Your browser does not fully support modern features. Please upgrade for a smoother experience.

Submitted Successfully!
Thank you for your contribution! You can also upload a video entry or images related to this topic.
For video creation, please contact our Academic Video Service.
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Rita Moretti | -- | 1895 | 2022-03-30 11:11:42 | | | |
| 2 | Amina Yu | + 7 word(s) | 1902 | 2022-03-31 03:06:00 | | | | |
| 3 | Amina Yu | Meta information modification | 1902 | 2022-03-31 03:15:14 | | |
Video Upload Options
We provide professional Academic Video Service to translate complex research into visually appealing presentations. Would you like to try it?
Cite
If you have any further questions, please contact Encyclopedia Editorial Office.
Moretti, R.; Caruso, P. Small Vessel Disease. Encyclopedia. Available online: https://encyclopedia.pub/entry/21168 (accessed on 21 May 2026).
Moretti R, Caruso P. Small Vessel Disease. Encyclopedia. Available at: https://encyclopedia.pub/entry/21168. Accessed May 21, 2026.
Moretti, Rita, Paola Caruso. "Small Vessel Disease" Encyclopedia, https://encyclopedia.pub/entry/21168 (accessed May 21, 2026).
Moretti, R., & Caruso, P. (2022, March 30). Small Vessel Disease. In Encyclopedia. https://encyclopedia.pub/entry/21168
Moretti, Rita and Paola Caruso. "Small Vessel Disease." Encyclopedia. Web. 30 March, 2022.
Copy Citation
Small vessel disease (SVD) is one of the most frequent pathological conditions which lead to dementia. Biochemical and neuroimaging might help correctly identify the clinical diagnosis of this relevant brain disease. The microvascular alterations which underlie SVD have common origins, similar cognitive outcomes, and common vascular risk factors.
small vessel disease
vascular damage
reactive oxygen species
metalloproteinases
1. Possible and Proved New Markers of Blood–Brain Barrier Leakage, Perivascular Enlargements, and Mitochondrial Alterations
Small vessel disease (SVD) has the small vessels (pial and the small penetrating) and white matter as a significant definite target. Nevertheless, growing attention has been dedicated to disrupting perivascular spaces, astrocytic end-feet, capillaries, and veins. As a final point, the blood–brain barrier (BBB) has been addressed as another potential target of the intrigued mechanisms that underlie the small vessel brain pathology complex. BBB is not only a solid defensive barrier but acts as an active and specific player of active selection crossover, possessing cell-cell signaling with the end-feet of astrocytes and disclosure a potential role of maintaining efflux pumps [1][2][3][4][5]. Thus, the disruption of the BBB is proportionately increased by normal aging but progresses as a hallmark in different pathologies, i.e., multiple sclerosis or in primary inflammatory disease. Nevertheless, it is an expression of white matter inflammation, even due to chronic hypoperfusion, such as the one which occurs in small vessel disease [SVD], accomplishing the progression and the extension of the white matter sufferance, named as white matter hyperintensities (WMH) [6][7][8][9][10][11][12], the confluency of which is synonymous with SVD progression, leading to subcortical vascular dementia (sVAD) [13][12]. In AD-prone patients, BBB disruption has been signaled even in hippocampal degeneration, which occurs after a major stroke [11][12].
A dynamic contrast-enhanced MRI (DCE-MRI) [14] has been employed for in-vivo quantification of the pathological passage of plasma through BBB [15][16]. Moreover, apart from the BBB leakage, the possibility of estimating the vascular permeability-surface area product (PS) and the plasma volume fraction (VP) in a given region of interest has also been described [17][18]. The model suggested that PS increased with WMH severity, aging, and other vascular risk factors, and at the same time, a lower blood vP [16]. The most promising in-vivo demonstration is that BBB integrity is compromised in more severe WMH, even beyond visible lesions [14] (Insert Figure 1).
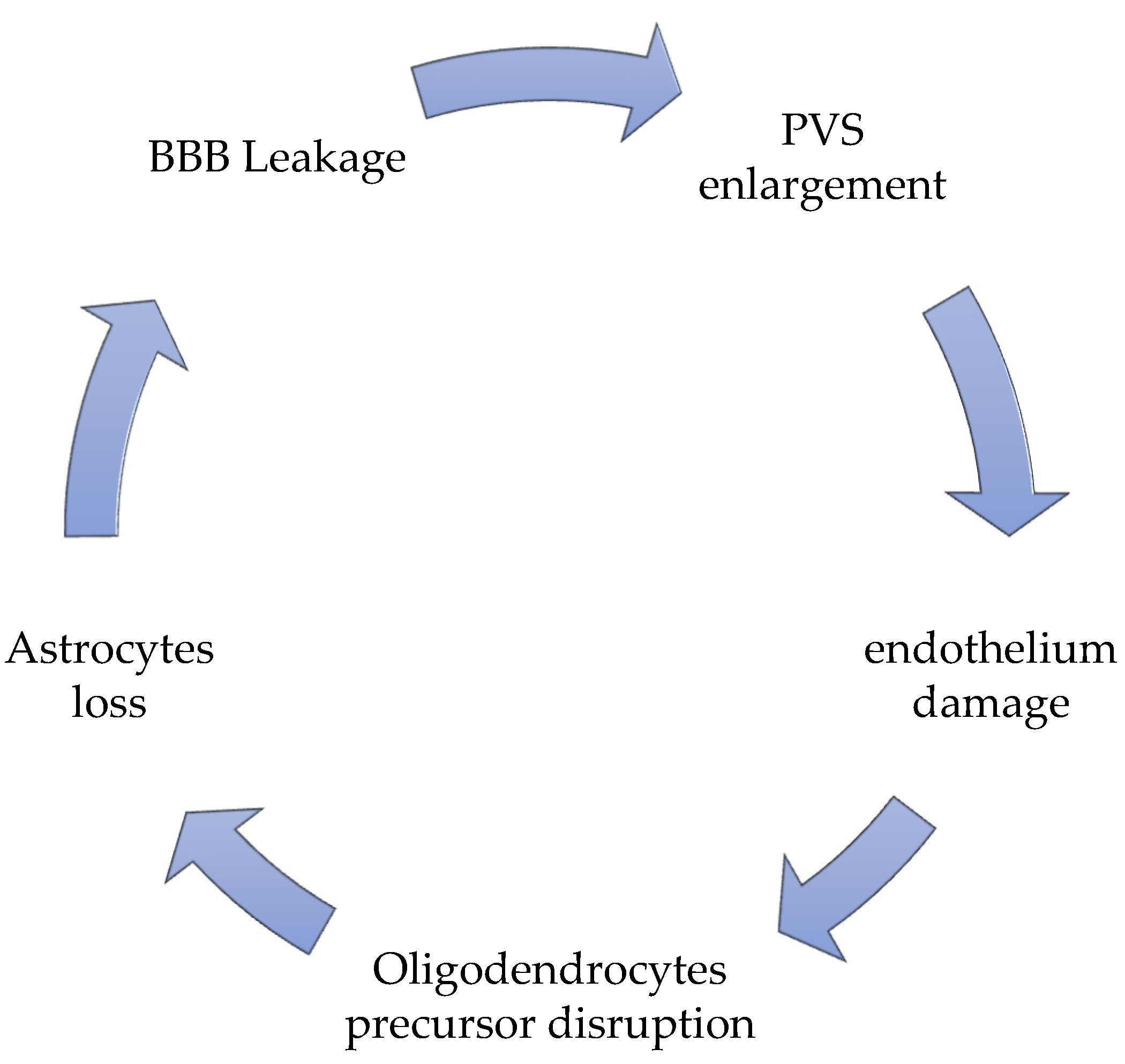
Figure 1. The vicious circle of SVD pathology.
Even if it was known that BBB is disrupted in SVD, it is still unknown that the reasons for BBB leakage in this condition. The most disputed involvement is one of the pericytes. Pericytes are capillary mural cells that stabilize newly formed vessels and induce repair. When a pericyte-deficient adult mouse model has been employed [19], different transcriptional changes in brain endothelial cells have been mapped due to a defective pericyte contact at a single-cell level. In that conformation, endothelial cells, deprived of pericyte contacts, seem to exhibit a “venous-shifted molecular pattern,” and therefore lack any capillary specialization, and upregulate proteins which are typically expressed during developmental stages, such as the Fibroblast Growth Factor Binding Protein (Fgfbp1), or those expressed during pathological angiogenesis, such as Angiopoietin 2 (Angpt2). These aspects permit a possible cell proliferation, with a very flawed arteriolar BBB regulation system, and reduction of the angiogenesis process [19]. Fgfbp1 and Angpt2 levels could probably be crucial markers of BBB leakage during SVD. More studies will be necessary to prove that.
Perivascular spaces (PVS) have gained an essential role in SVD pathogenesis; they are no longer considered as virtual empty spaces, but as the most efficacious catabolites clearance system [13]; they are resident sites of perivascular macrophages, pial cells, mast cells, nerve fibers, and collagen fibers [20]. PVS are virtual spaces intimately connected to deep arterioles [21]. Even in these conditions, they act as a lymphatic net, defined as a glymphatic-perivascular territory [22].
Their malfunction, the hallmarks of which are the combined enlargement and widening, is the principal responsibility for perivascular accumulation of catabolites and toxic substances, which is determinant for enhancing ongoing neural damage until starvation [23][24]. The perivascular debris accumulation, together with the BBB leakage, potentiates and accelerates the perivascular inflammation, strongly favored by the stagnation-induced process and by medical conditions which influence it, such as hypertension and diabetes [25][26][27][28][29]. PVS enlargement is responsible for an altered cerebrovascular reactivity (CVR) [13], due to the extension of the constant inflammatory response [30] present as a constant marker in SVD, due to the chronic hypoperfusion state. The PVS is never an isolated situation, but it is accompanied by an altered BBB disruption and a significant perivascular inflammation [26][31][32][33][34]. More recently, new actors contribute with BBB leakage and PVS enlargement to help the progress of SVD [35][36][37][38], such as the oligodendrocyte precursor cells (OPCs), which generally help BBB stabilization [38][39] and the astrocytes, which exert their fundamental role as regulating the signal of neuro-vascular coupling [13]. Oligodendrocytes are the first victims of chronic models of chronic cerebral hypoperfusion (CCH), together with the precocious sufferance of the perineural space [40][41][42], and with a hyper-activation of microglia, firstly in the hippocampus [43][44], then in the thalamus, up to in the cortical neuronal population [45]. Secondary to oligodendrocytes, astrocyte death occurs in proportion to the chronic ischemia condition’s length and severity [46][47], due to the ongoing modifications of general and neuronal metabolic requests. Their death is a consequence of chronic hypoxia, but it worsens neuronal death due to a lack of functions, regulating the neurovascular coupling signal [48]. The process by which this occurs is that during the entire process of chronic ischemia, microglia retract its branches, with a consequent reduction of the length and strength of the microglial ramification, with a concomitant degeneration of the soma [49]. The frontal activation of microglia occurs in a two-step pattern: at the beginning, M1 activation upregulates TNF alpha, Il-23, IL-1beta, and Il12 production, which attack neurons, and directly contribute to their injury; only after M2 activation occurs can the reparation process can begin [50]. In the SVD, due to the chronic hypoxia-hypoperfusion condition [13], the passage through M1 towards M2 activation does not occur [50]. In SVD, there is a substantial augmentation of M1 activation, together with a heavy reduction of M2 promotion [51][52]. The brisk oligodendrocyte degeneration, associated with M1 activation, increases calcium currents and induces a severe apoptosis process. The calcium increases, and the severe apoptosis is accompanied by an augmentation of caspase-3 RNA and matrix-metalloprotease 2 (MMP-2) [53]. At the beginning of the SVD process, these markers reflect the temptation reparation process induced by a standard M1/M2 passage, as described above. Nevertheless, until the chronic inflammatory condition occurs in SVD ongoing development, there is an alteration of the M1/M2 passage, with a predominant M1 event; therefore, in SVD patients’ cerebrospinal fluid (CSF), there is a constant growth of oligodendrocyte-derived myelin sheath-like myelin lipid sulfatide (ODMSMS) and myelin essential protein (MBP) due to the massive oligodendrocytes death [54][55][56][57]. For similar reasons, markers of axonal damage, i.e., neurofilament light chain (NFL), together with CSF α-1 antitrypsin, tissue inhibitor of metalloproteinase-1 (TIMP-1), plasminogen activator inhibitor-1 (PAI-1), and apolipoprotein H (ApoH) have been found to increase very early in the CSF in SVD [58][59][60]. Finally, due to the BBB leakage, ultrastructural studies find that in older animals as well as in those affected by SVD, there are severe alterations of the capillary basement membrane of the deeper arterioles, inside the white matter, filling plasma proteins into vascular bagging and collagen deposition inside PVS, in a phenomenon described as microvascular fibrosis [5][50][61]. Many studies have testified that microvascular fibrosis and BBB splitting have a higher CSF/serum albumin (SA) ratio in patients with SVD [61]. Matrix remodeling pathway (TIMP-1 and matrix metalloproteinases) as an expression of endothelium disruption in SVD has been described [61].
2. Markers of Oxidative Damages in SVD
Reactive oxygen species (ROS) is an umbrella term for many ordinary derivatives of molecular oxygen, and their accumulation leads to a complex phenomenon called oxidative distress. There are two species, hydrogen peroxide (H2O2) and the superoxide anion radical (O2−), which are key redox signaling agents generated under the control of growth factors and cytokines by more than 40 enzymes, prominently including nicotinamide adenine dinucleotide phosphate (NADPH) oxidases [13] and the mitochondrial electron transport chain [62]. When mitochondrial cells usually function, the active process of oxidative phosphorylation converts oxygen to superoxide by oxidase enzymes, and superoxide can be transformed by superoxide dismutase (SOD) or to non-radical hydrogen peroxide [62][63][64], for example, from glutathione peroxidase (Gpx), or when catalase enzymatically metabolizes hydrogen peroxide to water and oxygen [63].
Chronic cerebral conditions of constant hypoxia are the principal inductors of the uncontrolled production of ROS [65][66].
NADPH oxidase activity and mitochondrial are significantly higher in cerebral arteries when compared with systemic arteries in blood vessels from healthy animals (mouse, rat, pig, and rabbit) [67][68]. Thus, brain vessels are one of the most prominent productions of ROS, suggesting that there could be fundamental ROS-dependent signaling in cerebral arteries, which might be indispensable for vasoactive regulation properties.
Thus, the accumulation of ROS species, associated with mitochondrial dysfunction, BBB disruption, and chronic inflammatory status are three conditions in SVD and are proportionate to WMH extension. They lead to an altered endothelial further altered activation, which is reflected in a decoupling of the neurovascular coupling system, with significant sub-cortical and cortical signal alteration, with consequent reflex in oligodendrocytes astrocytes and finally to neurons [13][69]. An active role of flow-dependent responses in rat cerebral arteries has been recently demonstrated in vivo, directly exerted by the NADPH-oxidase reactions [70]. Specifically, Nox2-NADPH oxidase dysfunction is related to the propagation of the ischemic brain injury, derived by the occlusion of larger pial arteries; Nox2/NOx2 knock-out mice, in the same condition, show the minor extension of brain injury after an ischemic infarct [71].
The induced alterations of mitochondrial DNA by ROS attacks and chronic ischemic conditions are some of the most critical contributors to neuronal aging and degeneration, either considering oxidative damage as a promoter or as a consequence of it [72][73][74].
The decline of mitochondrial functioning has been largely implicated in the aging process and is characterized by a reduced density of mitochondria and reduced mitogenesis [75][76][77][78][79]. Such changes, which originate as replication errors, accumulate in postmitotic tissues during aging, leading to increased proportions of impaired mitochondria [79]. In the aging brain, there has been a sufficient demonstration of impairment of synaptic mitochondria leading to impaired neurotransmission and cognitive failure [76][77][78][79][80][81][82]. Precocious forms of small vessel disease, leading to vascular dementia, have been described in specific mitochondrial point mutation [83]. Other mitochondrial mutation phenotypes have been described as pure brain involvement, including fluctuating encephalopathy, seizures, dementia, migraine, stroke-like episodes, ataxia, and spasticity [76][80][81][82]. Growing attention should be paid to mitochondrial DNA mutations for brain pathologies, in order to gain more robust data on their possible relevance, and their correlation with postmortem neuropathologic features, to advance the understanding [83][84][85][86][87][88].
Oxidative stress potentiates the disorders of the endothelium-dependent NO signaling [89][90]. Uncoupling endothelial NO synthase (eNOS) (for example, in relation with lower levels of tetrahydrobiopterin) switches the production of NO to that of superoxide, causing an overwhelming potentiation of ROS production, accelerating the oxidative stress, lowering the NO anti-inflammatory properties [91][92], and reducing NO modulation of Rho-kinase activity, inhibiting vascular tone control [93]. Rho-kinase, as a counterpart, influences mRNA-stability of eNOS [94].
The induction of oxidative stress is one of the most important promoters of pathological angiogenesis, by lipid oxygenation, thickening the blood vessel walls [95][96]. Moreover, the ApoE4 allele and the AD process seem to be involved in promoting vascular alterations independently of other recognized factors, i.e., age, diabetes, hypertension, and obesity, and so on. However, it is supposed to worsen the confluency of WMH, probably somehow linked to ROS augmentation, without any other positive data [97][98][99].
References
- Abbott, N.J.; Patabendige, A.A.K.; Dolman, D.E.M.; Yusof, S.R.; Begley, D.J. Structure and function of the blood-brain barrier. Neurobiol. Dis. 2010, 37, 13–25.
- Zlokovic, B.V. The Blood-Brain Barrier in Health and Chronic Neurodegenerative Disorders. Neuron 2008, 57, 178–201.
- Cserr, H.F.; DePasquale, M.; Patlak, C.S. Regulation of brain water and electrolytes during acute hyperosmolality in rats. Am. J. Physiol. 1987, 253, F522–F529.
- Fraser, P.A.; Dallas, A.D. Permeability of disrupted cerebral microvessels in the frog. J. Physiol. 1993, 461, 619–663.
- Bridges, L.R.; Andoh, J.; Lawrence, A.; Khoong, C.H.; Poon, W.W.; Esiri, M.M.; Markus, H.S.; Hainsworth, A.H. Blood-Brain Barrier Dysfunction and Cerebral Small Vessel Disease (Arteriolosclerosis) in Brains of Older People. J. Neuropathol. Exp. Neurol. 2014, 73, 1026–1033.
- Seo, J.H.; Miyamoto, N.; Hayakawa, K.; Pham, L.-D.D.; Maki, T.; Ayata, C.; Kim, K.-W.; Lo, E.H.; Arai, K. Oligodendrocyte precursors induce early blood-brain barrier opening after white matter injury. J. Clin. Investig. 2013, 123, 782–786.
- Wardlaw, J.M.; Sandercock, P.A.G.; Dennis, M.S.; Starr, J. Ithe s breakdown of the blood-brain barrier responsible for lacunar stroke, leukoaraiosis, and dementia? Stroke 2003, 34, 806–812.
- Wardlaw, J.M.; Doubal, F.N.; Valdes-Hernandez, M.; Wang, X.; Chappell, F.M.; Shuler, K.; Armitage, P.A.; Carpenter, T.C.; Dennis, M.S. Blood–Brain Barrier Permeability and Long-Term Clinical and Imaging Outcomes in Cerebral Small Vessel Disease. Stroke 2013, 44, 525–527.
- Erdo, F.; Denes, L.; de Lange, E. Age-associated physiological and pathological changes at the blood-brain barrier: A review. J. Cereb. Blood Flow Metab. 2017, 37, 4–24.
- Farrall, A.J.; Wardlaw, J.M. Blood-brain barrier: Aging and microvascular disease–systematic review and meta-analysis. Neurobiol. Aging 2009, 30, 337–352.
- Li, Y.; Li, M.; Zhang, X.; Shi, Q.; Yang, S.; Fan, H.; Qin, W.; Yang, L.; Yuan, J.; Jiang, T.; et al. Higher blood-brain barrier permeability is associated with ha igher white matter hyperintensities burden. J. Neurol. 2017, 264, 1474–1481.
- Li, Y.; Li, M.; Zuo, L.; Shi, Q.; Qin, W.; Yang, L.; Jiang, T.; Hu, W. Compromised blood-brain barrier integrity is associated with the total magnetic resonance imaging burden of cerebral small vessel disease. Front. Neurol. 2018, 9, 221.
- Moretti, R.; Caruso, P. Small Vessel Disease-Related Dementia: An Invalid Neurovascular Coupling? Int. J. Mol. Sci. 2020, 21, 1095.
- Stringer, M.S.; Heye, A.K.; Armitage, P.A.; Chappell, F.; Hernández, M.D.C.V.; Makin, S.D.J.; Sakka, E.; Thrippleton, M.J.; Wardlaw, J.M. Tracer kinetic assessment of blood-brain barrier leakage and blood volume in cerebral small vessel disease: Associations with disease burden and vascular risk factors. NeuroImage 2021, 32, 102883.
- Thrippleton, M.J.; Backes, W.H.; Sourbron, S.; Ingrisch, M.; Osch, M.J.P.; Dichgans, M.; Fazekas, F.; Ropele, S.; Frayne, R.; Oostenbrugge, R.J.; et al. Quantifying blood-brain barrier leakage in small vessel disease: Review and consensus recommendations. Alzheimer’s Dement. 2019, 15, 840–858.
- Manning, C.; Stringer, M.; Dickie, B.; Clancy, U.; Hernandez, M.C.V.; Wiseman, S.J.; Garcia, D.J.; Sakka, E.; Backes, W.H.; Ingrisch, M.; et al. Sources of systematic error in DCE-MRI estimation of low-level blood-brain barrier leakage. Magn. Reason. Med. 2021, 86, 1888–1903.
- Stewart, C.R.; Stringer, M.S.; Shi, Y.; Thrippleton, M.J.; Wardlaw, J.M. Associations Between White Matter Hyperintensity Burden, Cerebral Blood Flow and Transit Time in Small Vessel Disease: An Updated Meta-Analysis. Front. Neurol. 2021, 12, 647848.
- Heye, A.K.; Thrippleton, M.J.; Armitage, P.A.; Hernandez, M.D.C.V.; Makin, S.D.; Glatz, A.; Sakka, E.; Wardlaw, J.M. Tracer kinetic modelling for DCE-MRI quantification of subtle blood-brain barrier permeability. Neuroimage 2016, 125, 446–455.
- Mäe, M.A.; He, L.; Nordling, S.; Vazquez-Liebanas, E.; Nahar, K.; Jung, B.; Li, X.; Tan, B.C.; Chin Foo, J.; Cazenave-Gassiot, A.; et al. Single-Cell Analysis of Blood-Brain Barrier Response to Pericyte Loss. Circ. Res. 2021, 128, e46–e62.
- Zhang, E.T.; Inman, C.B.; Weller, R.O. Interrelationships of the pia mater and the perivascular (Wirchov- Robin) spaces in the human cerebrum. J. Anat. 1990, 170, 111–123.
- Iadecola, C. The neurovascular Unit coming of age: A journey through neurovascular coupling in health and disease. Neuron 2017, 96, 17–42.
- Hendrikx, D.; Smits, A.; Lavanga, M.; De Wel, O.; Thewissen, L.; Jansen, K.; Caicedo, A.; Van Huffe, S.; Naulaers, G. Measurement of Neurovascular Coupling in Neonates. Front. Physiol. 2019, 10, 65.
- Sweeney, M.D.; Sagare, A.P.; Zlokovic, B.V. Blood-brain barrier breakdown in Alzheimer disease and other neurodegenerative disorders. Nat. Rev. Neurol. 2018, 14, 133–150.
- Abbott, N.J.; Pizzo, M.E.; Preston, J.E.; Janigro, D.; Thorne, R.G. The role of brain barriers in fluid movement in the CNS: Is there a ‘glymphatic’ system? Acta Neuropathol. 2018, 135, 387–407.
- Huijts, M.; Duits, A.; Staals, J.; Kroon, A.A.; De Leeuw, P.W.; Van Oostenbrugge, R.J. Basal ganglia enlarged perivascular spaces are linked to cognitive function in patients with cerebral small vessel disease. Curr. Neurovasc. Res. 2014, 11, 136–141.
- Dalkara, T.; Alarcon-Martinez, L. Cerebral micro-vascular signaling in health and disease. Brain Res. 2015, 1623, 3–17.
- Wardlaw, J.M.; Benveniste, H.; Nedergaard, M.; Zlokovic, B.V.; Mestre, H.; Lee, H.; Doubal, F.N.; Brown, R.; Ramirez, J.; MacIntosh, B.J.; et al. Perivascular spaces in the brain: Anatomy, physiology and pathology. Nat. Rev. Neurol. 2020, 16, 137–153.
- Zhang, W.; Zhou, Y.; Wang, J.; Gong, X.; Chen, Z.; Zhang, X.; Cai, J.; Chen, S.; Fang, L.; Sun, J.; et al. Glymphatic clearance function in patients with cerebral small vessel disease. Neuroimage 2021, 238, 118257.
- Benveniste, H.; Nedergaard, M. Cerebral small vessel disease: A glymphopathy? Curr. Opin. Neurobiol. 2022, 72, 15–21.
- Erkinjunnti, T.; Gauthier, S. Diagnosing vascular cognitive impairment and dementia. In Concepts and Controversies in Vascular Cognitive Impairment in Clinical Practice; Wahlund, L.O., Erkinjunnti, T., Gauthier, S., Eds.; Cambridge University Press: Cambridge, UK, 2009; pp. 3–9.
- Jiménez-Balado, J.; Riba-Llena, I.; Garde, E.; Valor, M.; Gutiérrez, B.; Pujadas, F.; Delgado, P. Prevalence of hippocampal enlarged perivascular spaces in a sample of patients with hypertension and their relation with vascular risk factors and cognitive function. J. Neurol. Neurosurg. Psychiatry 2018, 89, 651–656.
- Giannakopoulos, P.; Gold, G.; Kowaru, E.; von Gunten, A.; Imhof, A.; Bouras, C. Assessing the cognitive impact of Alzheimer disease pathology and vascular burden in the aging brain: The Geneva experience. Acta Neuropathol. 2007, 113, 1–12.
- Van der Veen, P.H.; Muller, M.; Vinken, K.L.; Hendrikse, J.; Mali, W.P.; van der Graaf, Y.; Geerlings, M.I.; SMART Study Group. Longitudinal relationship between cerebral small vessel disease and cerebral blood flow. The second manifestations of arterial disease-magnetic resonance study. Stroke 2015, 46, 1233–1238.
- Cuadrado-Godia, E.; Dwivedi, P.; Sharma, S.; Santiago, A.O.; Roquer Gonzalez, J.; Balcells, M.; Laird, J.; Turk, M.; Suri, H.S.; Nicolaides, A.; et al. Cerebral Small Vessel Disease: A Review Focusing on Pathophysiology, Biomarkers, and Machine Learning Strategies. J. Stroke 2018, 20, 302–320.
- Zhang, C.E.; Wong, S.M.; van de Haar, H.J.; Staals, J.; Jansen, J.F.; Jeukens, C.R.; Hofman, P.A.; van Oostenbrugge, R.J.; Backes, W.H. Blood-brain barrier leakage is more widespread in patients with cerebral small vessel disease. Neurology 2017, 88, 426–432.
- Huisa, B.N.; Caprihan, A.; Thompson, J.; Prestopnik, J.; Qualls, C.R.; Rosenberg, G.A. Long-term blood-brain barrier permeability changes in Binswanger disease. Stroke 2015, 46, 2413–2418.
- Wardlaw, J.M.; Makin, S.J.; Hernández, M.C.V.; Armitage, P.A.; Heye, A.K.; Chappell, F.M.; Muñoz-Maniega, S.; Sakka, E.; Shuler, K.; Dennis, M.S.; et al. Blood-brain barrier failure as a core mechanism in cerebral small vessel disease and dementia: Evidence from a cohort study. Alzheimer’s Dement. 2017, 13, 634–643.
- Ihara, M.; Yamamoto, Y. Emerging evidence for pathogenesis of sporadic cerebral small vessel disease. Stroke 2016, 47, 554–560.
- Rajani, R.M.; Williams, A. Endothelial cell-oligodendrocyte interactions in small vessel disease and aging. Clin. Sci. 2017, 131, 369–379.
- Furukawa, S.; Sameshima, H.; Yang, L.; Hariskuma, M.; Ikenoue, T. Regional differences of microglial accumulation within 72 hours of hypoxia-ischemia and the effect of acetylcholine receptor agonist on brain damage and microglial activation in newborn rats. Brain Res. 2014, 1562, 52–58.
- Petito, C.K. Transformation of postisichemic perineuronal glial cells. J. Cereb. Blood Flow Metabol. 1986, 6, 616–624.
- Petito, C.K.; Olarte, J.P.; Roberts, B.; Nowak, T.S.; Pulsinelli, W.A. Selective glial vulnerability following transient global ischemia in rat brain. J. Neuropathol. Exp. Neurol. 1998, 57, 231–238.
- Masuda, T.; Croom, D.; Hida, H.; Kirov, S.A. Capillary blood flow around microglial somata determines dynamics of microglial processes in ischemic conditions. Glia 2011, 59, 1744–1753.
- Ju, F.; Ran, Y.; Zhu, L.; Cheng, X.; Gao, H.; Xi, X.; Yang, Z.; Zhang, S. Increased BBB Permeability Enhances Activation of Microglia and Exacerbates Loss of Dendritic Spines after Transient Global Cerebral Ischemia. Front. Cell Neurosci. 2018, 12, 236.
- Zhang, S. Microglial activation after ischaemic stroke. Stroke Vasc. Neurol. 2019, 4, 71–74.
- Iadecola, C. The pathobiology of vascular dementia. Neuron 2013, 80, 844–866.
- Filous, A.S.; Silver, J. Targeting astrocytes in CNS injury and disease: A translational research approach. Prog. Neurobiol. 2016, 144, 173–187.
- Forsberg, K.M.E.; Zhang, Y.; Reiners, J.; Ander, M.; Niedermayer, A.; Fang, L.; Neugebauer, H.; Kassubek, J.; Katona, I.; Weis, J.; et al. Endothelial damage, vascular bagging and remodeling of the microvascular bed in human microangiopathy with deep white matter lesions. Acta Neuropathol. Commun. 2018, 6, 128.
- Szalay, G.; Martinecz, B.; Lénárt, N.; Környei, Z.; Orsolits, B.; Judák, L.; Császár, E.; Fekete, R.; West, B.L.; Katona, G.; et al. Microglia protect against brain injury and their selective elimination dysregulates neuronal network activity after stroke. Nat. Commun. 2016, 7, 11499.
- Zhao, S.-C.; Ma, L.-S.; Chu, Z.-H.; Xu, H.; Wu, W.-Q.; Liu, F. Regulation of microglial activation in stroke. Acta Pharmacol. Sin. 2017, 38, 445–458.
- Morrison, H.W.; Filosa, J.A. A quantitative spatiotemporal analysis of microglia morphology during ischemic stroke and reperfusion. J. Neuroinflamm. 2013, 10, 4.
- Perego, C.; Fumagalli, S.; De Simoni, M.G. Temporal pattern of expression and colocalization of microglia/macrophage phenotype markers following brain ischemic injury in mice. J. Neuroinflamm. 2011, 8, 174.
- Farkas, E.; Donka, G.; de Vous, R.A.I.; Mihaly, A.; Bari, F.; Luiten, P.G.M. Experimental cerebral hypoperfusion induces white matter injury and microglial activation in the rat brain. Acta Neuropathol. 2004, 108, 57–64.
- Paolini Paoletti, F.; Simoni, S.; Parnetti, L.; Gaetani, L. The Contribution of Small Vessel Disease to Neurodegeneration: Focus on Alzheimer’s Disease, Parkinson’s Disease and Multiple Sclerosis. Int. J. Mol. Sci. 2021, 22, 4958.
- Fredman, P.; Wallin, A.; Blennow, K.; Davidsson, P.; Gottfries, C.; Svennerholm, L. Sulfatide as a biochemical marker in cerebrospinal fluid of patients with vascular dementia. Acta Neurol. Scand. 1992, 85, 103–106.
- Tullberg, M.; Månsson, J.E.; Fredman, P.; Lekman, A.; Blennow, K.; Ekman, R.; Rosengren, L.E.; Tisell, M.; Wikkelso, C. CSF sulfatide distinguishes between normal pressure hydrocephalus and subcortical arteriosclerotic encephalopathy. J. Neurol. Neurosurg. Psychiatry 2000, 69, 74–81.
- Thibert, K.A.; Raymond, G.V.; Nascene, D.R.; Miller, W.P.; Tolar, J.; Orchard, P.J.; Lund, T.C. Cerebrospinal fluid matrix metalloproteinases are elevated in cerebral adrenoleukodystrophy and correlate with MRI severity and neurologic dysfunction. PLoS ONE 2012, 7, e50430.
- Jonsson, M.; Zetterberg, H.; Van Straaten, E.; Lind, K.; Syversen, S.; Edman, Å.; Blennow, K.; Rosengren, L.; Pantoni, L.; Inzitari, D.; et al. Cerebrospinal fluid biomarkers of white matter lesions—Cross-sectional results from the LADIS study. Eur. J. Neurol. 2010, 17, 377–382.
- Bjerke, M.; Andreasson, U.; Rolstad, S.; Nordlund, A.; Lind, K.; Zetterberg, H.; Edman, Å.; Blennow, K.; Wallin, A. Subcortical vascular dementia biomarker pattern in mild cognitive impairment. Dement. Geriatr. Cogn. Disord. 2009, 28, 348–356.
- Öhrfelt, A.; Andreasson, U.; Simon, A.; Zetterberg, H.; Edman, Å.; Potter, W.; Holder, D.; Devanarayan, V.; Seeburger, J.; Smith, A.D.; et al. Screening for New Biomarkers for Subcortical Vascular Dementia and Alzheimer’s Disease. Dement. Geriatr. Cogn. Dis. Extra 2011, 1, 31–42.
- Peters, A.; Sethares, C. Age-related changes in the morphology of cerebral capillaries do not correlate with cognitive decline. J. Comp. Neurol. 2012, 520, 1339–1347.
- Sies, H.; Jones, D.P. Reactive oxygen species (ROS) as pleiotropic physiological signalling agents. Nat. Rev. Mol. Cell Biol. 2020, 21, 363–383.
- Fernando, M.S.; Simpson, J.E.; Matthews, F.; Brayne, C.; Lewis, C.E.; Barber, R.; Kalaria, R.N.; Forster, G.; Esteves, F.; Wharton, S.B.; et al. White matter lesions in an unselected cohort of the elderly: Molecular pathology suggests origin from chronic hypoperfusion injury. Stroke 2006, 37, 1391–1398.
- Egea, J.; Fabregat, I.; Frapart, Y.M.; Ghezzi, P.; Görlach, A.; Kietzmann, T.; Kubaichuk, K.; Knaus, U.G.; Lopez, M.G.; Olaso-Gonzalez, G.; et al. European contribution to the study of ROS: A summary of the findings and prospects for the future from the COST action BM1203 (EU-ROS). Redox Biol. 2017, 13, 94–162, Erratum in Redox Biol. 2018, 14, 694–696.
- Dikalov, S.I.; Dikalova, A.E.; Bikineyeva, A.T.; Schmidt, H.H.; Harrison, D.G.; Griendling, K.K. Distinct roles of Nox1 and Nox4 in basal and angiotensin II-stimulated superoxide and hydrogen peroxide production. Free Radic. Biol. Med. 2008, 45, 1340–1351.
- Zhang, X.; Wu, B.; Nie, K.; Jia, Y.; Yu, J. Effects of acupuncture on declined cerebral blood flow, impaired mitochondrial respiratory function and oxidative stress in multi-infarct dementia rats. Neurochem. Int. 2014, 65, 23–29.
- Huang, J.L.; Fu, S.T.; Jiang, Y.Y.; Cao, Y.B.; Guo, M.L.; Wang, Y.; Xu, Z. Protective effects of Nicotiflorin on reducing memory dysfunction, energy metabolism failure, and oxidative stress in multi-infarct dementia model rats. Pharmacol. Biochem. Behav. 2007, 86, 741–748.
- Takac, I.; Schröder, K.; Brandes, R.P. The Nox family of NADPH oxidases: Friend or foe of the vascular system? Curr. Hypertens. Rep. 2012, 14, 70–78.
- Miller, A.A.; Drummond, G.R.; DeSilva, T.M.; Mast, A.E.; Hickey, H.; Williams, J.P.; Broughton, B.R.; Sobey, C.G. NADPH oxidase activity is higher in cerebral versus systemic arteries of four animal species: Role of Nox2. Am. J. Physiol. Heart Circ. Physiol. 2009, 296, H220–H225.
- Liu, H.; Zhang, J. Cerebral hypoperfusion and cognitive impairment: The pathogenic role of vascular oxidative stress. Int. J. Neurosci. 2012, 122, 494–499.
- Paravicini, T.M.; Miller, A.A.; Drummond, G.R.; Sobey, C.G. Flow-induced cerebral vasodilatation in vivo involves activation of phosphatidylinositol-3kinase, NADPH-oxidase, and nitric oxide synthase. J. Cereb. Blood Flow Metab. 2006, 26, 836–845.
- De Silva, T.M.; Brait, V.H.; Drummond, G.R.; Sobey, C.G.; Miller, A.A. Nox2 oxidase activity accounts for the oxidative stress and vasomotor dysfunction in mouse cerebral arteries following ischemic stroke. PLoS ONE 2011, 6, e28393.
- Lin, M.T.; Beal, M.F. Mitochondrial dysfunction and oxidative stress in neurodegenerative diseases. Nature 2006, 443, 787–795.
- Raz, N. The aging brain: Structural changes and their implications for cognitive aging. In New Frontiers in Cognitive Aging; Dixon, R., Bäckman, L., Nilsson, L., Eds.; Oxford University Press: Telangana, India, 2004; pp. 115–134.
- Sun, N.; Youle, R.J.; Finkel, T. The mitochondrial basis of aging. Mol. Cell 2016, 61, 654–666.
- Valiente-Pallejà, A.; Tortajada, J.; Bulduk, B.K.; Vilella, E.; Garrabou, G.; Muntané, G.; Martorell, L. Comprehensive summary of mitochondrial DNA alterations in the postmortem human brain: A systematic review. EBioMedicine 2022, 76, 103815.
- Corral-Debrinski, M.; Horton, T.; Lott, M.T.; Shoffner, J.M.; Beal, M.F.; Wallace, D.C. Mitochondrial DNA deletions in human brain: Regional variability and increase with advanced age. Nat. Genet. 1992, 2, 324–329.
- Taylor, S.D.; Ericson, N.G.; Burton, J.N.; Prolla, T.A.; Silber, J.R.; Shendure, J.; Bielas, J.H. Targeted enrichment and high-resolution digital profiling of mitochondrial DNA deletions in human brain. Aging Cell 2014, 13, 29–38.
- Kennedy, S.R.; Salk, J.J.; Schmitt, M.W.; Loeb, L.A. Ultra-sensitive sequencing reveals an age-related increase in somatic mitochondrial mutations that are inconsistent with oxidative damage. PLoS Genet. 2013, 9, 1003794.
- Diaz, F.; Bayona-Bafaluy, M.P.; Rana, M.; Mora, M.; Hao, H.; Moraes, C.T. Human mitochondrial DNA with large deletions repopulates organelles faster than full-length genomes under relaxed copy number control. Nucleic Acids Res. 2002, 30, 4626–4633.
- Guo, L.; Tian, J.; Du, H. Mitochondrial dysfunction and synaptic transmission failure in Alzheimer’s disease. J. Alzheimers Dis. 2017, 57, 1071–1086.
- Ballif, B.C.; Theisen, A.; Coppinger, J.; Gowans, G.C.; Hersh, J.H.; Madan-Khetarpal, S.; Schmidt, K.R.; Tervo, R.; Escobar, L.F.; Friedrich, C.A.; et al. Expanding the clinical phenotype of the 3q29 microdeletion syndrome and characterization of the reciprocal microduplication. Mol. Cytogenet. 2008, 1, 8.
- Lanza, G.; Cantone, M.; Musso, S.; Borgione, E.; Scuderi, C.; Ferri, R. Early-onset subcortical ischemic vascular dementia in an adult with mtDNA mutation 3316G>A. J. Neurol. 2018, 265, 968–969.
- Campbell, G.R.; Ziabreva, I.; Reeve, A.K.; Krishnan, K.J.; Reynolds, R.; Howell, O.; Lassmann, H.; Turnbull, D.M.; Mahad, D.J. Mitochondrial DNA deletions and neurodegeneration in multiple sclerosis. Ann. Neurol. 2011, 69, 481–492.
- Chinnery, P.F. Mitochondrial disorders overview. In GeneReviews ; Margaret, P.A., Ardinger, H.H., Pagon, R.A., Wallace, S.E., Bean, L.J.H., Karen, S.A.A., Eds.; University of Washington, Seattle: Seattle, WA, USA, 2020; p. 1993.
- Basel, D. Mitochondrial DNA Depletion Syndromes. Clin. Perinatol. 2020, 47, 123–141.
- Coskun, P.E.; Wyrembak, J.; Derbereva, O.; Melkonian, G.; Doran, E.; Lott, I.T.; Head, E.; Cotman, C.W.; Wallace, D.C. Systemic mitochondrial dysfunction and the etiology of Alzheimer’s disease and down syndrome dementia. J. Alzheimers Dis. 2010, 20, 293–310.
- Roca-Bayerri, C.; Robertson, F.; Pyle, A.; Hudson, G.; Payne, B.A.I. Mitochondrial DNA damage and brain aging in human immunodeficiency virus. Clin. Infect. Dis. 2021, 73, e466–e473.
- Touyz, R.M.; Briones, A.M. Reactive oxygen species and vascular biology: Implications in human hypertension. Hypertens. Res. 2011, 34, 5–14.
- Mayhan, W.G.; Arrick, D.M.; Sharpe, G.M.; Sun, H. Age-related alterations in reactivity of cerebral arterioles: Role of oxidative stress. Microcirculation 2008, 15, 225–236.
- Dong, Y.F.; Kataoka, K.; Toyama, K.; Sueta, D.; Koibuchi, N.; Yamamoto, E.; Yata, K.; Tomimoto, H.; Ogawa, H.; Kim-Mitsuyama, S. Attenuation of brain damage and cognitive impairment by direct renin inhibition in mice with chronic cerebral hypoperfusion. Hypertension 2011, 58, 635–642.
- Santhanam, A.V.; d’Uscio, L.V.; Katusic, Z.S. Erythropoietin increases bioavailability of tetrahydrobiopterin and protects cerebral microvasculature against oxidative stress induced by eNOS uncoupling. J. Neurochem. 2014, 131, 521–529.
- Xie, H.; Ray, P.E.; Short, B.L. NF-kappa B activation plays a role in superoxide-mediated cerebral dysfunction after hypoxia/reoxygenation. Stroke 2005, 36, 1047–1052.
- Aghajanian, A.; Wittchen, E.S.; Campbell, S.L.; Burridge, K. Direct activation of RhoA by reactive oxygen species requires a redox-sensitive motif. PLoS ONE 2009, 4, e8045.
- Faraco, G.; Moraga, A.; Moore, J.; Anrather, J.; Pickel, V.M.; Iadecola, C. Circulating endothelin-1 alters critical mechanisms regulating cerebral microcirculation. Hypertension 2013, 62, 759–766.
- Bochkov, V.N.; Philippova, M.; Oskolkova, O.; Kadl, A.; Furnkranz, A.; Karabeg, E.; Afonyushkin, T.; Gruber, F.; Breuss, J.; Minchenko, A.; et al. Oxidized phospholipids stimulate angiogenesis via autocrine mechanisms, implicating a novel role for lipid oxidation in the evolution of atherosclerotic lesions. Circ. Res. 2006, 99, 900–908.
- Tai, L.M.; Thomas, R.; Marottoli, F.M.; Koster, K.P.; Kanekiyo, T.; Morris, A.W.; Bu, G. The role of APOE in cerebrovascular dysfunction. Acta Neuropathol. 2016, 131, 709–723.
- Han, B.H.; Zhou, M.L.; Johnson, A.W.; Singh, I.; Liao, F.; Vellimana, A.K.; Nelson, J.W.; Milner, E.; Cirrito, J.R.; Basak, J.; et al. Contribution of reactive oxygen species to cerebral amyloid angiopathy, vasomotor dysfunction, and micro-hemorrhage in aged Tg2576 mice. Proc. Natl. Acad. Sci. USA 2015, 112, E881–E890.
- Grochowski, C.; Litak, J.; Kamieniak, P.; Maciejewski, R. Oxidative stress in cerebral small vessel disease. Role of reactive species. Free Radic. Res. 2018, 52, 1–13.
More
Information
Subjects:
Neurosciences
Contributors
MDPI registered users' name will be linked to their SciProfiles pages. To register with us, please refer to https://encyclopedia.pub/register
:
View Times:
816
Revisions:
3 times
(View History)
Update Date:
31 Mar 2022
Notice
You are not a member of the advisory board for this topic. If you want to update advisory board member profile, please contact office@encyclopedia.pub.
OK
Confirm
Only members of the Encyclopedia advisory board for this topic are allowed to note entries. Would you like to become an advisory board member of the Encyclopedia?
Yes
No
${ textCharacter }/${ maxCharacter }
Submit
Cancel
Back
Comments
${ item }
|
More
No more~
There is no comment~
${ textCharacter }/${ maxCharacter }
Submit
Cancel
${ selectedItem.replyTextCharacter }/${ selectedItem.replyMaxCharacter }
Submit
Cancel
Confirm
Are you sure to Delete?
Yes
No





