Your browser does not fully support modern features. Please upgrade for a smoother experience.

Submitted Successfully!
Thank you for your contribution! You can also upload a video entry or images related to this topic.
For video creation, please contact our Academic Video Service.
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Ioannis Ilias | + 2181 word(s) | 2181 | 2022-03-21 09:05:42 | | | |
| 2 | Lindsay Dong | Meta information modification | 2181 | 2022-03-22 03:01:03 | | |
Video Upload Options
We provide professional Academic Video Service to translate complex research into visually appealing presentations. Would you like to try it?
Cite
If you have any further questions, please contact Encyclopedia Editorial Office.
Ilias, I. Sex/Gender Differences in Uses and Effects of Metformin. Encyclopedia. Available online: https://encyclopedia.pub/entry/20809 (accessed on 09 May 2026).
Ilias I. Sex/Gender Differences in Uses and Effects of Metformin. Encyclopedia. Available at: https://encyclopedia.pub/entry/20809. Accessed May 09, 2026.
Ilias, Ioannis. "Sex/Gender Differences in Uses and Effects of Metformin" Encyclopedia, https://encyclopedia.pub/entry/20809 (accessed May 09, 2026).
Ilias, I. (2022, March 21). Sex/Gender Differences in Uses and Effects of Metformin. In Encyclopedia. https://encyclopedia.pub/entry/20809
Ilias, Ioannis. "Sex/Gender Differences in Uses and Effects of Metformin." Encyclopedia. Web. 21 March, 2022.
Copy Citation
Metformin (MTF) occupies a major and fundamental position in the therapeutic management of type 2 diabetes mellitus (T2DM). Gender differences in some effects and actions of MTF have been reported. Women are usually prescribed lower MTF doses compared to men and report more gastrointestinal side effects. The incidence of cardiovascular events in women on MTF has been found to be lower to that of men on MTF.
Metformin
gender
insulin resistance
1. Introduction: Metformin—Gender Medicine
Metformin (MTF) occupies a major and fundamental position in the therapeutic management of type 2 diabetes mellitus (T2DM) [1][2][3]. Sex pertains to “the different biological and physiological characteristics of males and females, such as reproductive organs, chromosomes or hormones”, whereas gender pertains to “the socially constructed characteristics of women and men—such as norms, roles and relationships of and between groups of women and men", quoting the relevant definitions from the Council of Europe (https://www.coe.int/en/web/gender-matters/sex-and-gender, accessed on 7 March 2022). Gender medicine is the medical discipline that integrates any effect of sex and/or gender on the overall level of health (prevention, diagnosis and treatment/management of diseases), taking into account biological as well as social sex differences [4]. Its aim is to improve health for any gender. Gender medicine may be a neglected dimension of medicine.
2. Pharmacokinetics, Pharmacodynamics and Metabolism of MTF
MTF is a weak base, and is very polar and extremely soluble in water [5]. It is absorbed from the small intestine, leading to a peak in concentration in one to two hours after oral intake. Its bioavailability is from 50% to 60% [6]. MTF is weakly bound to proteins. Its plasma half-life is estimated to be 1.5 to 5.0 h and it is practically unmetabolized after being distributed mainly in the liver, kidneys and intestine [7][8][9]. Excretion occurs via the kidneys, with a clearance of 933–1317 mL/min, involving glomerular filtration and tubular secretion [9]. The mechanisms of cellular action of MTF are still poorly understood. Various molecular responses are elicited by MTF and apparently some are influenced by sex hormones (Figure 1) [10][11][12][13]. The normoglycemic effect of MTF results mainly from a decrease in hepatic glucose production by inhibition of gluconeogenesis and by an action on glucose-6-phosphatase [2]. In addition to this action on the liver, which results mainly in a decrease in fasting blood sugar, MTF also potentiates the effect of insulin on muscle glucose uptake.
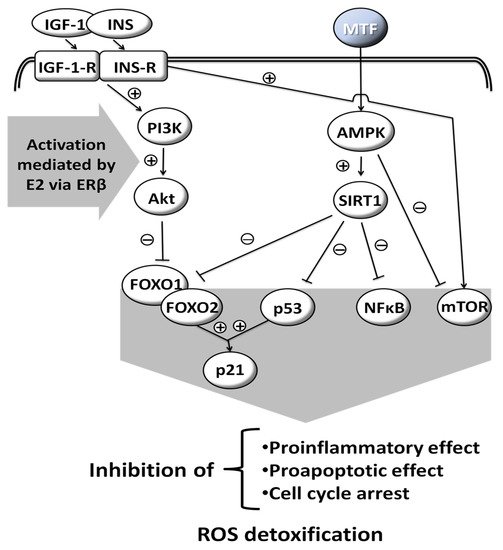
Figure 1. Molecular responses that are elicited by MTF; some are influenced by sex hormones. Insulin-like Growth Factor-1, INS: Insulin, IGF-1-R: Insulin-like Growth Factor-1-Receptor, INS-R: Insulin Receptor, PI3K: Phosphoinositide 3-kinase, Akt: Serine/threonine protein kinase B, AMPK: AMP-activated protein kinase, SIRT1: Sirtuin 1, FOXO1: Forkhead box protein O1, FOXO2: Forkhead box protein O2, p53: Tumor protein p53, NFκB: Nuclear factor kappa-light-chain-enhancer of activated B cells, mTOR: Mammalian target of rapamycin, p21: Cyclin-dependent kinase inhibitor 1, ROS: Reactive oxygen species, E2: Estradiol, ERβ: Estrogen receptor β, (+): activation, (−): inhibition, drawn with data from [10][11][12][13].
3. Gender-Specific Use of MTF
3.1. MTF in Women with Polycystic Ovary Syndrome
Polycystic ovary syndrome (PCOS) is a common endocrine disease (with variable prevalence worldwide, ranging from 6% to 26%) [14], which is characterized by anovulation, clinical or biochemical hyperandrogenemia and polycystic ovary morphology [15]. Hyperinsulinemia as a result of tissue insulin resistance, is central to PCOS [16]. Insulin resistance is observed in 45–65% of patients with PCOS and is associated with excessive phosphorylation of insulin receptors. Hyperinsulinemia, impaired glucose tolerance, dyslipidemia and hypertension affect 40–45% of patients with PCOS [17]. Hyperinsulinemia adversely affects the hypothalamic-pituitary-ovarian axis, resulting in altered endocrine control, menstrual irregularity and infertility [15].
Many interventional studies have demonstrated the positive effect of MTF on both the reproductive as well as metabolic aspects of the syndrome [15]. However, the mechanisms, by which MTF exerts its effects in treating PCOS, are only partially understood. The rationale of MTF use in PCOS is based on the fact that hyperinsulinemia is the basis of the syndrome and adversely affects ovarian function. Insulin boosts 17OH-progesterone activity causing ovarian stroma hypertrophy, follicular atresia and anovulation [15]. Therefore, MTF directly or indirectly improves steroidogenesis (this has been noted by in vitro studies of granuloma cell response to follicle stimulating hormone (FSH) and insulin growth factor 1 (IGF-1) [18]).
In 2013, the American Endocrine Society issued guidelines on the management of PCOS which included MTF as a treatment [19]. Specifically, MTF was recommended for women with PCOS and T2DM or insulin resistance, after failure of lifestyle change, diet and exercise, as a daily routine. It was not advised as a first line treatment of skin manifestations of PCOS (hair loss, acne), complications of the syndrome in pregnancy or for obesity [15]. MTF can also be given to women with menstrual disorders in which contraceptive treatment has failed or in women who wish to have children, as a second choice of treatment. There is no straightforward answer to whether all women with PCOS should undergo MTF treatment [20]. Proponents of MTF consider it a necessary drug for women with PCOS not only to prevent its long-term complications (in the context of insulin resistance) but also because MTF has been shown to improve all of the syndrome’s parameters. The first line treatment that includes diet and physical exercise is a time-consuming process that requires the compliance of women on a strict, long-term schedule, and relapse is very common.
Another element that comes to add to the beneficial action of MTF is in the treatment of adolescent girls with obesity and hyperandrogenemia. It seems that 50% of adolescent women with hyperandrogenemia have already developed resistance to progesterone-mediated gonadotropin-releasing hormone (GnRH) pulse suppression. The abnormal regulation of GnRH and luteinizing hormone (LH) secretion by the persistence of increased frequency of GnRH pulses is already present in adolescent girls with hyperandrogenemia before menarche [21]. Thus, the correction of androgen overproduction in PCOS is deemed to be necessary.
3.2. MTF in Women with Gestational Diabetes
The use of MTF in pregnancy is considered to be safe overall, with favorable effects on maternal weight gain, the incidence of preeclampsia, the dosage in concomitant insulin administration, and the rate of fetal macrosomia and neonatal hypoglycemia; it may increase the rate of small-for-gestational-age infants [22][23][24][25][26][27][28]. Apparently, MTF lowers proinflammatory cytokines (tumor necrosis factor alpha (TNF-α), interleukin [IL]-1-alpha and IL-1-beta and IL-6) in serum, placenta and omental tissues [29]. Interestingly, although in a very critical assessment of metanalyses regarding MTF, most were deemed to be of low quality, the exception being those in obstetric/gynecological settings [30]. There are caveats in the use of MTF in pregnancy: it was found—in vitro, in human embryonic stem cells—to decrease the differentiation of pancreatic beta cells [31], and in mice to decrease or arrest early embryonic development [32].
4. Sex/Gender Differences Using MTF
4.1. Prescribing/Administering MTF for Diabetes
There may be a difference in T2DM prevalence by sex/gender [33]; this difference depends on the definition of diabetes per se: men tend to have higher fasting plasma glucose more often, whereas women tend to have abnormalities in the oral glucose tolerance test (both modalities are used in the diagnosis of the disease) [34]. Although MTF is widely prescribed worldwide as a first-choice medication for T2DM [35], few studies have seen the light regarding use by gender. Although women are more concerned than men about their body image [36], and MTF may show a modest effect on weight loss [37], women are usually prescribed lower MTF doses compared to men (and they report more gastrointestinal side effects) [38].
4.2. MTF and Vitamin B12/Homocysteine
The long-term administration of MTF significantly lowers vitamin B12 levels [39][40][41]. Vitamin B12 deficiency with MTF is rarely symptomatic; it is linked to a reduction in the intestinal absorption of cobalamin and can be reversed by the discontinuation of MTF or with oral B12 supplementation. Men have lower vitamin B12 levels compared to women [42]. In a study of patients with T2DM (without a control group), higher doses of MTF and male sex were factors associated with lower levels of vitamin B12 [43]. Nevertheless, the effect of MTF on B12 by sex/gender, has not been assessed adequately; this is of interest given the sex/gender differences presented above. Additionally, MTF may conditionally elevate or reduce homocysteine levels, which is critical for people with obesity [44][45].
4.3. MTF and Cardiovascular Disease
MTF is considered to be associated with some degree of cardioprotection [46][47]; the latter is apparently the net result of its beneficial actions on endothelial and smooth muscle cells, blood lipids and systemic chronic inflammation [48][49]. In experimental models, MTF was beneficial with regards to myocardial reperfusion, fibrosis and inflammation in post-experimental myocardial ischemia [50][51]. In an older, small scale, study, MTF was noted to have a favorable effect on cardiac metabolism in women (increasing myocardial glucose uptake and lowering fat metabolism), in contrast to having an unfavorable one (with opposite effects) in men [52]. In a study of 167,254 (46% women) patients with T2DM who were already using MTF and started newer anti-diabetic medications, the incidence of cardiovascular events, after a median observation time of 4.5 years, in women was lower compared to that of men (14.7 versus 16.7 per 1000-person-year) [53]. Nevertheless, a systematic review of MTF’s overall actions has not been conclusive regarding micro- and macrovascular complications in patients with T2DM [54].
4.4. MTF and Andrology/Urology
In small (and—apparently—underpowered) studies, the effects of MTF solely on men have been observed. This medication has been reported to be of benefit in non-diabetic men with erectile dysfunction who had not responded to sildenafil [55]. The mechanisms are obscure: they may be direct, via endothelium-dependent vasodilatation or attenuation of sympathetic nerve activity, and indirect, via MTF’s effect on blood pressure [55]. Indirect proof of the low power of studies is that erectile dysfunction, low sex drive and low testosterone (total, free and bioavailable) have also been attributed to MTF use in men with T2DM [56]. Furthermore, the use of MTF for T2DM, in men with prostate cancer, has been associated with lower prostate-specific antigen levels and improved survival [55][57][58][59].
4.5. Musculoskeletal Effects of MTF
From in vitro studies, a role for MTF has been proposed in the stimulation of osteogenesis; in vivo studies are less conclusive [60]. Furthermore, MTF activates adenosine monophosphate-activated protein kinase (AMPK) signaling pathways. The activation of AMPK has been implicated in muscle repair [60]. Thus, it is not surprising that patients using MTF report less musculoskeletal pain vis-à-vis patients not on MTF [61]. The beneficial musculoskeletal effects of MTF were recently found to be more pronounced in women compared to men [61].
4.6. MTF and Experimentally-Induced Neurological Disease
An interesting dimorphism has been observed in mice regarding experimentally induced neuropathic pain and spinal cord microglial activation. MTF was shown to prevent and reverse neuropathic pain and spinal cord microglial activation only in male mice [62]. The researchers presume that the known activation of AMPK may be implicated, although no firm etiology for the sex difference in observations has been formulated. On the other hand, in another experimental study of brain injury in mice, MTF was beneficial for cognitive recovery in females but not males [63], pointing to a crucial relevant role for estradiol/testosterone [63].
4.7. MTF and Aging/Life Span (Experimental)
Dimorphic sex responses to MTF regarding life span have been described: chronic administration of MTF extended the lifespan of female mice and curtailed the lifespan of male mice [64]. Yet, more recent studies show that the positive effect of MTF on longevity is more prominent in male mice [65]. In the Mexican fruit fly the effect of MTF on longevity is dose-dependent, and is beneficial in higher doses for females and in lower doses for males [66].
4.8. MTF and Cancer
In older and newer studies, MTF in subjects with T2DM (in the older studies at low doses of 500 mg/day or less) was shown to be more beneficial vis-à-vis the incidence of colorectal cancer in women compared to men [67][68]. In men, MTF use may lower the risk of prostate cancer, but the effect—if any—is apparently slight and statistically non-significant [69]. In a Lithuanian cohort, the lowest risk for endometrial cancer was observed in diabetic women who used only MTF (with a standardized incidence ratio [SIR] of 1.69 and 95% confidence interval [CI] of 1.49 to 1.92) [70]. MTF was found to lower the markers of proliferation in endometrial cancer cells [71][72].
4.9. MTF and the Microbiome
Subtle differences have been reported in the gut microbiome between the male and female offspring of MTF-treated mice [73], as well as after MTF treatment in adult mice [74]. Before treatment with MTF and a high-fat diet (HFD), female mice had a preponderance of Lactobacillus species, whereas male mice had a preponderance of Proteobacteria species. After ten weeks of HFD and MTF, the bacterial species were different in males and females: a more pronounced increase in Bacteroides was noted in female mice compared to male ones [74].
4.10. MTF and COVID-19
Currently, there is a global effort to fight and win against the new severe acute respiratory syndrome coronvirus-2 (SARS-CoV-2) pandemic and its related coronavirus disease 2019 (COVID-19); proper management of T2DM is of even greater importance, since the presence of diabetes is associated with the most severe forms of COVID-19 and related mortality [75][76], and glycemic control is crucial [77][78]. In addition, a significant increase of cardiometabolic complications has been reported in many geographical areas, highlighting the need of a comprehensive and multidisciplinary approach to this terrible pandemic [79][80].
References
- Kaneto, H.; Kimura, T.; Obata, A.; Shimoda, M.; Kaku, K. Multifaceted Mechanisms of Action of Metformin Which Have Been Unraveled One after Another in the Long History. Int. J. Mol. Sci. 2021, 22, 2596.
- Lv, Z.; Guo, Y. Metformin and Its Benefits for Various Diseases. Front. Endocrinol. 2020, 11, 191.
- Vaughan, E.M.; Rueda, J.J.; Samson, S.L.; Hyman, D.J. Reducing the Burden of Diabetes Treatment: A Review of Low-cost Oral Hypoglycemic Medications. Curr. Diabetes Rev. 2020, 16, 851–858.
- Regitz-Zagrosek, V. Why Do We Need Gender Medicine? In Sex and Gender Aspects in Clinical Medicine; Oertelt-Prigione, S., Regitz-Zagrosek, V., Eds.; Springer: London, UK, 2012; pp. 1–4.
- Yendapally, R.; Sikazwe, D.; Kim, S.S.; Ramsinghani, S.; Fraser-Spears, R.; Witte, A.P.; La-Viola, B. A review of phenformin, metformin, and imeglimin. Drug Dev. Res. 2020, 81, 390–401.
- DeFronzo, R.A.; Buse, J.B.; Kim, T.; Burns, C.; Skare, S.; Baron, A.; Fineman, M. Once-daily delayed-release metformin lowers plasma glucose and enhances fasting and postprandial GLP-1 and PYY: Results from two randomised trials. Diabetologia 2016, 59, 1645–1654.
- Sundelin, E.; Jensen, J.B.; Jakobsen, S.; Gormsen, L.C.; Jessen, N. Metformin Biodistribution: A Key to Mechanisms of Action? J. Clin. Endocrinol. Metab. 2020, 105, 3374–3383.
- Glossmann, H.H.; Lutz, O.M.D. Pharmacology of metformin—An update. Eur. J. Pharmacol. 2019, 865, 172782.
- Mariano, F.; Biancone, L. Metformin, chronic nephropathy and lactic acidosis: A multi-faceted issue for the nephrologist. J. Nephrol. 2021, 34, 1127–1135.
- Wang, C.; Chen, B.; Feng, Q.; Nie, C.; Li, T. Clinical perspectives and concerns of metformin as an anti-aging drug. Aging Med. 2020, 3, 266–275.
- Hampsch, R.A.; Wells, J.D.; Traphagen, N.A.; McCleery, C.F.; Fields, J.L.; Shee, K.; Dillon, L.M.; Pooler, D.B.; Lewis, L.D.; Demidenko, E.; et al. AMPK Activation by Metformin Promotes Survival of Dormant ER(+) Breast Cancer Cells. Clin Cancer Res 2020, 26, 3707–3719.
- Peng, M.; Darko, K.O.; Tao, T.; Huang, Y.; Su, Q.; He, C.; Yin, T.; Liu, Z.; Yang, X. Combination of metformin with chemotherapeutic drugs via different molecular mechanisms. Cancer Treat. Rev. 2017, 54, 24–33.
- Zou, J.; Hong, L.; Luo, C.; Li, Z.; Zhu, Y.; Huang, T.; Zhang, Y.; Yuan, H.; Hu, Y.; Wen, T.; et al. Metformin inhibits estrogen-dependent endometrial cancer cell growth by activating the AMPK-FOXO1 signal pathway. Cancer Sci. 2016, 107, 1806–1817.
- Rao, M.; Broughton, K.S.; LeMieux, M.J. Cross-sectional Study on the Knowledge and Prevalence of PCOS at a Multiethnic University. Prog. Prev. Med. 2020.
- Bulsara, J.; Patel, P.; Soni, A.; Acharya, S. A review: Brief insight into Polycystic Ovarian syndrome. Endocr. Metab. Sci. 2021, 3, 100085.
- Dumesic, D.A.; Oberfield, S.E.; Stener-Victorin, E.; Marshall, J.C.; Laven, J.S.; Legro, R.S. Scientific Statement on the Diagnostic Criteria, Epidemiology, Pathophysiology, and Molecular Genetics of Polycystic Ovary Syndrome. Endocr. Rev. 2015, 36, 487–525.
- Mahmood, K.; Naeem, M.; Rahimnajjad, N.A. Metformin: The hidden chronicles of a magic drug. Eur. J. Intern. Med. 2013, 24, 20–26.
- Zhou, J.; Kumar, T.R.; Matzuk, M.M.; Bondy, C. Insulin-like growth factor I regulates gonadotropin responsiveness in the murine ovary. Mol. Endocrinol. 1997, 11, 1924–1933.
- Legro, R.S.; Arslanian, S.A.; Ehrmann, D.A.; Hoeger, K.M.; Murad, M.H.; Pasquali, R.; Welt, C.K. Diagnosis and Treatment of Polycystic Ovary Syndrome: An Endocrine Society Clinical Practice Guideline. J. Clin. Endocrinol. Metab. 2013, 98, 4565–4592.
- Marshall, J.C.; Dunaif, A. Should all women with PCOS be treated for insulin resistance? Fertil. Steril. 2012, 97, 18–22.
- Apter, D.; Bützow, T.; Laughlin, G.A.; Yen, S.S. Accelerated 24-hour luteinizing hormone pulsatile activity in adolescent girls with ovarian hyperandrogenism: Relevance to the developmental phase of polycystic ovarian syndrome. J. Clin. Endocrinol. Metab. 1994, 79, 119–125.
- Musa, O.A.H.; Syed, A.; Mohamed, A.M.; Chivese, T.; Clark, J.; Furuya-Kanamori, L.; Xu, C.; Toft, E.; Bashir, M.; Abou-Samra, A.B.; et al. Metformin is comparable to insulin for pharmacotherapy in gestational diabetes mellitus: A network meta-analysis evaluating 6046 women. Pharmacol. Res. 2021, 167, 105546.
- Tarry-Adkins, J.L.; Ozanne, S.E.; Aiken, C.E. Impact of metformin treatment during pregnancy on maternal outcomes: A systematic review/meta-analysis. Sci. Rep. 2021, 11, 9240.
- Wang, X.; Liu, W.; Chen, H.; Chen, Q. Comparison of Insulin, Metformin, and Glyburide on Perinatal Complications of Gestational Diabetes Mellitus: A Systematic Review and Meta-Analysis. Gynecol. Obstet. Investig. 2021, 86, 218–230.
- Bidhendi Yarandi, R.; Amiri, M.; Ramezani Tehrani, F.; Behboudi-Gandevani, S. Effectiveness of antidiabetic agents for treatment of gestational diabetes: A methodological quality assessment of meta-analyses and network meta-analysis. J. Diabetes Investig. 2021, 12, 2247–2258.
- He, K.; Guo, Q.; Ge, J.; Li, J.; Li, C.; Jing, Z. The efficacy and safety of metformin alone or as an add-on therapy to insulin in pregnancy with GDM or T2DM: A systematic review and meta-analysis of 21 randomized controlled trials. J. Clin. Pharm. Ther. 2021, 47, 168–177.
- Benham, J.L.; Donovan, L.E.; Yamamoto, J.M. Metformin in Pregnancy for Women with Type 2 Diabetes: A Review. Curr. Diab. Rep. 2021, 21, 36.
- Newman, C.; Dunne, F.P. Metformin for pregnancy and beyond; the pros and cons. Diabet. Med. 2021, 39, e14700.
- Anness, A.R.; Baldo, A.; Webb, D.R.; Khalil, A.; Robinson, T.G.; Mousa, H.A. Effect of metformin on biomarkers of placental-mediated disease: A systematic review and meta-analysis. Placenta 2021, 107, 51–58.
- Li, X.; Celotto, S.; Pizzol, D.; Gasevic, D.; Ji, M.M.; Barnini, T.; Solmi, M.; Stubbs, B.; Smith, L.; López Sánchez, G.F.; et al. Metformin and health outcomes: An umbrella review of systematic reviews with meta-analyses. Eur. J. Clin. Investig. 2021, 51, e13536.
- Nguyen, L.; Lim, L.Y.; Ding, S.S.L.; Amirruddin, N.S.; Hoon, S.; Chan, S.Y.; Teo, A.K.K. Metformin Perturbs Pancreatic Differentiation From Human Embryonic Stem Cells. Diabetes 2021, 70, 1689–1702.
- Nayak, G.; Salian, S.R.; Agarwal, P.; Suresh Poojary, P.; Rao, A.; Kumari, S.; Kalthur, S.G.; Shreya, A.B.; Mutalik, S.; Adiga, S.K.; et al. Antidiabetic drug metformin affects the developmental competence of cleavage-stage embryos. J. Assist. Reprod. Genet. 2020, 37, 1227–1238.
- Kautzky-Willer, A. Sex and Gender Differences in Endocrinology. In Sex and Gender Aspects in Clinical Medicine; Oertelt-Prigione, S., Regitz-Zagrosek, V., Eds.; Springer: London, UK, 2012; pp. 125–149.
- Mauvais-Jarvis, F. Epidemiology of Gender Differences in Diabetes and Obesity. In Sex and Gender Factors Affecting Metabolic Homeostasis, Diabetes and Obesity; Springer International Publishing AG: Cham, Switzerland, 2017; pp. 3–8.
- Flory, J.; Lipska, K. Metformin in 2019. JAMA 2019, 321, 1926–1927.
- Nomura, K.; Itakura, Y.; Minamizono, S.; Okayama, K.; Suzuki, Y.; Takemi, Y.; Nakanishi, A.; Eto, K.; Takahashi, H.; Kawata, Y.; et al. The Association of Body Image Self-Discrepancy with Female Gender, Calorie-Restricted Diet, and Psychological Symptoms among Healthy Junior High School Students in Japan. Front. Psychol. 2021, 12, 576089.
- Grandone, A.; Di Sessa, A.; Umano, G.R.; Toraldo, R.; Miraglia Del Giudice, E. New treatment modalities for obesity. Best Pract. Res. Clin. Endocrinol. Metab. 2018, 32, 535–549.
- de Vries, S.T.; Denig, P.; Ekhart, C.; Mol, P.G.M.; van Puijenbroek, E.P. Sex Differences in Adverse Drug Reactions of Metformin: A Longitudinal Survey Study. Drug Saf. 2020, 43, 489–495.
- Chapman, L.E.; Darling, A.L.; Brown, J.E. Association between metformin and vitamin B(12) deficiency in patients with type 2 diabetes: A systematic review and meta-analysis. Diabetes Metab. 2016, 42, 316–327.
- Infante, M.; Leoni, M.; Caprio, M.; Fabbri, A. Long-term metformin therapy and vitamin B12 deficiency: An association to bear in mind. World J. Diabetes 2021, 12, 916–931.
- Yang, W.; Cai, X.; Wu, H.; Ji, L. Associations between metformin use and vitamin B(12) levels, anemia, and neuropathy in patients with diabetes: A meta-analysis. J. Diabetes 2019, 11, 729–743.
- Margalit, I.; Cohen, E.; Goldberg, E.; Krause, I. Vitamin B12 Deficiency and the Role of Gender: A Cross-Sectional Study of a Large Cohort. Ann. Nutr. Metab. 2018, 72, 265–271.
- Alvarez, M.; Sierra, O.R.; Saavedra, G.; Moreno, S. Vitamin B12 deficiency and diabetic neuropathy in patients taking metformin: A cross-sectional study. Endocr. Connect. 2019, 8, 1324–1329.
- Wang, J.; You, D.; Wang, H.; Yang, Y.; Zhang, D.; Lv, J.; Luo, S.; Liao, R.; Ma, L. Association between homocysteine and obesity: A meta-analysis. J. Evid. Based Med. 2021, 14, 208–217.
- Zhang, Q.; Li, S.; Li, L.; Li, Q.; Ren, K.; Sun, X.; Li, J. Metformin Treatment and Homocysteine: A Systematic Review and Meta-Analysis of Randomized Controlled Trials. Nutrients 2016, 8, 798.
- Monami, M.; Candido, R.; Pintaudi, B.; Targher, G.; Mannucci, E. Effect of metformin on all-cause mortality and major adverse cardiovascular events: An updated meta-analysis of randomized controlled trials. Nutr. Metab. Cardiovasc. Dis. 2021, 31, 699–704.
- Wu, L.; Gunton, J.E. The Changing Landscape of Pharmacotherapy for Diabetes Mellitus: A Review of Cardiovascular Outcomes. Int. J. Mol. Sci. 2019, 20, 5853.
- Luo, F.; Das, A.; Chen, J.; Wu, P.; Li, X.; Fang, Z. Metformin in patients with and without diabetes: A paradigm shift in cardiovascular disease management. Cardiovasc. Diabetol. 2019, 18, 54.
- Mbara, K.C.; Mofo Mato, P.E.; Driver, C.; Nzuza, S.; Mkhombo, N.T.; Gcwensa, S.K.; McObothi, E.N.; Owira, P.M. Metformin turns 62 in pharmacotherapy: Emergence of non-glycaemic effects and potential novel therapeutic applications. Eur. J. Pharmacol. 2021, 898, 173934.
- Jo, W.; Kang, K.-K.; Chae, S.; Son, W.-C. Metformin Alleviates Left Ventricular Diastolic Dysfunction in a Rat Myocardial Ischemia Reperfusion Injury Model. Int. J. Mol. Sci. 2020, 21, 1489.
- Loi, H.; Kramar, S.; Laborde, C.; Marsal, D.; Pizzinat, N.; Cussac, D.; Roncalli, J.; Boal, F.; Tronchere, H.; Oleshchuk, O.; et al. Metformin Attenuates Postinfarction Myocardial Fibrosis and Inflammation in Mice. Int. J. Mol. Sci. 2021, 22, 9393.
- Lyons, M.R.; Peterson, L.R.; McGill, J.B.; Herrero, P.; Coggan, A.R.; Saeed, I.M.; Recklein, C.; Schechtman, K.B.; Gropler, R.J. Impact of sex on the heart’s metabolic and functional responses to diabetic therapies. Am. J. Physiol. Heart Circ. Physiol. 2013, 305, H1584–H1591.
- Raparelli, V.; Elharram, M.; Moura, C.S.; Abrahamowicz, M.; Bernatsky, S.; Behlouli, H.; Pilote, L. Sex Differences in Cardiovascular Effectiveness of Newer Glucose-Lowering Drugs Added to Metformin in Type 2 Diabetes Mellitus. J. Am. Heart Assoc. 2020, 9, e012940.
- Gnesin, F.; Thuesen, A.C.B.; Kähler, L.K.A.; Madsbad, S.; Hemmingsen, B. Metformin monotherapy for adults with type 2 diabetes mellitus. Cochrane Database Syst. Rev. 2020, 6, CD012906.
- Tseng, C.H. The Effect of Metformin on Male Reproductive Function and Prostate: An Updated Review. World J. Men’s Health 2021, 40, 11–29.
- Al-Kuraishy, H.M.; Al-Gareeb, A.I. Erectile Dysfunction and Low Sex Drive in Men with Type 2 DM: The Potential Role of Diabetic Pharmacotherapy. J. Clin. Diagn. Res. 2016, 10, FC21–FC26.
- Atalay, E.; Demir, A.; Eroglu, H.A. The Influences of Metformin on Prostate in Terms of PSA Level and Prostate Volume. Urol. J. 2020, 18, 181–185.
- Kincius, M.; Patasius, A.; Linkeviciute-Ulinskiene, D.; Zabuliene, L.; Smailyte, G. Reduced risk of prostate cancer in a cohort of Lithuanian diabetes mellitus patients. Aging Male 2020, 23, 1333–1338.
- Linkeviciute-Ulinskiene, D.; Patasius, A.; Kincius, M.; Zabuliene, L.; Smailyte, G. Preexisting diabetes, metformin use and long-term survival in patients with prostate cancer. Scand. J. Urol. 2020, 54, 401–407.
- Kalaitzoglou, E.; Fowlkes, J.L.; Popescu, I.; Thrailkill, K.M. Diabetes pharmacotherapy and effects on the musculoskeletal system. Diabetes Metab. Res. Rev. 2019, 35, e3100.
- Carvalho, E.S.A.P.; Harmer, A.R.; Ferreira, M.L.; Ferreira, P.H. The effect of the anti-diabetic drug metformin on musculoskeletal pain: A cross-sectional study with 21,889 individuals from the UK biobank. Eur. J. Pain 2021, 25, 1264–1273.
- Inyang, K.E.; Szabo-Pardi, T.; Wentworth, E.; McDougal, T.A.; Dussor, G.; Burton, M.D.; Price, T.J. The antidiabetic drug metformin prevents and reverses neuropathic pain and spinal cord microglial activation in male but not female mice. Pharmacol. Res. 2019, 139, 1–16.
- Ruddy, R.M.; Adams, K.V.; Morshead, C.M. Age- and sex-dependent effects of metformin on neural precursor cells and cognitive recovery in a model of neonatal stroke. Sci. Adv. 2019, 5, eaax1912.
- Anisimov, V.N.; Piskunova, T.S.; Popovich, I.G.; Zabezhinski, M.A.; Tyndyk, M.L.; Egormin, P.A.; Yurova, M.V.; Rosenfeld, S.V.; Semenchenko, A.V.; Kovalenko, I.G.; et al. Gender differences in metformin effect on aging, life span and spontaneous tumorigenesis in 129/Sv mice. Aging 2010, 2, 945–958.
- Zhu, X.; Shen, W.; Liu, Z.; Sheng, S.; Xiong, W.; He, R.; Zhang, X.; Ma, L.; Ju, Z. Effect of Metformin on Cardiac Metabolism and Longevity in Aged Female Mice. Front. Cell Dev. Biol. 2020, 8, 626011.
- Aceves-Aparicio, E.; Pérez-Staples, D.; Arredondo, J.; Corona-Morales, A.; Morales-Mávil, J.; Díaz-Fleischer, F. Combined Effects of Methoprene and Metformin on Reproduction, Longevity, and Stress Resistance in Anastrepha ludens (Diptera: Tephritidae): Implications for the Sterile Insect Technique. J. Econ. Entomol. 2021, 114, 142–151.
- Lee, M.-S.; Hsu, C.-C.; Wahlqvist, M.L.; Tsai, H.-N.; Chang, Y.-H.; Huang, Y.-C. Type 2 diabetes increases and metformin reduces total, colorectal, liver and pancreatic cancer incidences in Taiwanese: A representative population prospective cohort study of 800,000 individuals. BMC Cancer 2011, 11, 20.
- Wang, Y.; Xiao, J.; Zhao, Y.; Du, S.; Du, J. Effect of metformin on the mortality of colorectal cancer patients with T2DM: Meta-analysis of sex differences. Int. J. Colorectal. Dis. 2020, 35, 827–835.
- Ghiasi, B.; Sarokhani, D.; Najafi, F.; Motedayen, M.; Dehkordi, A.H. The Relationship Between Prostate Cancer and Metformin Consumption: A Systematic Review and Meta-analysis Study. Curr. Pharm. Des. 2019, 25, 1021–1029.
- Zabuliene, L.; Kaceniene, A.; Steponaviciene, L.; Linkeviciute-Ulinskiene, D.; Stukas, R.; Arlauskas, R.; Vanseviciute-Petkeviciene, R.; Smailyte, G. Risk of Endometrial Cancer in Women with Diabetes: A Population-Based Retrospective Cohort Study. J. Clin. Med. 2021, 10, 3453.
- Pabona, J.M.P.; Burnett, A.F.; Brown, D.M.; Quick, C.M.; Simmen, F.A.; Montales, M.T.E.; Liu, S.J.; Rose, T.; Alhallak, I.; Siegel, E.R.; et al. Metformin Promotes Anti-tumor Biomarkers in Human Endometrial Cancer Cells. Reprod. Sci. 2020, 27, 267–277.
- Petchsila, K.; Prueksaritanond, N.; Insin, P.; Yanaranop, M.; Chotikawichean, N. Effect of Metformin for Decreasing Proliferative Marker in Women with Endometrial Cancer: A Randomized Double-blind Placebo-Controlled Trial. Asian Pac. J. Cancer Prev. 2020, 21, 733–741.
- Salomäki-Myftari, H.; Vähätalo, L.H.; Ailanen, L.; Pietilä, S.; Laiho, A.; Hänninen, A.; Pursiheimo, J.P.; Munukka, E.; Rintala, A.; Savontaus, E.; et al. Neuropeptide Y Overexpressing Female and Male Mice Show Divergent Metabolic but Not Gut Microbial Responses to Prenatal Metformin Exposure. PLoS ONE 2016, 11, e0163805.
- Silamiķele, L.; Silamiķelis, I.; Ustinova, M.; Kalniņa, Z.; Elbere, I.; Petrovska, R.; Kalniņa, I.; Kloviņš, J. Metformin Strongly Affects Gut Microbiome Composition in High-Fat Diet-Induced Type 2 Diabetes Mouse Model of Both Sexes. Front. Endocrinol. 2021, 12, 626359.
- Stoian, A.P.; Kempler, P.; Stulnig, T.; Rizvi, A.A.; Rizzo, M. Diabetes and COVID-19: What 2 Years of the Pandemic Has Taught Us. Metab. Syndr. Relat. Disord. 2021.
- Ceriello, A.; Stoian, A.P.; Rizzo, M. COVID-19 and diabetes management: What should be considered? Diabetes Res. Clin. Pract. 2020, 163, 108151.
- Ilias, I.; Jahaj, E.; Kokkoris, S.; Zervakis, D.; Temperikidis, P.; Magira, E.; Pratikaki, M.; Vassiliou, A.G.; Routsi, C.; Kotanidou, A.; et al. Clinical Study of Hyperglycemia and SARS-CoV-2 Infection in Intensive Care Unit Patients. Vivo 2020, 34, 3029–3032.
- Michalakis, K.; Ilias, I. COVID-19 and hyperglycemia/diabetes. World J. Diabetes 2021, 12, 642–650.
- Rizvi, A.A.; Janez, A.; Rizzo, M. Cardiometabolic Alterations in the Interplay of COVID-19 and Diabetes: Current Knowledge and Future Avenues. Int. J. Mol. Sci. 2021, 22, 2311.
- Al Mahmeed, W.; Al-Rasadi, K.; Banerjee, Y.; Ceriello, A.; Cosentino, F.; Galia, M.; Goh, S.Y.; Kempler, P.; Lessan, N.; Papanas, N.; et al. Promoting a Syndemic Approach for Cardiometabolic Disease Management During COVID-19: The CAPISCO International Expert Panel. Front. Cardiovasc. Med. 2021, 8, 787761.
More
Information
Subjects:
Medicine, General & Internal
Contributor
MDPI registered users' name will be linked to their SciProfiles pages. To register with us, please refer to https://encyclopedia.pub/register
:
View Times:
1.8K
Revisions:
2 times
(View History)
Update Date:
22 Mar 2022
Notice
You are not a member of the advisory board for this topic. If you want to update advisory board member profile, please contact office@encyclopedia.pub.
OK
Confirm
Only members of the Encyclopedia advisory board for this topic are allowed to note entries. Would you like to become an advisory board member of the Encyclopedia?
Yes
No
${ textCharacter }/${ maxCharacter }
Submit
Cancel
Back
Comments
${ item }
|
More
No more~
There is no comment~
${ textCharacter }/${ maxCharacter }
Submit
Cancel
${ selectedItem.replyTextCharacter }/${ selectedItem.replyMaxCharacter }
Submit
Cancel
Confirm
Are you sure to Delete?
Yes
No





