Your browser does not fully support modern features. Please upgrade for a smoother experience.

Submitted Successfully!
Thank you for your contribution! You can also upload a video entry or images related to this topic.
For video creation, please contact our Academic Video Service.
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Liping Qiu | + 1650 word(s) | 1650 | 2022-01-25 03:36:38 | | | |
| 2 | Camila Xu | -133 word(s) | 1517 | 2022-02-10 08:48:45 | | | | |
| 3 | Camila Xu | -19 word(s) | 1498 | 2022-02-11 01:29:16 | | | | |
| 4 | Camila Xu | -19 word(s) | 1498 | 2022-02-11 01:40:37 | | |
Video Upload Options
We provide professional Academic Video Service to translate complex research into visually appealing presentations. Would you like to try it?
Cite
If you have any further questions, please contact Encyclopedia Editorial Office.
Qiu, L. DNA-Based Molecular Engineering of Cell Membrane. Encyclopedia. Available online: https://encyclopedia.pub/entry/19081 (accessed on 26 May 2026).
Qiu L. DNA-Based Molecular Engineering of Cell Membrane. Encyclopedia. Available at: https://encyclopedia.pub/entry/19081. Accessed May 26, 2026.
Qiu, Liping. "DNA-Based Molecular Engineering of Cell Membrane" Encyclopedia, https://encyclopedia.pub/entry/19081 (accessed May 26, 2026).
Qiu, L. (2022, February 02). DNA-Based Molecular Engineering of Cell Membrane. In Encyclopedia. https://encyclopedia.pub/entry/19081
Qiu, Liping. "DNA-Based Molecular Engineering of Cell Membrane." Encyclopedia. Web. 02 February, 2022.
Copy Citation
DNA, with intrinsic advantages of high versatility, programmability, and biocompatibility, has gained intense attention as a molecular tool for cell-surface engineering.
functional nucleic acids
DNA nanotechnology
cell membrane engineering
1. DNA-Based Intercellular Communication
In multicellular organisms, cell–cell communication was mainly mediated by membrane receptors. The development of artificial receptors to monitor and control intercellular reactions would be highly desired. Cell membrane-anchored DNA platforms, with high programmability and flexibility, showed great promise in mimicry and manipulation of cellular interaction.
Based on DNA hybridization with predictable thermodynamics, Zhao et al. reported DNA tension probes for quantifying the tensile forces at the junction between cells. As shown in Figure 1A,B, the tension probe was inset into the cellular junction through both the hydrophobic interaction and the ligand–receptor interaction [1]. Upon sensing the intercellular tension force, the DNA probe would undergo a conformation switch to separate the fluorophore pair, thus allowing dynamic detection of the intercellular force via FRET. Moreover, the capability to modulate the intercellular reaction would offer us a straightforward strategy for analyzing related biological processes. Xiong et al. mimicked the natural cell–cell adhesion by modifying the cell surface with specific aptamers (Figure 1C,D) [2]. Based on the aptamer-mediated molecular recognition, the effector T cells could specifically bind with target cancer cells, resulting in enhanced cancer-killing efficacy. To achieve intelligent control over the cellular interaction, DNA-based Boolean logic operation was utilized [3]. Li et al. engineered an amphiphilic DNA tetrahedral construct on the cell surface, which could undergo a conformation switch and assemble functional modules for sensing the cell’s adaptive response to the external environment [4]. As such, this membrane-anchored DNA construct allowed regulation of the cellular interaction coordinated with the cellular activity.

Figure 1. DNA-based probes mediated non-destructive modification on the cell surface and cell–cell interaction. (A,B) Lipid-modified DNA probes to visualize tensile forces at cell junctions. The intercellular tension force can open the DNA probes and large increase in the fluorescence intensity. Reprinted with permission from Zhao et al. [5], Copyright 2017, with permission from Elsevier. (C,D) Immune cells were modified with lipo-aptamer probes to enhance the cancer cell targeting and killing efficiency (The single asterisk indicates a significant difference between aptamer-modified and unmodified or Lib-modified groups determined by the one-tailed t-test at * p < 0.01, ** p < 0.001. The double asterisks indicate a significant difference between aptamer-modified and anti-Perforin treated groups determined by the one-tailed t-test at * p < 0.01). Reprinted with permission from Xiong et al. [2], Copyright 2013, with permission from Elsevier.
Another application for membrane anchored-DNA probes was to manipulate the morphology of cells with spatial resolution. The cell growth could be controlled via hybridization of the membrane-anchored DNA with the complementary DNA on the substrate interface [6]. Meanwhile, a 3D cell microsphere could be constructed by stepwise and programmable cell assembling, which displayed great promise for the construction of 3D artificial tissues [7]. In addition to single-stranded DNA probes, DNA nanostructures provided versatile tools for controllable cell assembly [8]. For example, the assembly between stem cells and niche cells was mediated through the mechanical and morphogen signaling from niche cells, providing a versatile strategy for the generation of stem cells in vitro [4].
2. DNA-Based Receptor Monitoring and Regulating
Protein receptors expressed on the cell surface played a pivotal role in regulating cellular function and behavior. Abnormal expression and dysfunction of surface receptors were closely related with the occurrence of many diseases. As previously reported, over 50% of commercially available therapeutic agents targeted cell surface receptors. DNA nanotechnology, taking advantage of high programmability, high addressability, and good biocompatibility, offered promising strategies for the detection and regulation of surface receptors. Particularly, aptamers, selected by the cell-SELEX technology, offered a panel of molecular tools for specific recognition of surface receptors [9][10][11].
Widespread application of aptamers for targeted cell imaging has been realized in the past two decades. [12][13] Their application for super-resolution imaging was emerging as an interesting topic. To image the membrane receptor at the single-molecule level, Delcanale et al. used aptamers as the molecular recognition ligand and developed DNA-based point accumulation for imaging in nanoscale topography (DNA-PAINT) (Figure 2A) [14]. The stochastic and transient binding against target receptors could be realized through regulating the binding affinity of aptamers, providing twinkling fluorescence signals for super-resolution imaging of the cell membrane. Recently, this strategy was further expanded to visualize the spatial proximity of membrane receptors using a split-docking site configuration [15].
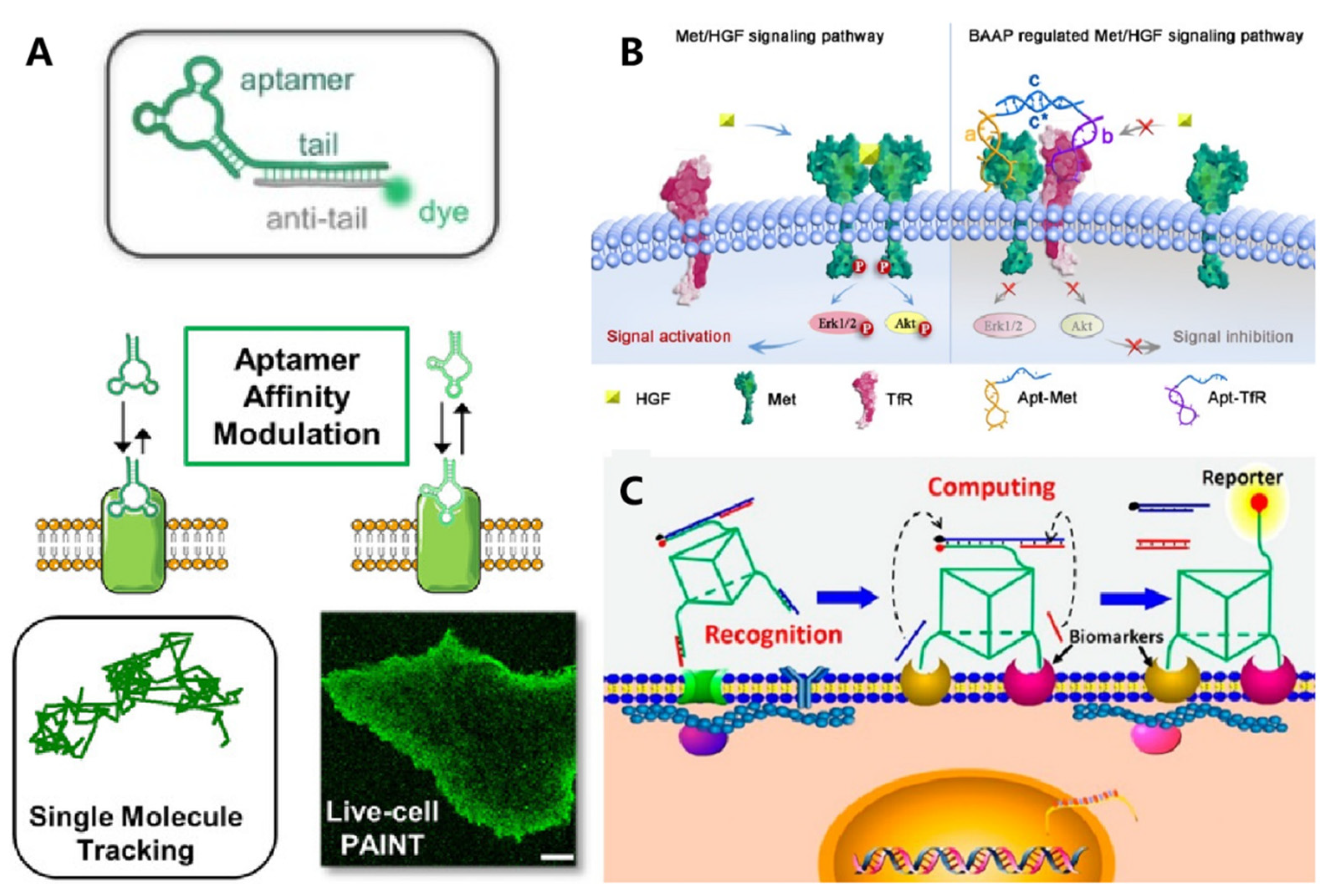
Figure 2. DNA-based molecule probes to monitor and regulate cell surface receptors. (A) Aptamer probes were used for single-molecule PAINT imaging to track and localize the membrane receptors on living cancer cells. Reprinted with permission from Delcanale et al. [16], Copyright 2020, with permission from Elsevier. (B) Bispecific aptamer induced protein dimerization to specifically regulate Met receptor function and downstream signaling pathways. Reprinted with permission from Wang et al. [17], Copyright 2019, with permission from Elsevier. (C) 3D DNA-logic gate nanomachine was constructed to recognize and compute the protein receptors on target cell surfaces. Reprinted with permission from Peng et al. [18], Copyright 2018, with permission from Elsevier.
Cell identification with high accuracy was fundamental in the perspective of precise medicine. Conversely, due to their high heterogeneity, it was rather challenging to identify specific cells with single makers. Exploiting multiple membrane biomarkers served as one of the promising strategies for improving the accuracy of cell identification. Meanwhile, to achieve intelligent cellular identification, DNA circuits have been incorporated to operate smart computation of multiple biomarkers on the cell membrane. As one of the typical examples, Rudchenko et al. reported an autonomous molecular machine based on DNA strand replacement reactions for precise cellular identification, which allowed distinguishing the subpopulation of lymphocytes from human blood cells [19]. To improve the efficiency and accuracy of DNA computation, Peng et al. designed a 3D DNA nanomachine that integrated all logic gate elements on one DNA construct (Figure 2B) [20]. A decision signal output could be generated only upon recognizing target cancer cells synchronously expressed with two membrane markers.
In addition to detecting the expression level of surface receptors, Li et al. developed an aptamer-based fluorescence probe for dynamically monitoring the dimerization of Met receptors [21]. The same group then developed a bispecific aptamer for mediating the heterodimerization between Met and TfR receptors, which could inhibit the formation of a Met homodimer mainly via a steric-hindrance effect, thus regulating downstream signaling pathways (Figure 2C) [22]. Meanwhile, the cellular morphology could be regulated by membrane-anchored DNA nanostructures, which then affect the motion and function of cells. Furthermore, the addressability of DNA origami enabled precise control over the spatial organization of membrane ligands. In 2014, Shaw et al. used hollow tube-like DNA origami to develop a “nanocaliper” and proved its feasibility for regulating the activation process of EphA2 through manipulating the spatial distribution of ephrin-A5 on the cell membrane [23].
3. DNA-Based Biomimetic Membrane Constructs
Protein channels on the cell membrane are one of the most important organelles for substance exchange with the external matrix and play significant roles in cell–cell communication. The programmability and site addressability of DNA nanostructures make them ideal materials for mimicking the structure and function of membrane channels. In 2016, Howorka et al. reported a DNA nanopore with hollow barrel structures for regulating the transportation of small molecules across the plasma membrane (Figure 3A) [24]. Specifically, the modification of hydrophobic cholesterol at the outside of the constructed DNA origami enabled the insertion of nanopores into the lipidic membrane, and the “closed” and “open” state of nanopores could be regulated with “lock” and “key” strands. They demonstrated that these artificial ion channels were able to control the transportation of small organic molecules with high selectivity. Soon after, Diederichs et al. constructed a synthetic DNA nanopore with a larger size, which enabled transportation of folded proteins [25].
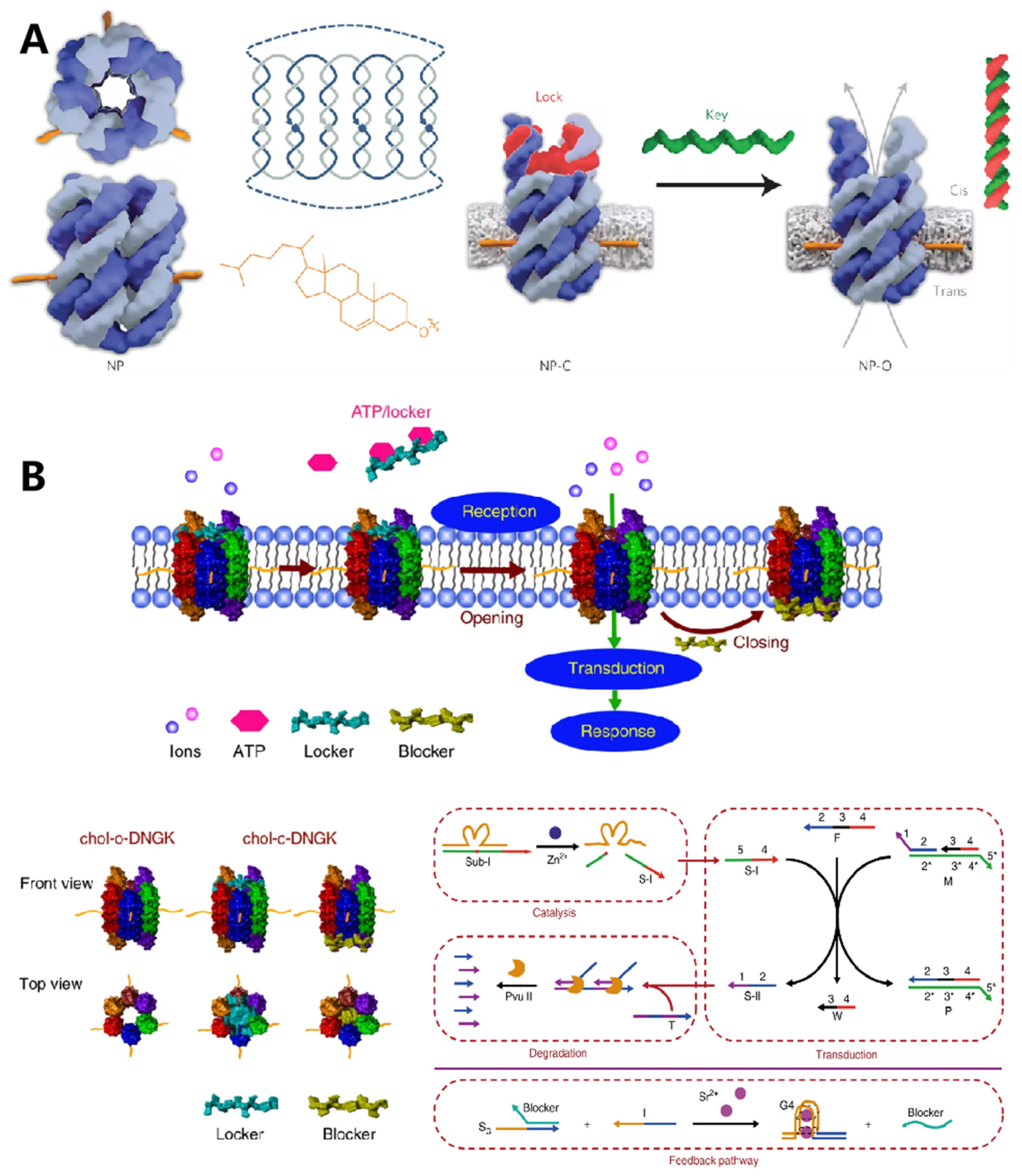
Figure 3. DNA nanostructures in constructing biomimetic membrane. (A) The typical DNA nanopore with a nanomechanical and sequence-specific gate to regulate the transportation of small molecules across the cell membrane. Reprinted with permission from Burns et al. [11], Copyright 2016, with permission from Elsevier. (B) DNA-based artificial signal transduction system with a cell-mimicking giant membrane vesicle. Reprinted with permission from Peng et al. [13], Copyright 2020, with permission from Elsevier.
In addition to constructing artificial nanopores for substance transporting, cell membrane-anchored DNA nanostructures could be designed as bio-mimic receptors to regulate the cell signaling process. In 2020, Peng et al. designed a DNA-based artificial signal transduction system with giant vesicles derived from living cells (Figure 3B) [26]. Adenosine triphosphate (ATP) responsive artificial nanopores were developed and anchored onto the vesicles through hydrophobic interactions. Upon exposure to ATP, the locker strand of nanopore would be released, leading to the import of metal ions, which then initiated a DNA circuit reaction to mimic the signaling cascades.
Meanwhile, specific DNA nanostructures anchored on the cell membrane could also display enzyme-like catalytic activity. For example, Göpfrich et al. reported that membrane-inserted dsDNA with porphyrin modification could locally induce the formation of hydrophilic pores [27]. They continued to construct a hollow barrel structure composed of four double helices. After being inserted into the cell membrane, this nanostructure displayed scramblase activity for promoting transportation of lipids between membrane leaflets, achieving a three-fold higher catalytic rate than protein enzymes [28].
References
- Zhao, B.; O’Brien, C.; Mudiyanselage, A.P.K.K.K.; Li, N.; Bagheri, Y.; Wu, R.; Sun, Y.; You, M. Visualizing Intercellular Tensile Forces by DNA-Based Membrane Molecular Probes. J. Am. Chem. Soc. 2017, 139, 18182–18185.
- Xiong, X.; Liu, H.; Zhao, Z.; Altman, M.B.; Lopez-Colon, D.; Yang, C.J.; Chang, L.J.; Liu, C.; Tan, W. DNA Aptamer-Mediated Cell Targeting. Angew. Chem. Int. Ed. 2013, 125, 1512–1516.
- Chandra, R.A.; Douglas, E.S.; Mathies, R.A.; Bertozzi, C.R.; Francis, M.B. Programmable Cell Adhesion Encoded by DNA Hybridization. Angew. Chem. Int. Ed. 2006, 45, 896–901.
- Li, X.; Xie, X.; Ma, Z.; Li, Q.; Liu, L.; Hu, X.; Liu, C.; Li, B.; Wang, H.; Chen, N.; et al. Programming Niche Accessibility and In Vitro Stemness with Intercellular DNA Reactions. Adv. Mater. 2018, 30, 1804861.
- Zeng, S.; Liu, D.; Li, C.; Yu, F.; Fan, L.; Lei, C.; Huang, Y.; Nie, Z.; Yao, S. Cell-Surface-Anchored Ratiometric DNA Tweezer for Real-Time Monitoring of Extracellular and Apoplastic pH. Anal. Chem. 2018, 90, 13459–13466.
- Wang, S.; Cai, X.; Wang, L.; Li, J.; Li, Q.; Zuo, X.; Shi, J.; Huang, Q.; Fan, C. DNA orientation-specific adhesion and patterning of living mammalian cells on self-assembled DNA monolayers. Chem. Sci. 2016, 7, 2722–2727.
- Todhunter, M.E.; Jee, N.Y.; Hughes, A.J.; Coyle, M.C.; Cerchiari, A.; Farlow, J.; Garbe, J.C.; LaBarge, M.A.; Desai, T.A.; Gartner, Z.J. Programmed synthesis of three-dimensional tissues. Nat. Methods 2015, 12, 975–981.
- Shi, P.; Zhao, N.; Lai, J.; Coyne, J.; Gaddes, E.R.; Wang, Y. Polyvalent Display of Biomolecules on Live Cells. Angew. Chem. Int. Ed. Engl. 2018, 57, 6800–6804.
- Sefah, K.; Shangguan, D.; Xiong, X.; O’Donoghue, M.B.; Tan, W. Development of DNA aptamers using Cell-SELEX. Nat. Protoc. 2010, 5, 1169–1185.
- Shangguan, D.; Li, Y.; Tang, Z.; Cao, Z.C.; Chen, H.W.; Mallikaratchy, P.; Sefah, K.; Yang, C.J.; Tan, W. Aptamers evolved from live cells as effective molecular probes for cancer study. Proc. Natl. Acad. Sci. USA 2006, 103, 11838–11843.
- Zhang, K.; Deng, R.; Sun, Y.; Zhang, L.; Li, J. Reversible control of cell membrane receptor function using DNA nano-spring multivalent ligands. Chem. Sci. 2017, 8, 7098–7105.
- Tan, W.; Donovan, M.J.; Jiang, J. Aptamers from Cell-Based Selection for Bioanalytical Applications. Chem. Rev. 2013, 113, 2842–2862.
- You, M.; Chen, Y.; Peng, L.; Han, D.; Yin, B.; Ye, B.; Tan, W. Engineering DNA aptamers for novel analytical and biomedical applications. Chem. Sci. 2011, 2, 1003–1010.
- Delcanale, P.; Porciani, D.; Pujals, S.; Jurkevich, A.; Chetrusca, A.; Tawiah, K.D.; Burke, D.H.; Albertazzi, L. Aptamers with Tunable Affinity Enable Single-Molecule Tracking and Localization of Membrane Receptors on Living Cancer Cells. Angew. Chem. Int. Ed. 2020, 59, 18546–18555.
- Schueder, F.; Lara-Gutiérrez, J.; Haas, D.; Beckwith, K.S.; Yin, P.; Ellenberg, J.; Jungmann, R. Super-Resolution Spatial Proximity Detection with Proximity-PAINT. Angew. Chem. Int. Ed. 2021, 60, 716–720.
- Jin, C.; He, J.; Zou, J.; Xuan, W.; Fu, T.; Wang, R.; Tan, W. Phosphorylated lipid-conjugated oligonucleotide selectively anchors on cell membranes with high alkaline phosphatase expression. Nat. Commun. 2019, 10, 2704.
- Liu, L.; Dou, C.-X.; Liu, J.-W.; Wang, X.-N.; Ying, Z.-M.; Jiang, J.-H. Cell Surface-Anchored DNA Nanomachine for Dynamically Tunable Sensing and Imaging of Extracellular pH. Anal. Chem. 2018, 90, 11198–11202.
- Zhao, W.; Schafer, S.; Choi, J.; Yamanaka, Y.J.; Lombardi, M.L.; Bose, S.; Carlson, A.L.; Phillips, J.A.; Teo, W.; Droujinine, I.A.; et al. Cell-surface sensors for real-time probing of cellular environments. Nat. Nanotechnol. 2011, 6, 524–531.
- Rudchenko, M.; Taylor, S.; Pallavi, P.; Dechkovskaia, A.; Khan, S.; Butler, V.P., Jr.; Rudchenko, S.; Stojanovic, M.N. Autonomous molecular cascades for evaluation of cell surfaces. Nat. Nanotechnol. 2013, 8, 580–586.
- Peng, R.; Zheng, X.; Lyu, Y.; Xu, L.; Zhang, X.; Ke, G.; Liu, Q.; You, C.; Huan, S.; Tan, W. Engineering a 3D DNA-Logic Gate Nanomachine for Bispecific Recognition and Computing on Target Cell Surfaces. J. Am. Chem. Soc. 2018, 140, 9793–9796.
- Li, H.; Wang, M.; Shi, T.; Yang, S.; Zhang, J.; Wang, H.H.; Nie, Z. A DNA-Mediated Chemically Induced Dimerization (D-CID) Nanodevice for Nongenetic Receptor Engineering To Control Cell Behavior. Angew. Chem. Int. Ed. 2018, 130, 10383–10387.
- Wang, L.; Liang, H.; Sun, J.; Liu, Y.; Li, J.; Li, J.; Li, J.; Yang, H. Bispecific Aptamer Induced Artificial Protein-Pairing: A Strategy for Selective Inhibition of Receptor Function. J. Am. Chem. Soc. 2019, 141, 12673–12681.
- Shaw, A.; Lundin, V.; Petrova, E.; Fördős, F.; Benson, E.; Al-Amin, A.; Herland, A.; Blokzijl, A.; Högberg, B.; Teixeira, A.I. Spatial control of membrane receptor function using ligand nanocalipers. Nat. Methods 2014, 11, 841–846.
- Burns, J.R.; Seifert, A.; Fertig, N.; Howorka, S. A biomimetic DNA-based channel for the ligand-controlled transport of charged molecular cargo across a biological membrane. Nat. Nanotechnol. 2016, 11, 152–156.
- Diederichs, T.; Pugh, G.; Dorey, A.; Xing, Y.; Burns, J.R.; Hung Nguyen, Q.; Tornow, M.; Tampé, R.; Howorka, S. Synthetic protein-conductive membrane nanopores built with DNA. Nat. Commun. 2019, 10, 5018.
- Peng, R.; Xu, L.; Wang, H.; Lyu, Y.; Wang, D.; Bi, C.; Cui, C.; Fan, C.; Liu, Q.; Zhang, X.; et al. DNA-based artificial molecular signaling system that mimics basic elements of reception and response. Nat. Commun. 2020, 11, 978.
- Göpfrich, K.; Li, C.-Y.; Mames, I.; Bhamidimarri, S.P.; Ricci, M.; Yoo, J.; Mames, A.; Ohmann, A.; Winterhalter, M.; Stulz, E.; et al. Ion Channels Made from a Single Membrane-Spanning DNA Duplex. Nano Lett. 2016, 16, 4665–4669.
- Ohmann, A.; Li, C.-Y.; Maffeo, C.; Al Nahas, K.; Baumann, K.N.; Göpfrich, K.; Yoo, J.; Keyser, U.F.; Aksimentiev, A. A synthetic enzyme built from DNA flips 107 lipids per second in biological membranes. Nat. Commun. 2018, 9, 2426.
More
Information
Subjects:
Chemistry, Analytical
Contributor
MDPI registered users' name will be linked to their SciProfiles pages. To register with us, please refer to https://encyclopedia.pub/register
:
View Times:
940
Revisions:
4 times
(View History)
Update Date:
11 Feb 2022
Notice
You are not a member of the advisory board for this topic. If you want to update advisory board member profile, please contact office@encyclopedia.pub.
OK
Confirm
Only members of the Encyclopedia advisory board for this topic are allowed to note entries. Would you like to become an advisory board member of the Encyclopedia?
Yes
No
${ textCharacter }/${ maxCharacter }
Submit
Cancel
Back
Comments
${ item }
|
More
No more~
There is no comment~
${ textCharacter }/${ maxCharacter }
Submit
Cancel
${ selectedItem.replyTextCharacter }/${ selectedItem.replyMaxCharacter }
Submit
Cancel
Confirm
Are you sure to Delete?
Yes
No





