
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Carlo Bertoldi | -- | 4046 | 2022-03-31 14:40:00 | | | |
| 2 | Vivi Li | -48 word(s) | 3998 | 2022-04-01 05:51:20 | | |
Video Upload Options
Bioactive glasses (BGs) were discovered by Larry L. Hench in 1969; the original composition, named Bioglass® 45S5 (45S5 from here on), displayed outstanding properties such as bone regeneration capabilities and antibacterial activity. In fact, such BG is an amorphous and biocompatible silica oxide-based inorganic material able to induce surface property responses resulting in the formation of a bond between the bone and the glass itself. In general, the surface of BGs, when soaked in physiological fluids, undergoes a complex ion exchange mechanism with the medium, inducing the formation of precipitates and subsequently hydroxyapatite deposition. This mechanism could explain the effectiveness of these BGs to bind to bones and the wide number of studies on BGs as supporting materials for bone tissue engineering and tooth remineralization.
1. Introduction
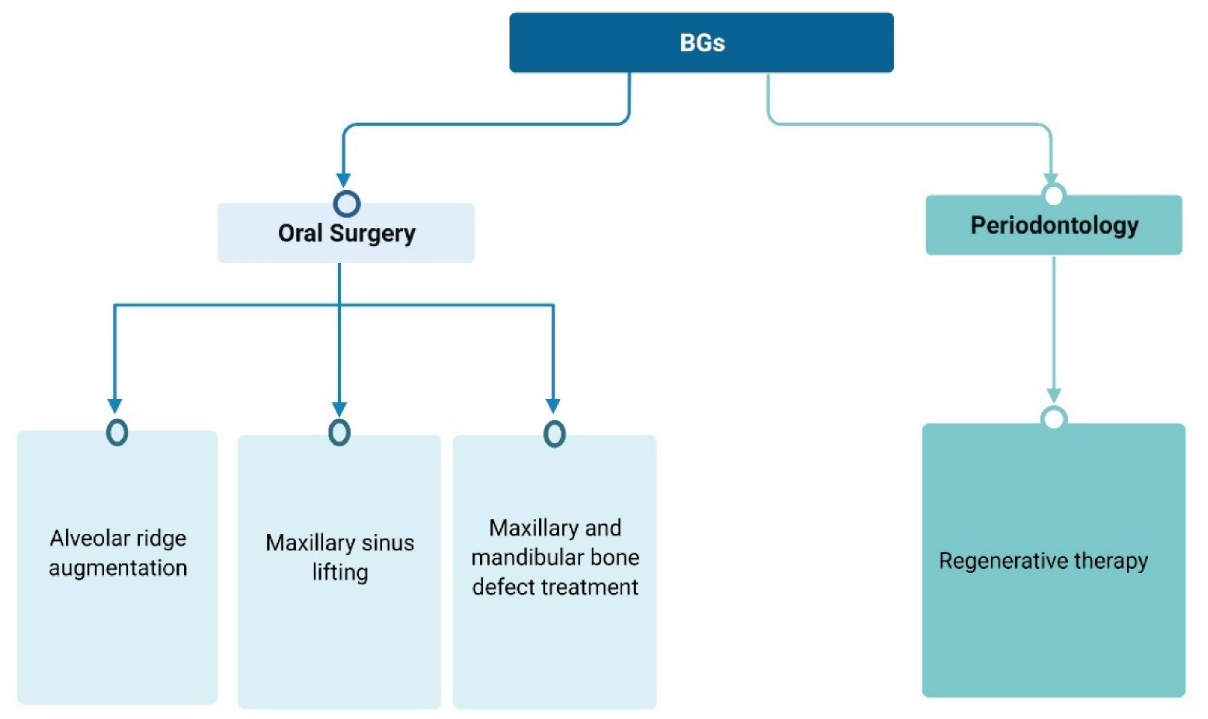

2. In Vitro Tests
3. In Vivo Tests
4. Clinical Studies
References
- Larry, R.; Hench, L. The story of Bioglass. J. Mater. Sci. Mater. Med. 2006, 17, 967–978.
- Jones, J.R. Reprint of: Review of bioactive glass: From Hench to hybrids. Acta Biomater. 2015, 23, S53–S82.
- Alves, E.G.L.; Serakides, R.; Rosado, I.R.; Pereira, M.M.; Ocarino, N.M.; Oliveira, H.P.; Góes, A.M.; Rezende, C.M.F. Effect of the ionic product of bioglass 60s on osteoblastic activity in canines. BMC Vet. Res. 2015, 11, 247.
- Taherkhani, S.; Moztarzadeh, F. Influence of strontium on the structure and biological properties of sol–gel-derived mesoporous bioactive glass powder. J. Sol-Gel Sci. Technol. 2016, 78, 539–549.
- Hoppe, A.; Güldal, N.S.; Boccaccini, A.R. A review of the biological response to ionic dissolution products from bioactive glasses and glass-ceramics. Biomaterials 2011, 32, 2757–2774.
- Zhu, H.; Zheng, K.; Boccaccini, A.R. Multi-functional silica-based mesoporous materials for simultaneous delivery of biologically active ions and therapeutic biomolecules. Acta Biomater. 2021, 129, 1–17.
- Mehrabi, T.; Mesgar, A.S.; Mohammadi, Z. Bioactive Glasses: A Promising Therapeutic Ion Release Strategy for Enhancing Wound Healing. ACS Biomater. Sci. Eng. 2020, 6, 5399–5430.
- Bellucci, D.; Veronesi, E.; Strusi, V.; Petrachi, T.; Murgia, A.; Mastrolia, I.; Dominici, M.; Cannillo, V. Human mesenchymal stem cell combined with a new strontium-enriched bioactive glass: An ex-vivo model for Bone Regeneration. Materials 2019, 12, 3633.
- Bellucci, D.; Veronesi, E.; Dominici, M.; Cannillo, V. A new bioactive glass with extremely high crystallization temperature and outstanding biological performance. Mater. Sci. Eng. C 2020, 110, 110699.
- Bellucci, D.; Cannillo, V. A novel bioactive glass containing strontium and magnesium with ultra-high crystallization temperature. Mater. Lett. 2018, 213, 67–70.
- Jell, G.; Stevens, M.M. Gene activation by bioactive glasses. J. Mater. Sci. Mater. Med. 2006, 17, 997–1002.
- Farano, V.; Maurin, J.C.; Attik, N.; Jackson, P.; Grosgogeat, B.; Gritsch, K. Sol–gel bioglasses in dental and periodontal regeneration: A systematic review. J. Biomed. Mater. Res. Part B Appl. Biomater. 2019, 107, 1210–1227.
- Abou Neel, E.A.; Ahmed, I.; Pratten, J.; Nazhat, S.N.; Knowles, J.C. Characterisation of antibacterial copper releasing degradable phosphate glass fibres. Biomaterials 2005, 26, 2247–2254.
- Abou, E.A.; Ae, N.; O’Austin, L.; Ae, D.; Ae, M.E.S.; Knowles, J.C. Processing, characterisation, and biocompatibility of zinc modified metaphosphate based glasses for biomedical applications. J. Mater. Sci. Mater. Med. 2008, 19, 1669–1679.
- Valappil, S.P.; Pickup, D.M.; Carroll, D.L.; Hope, C.K.; Pratten, J.; Newport, R.J.; Smith, M.E.; Wilson, M.; Knowles, J.C. Effect of silver content on the structure and antibacterial activity of silver-doped phosphate-based glasses. Antimicrob. Agents Chemother. 2007, 51, 4453–4461.
- Abou Neel, E.A.; Chrzanowski, W.; Knowles, J.C. Effect of increasing titanium dioxide content on bulk and surface properties of phosphate-based glasses. Acta Biomater. 2008, 4, 523–534.
- Lakhkar, N.J.; Abou Neel, E.A.; Salih, V.; Knowles, J.C. Strontium oxide doped quaternary glasses: Effect on structure, degradation and cytocompatibility. J. Mater. Sci. Mater. Med. 2009, 20, 1339–1346.
- Islam, M.T.; Felfel, R.M.; Abou Neel, E.A.; Grant, D.M.; Ahmed, I.; Hossain, K.M.Z. Bioactive calcium phosphate–based glasses and ceramics and their biomedical applications: A review. J. Tissue Eng. 2017, 8, 1–16.
- Nazhat, S.N.; Abou Neel, E.A.; Kidane, A.; Ahmed, I.; Hope, C.; Kershaw, M.; Lee, P.D.; Stride, E.; Saffari, N.; Knowles, J.C.; et al. Controlled microchannelling in dense collagen scaffolds by soluble phosphate glass fibers. Biomacromolecules 2007, 8, 543–551.
- Baino, F.; Verné, E. Production and characterization of glass-ceramic materials for potential use in dental applications: Thermal and mechanical properties, microstructure, and in vitro bioactivity. Appl. Sci. 2017, 7, 1330.
- Rahaman, M.N.; Day, D.E.; Sonny Bal, B.; Fu, Q.; Jung, S.B.; Bonewald, L.F.; Tomsia, A.P. Bioactive glass in tissue engineering. Acta Biomater. 2011, 7, 2355–2373.
- Salaria, S.K.; Ghuman, S.K.; Kumar, S.; Sharma, G. Management of localized advance loss of periodontal support associated Grade II furcation and intrabony defect in chronic periodontitis patient through amalgamation of platelet-rich fibrin and hydroxyapatite bioactive glass composite granules. Contemp. Clin. Dent. 2016, 7, 405–408.
- Salem, A.M.; Jones, S.J.; Ellis, I.R.; Chadwick, R.G. Investigating the addition of collagen and its integrin binding sequence (RGD) to glass polyalkenoate: In terms of material and cellular properties to explore a more biocompatible method of root caries restoration. J. Dent. 2016, 54, 68–76.
- Cannio, M.; Bellucci, D.; Roether, J.A.; Boccaccini, D.N.; Cannillo, V. Bioactive glass applications: A literature review of human clinical trials. Materials 2021, 14, 5440.
- Owens, G.J.; Singh, R.K.; Foroutan, F.; Alqaysi, M.; Han, C.M.; Mahapatra, C.; Kim, H.W.; Knowles, J.C. Sol–gel based materials for biomedical applications. Prog. Mater. Sci. 2016, 77, 1–79.
- Bertoldi, C.; Zaffe, D.; Consolo, U. Polylactide/polyglycolide copolymer in bone defect healing in humans. Biomaterials 2008, 29, 1817–1823.
- El-Rashidy, A.A.; Roether, J.A.; Harhaus, L.; Kneser, U.; Boccaccini, A.R. Regenerating bone with bioactive glass scaffolds: A review of in vivo studies in bone defect models. Acta Biomater. 2017, 62, 1–28.
- Miguez-Pacheco, V.; Hench, L.L.; Boccaccini, A.R. Bioactive glasses beyond bone and teeth: Emerging applications in contact with soft tissues. Acta Biomater. 2015, 13, 1–15.
- Cannillo, V.; Sola, A. Different approaches to produce coatings with bioactive glasses: Enamelling vs plasma spraying. J. Eur. Ceram. Soc. 2010, 30, 2031–2039.
- Bolelli, G.; Cannillo, V.; Gadow, R.; Killinger, A.; Lusvarghi, L.; Rauch, J. Microstructural and in vitro characterisation of high-velocity suspension flame sprayed (HVSFS) bioactive glass coatings. J. Eur. Ceram. Soc. 2009, 29, 2249–2257.
- Cannillo, V.; Colmenares-Angulo, J.; Lusvarghi, L.; Pierli, F.; Sampath, S. In vitro characterisation of plasma-sprayed apatite/wollastonite glass-ceramic biocoatings on titanium alloys. J. Eur. Ceram. Soc. 2009, 29, 1665–1677.
- Bellucci, D.; Sola, A.; Gentile, P.; Ciardelli, G.; Cannillo, V. Biomimetic coating on bioactive glass-derived scaffolds mimicking bone tissue. J. Biomed. Mater. Res. Part A 2012, 100, 3259–3266.
- Cattini, A.; Bellucci, D.; Sola, A.; Pawłowski, L.; Cannillo, V. Suspension plasma spraying of optimized functionally graded coatings of bioactive glass/hydroxyapatite. Surf. Coat. Technol. 2013, 236, 118–126.
- Cannillo, V.; Montorsi, M.; Siligardi, C.; Sola, A.; de Portu, G.; Micele, L.; Pezzotti, G. Microscale computational simulation and experimental measurement of thermal residual stresses in glass-alumina functionally graded materials. J. Eur. Ceram. Soc. 2006, 26, 1411–1419.
- Cannillo, V.; Leonelli, C.; Boccaccini, A.R. Numerical models for thermal residual stresses in Al2O3 platelets/borosilicate glass matrix composites. Mater. Sci. Eng. A 2002, 323, 246–250.
- Sergi, R.; Bellucci, D.; Cannillo, V. A review of bioactive glass/natural polymer composites: State of the art. Materials 2020, 13, 5560.
- Wu, C.; Zhou, Y.; Lin, C.; Chang, J.; Xiao, Y. Strontium-containing mesoporous bioactive glass scaffolds with improved osteogenic/cementogenic differentiation of periodontal ligament cells for periodontal tissue engineering. Acta Biomater. 2012, 8, 3805–3815.
- Zhu, M.; Li, J.; Chen, B.; Mei, L.; Yao, L.; Tian, J.; Li, H. The effect of calcium sodium phosphosilicate on dentin hypersensitivity: A systematic review and meta-analysis. PLoS ONE 2015, 10, e0140176.
- Zamani, D.; Moztarzadeh, F.; Bizari, D. Alginate-bioactive glass containing Zn and Mg composite scaffolds for bone tissue engineering. Int. J. Biol. Macromol. 2019, 137, 1256–1267.
- Lotfi, G.; Shokrgozar, M.A.; Mofid, R.; Abbas, F.M.; Ghanavati, F.; Baghban, A.A.; Yavari, S.K.; Pajoumshariati, S. Biological Evaluation of Bilayered Collagenous Coated Chitosan Membrane for Periodontal Guided Bone Regeneration. Ann. Biomed. Eng. 2016, 44, 2132–2144.
- Fakhri, E.; Eslami, H.; Maroufi, P.; Pakdel, F.; Taghizadeh, S.; Ganbarov, K.; Yousefi, M.; Tanomand, A.; Yousefi, B.; Mahmoudi, S.; et al. Chitosan biomaterials application in dentistry. Int. J. Biol. Macromol. 2020, 162, 956–974.
- Vagropoulou, G.; Trentsiou, M.; Georgopoulou, A.; Papachristou, E.; Prymak, O.; Kritis, A.; Epple, M.; Chatzinikolaidou, M.; Bakopoulou, A.; Koidis, P. Hybrid chitosan/gelatin/nanohydroxyapatite scaffolds promote odontogenic differentiation of dental pulp stem cells and in vitro biomineralization. Dent. Mater. 2021, 37, e23–e36.
- Yamamoto, T.; Ugawa, Y.; Kawamura, M.; Yamashiro, K.; Kochi, S.; Ideguchi, H.; Takashiba, S. Modulation of microenvironment for controlling the fate of periodontal ligament cells: The role of Rho/ROCK signaling and cytoskeletal dynamics. J. Cell Commun. Signal. 2018, 12, 369–378.
- Aydin, S.; Şahin, F. Stem Cells Derived from Dental Tissues. Adv. Exp. Med. Biol. 2019, 1144, 123–132.
- Hosoya, A.; Shalehin, N.; Takebe, H.; Fujii, S.; Seki, Y.; Mizoguchi, T.; Shimo, T.; Iijima, M.; Irie, K. Stem cell properties of Gli1-positive cells in the periodontal ligament. J. Oral Biosci. 2020, 62, 299–305.
- Li, C.; Guo, C.; Fitzpatrick, V.; Ibrahim, A.; Zwierstra, M.J.; Hanna, P.; Lechtig, A.; Nazarian, A.; Lin, S.J.; Kaplan, D.L. Design of biodegradable, implantable devices towards clinical translation. Nat. Rev. Mater. 2020, 5, 61–81.
- Zheng, K.; Niu, W.; Lei, B.; Boccaccini, A.R. Immunomodulatory bioactive glasses for tissue regeneration. Acta Biomater. 2021, 133, 168–186.
- Bertoldi, C.; Bellei, E.; Pellacani, C.; Ferrari, D.; Lucchi, A.; Cuoghi, A.; Bergamini, S.; Cortellini, P.; Tomasi, A.; Zaffe, D.; et al. Non-bacterial protein expression in periodontal pockets by proteome analysis. J. Clin. Periodontol. 2013, 40, 573–582.
- Socransky, S.S.; Haffajee, A.D. The nature of periodontal diseases. Ann. Periodontol. 1997, 2, 3–10.
- Meyle, J.; Iain Chapple, I. Molecular aspects of the pathogenesis of periodontitis. Periodontology 2000 2015, 69, 7–17.
- Melcher, A.H. Cells of periodontium: Their role in the healing of wounds. Ann. R. Coll. Surg. Engl. 1985, 67, 130–131.
- Sunandhakumari, V.J.; Vidhyadharan, A.K.; Alim, A.; Kumar, D.; Ravindran, J.; Krishna, A.; Prasad, M. Fabrication and in vitro characterization of bioactive glass/nano hydroxyapatite reinforced electrospun poly composite membranes for guided tissue regeneration. Bioengineering 2018, 5, 54.
- Esfahanizadeh, N.; Nourani, M.R.; Bahador, A.; Akhondi, N.; Montazeri, M. The Anti-biofilm Activity of Nanometric Zinc doped Bioactive Glass against Putative Periodontal Pathogens: An in vitro Study. Biomed. Glasses 2020, 4, 95–107.
- Varanasi, V.G.; Owyoung, J.B.; Saiz, E.; Marshall, S.J.; Marshall, G.W.; Loomer, P.M. The ionic products of bioactive glass particle dissolution enhance periodontal ligament fibroblast osteocalcin expression and enhance early mineralized tissue development. J. Biomed. Mater. Res. Part A 2011, 98, 177–184.
- Bai, N.; Chen, W.; Luo, L.; Tong, W.; Wen, C.; Zhan, X.; Sa, B. Effect of B2O3 on the structural and in vitro biological assessment of mesoporous bioactive glass nanospheres. J. Am. Ceram. Soc. 2021, 104, 3058–3072.
- Mota, J.; Yu, N.; Caridade, S.G.; Luz, G.M.; Gomes, M.E.; Reis, R.L.; Jansen, J.A.; Walboomers, X.F.; Mano, J.F. Chitosan/bioactive glass nanoparticle composite membranes for periodontal regeneration. Acta Biomater. 2012, 8, 4173–4180.
- Ruiz-Clavijo, A.; Hurt, A.P.; Kotha, A.K.; Coleman, N.J. Effect of calcium precursor on the bioactivity and biocompatibility of sol–gel-derived glasses. J. Funct. Biomater. 2019, 10, 13.
- Meneses, C.C.B.; Olivi, L.T.; Carvalho, C.N.; Gavini, G.; Sipert, C.R. Cytotoxic Effect of Niobium Phosphate Glass–based Gutta-Percha Points on Periodontal Ligament Fibroblasts In Vitro. J. Endod. 2020, 46, 1297–1301.
- Wen, C.; Bai, N.; Luo, L.; Ye, J.; Zhan, X.; Zhang, Y.; Sa, B. Structural behavior and in vitro bioactivity evaluation of hydroxyapatite-like bioactive glass based on the SiO2-CaO-P2O5 system. Ceram. Int. 2021, 47, 18094–18104.
- Uskoković, V.; Abuna, G.; Ferreira, P.; WU, V.M.; Gower, L.; Pires-de-Souza, F.C.P.; Murata, R.M.; Sinhoreti, M.A.C.; Geraldeli, S. Synthesis and characterization of nanoparticulate niobium- and zinc-doped bioglass-ceramic/chitosan hybrids for dental applications. J. Sol-Gel Sci. Technol. 2021, 97, 245–258.
- Caridade, S.G.; Merino, E.G.; Martins, G.V.; Luz, G.M.; Alves, N.M.; Mano, J.F. Membranes of poly/Bioglass® with asymmetric bioactivity for biomedical applications. J. Bioact. Compat. Polym. 2012, 27, 429–440.
- Beketova, A.; Poulakis, N.; Bakopoulou, A.; Zorba, T.; Papadopoulou, L.; Christofilos, D.; Kantiranis, N.; Zachariadis, G.A.; Kontonasaki, E.; Kourouklis, G.A. Inducing bioactivity of dental ceramic/bioactive glass composites by Nd:YAG laser. Dent. Mater. 2016, 32, e284–e296.
- Granel, H.; Bossard, C.; Collignon, A.-M.; Wauquier, F.; Lesieur, J.; Rochefort, G.Y.; Jallot, E.; Lao, J.; Wittrant, Y. Bioactive Glass/Polycaprolactone Hybrid with a Dual Cortical/Trabecular Structure for Bone Regeneration. ACS Appl. Bio Mater. 2019, 2, 3473–3483.
- Casarrubios, L.; Gómez-Cerezo, N.; Feito, M.J.; Vallet-Regí, M.; Arcos, D.; Portolés, M.T. Ipriflavone-loaded mesoporous nanospheres with potential applications for periodontal treatment. Nanomaterials 2020, 10, 2573.
- Shah, A.T.; Zahid, S.; Ikram, F.; Maqbool, M.; Chaudhry, A.A.; Rahim, M.I.; Schmidt, F.; Goerke, O.; Khan, A.S.; Ur Rehman, I. Tri-layered functionally graded membrane for potential application in periodontal regeneration. Mater. Sci. Eng. C Mater. Biol. Appl. 2019, 103, 109812.
- Jia, X.; Miron, R.J.; Yin, C.; Xu, H.; Luo, T.; Wang, J.; Jia, R.; Wu, M.; Zhang, Y.; Li, Y. HnRNPL inhibits the osteogenic differentiation of PDLCs stimulated by SrCl2 through repressing Setd2. J. Cell. Mol. Med. 2019, 23, 2667–2677.
- Sowmya, S.; Mony, U.; Jayachandran, P.; Reshma, S.; Kumar, R.A.; Arzate, H.; Nair, S.V.; Jayakumar, R. Tri-Layered Nanocomposite Hydrogel Scaffold for the Concurrent Regeneration of Cementum, Periodontal Ligament, and Alveolar Bone. Adv. Healthc. Mater. 2017, 6, 1601251.
- Moonesi Rad, R.; Atila, D.; Evis, Z.; Keskin, D.; Tezcaner, A. Development of a novel functionally graded membrane containing boron-modified bioactive glass nanoparticles for guided bone regeneration. J. Tissue Eng. Regen. Med. 2019, 13, 1331–1345.
- Balamurugan, A.; Balossier, G.; Laurent-Maquin, D.; Pina, S.; Rebelo, A.H.S.; Faure, J.; Ferreira, J.M.F. An in vitro biological and anti-bacterial study on a sol–gel derived silver-incorporated bioglass system. Dent. Mater. 2008, 24, 1343–1351.
- Felipe, M.E.M.C.; Andrade, P.F.; Novaes, A.B.J.; Grisi, M.F.M.; Souza, S.L.S.; Taba, M.; Palioto, D.B. Potential of bioactive glass particles of different size ranges to affect bone formation in interproximal periodontal defects in dogs. J. Periodontol. 2009, 80, 808–815.
- Lee, S.-B.; Jung, U.-W.; Choi, Y.; Jamiyandorj, O.; Kim, C.S.; Lee, Y.K.; Chai, J.K.; Choi, S.H. Investigation of bone formation using calcium phosphate glass cement in beagle dogs. J. Periodontal Implant. Sci. 2010, 40, 125–131.
- Carvalho, M.D.; Suaid, F.F.; Santamaria, M.P.; Casati, M.Z.; Nociti, F.M., Jr.; Sallum, A.W.; Sallum, E.A. Platelet-rich plasma plus bioactive glass in the treatment of intra-bony defects: A study in dogs. J. Appl. Oral Sci. Rev. FOB 2011, 19, 82–89.
- Zhang, Y.; Wei, L.; Wu, C.; Miron, R.J. Periodontal regeneration using strontium-loaded mesoporous bioactive glass scaffolds in osteoporotic rats. PLoS ONE 2014, 9, e104527.
- Lusvardi, G.; Zaffe, D.; Menabue, L.; Bertoldi, C.; Malavasi, G.; Consolo, U. In vitro and in vivo behaviour of zinc-doped phosphosilicate glasses. Acta Biomater. 2009, 5, 419–428.
- Samira, J.; Saoudi, M.; Abdelmajid, K.; Hassane, O.; Treq, R.; Hafed, E.; Abdelfatteh, E.; Hassib, K. Accelerated bone ingrowth by local delivery of Zinc from bioactive glass: Oxidative stress status, mechanical property, and microarchitectural characterization in an ovariectomized rat model. Libyan J. Med. 2015, 10, 28572.
- Bertoldi, C.; Venuta, M.; Guaraldi, G.; Lalla, M.; Guaitolini, S.; Generali, L.; Monzani, D.; Cortellini, P.; Zaffe, D. Are periodontal outcomes affected by personality patterns? A 18-month follow-up study. Acta Odontol. Scand. 2018, 76, 48–57.
- Bertoldi, C.; Pradelli, J.M.; Consolo, U.; Zaffe, D. Release of elements from retrieved maxillofacial plates and screws. J. Mater. Sci. Mater. Med. 2005, 16, 857–861.
- Bertoldi, C.; Zaffe, D. In vivo comparison of two bone substitutes in the distal femur of the rabbit. Int. J. Oral Maxillofac. Implant. 2012, 27, 119–127.
- Cortellini, P.; Tonetti, M.S. Clinical concepts for regenerative therapy in intrabony defects. Periodontology 2000 2015, 68, 282–307.
- Bertoldi, C.; Ferrari, M.; Giannetti, L. The use of only enamel matrix derivative allows outstanding regeneration results in periodontal intrabony defect treatment: A retrospective study. J. Biol. Regul. Homeost. Agents 2019, 33, 633–636.
- Tavelli, L.; McGuire, M.K.; Zucchelli, G.; Rasperini, G.; Feinberg, S.E.; Wang, H.L.; Giannobile, W.V. Biologics-based regenerative technologies for periodontal soft tissue engineering. J. Periodontol. 2020, 91, 147–154.
- Chambrone, L.; Ortega, M.A.S.; Sukekava, F.; Rotundo, R.; Kalemaj, Z.; Buti, J.; Pini Prato, G.P. Root coverage procedures for treating single and multiple recession-type defects: An updated Cochrane systematic review. J. Periodontol. 2019, 90, 1399–1422.
- Pellegrini, G.; Pagni, G.; Rasperini, G. Surgical approaches based on biological objectives: GTR versus GBR techniques. Int. J. Dent. 2013, 2013, 521547.
- Sculean, A.; Pietruska, M.; Arweiler, N.B.; Auschill, T.M.; Nemcovsky, C. Four-year results of a prospective-controlled clinical study evaluating healing of intra-bony defects following treatment with an enamel matrix protein derivative alone or combined with a bioactive glass. J. Clin. Periodontol. 2007, 34, 507–513.
- Mir-Mari, J.; Wui, H.; Jung, R.E.; Hämmerle, C.H.F.; Benic, G.I. Influence of blinded wound closure on the volume stability of different GBR materials: An in vitro cone-beam computed tomographic examination. Clin. Oral Implant. Res. 2016, 27, 258–265.
- Trombelli, L.; Farina, R. Clinical outcomes with bioactive agents alone or in combination with grafting or guided tissue regeneration. J. Clin. Periodontol. 2008, 35 (Suppl. 8), 117–135.
- Cortellini, P.; Tonetti, M.S. Improved wound stability with a modified minimally invasive surgical technique in the regenerative treatment of isolated interdental intrabony defects. J. Clin. Periodontol. 2009, 36, 157–163.
- Trombelli, L.; Simonelli, A.; Pramstraller, M.; Wikesjö, U.M.E.; Farina, R. Single Flap Approach with and without Guided Tissue Regeneration and a Hydroxyapatite Biomaterial in the Management of Intraosseous Periodontal Defects. J. Periodontol. 2010, 81, 1256–1263.
- Humagain, M.; Nayak, D.G.; Uppoor, A.S. A clinical evaluation of bioactive glass particulate in the treatment of mandibular class II furcation defects. Braz. J. Oral Sci. 2007, 6, 1450–1456.
- Kumar, P.G.; Kumar, J.A.; Anumala, N.; Reddy, K.P.; Avula, H.; Hussain, S.N. Volumetric analysis of intrabony defects in aggressive periodontitis patients following use of a novel composite alloplast: A pilot study. Quintessence Int. 2011, 42, 375–384.
- Subbaiah, R.; Thomas, B. Efficacy of a bioactive alloplast, in the treatment of human periodontal osseous defects-a clinical study. Med. Oral Patol. Oral Y Cir. Bucal 2011, 16, e239–e244.
- Mistry, S.; Kundu, D.; Datta, S.; Basu, D. Effects of bioactive glass, hydroxyapatite and bioactive glass-Hydroxyapatite composite graft particles in the treatment of infrabony defects. J. Indian Soc. Periodontol. 2012, 16, 241–246.
- Cayir Keles, G.; Ozkan Cetinkaya, B.; Albayrak, D.; Koprulu, H.; Acikgoz, G. Comparison of platelet pellet and bioactive glass in periodontal regenerative therapy. Acta Odontol. Scand. 2006, 64, 327–333.
- Demir, B.; Sengün, D.; Berberoğlu, A. Clinical evaluation of platelet-rich plasma and bioactive glass in the treatment of intra-bony defects. J. Clin. Periodontol. 2007, 34, 709–715.
- Cetinkaya, B.O.; Keles, G.C.; Pamuk, F.; Balli, U.; Keles, Z.P. Long-term clinical results on the use of platelet concentrate in the treatment of intrabony periodontal defects. Acta Odontol. Scand. 2014, 72, 92–98.
- Kaur, M.; Ramakrishnan, T.; Amblavanan, N.; Emmadi, P. Effect of platelet-rich plasma and bioactive glass in the treatment of intrabony defects—A split-mouth study in humans. Braz. J. Oral Sci. 2010, 9, 108–114.
- Biswas, S.; Sambashivaiah, S.; Kulal, R.; Bilichodmath, S.; Kurtzman, G.M. Comparative evaluation of bioactive glass and platelet rich fibrin in treating furcation Defects. J. Oral Implantol. 2016, 42, 411–415.
- Naqvi, A.; Gopalakrishnan, D.; Bhasin, M.T.; Sharma, N.; Haider, K.; Martande, S. Comparative evaluation of bioactive glass putty and platelet rich fibrin in the treatment of human periodontal intrabony defects: A randomized control trial. J. Clin. Diagn. Res. 2017, 11, ZC09–ZC13.
- Saravanan, D.; Rethinam, S.; Muthu, K.; Thangapandian, A. The Combined Effect of Bioactive Glass and Platelet-Rich Fibrin in Treating Human Periodontal Intrabony Defects—A Clinicoradiographic Study. Contemp. Clin. Dent. 2019, 10, 110–116.
- Slezák, R.; Paulusová, V. Use of the NovaBone augmentation material in the treatment of chronic periodontitis. Preliminary communication. Acta Med. /Univ. Carol. Fac. Med. Hradec Králové 2013, 56, 157–161.
- Grover, V.; Kapoor, A.; Malhotra, R.; Uppal, R.S. Evaluation of the efficacy of a bioactive synthetic graft material in the treatment of intrabony periodontal defects. J. Indian Soc. Periodontol. 2013, 17, 104–110.
- Gupta, V.; Bains, V.K.; Singh, G.P.; Jhingran, R. Clinical and cone beam computed tomography comparison of NovaBone dental putty and perioglas in the treatment of mandibular class II furcations. Indian J. Dent. Res. 2014, 25, 166–173.
- Lysenko, O.; Borysenko, A. Bioactive Glass-Ceramic Composition In Surgical Management of Periodontal Intrabony Defects. Georgian Med. News 2019, 295, 34–41.
- Koduru, S.; Aghanashini, S.; Nadiger, S.; Apoorva, S.; Bhat, D.; Puvvalla, B. A clinical and radiographic evaluation of the efficacy of nanohydroxyapatite versus bioactive calcium phosphosilicate putty in the treatment of human periodontal infrabony defects: A randomized clinical trial. Contemp. Clin. Dent. 2019, 10, 16–23.
- Katuri, K.; Kumar, P.; Swarna, C.; Swamy, D.; Arun, K. Evaluation of bioactive glass and demineralized freeze dried bone allograft in the treatment of periodontal intraosseous defects: A comparative clinico-radiographic study. J. Indian Soc. Periodontol. 2013, 17, 367–372.





