Video Upload Options
Foot-and-mouth disease (FMD) is an acute and highly contagious disease affecting the cloven-hoofed animals, such as pigs and cattle. The pathogen that causes FMD is known as FMD virus (FMDV), a single-stranded positive-sense RNA virus that is classified into the genus Aphthovirus in the family Picornaviridae.
1. Introduction
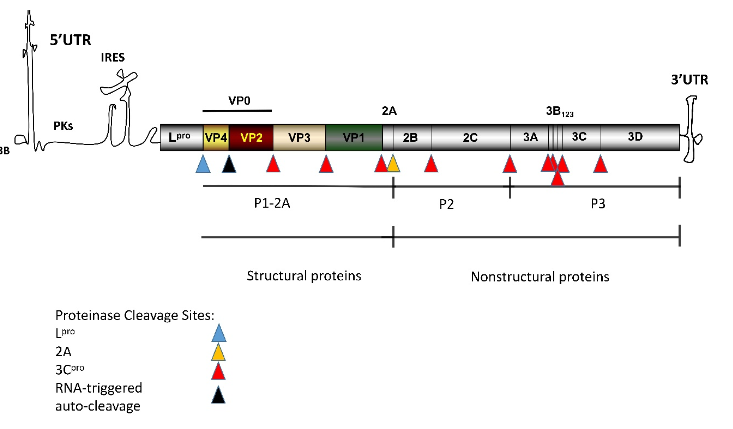
Foot-and-mouth disease (FMD) is an acute and highly contagious disease affecting thecloven-hoofed animals, such as pigs and cattle. The pathogen that causes FMD is known as FMD virus(FMDV), a single-stranded positive-sense RNA virus that is classified into the genus Aphthovirus inthe family Picornaviridae [1][2]. The pathogen causes vesicular disease of mouth and feet in susceptibleanimals [3]. The high mutation rate of the genome of FMDV and the rapid proliferation has led to therapid evolution of the virus and the formation of seven main serotypes [4][5][6]. The antigenic diversityamong the serotypes poses challenges to the research of efficient and cross-protective vaccines [8].The genome of FMDV contains an open reading frame (ORF) that encodes a polyprotein precursor,and it is cleaved into four structural proteins and 10 non-structural proteins by viral autoproteases andhost protease [7](Figure 1).Upon infection of the host, a virus will face the attack from the host’s immune response. In thelong-term battle with the host immune response, the virus has evolved and developed a series ofimmune escape mechanisms to overcome the killing and inhibition from the host immune system.The mechanism of virus immune escape can be divided into three categories: (1) enable the virus toavoid the recognition of humoral immune response; (2) interfere with the function of cellular immuneresponse; (3) interfere with the host’s immune response to the virus [8]. All these strategies would beexploited by the virus for replication and spreading to other hosts.As a highly contagious and fast-spreading virus, FMDV has multiple ways to evade the killing bythe immune system [9], which makes it difficult for controlling the virus. Viral capsid protein VP1 and leading protein Lpro can inhibit the production of interferon (IFN) and innate immune responseby interacting with soluble resistance-related calcium-binding protein (sorcin) or host transcriptionfactor ADNP [10][11]. Recently, new mechanisms and functions of FMDV proteins inhibiting innateimmunity have been discovered. DDX56 (a kind of RNA helicase), participate in RNA metabolism andribosome synthesis is reported to involve in this new mechanism. The interaction between FMDV3A and DDX56 suppresses the host innate immunity by reducing the phosphorylation of IRF3 [12].In addition, nucleotide-binding oligomerization domain 2 (NOD2), a member of the nucleotide-bindingoligomerization domain-like receptor (NLR) family [13], activates the NF-κB and IFN-βsignalingpathways during FMDV infection and inhibits the replication of FMDV in infected cells [14]. FMDV 2B,2C, and 3Cproinhibit the expression of NOD2 protein, which antagonizes the antiviral response [14].Reportedly, multiple structural and non-structural proteins of FMDV escape the killing of the hostimmune system. This review summrized the molecular mechanisms of immune evasion caused byFMDV proteins. The present study aimed to fill the gaps of knowledge on FMDV immune evasionmechanism, providing the basis for the prevention and control strategies for FMDV.
Figure 1.Schematic of the genome and polypeptide processing of FMDV [2][7]. The FMDV genomecontains an ORF of about 7 kbp, indicated by the shaded rectangle. Each region within the ORFrectangle represents a single protein. The flank of ORF is a long 5′untranslated region (5′-UTR) and ashort 3′-UTR. 3B covalently binds to the 5′-end.
2. Prospects and Future Directions
FMDV is a highly contagious virus that infects almost all cloven-hoofed animals, showingvesicles on the foot and mouth, skin erosion on the mucous membranes, fever, weight loss, pacing,and salivation, severely threatening the development of animal husbandry. However, in additionto causing acute infections and diseases, FMDV can be asymptomatic carriers in some cases, whichmight lead to another outbreak of FMD, making prevention and control challenging and costly. Highinfectivity, wide geographical distribution, wide host range, short-term immunity without serotypecross-protection, multiple modes of transmission, and persistent infection render the control and eradication of this disease rather difficult. Therefore, study the molecular mechanism underlyingFMDV evading immunity is imperative for the control of an epidemic situation.The immune system includes innate immunity and acquired immunity, which is a major protectivesystem against the invasion of pathogenic microorganisms, surveillance, and removal of foreign bodies.FMDV suppresses the function of the immune system at the initial stage of infection, such that thevirus can proliferate rapidly in the respiratory system and spread to its natural infection site [15].In terms of evading the humoral immune system, each serotype of FMDV is prone to antigenicvariation, which makes the virus escape from the neutralizing antibodies [16]. In the aspect ofinhibiting cellular immune response, FMDV infection can cause the decrease of host lymphocytes andis accompanied by severe viremia, which will eventually lead to the destruction of T cells and FMDVinfection inhibits the function of dendritic cells and weakens the ability of dendritic cells to processthem into antigens [143,144]. Previous studies have shown that, MHC class I molecule expression onthe surface of cells was suppressed at 30 min after FMDV infection, indicating that the cells infectedwith FMDV will immediately lose the ability to present MHC-I-related viral peptides to T lymphocytes.This mechanism would facilitate the virus escape from the host’s cytotoxic immune response. Limitingthe killing effect mediated by NK cells is also an important mechanism for FMDV to evade the cellularimmune response. Some studies have shown that the responsiveness of porcine NK cells decreasessignificantly 2–3 days after FMDV infection, and then returns to normal [17]. Strikingly, NK cellsisolated from infected pigs could not secrete IFN-γ [18]. The research on FMDV interference withimmune effect and suppression of innate immunity has been widely studied. Some proteins of FMDV(Lpro,2B, 3A, 3B, 3C) can directly or indirectly act on retinoic acid-induced gene I-like receptor (RLR) toinhibit innate immunity [19][20][21][22][23]. FMDV VP0, VP3, 3A, and 3B reduce the expression of junctionprotein VISA at the transcriptional or protein level [23][24][25]. FMDV Lpro, VP0, VP1, 2B, and 3A candirectly or indirectly target IRF3 to inhibit interferon production [21][23][26]. VP3 and 3C proteinsinhibit JAK-STAT signaling pathway, thus inhibiting ISGs production [27][28]. FMDV proteins Lproand 3C inhibit the synthesis of antiviral molecules by cutting related factors of host transcriptionand translation [11][29][30][31][32] . In addition, it is interesting that Lproprotein can not only induceapoptosis, but also inhibit host cell apoptosis and promote virus replication, which is achieved byblocking the translation ofα-IFN and inhibiting PKR synthesis [33]. FMDV protein VP2 and 2C canpromote virus replication by regulating autophagy [34][35]. These mechanisms provide opportunitiesfor rapid transcription and translation of FMDV.In the previous studies on FMDV, HEK293 cells have been widely used inin vitroexperimentsbecause of its highly transfected efficiency. However, HEK293 cells are not FMDV susceptible cells andthere are species differences between HEK293 cells and FMDV susceptible cells. Therefore, the use ofHEK293 cells for FMDV-related research has some limitations.
3. Conclusions
In summary, FMDV has evolved a variety of ways to evade the immune response in the long-termcombat with the host immune system. Although there are many breakthroughs in the research on theimmune escape of FMDV, many mechanisms underlying the FMDV-affected host immunity have notyet been elucidated, and the interaction between FMDV protein and host protein need to be exploredfurther. In addition to the interaction between the virus and host protein, exploring the mechanismof synergistic inhibition of immune response by multiple viral proteins is of great significance forthe development of specific drugs and new vaccines. Previous studies mainly focused on the effectof FMDV with respect to innate immunity. However, there are a few studies on acquired immunity,and these need to be supplemented further. Also, persistent infection of FMDV needs to be investigatedintensively in the future.
References
- Soren Alexandersen; N. Mowat; Foot-and-Mouth Disease: Host Range and Pathogenesis. Current Topics in Microbiology and Immunology / Ergebnisse der Microbiologie und Immunitätsforschung 2005, 288, 9-42, 10.1007/3-540-27109-0_2.
- Syed M. Jamal; Graham J. Belsham; Foot-and-mouth disease: past, present and future. Veterinary Research 2013, 44, 116-116, 10.1186/1297-9716-44-116.
- Nick J. Knowles; A R Samuel; Molecular epidemiology of foot-and-mouth disease virus. Virus Research 2003, 91, 65-80, 10.1016/s0168-1702(02)00260-5.
- Daniel T. Haydon; A.R Samuel; N.J Knowles; The generation and persistence of genetic variation in foot-and-mouth disease virus. Preventive Veterinary Medicine 2001, 51, 111-124, 10.1016/s0167-5877(01)00210-0.
- E. Domingo; Nonia Pariente; A. Airaksinen; C. González-Lopez; S. Sierra; M. Herrera; Ana Grande-Pérez; P. R. Lowenstein; S. C. Manrubia; E. Lázaro; et al.C. Escarmís Foot-and-Mouth Disease Virus Evolution: Exploring Pathways Towards Virus Extinction. Inducible Lymphoid Organs 2005, 288, 149-173, 10.1007/3-540-27109-0_7.
- E. Domingo; Carmen M Ruiz-Jarabo; Saleta Sierra; Armando Arias; Nonia Pariente; Eric Baranowski; Cristina Escarmis; Emergence and selection of RNA virus variants: memory and extinction.. Virus Research 2002, 82, 39-44, 10.1016/s0168-1702(01)00385-9.
- Peter W. Mason; Marvin J Grubman; Barry Baxt; Molecular basis of pathogenesis of FMDV. Virus Research 2003, 91, 9-32, 10.1016/s0168-1702(02)00257-5.
- Mireille T. Vossen; Ellen M. Westerhout; Cecilia Söderberg-Naucler; Emmanuel J. H. J. Wiertz; Viral immune evasion: a masterpiece of evolution. Immunogenetics 2002, 54, 527-542, 10.1007/s00251-002-0493-1.
- William T. Golde; Charles K. Nfon; Felix N. Toka; Immune evasion during foot‐and‐mouth disease virus infection of swine. Immunological Reviews 2008, 225, 85-95, 10.1111/j.1600-065X.2008.00672.x.
- Xiaying Li; Jianchang Wang; Jue Liu; Zhonghua Li; Yongqiang Wang; Yanfei Xue; Xiaoqi Li; Hong Cao; Shijun J. Zheng; Engagement of soluble resistance-related calcium binding protein (sorcin) with foot-and-mouth disease virus (FMDV) VP1 inhibits type I interferon response in cells. Veterinary Microbiology 2013, 166, 35-46, 10.1016/j.vetmic.2013.04.028.
- Gisselle N. Medina; Giselle M. Knudsen; Alexander L. Greninger; Anna Kloc; Fayna Díaz-San Segundo; Elizabeth Rieder; Marvin J. Grubman; Joseph L. DeRisi; Teresa De Los Santos; Interaction between FMDV Lpro and transcription factor ADNP is required for optimal viral replication. Virology 2017, 505, 12-22, 10.1016/j.virol.2017.02.010.
- Shao-Zu Fu; Wen-Ping Yang; Yi Ru; Ke-Shan Zhang; Yong Wang; Xiang-Tao Liu; Dan Li; Haixue Zheng; DDX56 cooperates with FMDV 3A to enhance FMDV replication by inhibiting the phosphorylation of IRF3.. Cellular Signalling 2019, 64, 109393, 10.1016/j.cellsig.2019.109393.
- Ahmed Sabbah; Te Hung Chang; Rosalinda Harnack; Victoria Frohlich; Kaoru Tominaga; Peter H. Dube; Yan Xiang; Santanu Bose; Activation of innate immune antiviral responses by Nod2. Nature Immunology 2009, 10, 1073-1080, 10.1038/ni.1782.
- Huisheng Liu; Zixiang Zhu; Qiao Xue; Fan Yang; Weijun Cao; Keshan Zhang; Xiangtao Liu; Haixue Zheng; Foot-and-Mouth Disease Virus Antagonizes NOD2-Mediated Antiviral Effects by Inhibiting NOD2 Protein Expression. Journal of Virology 2019, 93, 93, 10.1128/jvi.00124-19.
- Carolina Stenfeldt; Fayna Diaz-San Segundo; Teresa De Los Santos; Luis L. Rodriguez; Jonathan Arzt; The Pathogenesis of Foot-and-Mouth Disease in Pigs. Frontiers in Veterinary Science 2016, 3, 12, 10.3389/fvets.2016.00041.
- Carolina Stenfeldt; Juan M. Pacheco; M.V. Borca; Luis L. Rodriguez; Jonathan Arzt; Morphologic and phenotypic characteristics of myocarditis in two pigs infected by foot-and mouth disease virus strains of serotypes O or A. Acta Veterinaria Scandinavica 2014, 56, 42-42, 10.1186/s13028-014-0042-6.
- Felix N. Toka; Charles Nfon; Harry Dawson; William T. Golde; Natural Killer Cell Dysfunction during Acute Infection with Foot-and-Mouth Disease Virus. Clinical and Vaccine Immunology 2009, 16, 1738-1749, 10.1128/cvi.00280-09.
- Felix N. Toka; William T. Golde; Cell mediated innate responses of cattle and swine are diverse during foot-and-mouth disease virus (FMDV) infection: A unique landscape of innate immunity. Immunology Letters 2013, 152, 135-143, 10.1016/j.imlet.2013.05.007.
- Miguel Rodríguez-Pulido; María T. Sánchez-Aparicio; Encarnación Martínez-Salas; Adolfo García-Sastre; Francisco Sobrino; Margarita Sáiz; Innate immune sensor LGP2 is cleaved by the Leader protease of foot-and-mouth disease virus. PLOS Pathogens 2018, 14, e1007135, 10.1371/journal.ppat.1007135.
- Zixiang Zhu; Guoqing Wang; Fan Yang; Weijun Cao; Ruoqing Mao; Xiaoli Du; Xiangle Zhang; Chuntian Li; Dan Li; Keshan Zhang; et al.Hongbing ShuXiangtao LiuHaixue Zheng Foot-and-Mouth Disease Virus Viroporin 2B Antagonizes RIG-I-Mediated Antiviral Effects by Inhibition of Its Protein Expression. Journal of Virology 2016, 90, 11106-11121, 10.1128/jvi.01310-16.
- Zixiang Zhu; Chuntian Li; Xiaoli Du; Guoqing Wang; Weijun Cao; Fan Yang; Huanhuan Feng; Xiangle Zhang; Zhengwang Shi; Huanan Liu; et al.Hong TianDan LiKeshan ZhangXiangtao LiuHaixue Zheng Foot-and-mouth disease virus infection inhibits LGP2 protein expression to exaggerate inflammatory response and promote viral replication.. Cell Death & Disease 2017, 8, e2747-e2747, 10.1038/cddis.2017.170.
- Ming Li; Ting Xin; Xintao Gao; Jing Wu; Xixi Wang; Lichun Fang; Xiukun Sui; Hongfei Zhu; Shangjin Cui; Xiaoyu Guo; et al. Foot-and-mouth disease virus non-structural protein 2B negatively regulates the RLR-mediated IFN-β induction. Biochemical and Biophysical Research Communications 2018, 504, 238-244, 10.1016/j.bbrc.2018.08.161.
- Dan Li; Caoqi Lei; Zhisheng Xu; Fan Yang; Huanan Liu; Zixiang Zhu; Shu Li; Xiangtao Liu; Hong-Bing Shu; Haixue Zheng; et al. Foot-and-mouth disease virus non-structural protein 3A inhibits the interferon-β signaling pathway. Scientific Reports 2016, 6, 21888, 10.1038/srep21888.
- Dan Li; Jing Zhang; Wenping Yang; Yanchun He; Yi Ru; Shaozu Fu; Lulu Li; Xiangtao Liu; Haixue Zheng; Poly (rC) binding protein 2 interacts with VP0 and increases the replication of the foot-and-mouth disease virus.. Cell Death & Disease 2019, 10, 516, 10.1038/s41419-019-1751-6.
- Dan Li; Wenping Yang; Fan Yang; Huanan Liu; Zixiang Zhu; Kaiqi Lian; Caoqi Lei; Shu Li; Xiangtao Liu; Haixue Zheng; et al.Hong-Bing Shu The VP3 structural protein of foot‐and‐mouth disease virus inhibits the IFN‐β signaling pathway. The FASEB Journal 2016, 30, 1757-1766, 10.1096/fj.15-281410.
- Dang Wang; Liurong Fang; Rui Luo; Rui Ye; Ying Fang; Lilan Xie; Huanchun Chen; Shaobo Xiao; Foot-and-mouth disease virus leader proteinase inhibits dsRNA-induced type I interferon transcription by decreasing interferon regulatory factor 3/7 in protein levels. Biochemical and Biophysical Research Communications 2010, 399, 72-78, 10.1016/j.bbrc.2010.07.044.
- Dan Li; Jin Wei; Fan Yang; Hua-Nan Liu; Zi-Xiang Zhu; Wei-Jun Cao; Shu Li; Xiang-Tao Liu; Haixue Zheng; Hong-Bing Shu; et al. Foot-and-mouth disease virus structural protein VP3 degrades Janus kinase 1 to inhibit IFN-γ signal transduction pathways. Function of a membrane-embedded domain evolutionarily multiplied in the GPI lipid anchor pathway proteins PIG-B, PIG-M, PIG-U, PIG-W, PIG-V, and PIG-Z 2016, 15, 850-860, 10.1080/15384101.2016.1151584.
- Yijun Du; Jingshan Bi; Jiyu Liu; Xing Liu; Xiangju Wu; Ping Jiang; Dongwan Yoo; Yongguang Zhang; Jiaqiang Wu; Renzhong Wan; et al.Xiaomin ZhaoLihui GuoWenbo SunXiaoyan CongLei ChenJinbao Wang 3Cpro of Foot-and-Mouth Disease Virus Antagonizes the Interferon Signaling Pathway by Blocking STAT1/STAT2 Nuclear Translocation. Journal of Virology 2014, 88, 4908-4920, 10.1128/jvi.03668-13.
- Anne-Claude Gingras; Brian Raught; Nahum Sonenberg; eIF4 Initiation Factors: Effectors of mRNA Recruitment to Ribosomes and Regulators of Translation. Annual Review of Biochemistry 1999, 68, 913-963, 10.1146/annurev.biochem.68.1.913.
- Walter Glaser; Regina Cencic; Tim Skern; Robert P. Fadden; Amparo Ruiz; Timothy Haystead; Joaquı́n Ariño; Eulàlia De Nadal; Foot-and-Mouth Disease Virus Leader Proteinase. Journal of Biological Chemistry 2001, 276, 35473-35481, 10.1074/jbc.m104192200.
- Graham J. Belsham; Gerald M. McInerney; Natalie Ross-Smith; Foot-and-Mouth Disease Virus 3C Protease Induces Cleavage of Translation Initiation Factors eIF4A and eIF4G within Infected Cells. Journal of Virology 2000, 74, 272-280, 10.1128/JVI.74.1.272-280.2000.
- Paul Lawrence; Elizabeth A. Schafer; Elizabeth Rieder; The nuclear protein Sam68 is cleaved by the FMDV 3C protease redistributing Sam68 to the cytoplasm during FMDV infection of host cells. Virology 2012, 425, 40-52, 10.1016/j.virol.2011.12.019.
- Chelsea M. Hull; Philip C. Bevilacqua; Discriminating Self and Non-Self by RNA: Roles for RNA Structure, Misfolding, and Modification in Regulating the Innate Immune Sensor PKR. Accounts of Chemical Research 2016, 49, 1242-1249, 10.1021/acs.accounts.6b00151.
- Peng Sun; Shumin Zhang; Xiaodong Qin; Xingni Chang; Xiaorui Cui; Haitao Li; Shuaijun Zhang; Huanhuan Gao; Penghua Wang; Zhidong Zhang; et al.Jianxun LuoZhiyong Li Foot-and-mouth disease virus capsid protein VP2 activates the cellular EIF2S1-ATF4 pathway and induces autophagy via HSPB1.. Autophagy 2018, 14, 336-346, 10.1080/15548627.2017.1405187.
- Douglas P. Gladue; V. O'donnell; R. Baker-Branstetter; L. G. Holinka; Juan M. Pacheco; I. Fernandez-Sainz; Z. Lu; E. Brocchi; B. Baxt; M. E. Piccone; et al.L. RodríguezM.V. Borca Foot-and-Mouth Disease Virus Nonstructural Protein 2C Interacts with Beclin1, Modulating Virus Replication. Journal of Virology 2012, 86, 12080-12090, 10.1128/jvi.01610-12.