11493.
- Ang, M.; Devarajan, K.; Das, S.; Stanzel, T.; Tan, A.; Girard, M.; Schmetterer, L.; Mehta, J.S. Comparison of Anterior Segment Optical Coherence Tomography Angiography Systems for Corneal Vascularisation. Br. J. Ophthalmol. 2018, 102, 873–877.
- Kuckelkorn, R.; Remky, A.; Wolf, S.; Reim, M.; Redbrake, C. Video Fluorescein Angiography of the Anterior Eye Segment in Severe Eye Burns. Acta Ophthalmol. Scand. 1997, 75, 675–680.
- Cai, Y.; Alio Del Barrio, J.L.; Wilkins, M.R.; Ang, M. Serial Optical Coherence Tomography Angiography for Corneal Vascularization. Graefes Arch. Clin. Exp. Ophthalmol. 2017, 255, 135–139.
- Xu, K.; Tzankova, V.; Li, C.; Sharma, S. Intravenous Fluorescein Angiography-Associated Adverse Reactions. Can. J. Ophthalmol. 2016, 51, 321–325.
- Anijeet, D.R.; Zheng, Y.; Tey, A.; Hodson, M.; Sueke, H.; Kaye, S.B. Imaging and Evaluation of Corneal Vascularization Using Fluorescein and Indocyanine Green Angiography. Investig. Ophthalmol. Vis. Sci. 2012, 53, 650–658.
- Ang, M.; Tan, A.C.S.; Cheung, C.M.G.; Keane, P.A.; Dolz-Marco, R.; Sng, C.C.A.; Schmetterer, L. Optical Coherence Tomography Angiography: A Review of Current and Future Clinical Applications. Graefes Arch. Clin. Exp. Ophthalmol. 2018, 256, 237–245.
- Fung, S.S.M.; Stewart, R.M.K.; Dhallu, S.K.; Sim, D.A.; Keane, P.A.; Wilkins, M.R.; Tuft, S.J. Anterior Segment Optical Coherence Tomographic Angiography Assessment of Acute Chemical Injury. Am. J. Ophthalmol. 2019, 205, 165–174.
- Shu, X.; Beckmann, L.; Zhang, H.F. Visible-Light Optical Coherence Tomography: A Review. J. Biomed. Opt. 2017, 22, 121707.
- Akagi, T.; Uji, A.; Huang, A.S.; Weinreb, R.N.; Yamada, T.; Miyata, M.; Kameda, T.; Ikeda, H.O.; Tsujikawa, A. Conjunctival and Intrascleral Vasculatures Assessed Using Anterior Segment Optical Coherence Tomography Angiography in Normal Eyes. Am. J. Ophthalmol. 2018, 196, 1–9.
- Giarratano, Y.; Bianchi, E.; Gray, C.; Morris, A.; MacGillivray, T.; Dhillon, B.; Bernabeu, M.O. Automated Segmentation of Optical Coherence Tomography Angiography Images: Benchmark Data and Clinically Relevant Metrics. Transl. Vis. Sci. Technol. 2020, 9, 5.
- Siddiqui, Y.; Yin, J. Anterior Segment Applications of Optical Coherence Tomography Angiography. Semin. Ophthalmol. 2019, 34, 264–269.
- Luo, M.; Li, Y.; Zhuo, Y. Advances and Current Clinical Applications of Anterior Segment Optical Coherence Tomography Angiography. Front. Med. 2021, 8, 721442.
More
Information
Subjects:
Ophthalmology
Contributors
MDPI registered users' name will be linked to their SciProfiles pages. To register with us, please refer to https://encyclopedia.pub/register
:
View Times:
1.3K
Revisions:
5 times
(View History)
Update Date:
19 May 2022
Notice
You are not a member of the advisory board for this topic. If you want to update advisory board member profile, please contact office@encyclopedia.pub.
OK
Confirm
Only members of the Encyclopedia advisory board for this topic are allowed to note entries. Would you like to become an advisory board member of the Encyclopedia?
Yes
No
${ textCharacter }/${ maxCharacter }
Submit
Cancel
Back
Comments
${ item }
|
More
No more~
There is no comment~
${ textCharacter }/${ maxCharacter }
Submit
Cancel
${ selectedItem.replyTextCharacter }/${ selectedItem.replyMaxCharacter }
Submit
Cancel
Confirm
Are you sure to Delete?
Yes
No





