Your browser does not fully support modern features. Please upgrade for a smoother experience.

Submitted Successfully!
Thank you for your contribution! You can also upload a video entry or images related to this topic.
For video creation, please contact our Academic Video Service.
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Pedro Fonte | + 6749 word(s) | 6749 | 2022-03-15 04:28:22 | | | |
| 2 | Rita Xu | Meta information modification | 6749 | 2022-03-18 02:35:03 | | |
Video Upload Options
We provide professional Academic Video Service to translate complex research into visually appealing presentations. Would you like to try it?
Cite
If you have any further questions, please contact Encyclopedia Editorial Office.
Fonte, P. Nanomedicine for Head and Neck Cancer. Encyclopedia. Available online: https://encyclopedia.pub/entry/20699 (accessed on 21 May 2026).
Fonte P. Nanomedicine for Head and Neck Cancer. Encyclopedia. Available at: https://encyclopedia.pub/entry/20699. Accessed May 21, 2026.
Fonte, Pedro. "Nanomedicine for Head and Neck Cancer" Encyclopedia, https://encyclopedia.pub/entry/20699 (accessed May 21, 2026).
Fonte, P. (2022, March 17). Nanomedicine for Head and Neck Cancer. In Encyclopedia. https://encyclopedia.pub/entry/20699
Fonte, Pedro. "Nanomedicine for Head and Neck Cancer." Encyclopedia. Web. 17 March, 2022.
Copy Citation
Head and neck cancers rank sixth among the most common cancers today, and the survival rate has remained virtually unchanged over the past 25 years, due to late diagnosis and ineffective treatments. They have two main risk factors, tobacco and alcohol, and human papillomavirus infection is a secondary risk factor. These cancers affect areas of the body that are fundamental for the five senses. Therefore, it is necessary to treat them effectively and non-invasively as early as possible, in order to do not compromise vital functions, which is not always possible with conventional treatments (chemotherapy or radiotherapy).
head and neck cancer
nanomedicine
drug delivery
nanocarrier
1. Introduction
The words cancer, tumor, and neoplasia are generic terms that define a broad range of diseases characterized by unchecked division or uncontrolled proliferation of cells in a tissue or organ. Due to their high rate of division, these cells may invade the adjacent tissues or disseminate at a distance, in a process known as metastasis [1]. This disease can affect any organ or part of the body, and has many histological types. It has a great impact on the quality of life, not only for the patient but also for his family, particularly at a psychological and emotional level, and is often associated with financial hardship [2][3]. Moreover, it is the largest cause of morbidity and mortality worldwide. It is even the leading cause of death in developed countries, and the second in developing countries [3][4]. One of the main factors associated with the increase in the global cancer burden the level of human development, which includes the factors of population growth and aging, changes in lifestyle, economic variables, and social changes [5]. Head and neck cancer (HNC) is a complex and heterogeneous disease that encompasses a large number of cancer cell locations and is considered a leading cause of cancer death worldwide [4][6]. The American Cancer Society refers to HNC as one of the leading causes of death, along with cancers of the digestive system, respiratory system, breast, and reproductive system [7]. Though the various etiological risk factors are well defined, and advances have been made both in its diagnosis and therapy using different approaches, its morbidity has not significantly reduced over the last decades [8].
In this sense, early diagnosis is fundamental to reducing the involvement of other organs and structures, and to improving the prognosis of the disease once the success of the therapy is promoted by early initiation. Conventional cancer treatment protocols include local surgery combined with adjuvant systemic therapies—namely, the administration of cytotoxic drugs and the application of radiation. However, these are invasive methods and are associated with several serious side effects that compromise the patients’ quality of life, such as severe toxicity, mucositis, dysphagia, xerostomia, radiation dermatitis, hematologic toxicity, neurotoxicity, and ototoxicity [6][8]. The fact that these anticancer drugs cannot discriminate normal healthy cells from tumor cells results not only in side effects, but in, low concentrations reaching the target zones, compromising the therapeutic effect.
Nanomedicine using nanotechnology-based systems emerged as a new cancer management strategy. It involves a wide variety of nanostructures with small particle size distributions capable of crossing biological barriers and interacting with the target molecules of the therapy, among other structures present in the cells, reducing the effects of treatment on healthy cells [9][10].
Thus, nanotechnology-based systems for HNC management have the potential to emerge as alternatives to conventional treatments, as these systems can offer solutions to the problems encountered in conventional treatments: they not only minimize non-specific delivery failures and cell death, and reduce multidrug resistance, but also maximize the efficacy of therapeutic agents. Since they are nanometer-sized systems, they easily penetrate and permeate through the cell membrane, blood capillaries, and biological barriers [11][12][13][14]. These capabilities are very important in the case of HNC, because in some cases, to reach the therapeutic target it is necessary cross biological barriers such as the blood–brain barrier, which researchers know reduces drug delivery to the brain [15]. This technology also can help clinicians in the monitoring of the disease, because they allow less invasive, more sensitive, and more specific analysis of the affected structures, and facilitate the diagnosis of the disease at an earlier stage, providing greater chances of total remission and therapeutic success. Moreover, nanocarriers are not only biocompatible, but can deliver various diagnostic probes with the ability to target specific biomolecules. Therefore, they can help to provide important structural and metabolic information about a tumor more successfully than other imaging techniques, and identify hidden metastases. Moreover, they can also function as optical contrast agents during image-guided surgery [16][17].
2. Anatomophysiology of the Head and Neck
The head is the upper part of the body that is attached to the rest of the body through the neck. It is the structure that contains the encephalon, which is responsible for their entire state of consciousness (creativity, ideas, imagination, responses, decisions, and memory), along with its protective coatings, the ears, and the face that ensures their identity [18].
In the head (Figure 1), there are also the sensitive receptors (eyes, mouth, ears, and nose); the structures for transmission of voice and expression; and the entrances for nutrients, water, and oxygen and the exit of carbon dioxide. The skull, the bony part of the head, supports the face and protects the brain, and it is composed of two parts, the neurocranium (bone box of the encephalon and cranial meninges) and the viscerocranium (bones of the face). The neurocranium is covered with subcutaneous tissue and skin (part of this skin forms the scalp) [18]. For the face there is a set of bones, the facial bones, which form the nasal cavity, surround the eyeballs, and support the teeth of both jaws. The skull of an adult has 22 individual bones, of which only one is a moving bone, which is the mandible (lower jaw). The remaining 22 bones are immobile and are united in a single unit [19].
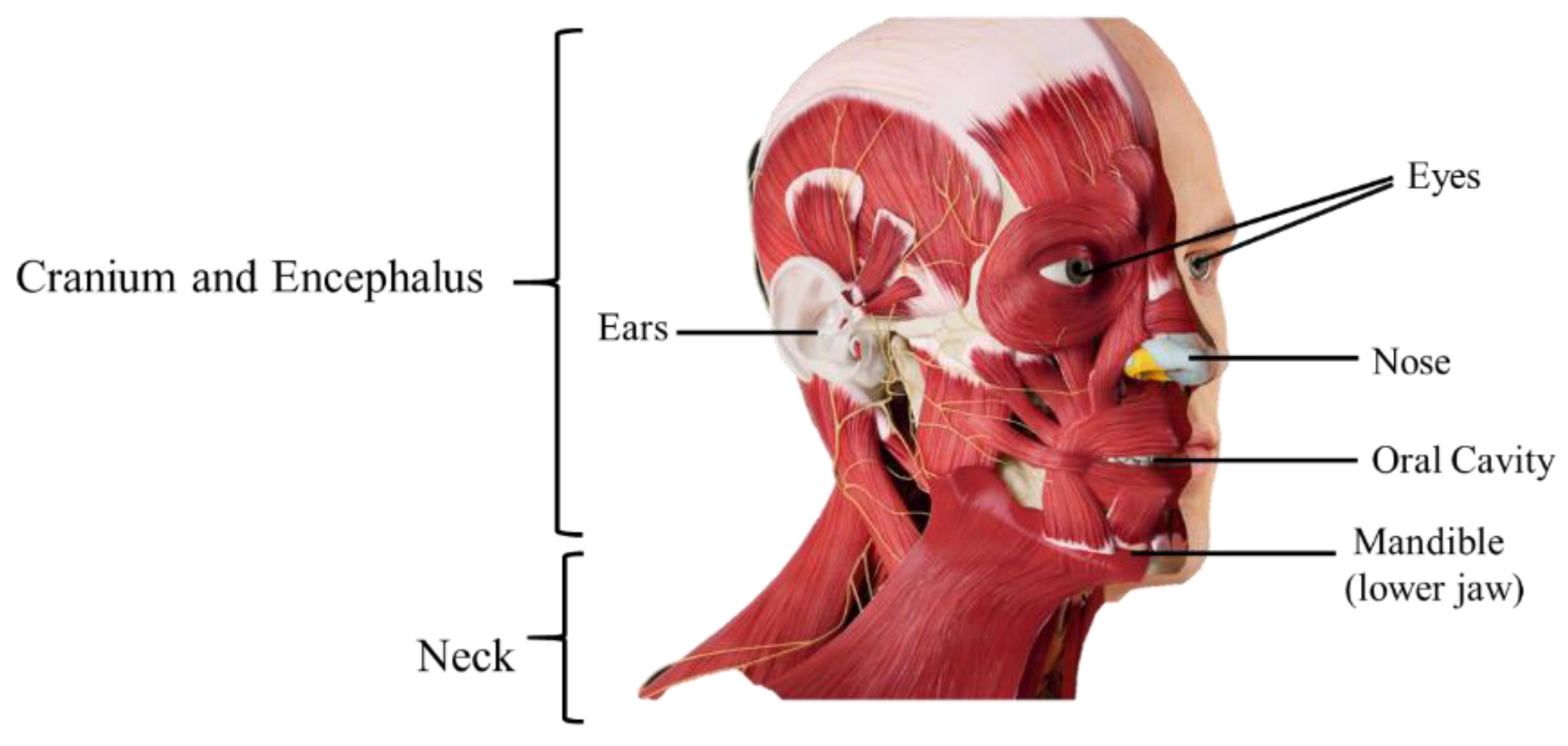
Figure 1. Anatomical structures of head and neck.
The neck is the transition zone between the base of the cranium above, and the clavicles and cervical spine below. It connects the head to the torso and limbs, being an important structure between them. It contains numerous vital structures with essential functions for normal physiology, such as breathing, speaking, swallowing, and the regulation of metabolism. In it are several important organs and tissues, such as the larynx, trachea, thyroid, parathyroid, esophagus, and vertebrae [18]. There are also the structures for circulatory and lymphatic inflow and outflow from the head. The main arterial blood flow to the head and neck comes through the carotid arteries, and the main venous drainage occurs via the jugular veins. Lymph from those structures drains into the cervical lymph nodes. The neck has important muscles, such as the platysma, a thin muscle spanning the upper chest to the cheek and lower lip, drawing the lower central lip. It is also the center of a multitude of nervous system structures, transmitting nervous signals from the brain to the body. On the front side of the neck is the thyroid cartilage, the largest cartilage around the larynx and trachea. The neck is slim to allow for the flexibility needed to position the head and maximize the efficiency of its sensitive organs (eyes, ears, mouth, and noise). It is therefore a region that is characterized by fragility and vulnerability. In addition, some vital structures, such as the trachea, esophagus, and glandule of the thyroid, have no bone protection [18][20].
The upper aerodigestive tract is located throughout the head and neck region. It includes the oral cavity, oropharynx, laryngopharynx, larynx, nasopharynx, salivary glands, and nasal cavity (Figure 2). The production and articulation of speech, swallowing, and respiration are functions controlled by the oral cavity, pharynx, and larynx. In addition, these structures also protect the airways [21][22]. Damage to one of these structures may affect the functioning of the others. For example, during swallowing it is essential to protect the airways; otherwise, aspiration may result. The oral cavity, besides being involved in phonation, also controls the voluntary phase of swallowing, namely, chewing, preparing the cake, and delivering it to the oropharynx [22].
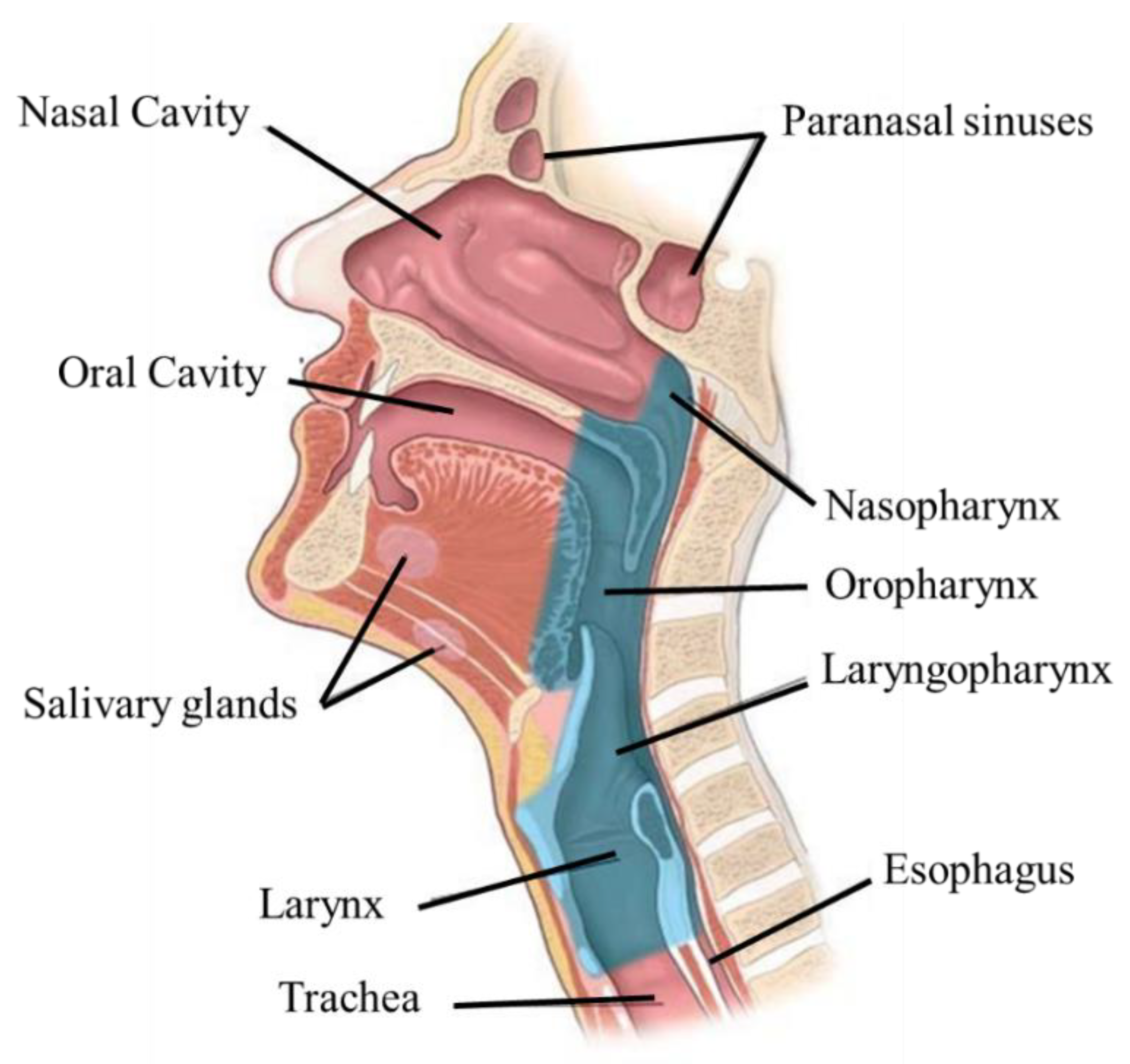
Figure 2. Structures and organs of the upper aerodigestive tract.
3. Head and Neck Cancer
HNC covers the set of diseases involving abnormal soft tissue growth located in these anatomical areas: deeper than the skull and above the clavicles, except those located in the eye, brain, and esophagus. In addition, it must be malignant, showing invasion of neighboring tissues and dissemination into the bloodstream. These cancers start in the epithelial layer of the mucosa of the upper aerodigestive tract, and consequently include malignant neoplasms originating in the oral cavity, nasopharynx, oropharynx, laryngopharynx, larynx, paranasal sinuses, and salivary glands [22][23][24]—for instance, sarcoma, lymphoma, and salivary gland carcinomas [21]. The pharyngeal area is one of the most common sites affected among the different parts of the oral cavity [13].
3.1. Epidemiology, Etiology, and Risk Factors
According to data from 2021, every year, about 932,000 new HNC cases are registered and there are about 467,000 HNC deaths [4]. Neoplasms originating in this anatomical area are some of the most common cancers worldwide, as HNC cases represent about 6% of all cases of cancer [23][24][25][26][27]. The incidence of HNC varies depending on geographic region, population, gender (more common in men), and exposure to diverse risk factors. The major risk factors include tobacco, alcohol consumption, and human papillomavirus (HPV) infection, these being recognized as the main causes of upper aerodigestive cancers in industrialized regions [11][13][24][26][28][29][30]. However, HNC results from several factors, including genetic predisposition, environmental exposure, and behavioral/lifestyle factors. The use of tobacco and alcohol is responsible for about 72% of all HNCs, 4% being due to the use of alcohol alone, 33% to tobacco alone, and 35% to the combined use of these. Thus, the cooccurrence of smoking and alcohol consumption increases the chance of developing HNC, though smoking is considered to be a more major risk factor than alcohol for this type of cancer [29][31]. That said, alcohol is a trigger for the tobacco promoter effect in neoplasm formation [21]. After smoking cessation, there is a reduction in relative risk; however, an individual who was a heavy smoker has triple the risk of a non-smoker, even after 10 years of cessation [21].
Some occupational and environmental contexts have been related to increased incidence of HNC, such as agricultural activities and working a as a cook, waiter, firefighter, butcher or meat preparer, knitter, or roofer. These associations exist because these work environments and occupations are more conducive to smoking and/or alcohol consumption [30]. HPV infection, particularly subtype 16, and to a slighter extent, subtype 18, is believed to be a risk factor for oropharyngeal cancer, based on results in a young non-smoking population [26][29]. Patients with oropharyngeal cancer initiated by the virus typically showed better therapeutic results, and consequently a higher overall survival rate; thus, it is possible to note that age may function as a protective factor, being the reason for the increased survival of this group. Additionally, the augmented expression of p16 protein in HPV-related tumors has significantly better disease-specific survival when compared with non-virus-related tumors that do not exhibit increased p16 protein expression [29]. Infection with this virus promotes an uncontrolled cell cycle which results in genetic instability which, over time, promotes the transformation of premalignant lesions into invasive squamous cell carcinomas. In the case of oropharyngeal squamous cell carcinoma, the stage of development of the virus is an independent prognostic factor for overall survival and progression-free survival [32][33].
Epidemiological studies show that, although the previously-mentioned risk factors are the main ones for most HNCs, nasopharyngeal cancers usually present a set of common etiological factors that include, in addition to those described above, Epstein–Barr virus (EBV) infection and processed food [24][34]. EBV is a DNA lymphotropic herpesvirus that is responsible for the presence of infectious mononucleosis and is highly prevalent in healthy individuals, affecting more than 90% of individuals worldwide [35]. This virus is not found in tumor cells exclusively; however, it is not present in normal cells of the nasopharyngeal epithelium, which implies a direct relationship between EBV activation and the pathogenesis of the tumor [34]. Regarding the co-presence of the above-mentioned viral infections and HNC, a study performed by Al-Thawadi et al. showed that HPV and EBV oncoviruses are co-present in squamous cell carcinomas, especially when they occur in the oral cavity, which may promote their initiation and/or progression; however, the mechanisms of this relationship need to be better elucidated [36].
3.2. Pathophysiology
Understanding the origin and pathophysiology of the HCN is fundamental to predicting and managing the course of the disease, and its impact on the patient’s quality of life. This process facilitates the choice of the most appropriate treatment or combination (surgery, radiation therapy, and chemotherapy) while also minimizing possible sequels, such as significant acute and chronic damage to the oral cavity, which is not limited to the hard tissue (teeth and alveolar bone) and the oral mucous membrane, but also affects the soft tissues of the head and neck [37]. Generally, all these malignancies are epithelial because they develop on the upper layers of the epidermis (mucosa) of the upper aerodigestive tract, squamous cell carcinoma being the most common histological type of head and neck tumor. These tumors can range from poorly to well differentiated, and in fact, about 90% of all HNC are squamous cell carcinomas and variants [23][28][38].
In the presence of premature lesions, leucoplakia and erythroplakia with histologic features of hyperplasia or dysplasia are evident. Both cases may deform into invasive tumors, but erythroplakia presents a higher risk of transformation [39]. As in other types of cancer, malignant cells also escape recognition and destruction by immune agents and inhibit or manipulate antitumor immune defenses. It is therefore common for patients with these types of cancer to have low concentrations of CD3+, CD4+, and CD8+ T cells, which may persist even several years after curative surgery. The main mechanisms of immune escape used by tumors to grow and target immune cells are immune destruction escape, tumor suppressor escape and cell regulation, reduction of T lymphocyte activity, immunosuppressive cells, and cytokines that control local and systemic effects [28].
Since the affected areas in these types of cancers are adjacent to the respiratory and digestive systems, the same agents that promote the development of cancer cells in HNCs also affect other organs throughout the body, including the lungs. Thus, these tumors may not appear in isolation, but rather in association with other secondary tumors [40]. As in other types of cancer, angiogenesis is also a determining factor in the development of neoplasm and progression of tumors and is regulated by several endogenous proangiogenic and antiangiogenic factors. Fundamental factors for the growth of cancer and metastasis are the vascular endothelial growth factor (VEGF) and its receptors. This receptor can be upregulated and has significant importance in the prognosis of several HNCs [22][28]. Another important marker that has high expression in HNC is the epidermal growth factor receptor (EGFR), which is expressed in more than 90% of tumors. EGFR is highly expressed in normal epithelial cells, so alterations in its pathways can promote a malignant transformation of HNC [32]. Their expression levels correlate with worsened disease-free survival and overall survival [22][32].
HNC can also result from mutations in various genes and pathways, including both tumor-suppressor genes and oncogenes. Some biomarkers aid in the screening, diagnosis, and management of the disease. TP53 and CDKN2A/P16 are mutated tumor-suppressor genes frequently observed in HNC that may confer growth advantages to cells and encourage the development of carcinoma. FAT1 is one of the latest genes implicated in HNC that participates in cell cycle regulation and proliferation, and it is described as a tumor suppressor gene. NOTCH1 is the most recent cancer gene associated with HNC development. Functionally, the gene signaling has both oncogenic and tumor-suppressive roles depending on the cellular context; however, its exact role in pathogenesis needs to be better elucidated. The RAS gene family involves three oncogenes whose mutations in their cell cascades are included in approximately one-quarter of all human cancers. The PIK3CA pathway is another critical pathway in HNC carcinogenesis [32]. Another marker that is sometimes overexpressed in HNC is programmed death-ligand 1 (PD-L1), a transmembrane protein that acts as a co-inhibitory factor of the immune response, reducing the patient’s immune response to tumor cells. Its presence is thus associated with a negative prognosis [41][42]. Advancements in the knowledge of these molecular structures (receptors) and genetic changes which are biomarkers for HNC are important, as they can be potential targets for therapy and help to define new diagnostic and therapeutic strategies, namely, those concerning personalized therapeutics.
3.3. Signs and Symptoms
The first symptoms that may indicate the presence or prognosis of HNC are complaints about the aerodigestive tract that are not resolved the conventional treatments. Usually, the tumors in the oral cavity initially start with lesions in the gingivobuccal sulcus tongue, buccal mucosa, or floor of the mouth. Some patients also present with a nonhealing ulcer, an area of irritation between adjacent teeth, pain, or bleeding that does not cease within a normal timeframe. Some other HNCs may be asymptomatic until they are advanced or have developed metastases.
In the presence of the suspicious signs and symptoms above, it is essential to perform a biopsy for a definitive diagnosis. Another sign that the development of tumor tissue can trigger is the presence of paranesthesia in the oral or nasal cavity because of nerve compression, one of the most common presentations being numbness of the lips and tongue. Lesions in the alveolar ridge can cause loss of malocclusion teeth, or poorly fitting dentures, and tongue lesions can make it difficult to chew. If these symptoms are found in patients with a history of tobacco or alcohol use, the tumor may already be advanced [22].
The lesions in the oropharynx have more insidious symptoms, which can be confused with other diseases or problems, since they can cause pain during swallowing or otalgia. Tumors in the pharynx or supraglottis can cause neck masses because of metastatic lymphadenopathy, though these can be too small to cause other symptoms and be easily detected by a physical examination. Another symptom of this type of cancer is weight loss, not only because of one’s difficulty in swallowing or dysphagia, but also because of the effects on the metabolism. Tumors in the laryngopharynx in advanced stages can reach the larynx, resulting in hoarseness or vocal cord paralysis. The tumors in the glottis are usually the ones that are easiest to access, thereby allowing diagnosis in earlier phases. Early diagnosis is also helped by the vocal cord infections and hoarseness which can occur. In advanced stages, these tumors can compromise breathing [22]. Thus, identifying recurrent symptoms at an early stage is essential for successful treatment.
3.4. Diagnosis and Treatment
For the diagnosis of a patient with upper aerodigestive tract complaints, it is essential to understand which areas of the head and neck may cause the symptoms. This procedure is fundamental because the extent of the tumor influences the prognosis and treatment [22][33]. An accurate and early diagnosis is one of the main strategies for successful management of HNC; however, most head and neck tumors are locally advanced at the time of diagnosis, even though they can be easily detected by simple physical examinations [11][22][24]. An evaluation by a multidisciplinary team of the patients medical history, lifelong tobacco and alcohol consumption habits, and the past existence of other cancers and their treatments, including radiotherapy should be questioned [21][33].
The discovery and control of HNC is not always an easy task, since often the affected structures are not accessible to objective clinical examination, resulting in late presentation of the disease. Thus, if any symptom is suspected, an objective inspection of all the structures that may be involved should be performed, including examination of the oral cavity and oropharynx, palpation of the neck, and examination of suspect areas in the mouth. These patients should also undergo transnasal fiber optic endoscopy to examine the pharynx, larynx, and vocal cord structure [21]. Diagnosis will allow evaluating the prognosis of the patient, since it helps to establish the TNM (tumor, node, metastasis) profile of the tumor. TNM staging is commonly used to assess the stages of tumors of the head and neck (although variations are depending on the site of the primary tumor) (Table 1). The “T” classifies the extent of the primary tumor, the “N” refers to the infected regional lymph nodes (it is important to note that the lymphatic drainage of each head and neck subsite is different, so this must be assessed according to the location of the primary lesion), and the “M” refers to distant metastases. With this analysis, it is also possible to establish the stage of the disease, which can vary from 0 to IV, the latter being the stage with the worst prognosis [11][43].
Table 1. Tumor classification according to TNM profile [39][43]. T—extent of the primary tumor, N—infected regional lymph nodes, M—distant metastases.
| Stage | T | N | M |
|---|---|---|---|
| 0 | Defined shape | No invasion | No distant metastasis |
| I | Defined shape, less than 2 cm Does not invade the submucosa |
No invasion | No distant metastasis |
| II | Between 2 and 4 cm Initial invasion of the submucosa |
No invasion | No distant metastasis |
| III | Cancer cells rapidly divide Tumors with more than 4 cm |
Invasion | No distant metastasis |
| IV | Cancer cells enter the bloodstream | Invasion | Distant metastasis |
In recent years there has been great progress in defining the staging of head and neck tumors and therapeutic strategies. However, despite all the advances made in the various treatment modalities, the survival rate has remained almost unchanged in the last 25 years. Additionally, since head and neck anatomy are extremely complex, both being composed of several interconnected and interdependent structures, and since typically HNC occurs near structures that are important both at functional and cosmetic levels, early diagnosis is imperative [43].
Despite recent advances in diagnosis, approximately 70% of patients with head and neck squamous cell carcinoma (HNSCC) present with advanced-stage disease, frequently involving regional lymph nodes at the time of diagnosis, leading to high associated mortality. The 5-year survival rate is about 60% [26][28]. Late diagnosis usually implies that the cancer has infiltrated surrounding tissues and spread to the regional nodes, this sometimes being the only clinical manifestation. Furthermore, the invasion of surrounding structures allows the entrance of cancer cells into the bloodstream, which may enable the appearance of distant metastasis and secondary sites; however, distant metastases are not usually present (only in about 10% of patients) [11][24][28][44]. One of the biggest problems associated with this disease is its rate of recurrence. Approximately 50% to 60% of patients with localized HNSCC have their disease progress within two years after diagnosis, which drastically decreases the survival rate (from 80% to 50–35%—depending on the degree of disease progression). Patients with recurrent or metastatic disease have an estimated survival of less than one year [24][26].
Late diagnosis requires aggressive treatments with high morbidity. Most of the time, the treatment will compromise the organs necessary to perform simple functions, such as eating, breathing, and speaking [24][44]. Given the vital importance of the structures involved in these tumors, the therapeutic strategies adopted should not only aim to improve the survival rate, but also preserve the functions of the organs, indicating the need for a multidisciplinary approach [26][28]. The standard treatment for HNC involves surgical resection and radiotherapy (in combination or as isolated treatments) in early stages, and chemotherapy is used in advanced stages of the disease [26][38]. Concurrent chemo-radiation allows preservation of organ function, and it is the main treatment for tumors arising in the oropharynx, nasopharynx, laryngopharynx, and larynx. For oral cavity cancers, the highest cure rates are achieved by using surgical techniques with adjuvant or post-operative radiotherapy (associated or not with chemotherapy) [25]. Radiation therapy is also important in the control and palliation of symptoms in patients with advanced/incurable HNC, allowing tumor reduction, prevention of ulceration and bleeding, and pain control [45]. However, due to the complex anatomy of this region, the conventional approach is always limited, as the treatments can result in severe functional impairment. Surgical resection is usually inadequate due to anatomical limitations, so despite various attempts to improve the existing treatments, they still have severe side effects. The traditional surgical approach is always the preferred treatment, as it removes all macroscopic tumors, yet there is always the concern of having a microscopic disease. With this approach, it is common that malignant cells persist in the tissue margins adjacent to the surgery, meaning that microscopic disease is normally present in the margins of the surgical area, which is often associated with local recurrence and a poor prognosis. Thus, in most cases, radiotherapy is used as well. In addition, surgery can cause severe side effects, resulting in the loss of basic functions and the need for tracheostomy and/or gastrostomy. If a tumor invades the carotid artery or pre-vertebral tissues, it cannot be removed [21][38]. Therefore, if possible, radiation therapy is ideal, due to the reduction in the associated morbidity [38]. This type of treatment can be applied both to the primary tumor and to the lymphatic nodes, and can be referred to as organ-preserving therapy, with or without chemotherapy. However, although radiotherapy is a non-invasive treatment, it is not innocuous, as it can lead to both acute and chronic toxicity in normal tissues. For example, radiotherapy to the head and neck region may cause undesirable radiotherapy-induced changes in the surrounding tissues; and side effects such as oral mucositis, hyposalivation, loss of taste, dental caries, dysphagia, dermatitis (acute) osteoradionecrosis, vessels stenosis, hypothyroidism, hearing loss (late), and trismus, thereby negatively impacting the patient’s quality of life [25][38].
Regarding chemotherapy, this treatment modality is not usually employed as an isolated treatment for HNC, but it can still be used in some scenarios and for different purposes [11][38]:
-
Radiation enhancement/synchronous chemotherapy, used in conjunction with radiation therapy—reduces the risk of lymph node metastasis;
-
Neo-adjuvant/induction chemotherapy—to reduce the tumor’s size before the main treatment;
-
Adjuvant—acts on the small lesions that cannot be removed by surgery, reduces the recurrence rate, and improves the survival rate;
-
Palliation—for distant metastases.
Currently, the main chemotherapy drugs used in the treatment of HNC are antimetabolites, platinum compounds, taxanes like fluorouracil (5-FU), methotrexate (MTX), cisplatin, carboplatin, docetaxel, and paclitaxel; and various therapeutic protocols can be used, depending on the evolution and stage of the disease [11][46]. One of the standard combinations used to treat recurrent/metastatic HNC non-expressing PD-L1 is cisplatin/5-FU/cetuximab, which allows an increase in the speed of response to therapy, although toxicity may also be higher than the alternatives [47][48]. In the case of chemotherapy induction, the standard regime is cisplatin (100 mg/m2) on days 1, 22, and 43 of concomitant radiotherapy [48]. The combination of docetaxel (75 mg/m2) and low doses of cisplatin (75 mg/m2) and 5-fluorouracil (750 mg/m2) each day, for five consecutive days, is also used. This strategy has been shown to reduce the progression of distant metastases, particularly in high-risk patients [49]. In addition to these classes of compounds, EGFR inhibitors have also emerged as a new treatment strategy for HNC. Another method is using certain antibodies that can recognize receptors in cancer cell membranes, leading targeted cell death. Cetuximab (2006) was the first monoclonal antibody to be approved, and demonstrated considerably improved overall survival in patients with locally advanced and recurrent or metastatic HNC tumors. It also showed the role EGFR signaling pathways play in the treatment of HNC [28][32][50]. Nivolumab (2016) was the second antibody approved by the Food and Drug Administration (FDA) for cases of metastatic or recurrent HNSCC. More recently, in 2019, came the approval of pembrolizumab as a first-line treatment for patients with unresectable metastatic or recurrent HNSCC [50].
When used in combination with radiotherapy, drugs have more severe and longer lasting side effects than when used alone. This strategy is used for advanced tumors and is reported to be superior to surgery or radiotherapy alone by 6–8% in terms of 5 year survival [21][25][51]. Despite the advances made recently, focused on advanced treatments to preserve organ function and improve quality of life, most of these treatments for HNC have low efficacy, detrimental side effects, and associated morbidities, such as systemic toxicity and cosmetic damage due to lack of selectivity of the therapeutic agents and the invasiveness of the surgical procedures [26][52]. Most of the chemotherapy drugs lack specificity to tumor cells. Thus, they have negative effects on healthy cells as well, resulting in severe side effects, and usually, the concentration of drug achievable at the target is limited, resulting in suboptimal treatment [11][52]. In this sense, the patients subjected to these therapies require significant support from a multidisciplinary team for psychological and physical rehabilitation, including a speech and language therapist, a dietician, a restorative dentist, and a hygienist. However, after treatment, some side effects can persist, despite intensive rehabilitation. A small number of patients never return to a normal oral diet [21]. For that reason, research has been focusing on targeted cancer therapies to prevent these effects.
The progress in immune checkpoint inhibitors (ICIs) for HNC came to change the therapeutic landscape of the disease, and led to a remarkable benefit for some patients. These are a class of drugs that bind to proteins present in cell membranes that are produced by immune cells such as T cells and some cancer cells, having the ability to block them. These proteins function as checkpoints, allowing the differentiation of self from non-self antigens, and when blocked, can facilitate signaling and mobilization for cell death by circulating T-lymphocytes. The main checkpoint proteins found in cancer cells involved in this type of response in HNC are PD-L1 and CTLA-4 [50]. This therapeutic strategy in combination with other conventional ones, can generate long-lasting immune responses and may lead to a significant improvement in therapeutic efficacy and survival in patients with advanced HNC. An example of this therapeutic approach is pembrolizumab, an anti-PD-L1 antibody that has been approved as a first-line treatment for patients with recurrent or metastatic HNC. It was beneficial in some cases; however, only 20% of patients with advanced HNC who received it showed effective responses [53]. Additionally, the efficacy of these monoclonal antibody therapies is higher when the patients have PD-L1-expressing tumors, making this therapeutic quite specific [48]. Additionally, the majority of patients present primary resistance to ICIs, have several immune-related adverse events, and do not benefit from the use of these agents, emphasizing the need for developing predictive biomarkers to better determine who will benefit from treatment with ICIs and to reduce severe systemic toxicity [53][54].
Moreover, like conventional cytotoxic therapy, immunotherapy is hampered by transport problems. Additionally, this difficulty of membrane permeation into solid tumor tissue by an ICI compromises the efficacy of such therapy. Therefore, it is urgently necessary to optimize the transport strategy of these therapeutic agents, namely, by using nanotechnology, as it can allow the transport of drugs selectively into tumor tissue, minimize toxicity in healthy tissues, and reduce immune-related side events.
Overall, given the drawbacks of these conventional treatments, the need has arisen to develop strategies and innovative technologies that improve the efficacy and safety of HNC therapies while reducing adverse effects and resistances.
4. Nanomedicine as a Therapeutic Approach for HNC
Nanotechnology is a multifaceted science that combines research fields (Figure 3). Its uses in healthcare come under nanomedicine, which uses molecular tools for diagnostic procedures, treatment, and prevention of diseases (e.g., cancer) [55][56]. Like nanotechnology, the definition of nanomedicine is not agreed on. It concerns a range of particles, which should be at the nanoscale, between 1 and 1000 nm [56][57][58][59][60]. The goals of nanomedicine are common to those of traditional medicine, being earlier diagnosis, the development of non-invasive and effective treatments, and the minimization of side effects [57].
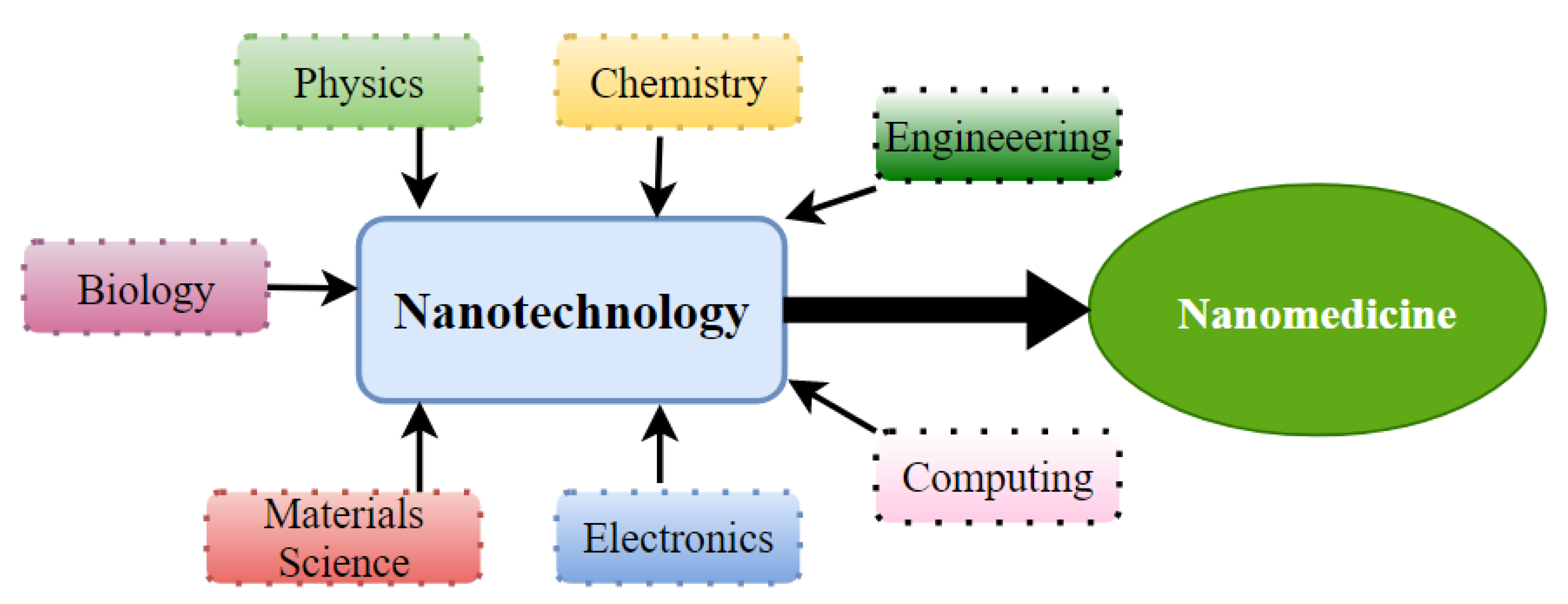
Figure 3. Scientific areas contributing to the use of nanotechnology to nanomedicine.
Nanomedicine offers not only improvements for existing techniques, but also the possibility of developing new techniques with superior efficacy, by manipulating drugs at the molecular level and altering their physicochemical properties, such as their solubility and permeability; or facilitating their sustained or controlled delivery [56][57]. Nanotechnology can be applied in numerous fields of medicine, such as imaging [61], drug delivery [62], DNA sequencing [63], and tissue engineering [64]. The arising of applications of nanotechnology in medicine offers new chances for dealing with the problems associated with commonly used therapies. Regardless of its varied applications in medicine, nanotechnology has played a particularly key role in the delivery of drugs using nanocarriers. With cancer being one of the most common diseases worldwide, the treatment of cancer has been the purpose of several studies in the field of nanomedicine. Most have aimed to improve of the existing treatments and at the same time reduce the associated side effects. This type of nanotechnology-based approach improves the overall efficacy and safety of drugs and increases the effectiveness of therapy, as it will condition their variable factors, such as pharmacokinetics, toxicity, targeted delivery, and stability [65].
The Nanomedicine Strategic Research and Innovation Agenda (2016–2030) from the European Nanomedicine Community points out the current needs for cancer treatment [65]:
-
The improvement of diagnosis and the development of novel strategies for early detection of tumors, circulating tumor cells, and metastases;
-
The improvement of treatment of solid tumors and chemo-resistant tumors;
-
The improved precision and efficacy of radiotherapy, immunotherapy, photodynamic, individualized, and hyperthermia therapies
-
Lower side effects through more targeted chemotherapy.
Therefore, nanomedicine enables us to address these challenges and improve cancer treatment. One of the main reasons for conventional HNC treatments’ failures is the inefficiency of the doses of therapeutic agents, as little of most drugs effectively reaches the tumor, partly caused by the dose-limiting profile of healthy tissues. The controlled delivery of drugs has become increasingly important because of the advantages that this can bring to the pharmaceutical industry and cancer treatment: it allows a higher drug concentration to reach the tumor cells, while reducing the dose of the drug being administered [26][66]. Drug delivery to a specific location has emerged as an approach to overcome the disadvantages of conventional treatments of HNC, including the absence of specificity of conventional cytotoxic agents [66][67][68][69]. The development of nanocarriers should consider several particulars—namely, influence the properties of the drug, such as its solubility, release profile, bioavailability, and immunogenicity. In addition, it allows overcoming barriers, such as phagocytic mononuclear opsonization. Additionally, four factors are taken into account for the effective design of nanocarriers, namely, the retention of drug-loaded nanoparticles inside of the human body, the escape from the immune system, the ability to reach the target site, and the effective release of the drug at the target site [13].
In addition, with drug delivery, resistance to drugs is avoided [70]. Nanocarriers are a class of versatile materials that can function as drug carriers, diagnostic agents, or targeting ligands for HNC. These carriers can improve the properties of drugs and their pharmacokinetics, depending on the intended use [66][68]. In addition, the use of nanocarriers for controlled delivery allows, due to their reduced size, overcoming certain biological barriers, such as the blood–brain barrier; however, they must fulfill some requirements to be used in HNC [66].
Firstly, the size and size distribution are among the most important characteristics of nanocarriers, as they are determinant factors for distribution, toxicity, circulation time, in vivo behavior, and targeting to the HNC [26][52]. The nanocarrier’s size is important because most of the properties of nanomaterials are owed to their small size, which gives them proportionally large surface areas, which is critical for drug delivery purposes [52]. Moreover, larger particles are recognized and cleared by the liver and the reticuloendothelial system [52], though this rapid clearance can be avoided through the use of polyethylene glycol (PEG) [26][52]. The small size can also facilitate cellular uptake; and small particles have large surface areas, exposing the bioactive molecules, leading to faster drug release [26].
Dosage is also an extremely important feature when addressing the targeting of drugs. Drugs used in HNC therapy are administered systemically, which requires high dosages and causes side effects in healthy tissues. Thus, it is necessary to decrease the drug concentration in healthy tissues without compromising the therapeutic dose at the site of interest. This reduction can be achieved by localized release of the drug in the target using controlled drug delivery systems [66][68][69]. With the progress made by studies on tumor biology, genetics, and nanotechnology, medicine has become more personalized. Great variability in therapeutic responses has been shown between patients and between head and neck tumors. In this sense, to personalize the therapeutic approach to be taken with each user, it is essential to understand the biological mechanisms related to the distribution and retention of nanocarriers in tumors [48][71].
Nanocarriers can be led to the site of interest by passive or active targeting strategies. These strategies aim at increasing the drug concentration in the target cells of HNC, retention, and reducing the toxicity to healthy/systemic tissues [67][68]. While passive targeting takes advantage of the unique characteristics of tumor pathophysiology to drive the nanocarriers to the site of interest without any stimulus or ligand, active targeting makes use of molecules or ligands specific to the site of interest (Figure 4).
4.1. Passive Targeting
Passive targeting consists of the systemic injection of the nanocarriers that will accumulate preferentially at the site of interest due to the enhanced permeability and retention (EPR) effect (Figure 4) [11]. It depends on the size of the nanocarriers, and if they are small enough, usually less than 100 nm, their circulation is extravagated through vascular defects typically present at the tumor sites due to accelerated angiogenesis. They should reach hepatic and spleen macrophages [68][70]. This passive targeting effect is also dependent on the degree of vascularization of the tumor, the porosity of the vessels, and the sizes of the pores on the vessels (which vary with the type and stage of the tumor). Solid tumors frequently have leaky vasculature in relation to normal tissues. There is also abnormal lymphatic drainage around these tumors. All these factors promote increased EPR by passive nanocarriers [72][73]. Thus, passive targeting depends on the anatomophysiological conditions of the target. The high production of blood vessels that occurs in tumoral tissues to promote their rapid growth allows carriers such as nanoparticles to be easily retained and accumulated in the tumor tissues [17].
The EPR effect can be influenced by a complex set of tumor microenvironment (TME) factors, including tumor characteristics, stage, vasculature, stroma, macrophages, lymphatics, and interstitial fluid pressure, and is therefore difficult to predict [17][74]. Concerning the EPR effect, there are still often some discrepancies between the experiences in animal models used for in vivo assays and human clinical treatments. There are significant size differences between mice and humans, and consequently, the pharmacokinetic profiles and the pharmacodynamic properties of the drugs in the tumors also differ. In this respect, larger animal models may be better benchmarks for estimating the EPR effects of targeted delivery of anti-cancer drugs in humans. Furthermore, in animal models, namely, xenograft mouse models, the effect of EPR may vary between formed and spontaneously implanted tumors. In the case of implanted tumors, the vessels are highly leaky and exaggerate the preferential leakage on nanodrugs. Thus, sometimes good results on solid tumors from animal models can result in disappointing effects when applied to humans, so it is important to take this effect into account when applying a candidate drug to humans [74][75].
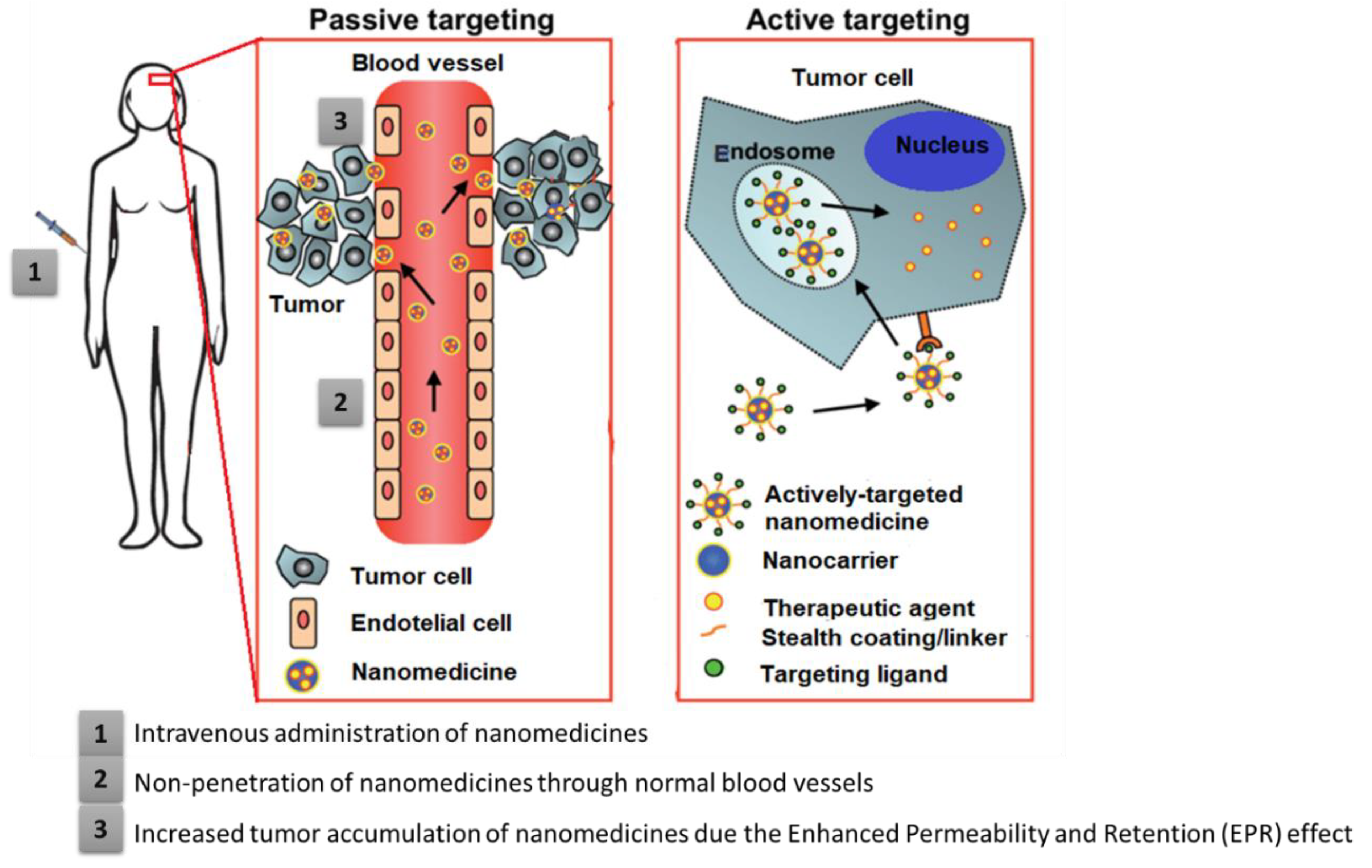
Figure 4. Drug delivery systems’ strategies for anticancer therapy: passive targeting and active targeting. Adapted with permission from [76].
4.2. Active Targeting
Active targeting consists of the use of a mechanism to increase the specificity of the nanocarrier to the action site [11]. One of the most effective ways to improve the specificity of a nanocarrier is by increasing its affinity for the target by using a molecule with the ability to recognize and bind to the target tissue [77]. Nanoparticles can be given an extra degree of tumor specificity by combining them with targeting ligands. These ligands bind to the nanoparticles’ target during tumor diagnosis/treatment once they interact with the right receptors on the surfaces of the target cells (are tumor-specific markers), and as a result of this interaction, cell internalization occurs [71][76]. All nanoparticles are capable of conjugation with target ligands, such as antibodies/antibody fragments, nucleic acids, sugars, vitamins, peptides, aptamers, and others small molecules. For example, to ensure that adequate contrast is provided during surgery in HNC, the target of interest must be highly expressed while expression is minimal in surrounding healthy tissues. The functionalization of nanoparticles has the advantage of leaving the fluorophore chemically unchanged, thereby limiting the possibility of optical performance [17].
The major challenge of active targeting is the choice of targeting agent to avoid toxicity to healthy cells. Another type of active targeting is the so-called physical/triggered targeting, which consists of the use of internal (pH, redox potential, enzymes) or external stimuli (UV light, temperature, ultrasound) to drive the nanocarrier to the site of interest before triggering the release of the drug [78][79]. This targeting is especially notable in the form of magnetic nanocarriers, which can be guided to the site of interest through an external magnetic field.
References
- World Health Organization (WHO)—Cancer Control Programme. Available online: http://www.who.int/cancer/en/ (accessed on 20 December 2021).
- Nayak, M.G.; George, A.; Vidyasagar, M.S.; Mathew, S.; Nayak, S.; Nayak, B.S.; Shashidhara, Y.N.; Kamath, A. Quality of life among cancer patients. Indian J. Palliat. Care 2017, 23, 445.
- Weizman, B.; Golan, N.; Ronen, O. Effect of socioeconomic status on survival in patients with head and neck cancer. Head Neck 2021, 43, 3001–3009.
- Sung, H.; Ferlay, J.; Siegel, R.L.; Laversanne, M.; Soerjomataram, I.; Jemal, A.; Bray, F. Global Cancer Statistics 2020: GLOBOCAN Estimates of Incidence and Mortality Worldwide for 36 Cancers in 185 Countries. CA Cancer J. Clin. 2021, 71, 209–249.
- Fidler, M.M.; Bray, F.; Soerjomataram, I. The global cancer burden and human development: A review. Scand. J. Public Health 2018, 46, 27–36.
- Rezende, T.M.; de Souza Freire, M.; Franco, O.L. Head and neck cancer: Proteomic advances and biomarker achievements. Cancer 2010, 116, 4914–4925.
- American Cancer Society. Cancer Facts & Figures. 2021. Available online: https://www.cancer.org/research/cancer-facts-statistics/all-cancer-facts-figures/cancer-facts-figures-2021.html (accessed on 11 January 2022).
- Birkeland, A.C.; Swiecicki, P.L.; Brenner, J.C.; Shuman, A.G. A review of drugs in development for the personalized treatment of head and neck squamous cell carcinoma. Expert Rev. Precis. Med. Drug Dev. 2016, 1, 379–385.
- de Lázaro, I.; Mooney, D.J. Obstacles and opportunities in a forward vision for cancer nanomedicine. Nat. Mater. 2021, 20, 1469–1479.
- Kurmi, B.D.; Patel, P.; Paliwal, R.; Paliwal, S.R. Molecular approaches for targeted drug delivery towards cancer: A concise review with respect to nanotechnology. J. Drug Deliv. Sci. Technol. 2020, 57, 101682.
- Zhao, Y.; Chen, H.; Chen, X.; Hollett, G.; Gu, Z.; Wu, J.; Liu, X. Targeted nanoparticles for head and neck cancers: Overview and perspectives. WIREs Nanomed. Nanobiotechnology 2017, 9, e1469.
- Gharat, S.A.; Momin, M.M.; Bhavsar, C. Oral squamous cell carcinoma: Current treatment strategies and nanotechnology-based approaches for prevention and therapy. Crit. Rev. Ther. Drug Carr. Syst. 2016, 33, 363–400.
- Bharadwaj, R.; Medhi, S. Oral squamous cell carcinoma and the cutting edge of nanotechnology. Multidiscip. Cancer Investig. 2020, 4, 36–45.
- El-Say, K.M.; El-Sawy, H.S. Polymeric nanoparticles: Promising platform for drug delivery. Int. J. Pharm. 2017, 528, 675–691.
- Dinda, S.C.; Pattnaik, G. Nanobiotechnology-based drug delivery in brain targeting. Curr. Pharm. Biotechnol. 2013, 14, 1264–1274.
- Chen, X.-J.; Zhang, X.-Q.; Liu, Q.; Zhang, J.; Zhou, G. Nanotechnology: A promising method for oral cancer detection and diagnosis. J. Nanobiotechnology 2018, 16, 52.
- Wojtynek, N.E.; Mohs, A.M. Image-guided tumor surgery: The emerging role of nanotechnology. Wiley Interdiscip. Rev. Nanomed. Nanobiotechnol. 2020, 12, e1624.
- Mooore, K.L.; Dalley, A.F.; Agur, A.M.R. Moore Clinically Oriented Anatomy; Wolters Kluwer India Pvt Ltd.: Gurugram, India, 2017.
- Betts, J.G.; Y, K.A.; Wise, J.A.; Johnson, E.; Poe, B.; Kruse, D.H.; Korol, O.; Johnson, J.E.; Womble, M.; DeSaix, P. 7.2 The skull. In Anatomy and Physiology; OpenStax: Houston, TX, USA, 2013.
- Roesch, Z.K.; Tadi, P. Anatomy of head and neck. In StatPearls; StatPearls Publishing Copyright © 2020; StatPearls Publishing LLC.: Treasure Island, FL, USA, 2020.
- Lester, S.; Yang, W.-Y. Principles and management of head and neck cancer. Surgery 2012, 30, 617–623.
- Crozier, E.; Sumer, B.D. Head and neck cancer. Med. Clin. N. Am. 2010, 94, 1031–1046.
- Bose, P.; Brockton, N.T.; Dort, J.C. Head and neck cancer: From anatomy to biology. Int. J. Cancer 2013, 133, 2013–2023.
- Johnson, N.W.; Amarasinghe, H.K. Epidemiology and aetiology of head and neck cancers. In Head and Neck Cancer; Bernier, J., Ed.; Springer: Geneva, Switzerland, 2011; p. 730.
- Kawashita, Y.; Soutome, S.; Umeda, M.; Saito, T. Oral management strategies for radiotherapy of head and neck cancer. Jpn. Dent. Sci. Rev. 2020, 56, 62–67.
- Wu, T.T.; Zhou, S.H. Nanoparticle-based targeted therapeutics in head-and-neck cancer. Int. J. Med. Sci. 2015, 12, 187–200.
- Gao, S.; Zheng, M.; Ren, X.; Tang, Y.; Liang, X. Local hyperthermia in head and neck cancer: Mechanism, application and advance. Oncotarget 2016, 7, 57367–57378.
- Argiris, A.; Karamouzis, M.V.; Raben, D.; Ferris, R.L. Head and neck cancer. Lancet 2008, 371, 1695–1709.
- Pezzuto, F.; Buonaguro, L.; Caponigro, F.; Ionna, F.; Starita, N.; Annunziata, C.; Buonaguro, F.M.; Tornesello, M.L. Update on head and neck cancer: Current knowledge on epidemiology, risk factors, molecular features and novel therapies. Oncology 2015, 89, 125–136.
- Aupérin, A. Epidemiology of head and neck cancers: An update. Curr. Opin. Oncol. 2020, 32, 178–186.
- Hashibe, M.; Brennan, P.; Chuang, S.C.; Boccia, S.; Castellsague, X.; Chen, C.; Curado, M.P.; Maso, L.D.; Daudt, A.W.; Fabianova, E.; et al. Interaction between tobacco and alcohol use and the risk of head and neck cancer: Pooled analysis in the international head and neck cancer epidemiology consortium. Cancer Epidemiol. Biomark. Prev. 2009, 18, 541–550.
- Chan, J.Y.K.; Zhen, G.; Agrawal, N. The role of tumor DNA as a diagnostic tool for head and neck squamous cell carcinoma. Semin. Cancer Biol. 2019, 55, 1–7.
- Zolkind, P.; Lee, J.J.; Jackson, R.S.; Pipkorn, P.; Massa, S.T. Untreated head and neck cancer: Natural history and associated factors. Head Neck 2021, 43, 89–97.
- Chua, M.L.K.; Wee, J.T.S.; Hui, E.P.; Chan, A.T.C. Nasopharyngeal carcinoma. Lancet 2016, 387, 1012–1024.
- Smatti, M.K.; Al-Sadeq, D.W.; Ali, N.H.; Pintus, G.; Abou-Saleh, H.; Nasrallah, G.K. Epstein–Barr virus epidemiology, serology, and genetic variability of LMP-1 oncogene among healthy population: An update. Front. Oncol. 2018, 8, 211.
- Al-Thawadi, H.; Gupta, I.; Jabeen, A.; Skenderi, F.; Aboulkassim, T.; Yasmeen, A.; Malki, M.I.; Batist, G.; Vranic, S.; Al Moustafa, A.-E. Co-presence of human papillomaviruses and Epstein–Barr virus is linked with advanced tumor stage: A tissue microarray study in head and neck cancer patients. Cancer Cell Int. 2020, 20, 361.
- Sroussi, H.Y.; Jessri, M.; Epstein, J. Oral assessment and management of the patient with head and neck cancer. Oral Maxillofac. Surg. Clin. 2018, 30, 445–458.
- Sahovaler, A.; Yeh, D.H.; Fung, K. General principles of head and neck cancer treatment. In Clinical Care and Rehabilitation in Head and Neck Cancer; Doyle, P.C., Ed.; Springer Nature: Cham, Switzerland, 2019; pp. 3–14.
- Vokes, E.E.; Weichselbaum, R.R.; Lippman, S.M.; Hong, W.K. Head and neck cancer. N. Engl. J. Med. 1993, 328, 184–194.
- Heroiu Cataloiu, A.-D.; Danciu, C.E.; Popescu, C.R. Multiple cancers of the head and neck. Maedica 2013, 8, 80–85.
- Chen, S.-W.; Li, S.-H.; Shi, D.-B.; Jiang, W.-M.; Song, M.; Yang, A.-K.; Li, Y.-D.; Bei, J.-X.; Chen, W.-K.; Zhang, Q. Expression of PD-1/PD-L1 in head and neck squamous cell carcinoma and its clinical significance. Int. J. Biol. Markers 2019, 34, 398–405.
- Lenouvel, D.; González-Moles, M.; Talbaoui, A.; Ramos-García, P.; González-Ruiz, L.; Ruiz-Ávila, I.; Gil-Montoya, J.A. An update of knowledge on PD-L1 in head and neck cancers: Physiologic, prognostic and therapeutic perspectives. Oral Dis. 2020, 26, 511–526.
- Stepnick, D.; Gilpin, D. Head and Neck Cancer—An Overview. Semin. Plast. Surg. 2009, 24, 107–116.
- Panikkanvalappil, S.R.; El-Sayed, M.A.; El-sayed, I.H. Advances in nanomedicine for head and neck cancer. In Head and Neck Cancer; Bernier, J., Ed.; Springer: Cham, Switzerland, 2016; pp. 827–843.
- Ordoñez, R.; Otero, A.; Jerez, I.; Medina, J.A.; Lupiañez-Pérez, Y.; Gomez-Millan, J. Role of radiotherapy in the treatment of metastatic head and neck cancer. OncoTargets Ther. 2019, 12, 677–683.
- Guidi, A.; Codecà, C.; Ferrari, D. Chemotherapy and immunotherapy for recurrent and metastatic head and neck cancer: A systematic review. Med. Oncol. 2018, 35, 37.
- Saloura, V.; Langerman, A.; Rudra, S.; Chin, R.; Cohen, E.E.W. Multidisciplinary care of the patient with head and neck cancer. Surg. Oncol. Clin. N. Am. 2013, 22, 179–215.
- Machiels, J.P.; René Leemans, C.; Golusinski, W.; Grau, C.; Licitra, L.; Gregoire, V. Squamous cell carcinoma of the oral cavity, larynx, oropharynx and hypopharynx: Clinical practice guidelines for diagnosis, treatment and follow-up. Ann. Oncol. 2020, 31, 1462–1475.
- Ferrari, D.; Ghi, M.G.; Franzese, C.; Codecà, C.; Gau, M.; Fayette, J. The slippery role of induction chemotherapy in head and neck cancer: Myth and reality. Front. Oncol. 2020, 10, 7.
- Al Qaraghuli, M.M. Biotherapeutic antibodies for the treatment of head and neck cancer: Current approaches and future considerations of photothermal therapies. Front. Oncol. 2020, 10, 2710.
- Herskovic, A.; Martz, K.; Al-Sarraf, M.; Leichman, L.; Brindle, J.; Vaitkevicius, V.; Cooper, J.; Byhardt, R.; Davis, L.; Emami, B. Combined Chemotherapy and Radiotherapy Compared with Radiotherapy Alone in Patients with Cancer of the Esophagus. N. Engl. J. Med. 1992, 326, 1593–1598.
- Pillai, G. Nanotechnology Toward Treating Cancer. In Applications of Targeted Nano Drugs and Delivery Systems; Elsevier Inc.: Amsterdam, The Netherlands, 2019; pp. 221–256.
- Xu, Q.; Fang, M.; Zhu, J.; Dong, H.; Cao, J.; Yan, L.; Leonard, F.; Oppel, F.; Sudhoff, H.; Kaufmann, A.M. Insights into nanomedicine for immunotherapeutics in squamous cell carcinoma of the head and neck. Int. J. Biol. Sci. 2020, 16, 2506.
- Napolitano, M.; Schipilliti, F.M.; Trudu, L.; Bertolini, F. Immunotherapy in head and neck cancer: The great challenge of patient selection. Crit. Rev. Oncol./Hematol. 2019, 144, 102829.
- Wilczewska, A.Z.; Niemirowicz, K.; Markiewicz, K.H.; Car, H. Nanoparticles as drug delivery systems. Pharmacol. Rep. 2012, 64, 1020–1037.
- Chang, E.H.; Harford, J.B.; Eaton, M.A.W.; Boisseau, P.M.; Dube, A.; Hayeshi, R.; Swai, H.; Lee, D.S. Nanomedicine: Past, present and future—A global perspective. Biochem. Biophys. Res. Commun. 2015, 468, 511–517.
- Caruthers, S.D.; Wickline, S.A.; Lanza, G.M. Nanotechnological applications in medicine. Curr. Opin. Biotechnol. 2007, 18, 26–30.
- Soares, S.; Sousa, J.; Pais, A.; Vitorino, C. Nanomedicine: Principles, properties, and regulatory issues. Front. Chem. 2018, 6, 360.
- Muthu, M.S.; Mei, L.; Feng, S.-S. Nanotheranostics: Advanced nanomedicine for the integration of diagnosis and therapy. Nanomedicine 2014, 9, 1277–1280.
- Astruc, D. Introduction to Nanomedicine. Molecules 2016, 21, 4.
- Cormode, D.P.; Skajaa, T.; Fayad, Z.A.; Mulder, W.J.M. Nanotechnology in medical imaging: Probe design and applications. Arterioscler. Thromb. Vasc. Biol. 2010, 29, 992–1000.
- Rodrigues, A.R.O.; Almeida, B.G.; Rodrigues, J.M.; Queiroz, M.J.R.P.; Calhelha, R.C.; Ferreira, I.C.F.R.; Pires, A.; Pereira, A.M.; Araújo, J.P.; Coutinho, P.J.G.; et al. Magnetoliposomes as carriers for promising antitumor thienopyridin-7-arylamines: Photophysical and biological studies. R. Soc. Chem. Adv. 2017, 7, 15352–15361.
- Clarke, J.; Wu, H.C.; Jayasinghe, L.; Patel, A.; Reid, S.; Bayley, H. Continuous base identification for single-molecule nanopore DNA sequencing. Nat. Nanotechnol. 2009, 4, 265–270.
- Danie Kingsley, J.; Ranjan, S.; Dasgupta, N.; Saha, P. Nanotechnology for tissue engineering: Need, techniques and applications. J. Pharm. Res. 2013, 7, 200–204.
- Nanomedicine, E.T.P. Nanomedicine Strategic Research & Innovation Agenda 2016–2030: Creating Junctions for Healthcare; European Technology Platform for Nanomedicine: Paris, France, 2016; pp. 1–31.
- Caban, S.; Aytekin, E.; Sahin, A.; Capan, Y. Nanosystems for drug delivery. OA Drug Des. Deliv. 2014, 2, 1–7.
- Cho, K.; Wang, X.; Nie, S.; Chen, Z.; Shin, D.M. Therapeutic nanoparticles for drug delivery in cancer. Clin. Cancer Res. 2008, 14, 1310–1316.
- Nikalje, A.P. Nanotechnology and its applications in medicine. Med. Chem. 2015, 5, 81–89.
- Pradhan, P.; Giri, J.; Rieken, F.; Koch, C.; Mykhaylyk, O.; Döblinger, M.; Banerjee, R.; Bahadur, D.; Plank, C. Targeted temperature sensitive magnetic liposomes for thermo-chemotherapy. J. Control. Release 2010, 142, 108–121.
- Aghebati-Maleki, A.; Dolati, S.; Ahmadi, M.; Baghbanzhadeh, A.; Asadi, M.; Fotouhi, A.; Yousefi, M.; Aghebati-Maleki, L. Nanoparticles and cancer therapy: Perspectives for application of nanoparticles in the treatment of cancers. J. Cell. Physiol. 2020, 235, 1962–1972.
- Bertrand, N.; Wu, J.; Xu, X.; Kamaly, N.; Farokhzad, O.C. Cancer nanotechnology: The impact of passive and active targeting in the era of modern cancer biology. Adv. Drug Deliv. Rev. 2014, 66, 2–25.
- Peer, D.; Karp, J.M.; Hong, S.; Farokhzad, O.C.; Margalit, R.; Langer, R. Nanocarriers as an emerging platform for cancer therapy. Nat. Nanotechnol. 2007, 2, 751–760.
- Khan, D.R. The use of nanocarriers for drug delivery in cancer therapy. J. Cancer Sci. Ther. 2010, 2, 58–62.
- Inagaki, F.F.; Furusawa, A.; Choyke, P.L.; Kobayashi, H. Enhanced nanodrug delivery in tumors after near-infrared photoimmunotherapy. Nanophotonics 2019, 8, 1673–1688.
- Wu, J. The Enhanced Permeability and Retention (EPR) Effect: The Significance of the Concept and Methods to Enhance Its Application. J. Pers. Med. 2021, 11, 771.
- van der Meel, R. Targeted Inhibition of Tumor Growth and Angiogenesis. Ph.D. Thesis, Universiteit Utrecht, Utrecht, The Netherlands, 2013.
- Torchilin, V.P. Passive and active drug targeting: Drug delivery to tumors as an example. In Handbook of Experimental Pharmacology, 197th ed.; Springer: Berlin, Germany, 2010; Volume 197, pp. 3–53.
- Kudr, J.; Haddad, Y.; Richtera, L.; Heger, Z.; Cernak, M.; Adam, V.; Zitka, O. Magnetic nanoparticles: From design and synthesis to real world applications. Nanomaterials 2017, 7, 243.
- Pawar, P.V.; Domb, A.J.; Kumar, N. Systemic targeting systems. In Advances in Delivery Science and Technology; Springer: Boston, MA, USA, 2014; pp. 61–91.
More
Information
Subjects:
Oncology
Contributor
MDPI registered users' name will be linked to their SciProfiles pages. To register with us, please refer to https://encyclopedia.pub/register
:
View Times:
738
Revisions:
2 times
(View History)
Update Date:
18 Mar 2022
Notice
You are not a member of the advisory board for this topic. If you want to update advisory board member profile, please contact office@encyclopedia.pub.
OK
Confirm
Only members of the Encyclopedia advisory board for this topic are allowed to note entries. Would you like to become an advisory board member of the Encyclopedia?
Yes
No
${ textCharacter }/${ maxCharacter }
Submit
Cancel
Back
Comments
${ item }
|
More
No more~
There is no comment~
${ textCharacter }/${ maxCharacter }
Submit
Cancel
${ selectedItem.replyTextCharacter }/${ selectedItem.replyMaxCharacter }
Submit
Cancel
Confirm
Are you sure to Delete?
Yes
No





