
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Christian Jannete Stevens-Hernandez | + 1263 word(s) | 1263 | 2022-03-16 07:28:10 | | | |
| 2 | Nora Tang | -76 word(s) | 1187 | 2022-03-29 08:51:12 | | |
Video Upload Options
Reticulocyte maturation begins after enucleation in the bone marrow. In rats, reticulocytes reside in the bone marrow from 6.5–17 hrs depending on the blood demand. These reticulocytes are termed as R1 and are characterized for their multi-lobular shape and their motility. The final stages of maturation occur during circulation where macrophages residing in the spleen may facilitate the process. These reticulocytes in circulation are termed as R2, are non-motile and have a “deep-dish” shape”. As part of their maturation, reticulocytes need to remove or degrade residual organelles and RNA. In addition, the reticulocyte must reduce its surface area and volume. On average, labelled baboon reticulocytes showed a reduction of 20% of their surface area and 15% of their volume after the first 24 h in circulation; at this point they showed a similar size distribution to that of mature RBC.
1. Protein Removal through Exosome Release
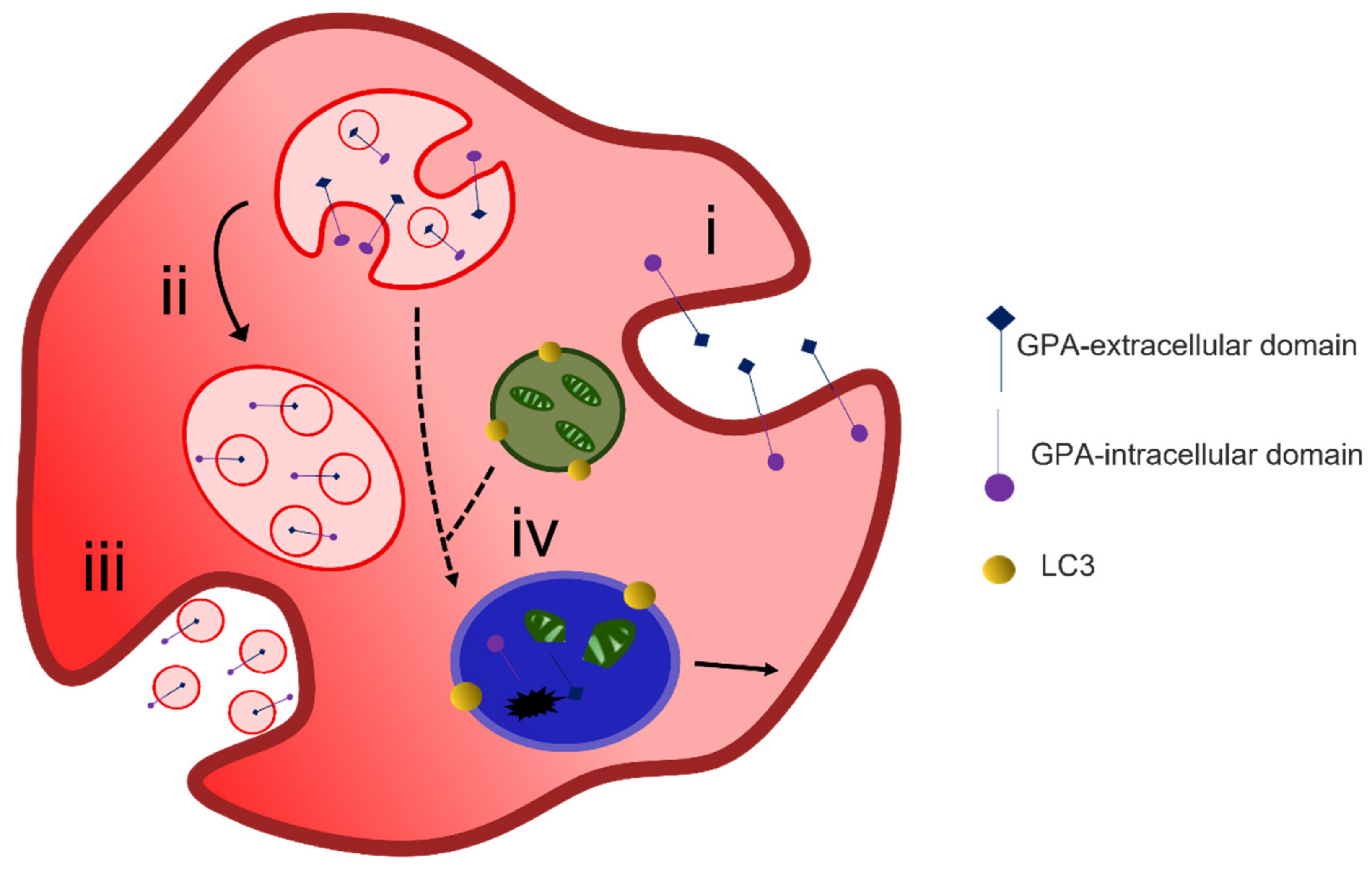
2. Alternative Methods of Reticulocyte Maturation
3. Organelle Clearance
References
- Johnstone, R.M.; Bianchini, A.; Teng, K. Reticulocyte maturation and exosome release: Transferrin receptor containing exosomes shows multiple plasma membrane functions. Blood 1989, 74, 1844–1851.
- Pan, B.-T.; Johnstone, R.M. Fate of the transferrin receptor during maturation of sheep reticulocytes in vitro: Selective externalization of the receptor. Cell 1983, 33, 967–978.
- Johnstone, R.M.; Adam, M.; Hammond, J.R.; Orr, L.; Turbide, C. Vesicle formation during reticulocyte maturation. Association of plasma membrane activities with released vesicles (exosomes). J. Biol. Chem. 1987, 262, 9412–9420.
- Díaz-Varela, M.; Menezes-Neto, A.; Perez-Zsolt, D.; Gámez-Valero, A.; Barber, J.S.; Izquierdo-Useros, N.; Martinez-Picado, J.; Fernández-Becerra, C.; Del Portillo, H.A. Proteomics study of human cord blood reticulocyte-derived exosomes. Sci. Rep. 2018, 8, 14046.
- Carayon, K.; Chaoui, K.; Ronzier, E.; Lazar, I.; Bertrand-Michel, J.; Roques, V.; Balor, S.; Terce, F.; Lopez, A.; Salomé, L.; et al. Proteolipidic Composition of Exosomes Changes during Reticulocyte Maturation. J. Biol. Chem. 2011, 286, 34426–34439.
- Géminard, C.; de Gassart, A.; Blanc, L.; Vidal, M. Degradation of AP2 During Reticulocyte Maturation Enhances Binding of Hsc70 and Alix to a Common Site on TfR for Sorting into Exosomes. Traffic 2004, 5, 181–193.
- Trajkovic, K.; Hsu, C.; Chiantia, S.; Rajendran, L.; Wenzel, D.; Wieland, F.; Schwille, P.; Brügger, B.; Simons, M. Ceramide Triggers Budding of Exosome Vesicles into Multivesicular Endosomes. Science 2008, 319, 1244–1247.
- Vidal, M.; Mangeat, P.; Hoekstra, D. Aggregation reroutes molecules from a recycling to a vesicle-mediated secretion pathway during reticulocyte maturation. J. Cell Sci. 1997, 110, 1867–1877.
- Barrès, C.; Blanc, L.; Bette-Bobillo, P.; André, S.; Mamoun, R.; Gabius, H.-J.; Vidal, M. Galectin-5 is bound onto the surface of rat reticulocyte exosomes and modulates vesicle uptake by macrophages. Blood 2010, 115, 696–705.
- Griffiths, R.E.; Kupzig, S.; Cogan, N.; Mankelow, T.J.; Betin, V.M.S.; Trakarnsanga, K.; Massey, E.J.; Lane, J.D.; Parsons, S.F.; Anstee, D.J. Maturing reticulocytes internalize plasma membrane in glycophorin A—Containing vesicles that fuse with autophagosomes before exocytosis. Blood 2012, 119, 6296–6306.
- Mankelow, T.; Griffiths, R.E.; Trompeter, S.; Flatt, J.F.; Cogan, N.M.; Massey, E.J.; Anstee, D.J. The ins and outs of reticulocyte maturation revisited: The role of autophagy in sickle cell disease. Autophagy 2016, 12, 590–591.
- Griffiths, R.E.; Kupzig, S.; Cogan, N.; Mankelow, T.J.; Betin, V.M.; Trakarnsanga, K.; Massey, E.J.; Parsons, S.F.; Anstee, D.J.; Lane, J.D. The ins and outs of human reticulocyte maturation. Autophagy 2012, 8, 1150–1151.
- Moura, P.; Hawley, B.R.; Mankelow, T.; Griffiths, R.E.; Dobbe, J.; Streekstra, G.J.; Anstee, D.J.; Satchwell, T.J.; Toye, A. Non-muscle myosin II drives vesicle loss during human reticulocyte maturation. Haematologica 2018, 103, 1997–2007.
- Minetti, G.; Bernecker, C.; Dorn, I.; Achilli, C.; Bernuzzi, S.; Perotti, C.; Ciana, A. Membrane Rearrangements in the Maturation of Circulating Human Reticulocytes. Front. Physiol. 2020, 11, 215.
- De Gassart, A.; Géminard, C.; Février, B.; Raposo, G.; Vidal, M. Lipid raft-associated protein sorting in exosomes. Blood 2003, 102, 4336–4344.
- Zhang, J.; Wu, K.; Xiao, X.; Liao, J.; Hu, Q.; Chen, H.; Liu, J.; An, X. Autophagy as a Regulatory Component of Erythropoiesis. Int. J. Mol. Sci. 2015, 16, 4083–4094.
- Honda, S.; Arakawa, S.; Nishida, Y.; Yamaguchi, H.; Ishii, E.; Shimizu, S. Ulk1-mediated Atg5-independent macroautophagy mediates elimination of mitochondria from embryonic reticulocytes. Nat. Commun. 2014, 5, 4004.
- Hammerling, B.C.; Najor, R.H.; Cortez, M.Q.; Shires, S.E.; Leon, L.J.; Gonzalez, E.R.; Boassa, D.; Phan, S.; Thor, A.; Jimenez, R.E.; et al. A Rab5 endosomal pathway mediates Parkin-dependent mitochondrial clearance. Nat. Commun. 2017, 8, 14050.
- Grosso, R.; Fader, C.M.; Colombo, M.I. Autophagy: A necessary event during erythropoiesis. Blood Rev. 2017, 31, 300–305.
- Fader, C.; Colombo, M. Multivesicular Bodies and Autophagy in Erythrocyte Maturation. Autophagy 2006, 2, 122–125.
- Betin, V.M.; Singleton, B.K.; Parsons, S.F.; Anstee, D.J.; Lane, J.D. Autophagy facilitates organelle clearance during differentiation of human erythroblasts: Evidence for a role for ATG4 paralogs during autophagosome maturation. Autophagy 2013, 9, 881–893.
- Dardalhon, V.; Géminard, C.; Reggio, H.; Vidal, M.; Sainte-Marie, J. Fractionation analysis of the endosomal compartment during rat reticulocyte maturation. Cell Biol. Int. 2002, 26, 669–678.





