
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Evgenia Kotsifa | + 2864 word(s) | 2864 | 2022-03-15 03:11:01 | | | |
| 2 | Peter Tang | -88 word(s) | 2776 | 2022-03-24 03:46:34 | | |
Video Upload Options
Hepatocellular carcinoma (HCC) is the most common primary liver malignancy. It is principally associated with liver cirrhosis and chronic liver disease. The major risk factors for the development of HCC include viral infections (HBV, HCV), alcoholic liver disease (ALD,) and non-alcoholic fatty liver disease (NAFLD). The optimal treatment choice is dictated by multiple variables such as tumor burden, liver function, and patient’s health status. Surgical resection, transplantation, ablation, transarterial chemoembolization (TACE), and systemic therapy are potentially useful treatment strategies. TACE is considered the first-line treatment for patients with intermediate stage HCC.
1. Introduction
2. Treatment Procedure
2.1. cTACE vs. DEB-TACE
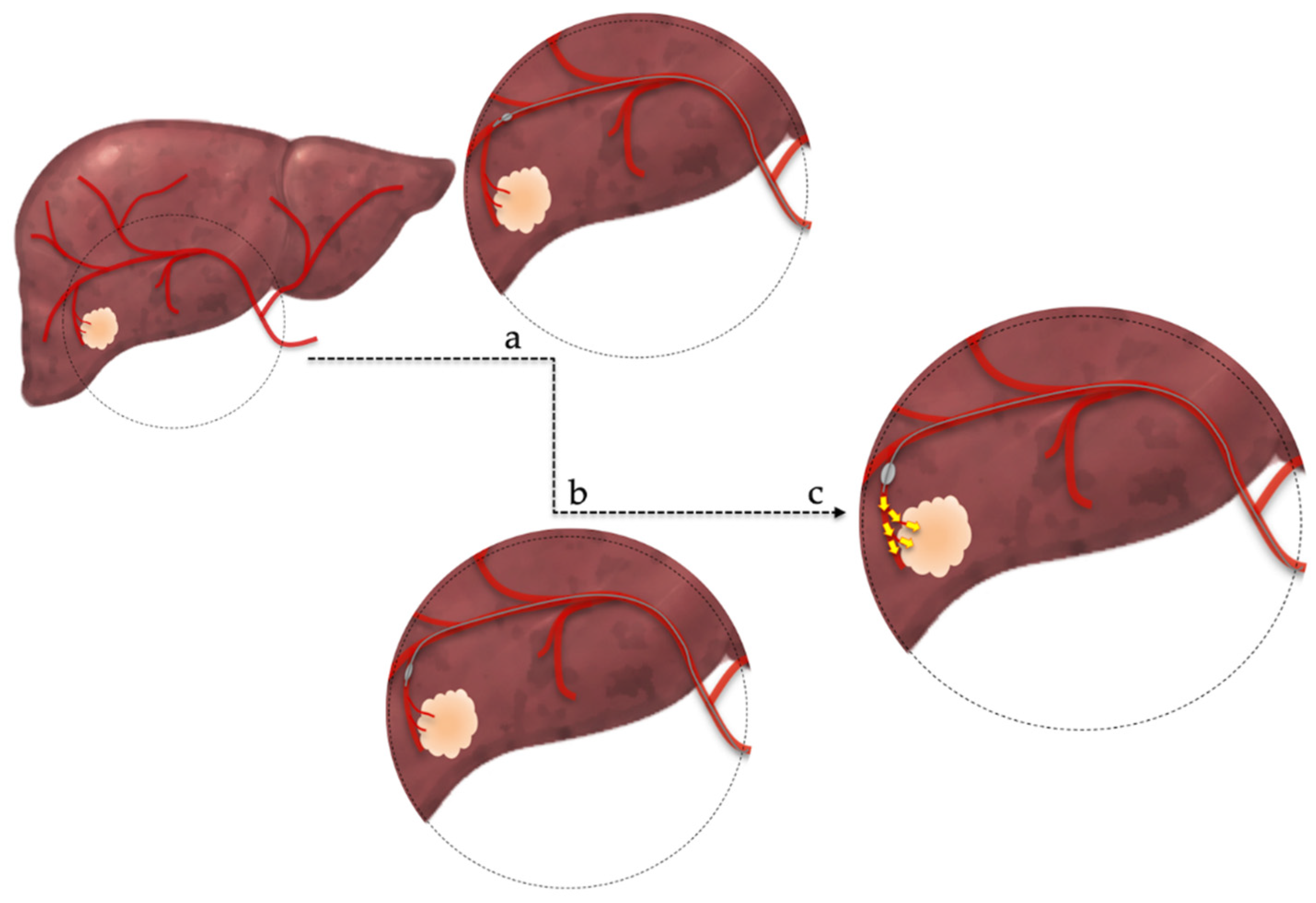
2.2. Patient Selection
- 2.3. Contraindications and Adverse Effects
3. Prognostic Scores
|
Score |
Parameters Used |
Prognostic Value |
Demerits |
|---|---|---|---|
|
HAP [33] |
Albumin, Bilirubin, AFP, Size of dominant tumor |
Prognosis of HCC patients undergoing TACE |
|
|
ART [34] |
Radiological response after the first TACE, increase of serum AST, increase of Child-Pugh score |
Differentiation of patients who would benefit from a second TACE |
Failed to predict overall survival in patients who received repeated TACE |
|
STATE [35] |
Albumin, CRP, Size of the largest tumor, Number of tumors |
Identification of patients unsuitable for first-time TACE |
|
|
ABCR [36] |
AFP, BCLC, Child-Pugh increase, Radiological response |
Differentiation of patients who would benefit from a second TACE |
Failed to show sufficient prognostic ability to guide the decision-making process regarding subsequent TACE |
|
CHIP [37] |
Child-Pugh, number of lesions, HCV-RNA positivity |
Stratification of patients within BCLC Stage B |
|
|
M-TACE [38] |
Bilirubin, INR, CRP, creatinine, AFP, tumor extension |
Identification of patients most likely to benefit from TACE |
|
|
Six & Twelve [39] |
Tumor size, tumor number |
Outcome prediction and risk stratification of recommended TACE candidates |
Only “ideal” TACE candidates included |
|
Pre-TACE & Post-TACE predict [40] |
Tumor size, tumor number, AFP, albumin, bilirubin, vascular invasion, cause, radiological response |
Prediction of survival among patients receiving TACE |
Calculator needed |
4. Combined Treatments
5. Conclusions
Even though TACE is currently considered the first-line treatment for patients with HCC in the intermediate stage, recent studies have showed that it can beneficial for patients beyond stage B. Moreover, since BCLC stage B represents a heterogeneous group, not all intermediate-stage HCC patients benefit the same from TACE. Therefore, treatment allocation should be decided by a tumor board of specialists and each HCC patient should receive personalized treatment according to his/her individual features. Unfortunately, in many countries, a tumor board is not available in every hospital and physicians should address virtual boards remotely via telemedicine (Figure 2). In conclusion, TACE is an established procedure with proven efficacy and known adverse effects and contraindications. Nevertheless, additional studies and clinical trials are warranted to redefine patient selection criteria, introduce new indications, and stratify patients according to their individual prognostic evaluation.
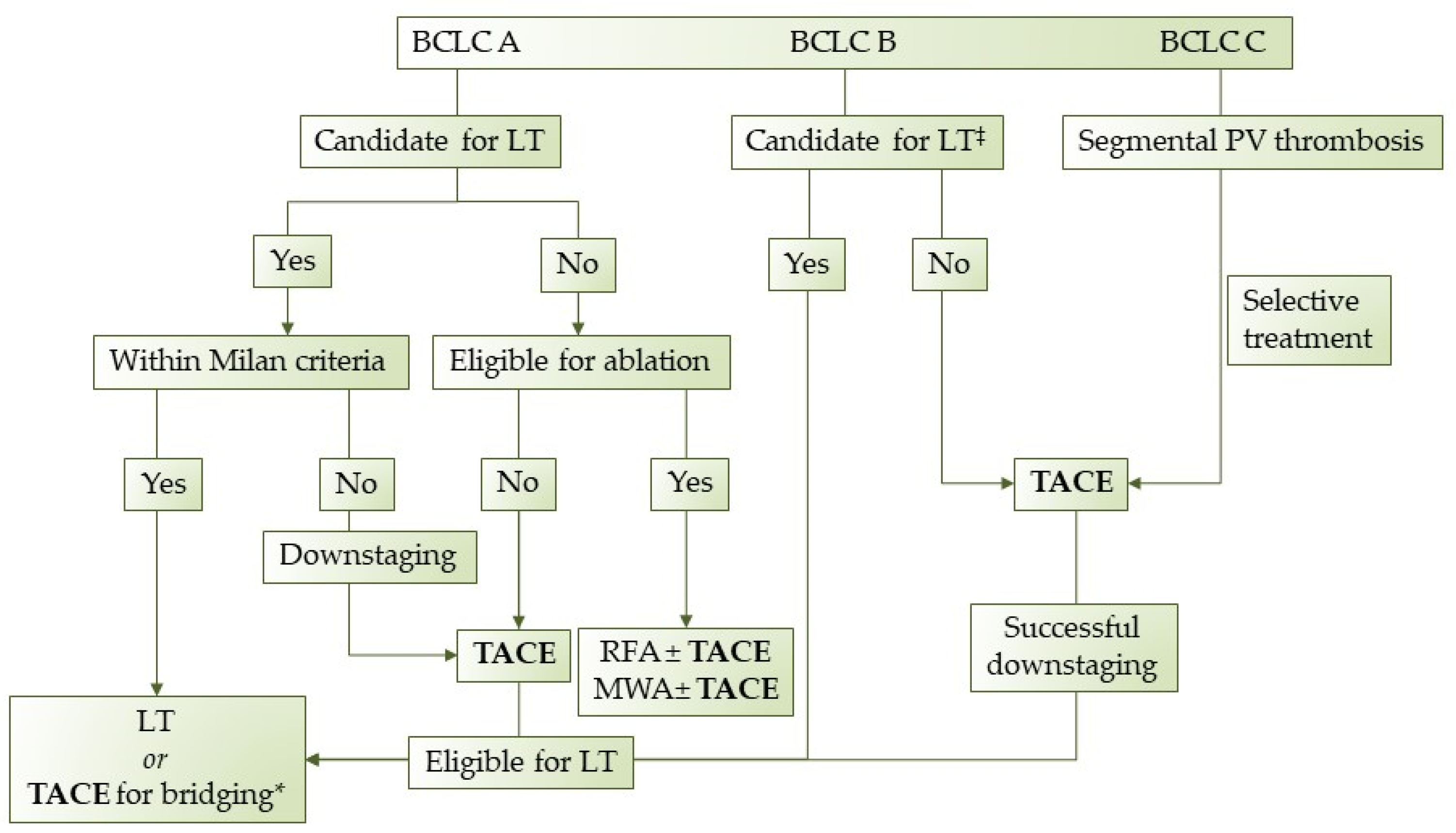
Figure 2. Proposed treatment algorithm regarding TACE in HCC patients. LT: Liver transplantation, TACE: transarterial chemoembolization, RFA: radiofrequency ablation, MWA: microwave ablation, *when in waiting list >6 months, ‡extended liver transplant criteria (size, AFP).
References
- Forner, A.; Reig, M.; Bruix, J. Hepatocellular carcinoma. Lancet 2018, 391, 1301–1314.
- Ioannou, G.N.; Green, P.; Lowy, E.; Mun, E.J.; Berry, K. Differences in hepatocellular carcinoma risk, predictors and trends over time according to etiology of cirrhosis. PLoS ONE 2018, 13, e0204412.
- Frager, S.Z.; Schwartz, J.M. Hepatocellular Carcinoma: Epidemiology, Screening, and Assessment of Hepatic Reserve. Curr. Oncol. 2020, 27, 138–143.
- Reig, M.; Forner, A.; Rimola, J.; Ferrer-Fábrega, J.; Burrel, M.; Garcia-Criado, A.; Kelley, R.K.; Galle, P.R.; Mazzaferro, V.; Salem, R.; et al. BCLC strategy for prognosis prediction and treatment recommendation Barcelona Clinic Liver Cancer (BCLC) staging system. The 2022 update. J. Hepatol. 2021, 76, 681–693.
- Llovet, J.M.; Brú, C.; Bruix, J. Prognosis of hepatocellular carcinoma: The BCLC staging classification. In Seminars in Liver Disease; Thieme Medical Publishers, Inc.: New York, NY, USA, 1999; Volume 19, pp. 329–338.
- Chang, Y.; Jeong, S.W.; Jang, J.Y.; Kim, Y.J. Recent Updates of TransarterialChemoembolilzation in Hepatocellular Carcinoma. Int. J. Mol. Sci. 2020, 21, 8165.
- Tsurusaki, M.; Murakami, T. Surgical and Locoregional Therapy of HCC: TACE. Liver Cancer 2015, 4, 165–175.
- Raoul, J.-L.; Forner, A.; Bolondi, L.; Cheung, T.T.; Kloeckner, R.; de Baere, T. Updated use of TACE for hepatocellular carcinoma treatment: How and when to use it based on clinical evidence. Cancer Treat. Rev. 2019, 72, 28–36.
- Sieghart, W.; Hucke, F.; Peck-Radosavljevic, M. Transarterial chemoembolization: Modalities, indication, and patient selection. J. Hepatol. 2015, 62, 1187–1195.
- Zou, J.H.; Zhang, L.; Ren, Z.G.; Ye, S.L. Efficacy and safety of cTACE versus DEB-TACE in patients with hepatocellular carcinoma: A meta-analysis. J. Dig. Dis. 2016, 17, 510–517.
- Chen, P.; Yuan, P.; Chen, B.; Sun, J.; Shen, H.; Qian, Y. Evaluation of drug-eluting beads versus conventional transcatheter arterial chemoembolization in patients with unresectable hepatocellular carcinoma: A systematic review and meta-analysis. Clin. Res. Hepatol. Gastroenterol. 2017, 41, 75–85.
- Han, T.; Yang, X.; Zhang, Y.; Li, G.; Liu, L.; Chen, T.; Zheng, Z. The clinical safety and efficacy of conventional transcatheter arterial chemoembolization and drug-eluting beads-transcatheter arterial chemoembolization for unresectable hepatocellular carcinoma: A meta-analysis. Biosci. Trends 2019, 13, 374–381.
- Facciorusso, A.; Di Maso, M.; Muscatiello, N. Drug-eluting beads versus conventional chemoembolization for the treatment of unresectable hepatocellular carcinoma: A meta-analysis. Dig. Liver Dis. 2016, 48, 571–577.
- Wang, H.; Cao, C.; Wei, X.; Shen, K.; Shu, Y.; Wan, X.; Sun, J.; Ren, X.; Dong, Y.; Liu, Y.; et al. A comparison between drug-eluting bead-transarterial chemoembolization and conventional transarterial chemoembolization in patients with hepatocellular carcinoma: A meta-analysis of six randomized controlled trials. J. Cancer Res. Ther. 2020, 16, 243–249.
- Irie, T.; Kuramochi, M.; Takahashi, N. Dense Accumulation of Lipiodol Emulsion in Hepatocellular Carcinoma Nodule during Selective Balloon-occluded Transarterial Chemoembolization: Measurement of Balloon-occluded Arterial Stump Pressure. Cardiovasc. Interv. Radiol. 2013, 36, 706–713.
- Golfieri, R.; Bezzi, M.; Verset, G.; Fucilli, F.; Mosconi, C.; Cappelli, A.; Paccapelo, A.; Lucatelli, P.; Magand, N.; Rode, A.; et al. Balloon-Occluded Transarterial Chemoembolization: In Which Size Range Does It Perform Best? A Comparison of Its Efficacy versus Conventional Transarterial Chemoembolization, Using Propensity Score Matching. Liver Cancer 2021, 10, 522–534.
- Hatanaka, T.; Arai, H.; Kakizaki, S. Balloon-occluded transcatheter arterial chemoembolization for hepatocellular carcinoma. World J. Hepatol. 2018, 10, 485–495.
- Lucatelli, P.; de Rubeis, G.; Rocco, B.; Basilico, F.; Cannavale, A.; Abbatecola, A.; Nardis, P.G.; Corona, M.; Brozzetti, S.; Catalano, C.; et al. Balloon occluded TACE (B-TACE) vs DEM-TACE for HCC: A single center retrospective case control study. BMC Gastroenterol. 2021, 21, 51.
- Raoul, J.-L.; Sangro, B.; Forner, A.; Mazzaferro, V.M.; Piscaglia, F.; Bolondi, L.; Lencioni, R. Evolving strategies for the management of intermediate-stage hepatocellular carcinoma: Available evidence and expert opinion on the use of transarterial chemoembolization. Cancer Treat. Rev. 2011, 37, 212–220.
- Mazzaferro, V.; Llovet, J.M.; Miceli, R.; Bhoori, S.; Schiavo, M.; Mariani, L.; Camerini, T.; Roayaie, S.; Schwartz, M.E.; Grazi, G.L.; et al. Predicting survival after liver transplantation in patients with hepatocellular carcinoma beyond the Milan criteria: A retrospective, exploratory analysis. Lancet Oncol. 2009, 10, 35–43.
- Kimura, H.; Ohkawa, K.; Miyazaki, M.; Sakakibara, M.; Imanaka, K.; Tamura, T.; Sueyoshi, H.; Takada, R.; Fukutake, N.; Uehara, H.; et al. Subclassification of patients with intermediate-stage (Barcelona Clinic Liver Cancer stage-B) hepatocellular carcinoma using the up-to-seven criteria and serum tumor markers. Hepatol. Int. 2017, 11, 105–114.
- Bargellini, I.; Sacco, R.; Bozzi, E.; Bertini, M.; Ginanni, B.; Romano, A.; Cicorelli, A.; Tumino, E.; Federici, G.; Cioni, R.; et al. Transarterial chemoembolization in very early and early-stage hepatocellular carcinoma patients excluded from curative treatment: A prospective cohort study. Eur. J. Radiol. 2012, 81, 1173–1178.
- Xue, T.-C.; Xie, X.-Y.; Zhang, L.; Yin, X.; Zhang, B.-H.; Ren, Z.-G. Transarterial chemoembolization for hepatocellular carcinoma with portal vein tumor thrombus: A meta-analysis. BMC Gastroenterol. 2013, 13, 60.
- Vogl, T.J.; Gruber-Rouh, T. HCC: Transarterial Therapies—What the Interventional Radiologist Can Offer. Dig. Dis. Sci. 2019, 64, 959–967.
- Lencioni, R.; de Baere, T.; Soulen, M.C.; Rilling, W.S.; Geschwind, J.F.H. Lipiodol transarterial chemoembolization for hepatocellular carcinoma: A systematic review of efficacy and safety data. Hepatology 2016, 64, 106–116.
- Mason, M.C.; Massarweh, N.N.; Salami, A.; Sultenfuss, M.A.; Anaya, D.A. Post-embolization syndrome as an early predictor of overall survival after transarterial chemoembolization for hepatocellular carcinoma. HPB 2015, 17, 1137–1144.
- Agrawal, R.; Majeed, M.; Aqeel, S.B.; Wang, Y.; Haque, Z.; Omar, Y.A.; Upadhyay, S.B.; Gast, T.; Attar, B.M.; Gandhi, S. Identifying predictors and evaluating the role of steroids in the prevention of post-embolization syndrome after transarterial chemoembolization and bland embolization. Ann. Gastroenterol. 2021, 34, 241–246.
- Ogasawara, S.; Chiba, T.; Ooka, Y.; Kanogawa, N.; Motoyama, T.; Suzuki, E.; Tawada, A.; Nagai, K.; Nakagawa, T.; Sugawara, T.; et al. A randomized placebo-controlled trial of prophylactic dexamethasone for transcatheter arterial chemoembolization. Hepatology 2018, 67, 575–585.
- Lu, H.; Zheng, C.; Liang, B.; Xiong, B. Efficacy and safety analysis of dexamethasone-lipiodol emulsion in prevention of post-embolization syndrome after TACE: A retrospective analysis. BMC Gastroenterol. 2021, 21, 256.
- Kudo, M.; Izumi, N.; Kokudo, N.; Matsui, O.; Sakamoto, M.; Nakashima, O.; Kojiro, M.; Makuuchi, M.; for the HCC Expert Panel of Japan Society of Hepatology. Management of Hepatocellular Carcinoma in Japan: Consensus-Based Clinical Practice Guidelines Proposed by the Japan Society of Hepatology (JSH) 2010 Updated Version. Dig. Dis. 2011, 29, 339–364.
- Cheng, A.-L.; Amarapurkar, D.; Chao, Y.; Chen, P.-J.; Geschwind, J.-F.; Goh, K.L.; Han, K.-H.; Kudo, M.; Lee, H.C.; Lee, R.-C.; et al. Re-evaluating transarterial chemoembolization for the treatment of Hepatocellular Carcinoma: Consensus recommendations and review by an International Expert Panel. Liver Int. 2014, 34, 174–183.
- Zhong, B.-Y.; Wang, W.-S.; Zhang, S.; Zhu, H.-D.; Zhang, L.; Shen, J.; Zhu, X.-L.; Teng, G.-J.; Ni, C.-F. Re-evaluating Transarterial Chemoembolization Failure/Refractoriness: A Survey by Chinese College of Interventionalists. J. Clin. Transl. Hepatol. 2021, 9, 521–527.
- Kadalayil, L.; Benini, R.; Pallan, L.; O’Beirne, J.; Marelli, L.; Yu, D.; Hackshaw, A.; Fox, R.; Johnson, P.; Burroughs, A.K.; et al. A simple prognostic scoring system for patients receiving transarterialembolisation for hepatocellular cancer. Ann. Oncol. 2013, 24, 2565–2570.
- Sieghart, W.; Hucke, F.; Pinter, M.; Graziadei, I.; Vogel, W.; Müller, C.; Heinzl, H.; Trauner, M.; Peck-Radosavljevic, M. The ART of decision making: Retreatment with transarterial chemoembolization in patients with hepatocellular carcinoma. Hepatology 2013, 57, 2261–2273.
- Hucke, F.; Pinter, M.; Graziadei, I.; Bota, S.; Vogel, W.; Müller, C.; Heinzl, H.; Waneck, F.; Trauner, M.; Peck-Radosavljevic, M.; et al. How to STATE suitability and START transarterial chemoembolization in patients with intermediate stage hepatocellular carcinoma. J. Hepatol. 2014, 61, 1287–1296.
- Adhoute, X.; Penaranda, G.; Naude, S.; Raoul, J.L.; Perrier, H.; Bayle, O.; Monnet, O.; Beaurain, P.; Bazin, C.; Pol, B.; et al. Retreatment with TACE: The ABCR SCORE, an aid to the decision-making process. J. Hepatol. 2015, 62, 855–862.
- Ogasawara, S.; Chiba, T.; Ooka, Y.; Kanogawa, N.; Motoyama, T.; Suzuki, E.; Tawada, A.; Azemoto, R.; Shinozaki, M.; Yoshikawa, M.; et al. A Prognostic Score for Patients with Intermediate-Stage Hepatocellular Carcinoma Treated with Transarterial Chemoembolization. PLoS ONE 2015, 10, e0125244.
- Op den Winkel, M.; Nagel, D.; Op den Winkel, P.; Trojan, J.; Paprottka, P.M.; Steib, C.J.; Schmidt, L.; Göller, M.; Stieber, P.; Göhring, P.; et al. Transarterial chemoembolization for hepatocellular carcinoma: Development and external validation of the Munich-TACE score. Eur. J. Gastroenterol. Hepatol. 2018, 30, 44–53.
- Wang, Q.; Xia, D.; Bai, W.; Wang, E.; Sun, J.; Huang, M.; Mu, W.; Yin, G.; Li, H.; Zhao, H.; et al. Development of a prognostic score for recommended TACE candidates with hepatocellular carcinoma: A multicentre observational study. J. Hepatol. 2019, 70, 893–903.
- Han, G.; Berhane, S.; Toyoda, H.; Bettinger, D.; Elshaarawy, O.; Chan, A.W.H.; Kirstein, M.; Mosconi, C.; Hucke, F.; Palmer, D.; et al. Prediction of Survival Among Patients Receiving Transarterial Chemoembolization for Hepatocellular Carcinoma: A Response-Based Approach. Hepatology 2020, 72, 198–212.
- European Association for the Study of the Liver. Electronic address eee, European Association for the Study of the L. EASL Clinical Practice Guidelines: Management of hepatocellular carcinoma. J. Hepatol. 2018, 69, 182–236.
- Gervais, D.A.; Arellano, R.S. Percutaneous Tumor Ablation for Hepatocellular Carcinoma. Am. J. Roentgenol. 2011, 197, 789–794.
- Xu, Z.; Xie, H.; Zhou, L.; Chen, X.; Zheng, S. The Combination Strategy of Transarterial Chemoembolization and Radiofrequency Ablation or Microwave Ablation against Hepatocellular Carcinoma. Anal. Cell. Pathol. 2019, 2019, 8619096.
- Pillai, K.; Akhter, J.; Chua, T.C.; Shehata, M.; Alzahrani, N.; Al-Alem, I.; Morris, D.L. Heat Sink Effect on Tumor Ablation Characteristics as Observed in Monopolar Radiofrequency, Bipolar Radiofrequency, and Microwave, Using Ex Vivo Calf Liver Model. Medicine 2015, 94, e580.
- Dhir, M.; Melin, A.A.; Douaiher, J.; Lin, C.; Zhen, W.; Hussain, S.M.; Geschwind, J.-F.H.; Doyle, M.B.M.; Abou-Alfa, G.K.; Are, C. A Review and Update of Treatment Options and Controversies in the Management of Hepatocellular Carcinoma. Ann. Surg. 2016, 263, 1112–1125.





