
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Hiroko Isoda | + 3620 word(s) | 3620 | 2022-01-10 08:17:12 | | | |
| 2 | Conner Chen | Meta information modification | 3620 | 2022-01-21 02:16:16 | | |
Video Upload Options
Diabetes mellitus, especially type 2 (T2DM), is a major public health problem globally. DM is characterized by high levels of glycemia and insulinemia due to impaired insulin secretion and insulin sensitivity of the cells, known as insulin resistance. T2DM causes multiple and severe complications such as nephropathy, neuropathy, and retinopathy causing cell oxidative damages in different internal tissues, particularly the pancreas, heart, adipose tissue, liver, and kidneys. Isorhamnetin, a plant flavonoid, has long been studied for its potential anti-diabetic effects. Isorhamnetin is a monomethoxyflavone or an O-methylated flavonol from the class of flavonoids. It is quercetin in which a methoxy group replaces the hydroxy group at position 3’. Some isorhamnetin derivatives are present in nature, such as isorhamnetin 3-O-β-d-glucopyranoside, isorhamnetin 3-O-neohesperidoside, and isorhamnetin 3-O-rutinoside from Calendula officinalis L.. Isorhamnetin presents significant biological properties such as antioxidant, anticancer, antimicrobial, antiviral, anti-inflammatory and anti-diabetic effects.
1. General Overview of Biological Activities of Isorhamnetin
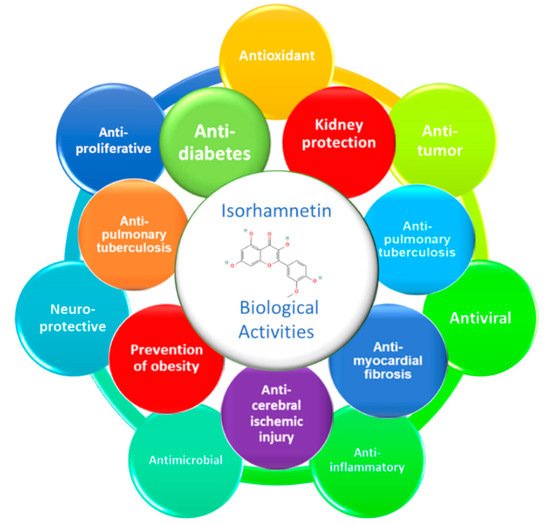
2. Anti-Diabetic Effect of Isorhamnetin
2.1. General Overview on Diabetes and Its Link with Metabolic Syndrome
2.2. Effect of Isorhamnetin on Associated Metabolic Pathways
| Plant Sources | Extraction Methods | Action Modes | References |
|---|---|---|---|
| Vaccinium vitis-idaea | Fractionation guided 80% ethanol > ethyl acetate > water |
Enhances muscle cell glucose uptake | [48] |
| Nitraria retusa | Maceration in water-ethanol solution | Modulation of lipogenesis–lipolysis balance | [49] |
| Nitraria retusa | Maceration in water-ethanol solution | 3T3-L1 preadipocyte differentiation regulation | [50] |
| Corchorus olitorius | Soxhlet extraction with methanol, the residue from the above soluble extraction was hydrolyzed directly with 200 mL of 4 M NaOH solution. Then, the mixture was adjusted to pH 2 with concentrated HCl and the bound phytochemicals were extracted with ethyl acetate. | - α-Amylase inhibition - α-Glucosidase inhibition - Angiotensin I converting enzyme (ACE) inhibition - Degradation of deoxyribose |
[44] |
| Salicornia herbacea | Fractionation guided ethanol under reflux > ethyl acetate ethyl acetate fraction was eluted on a silica gel chromatography by gradient elution with chloroform and methanol |
- α-Glucosidase - Regulating the expression of ERK, PI3K, AKT, IRS-2, and PDX-1 |
[45] |
| Eruca sativa | Extraction with ethanol by the accelerated solvent extractor, at 50 °C, 1500 psi for 20 min | Activation of PPAR-α and suppression of inflammatory cytokines | [51] |
| Oenanthe javanica | Isorhamnetin was isolated from the aerial part of O. javanica and its purity was higher than 95.0% | Attenuation of fibrosis in rat hepatic stellate cells via inhibition of ERK signaling pathway | [52] |
2.2.1. Effect of Isorhamnetin on Glucose Transporters
2.2.2. Effect of Isorhamnetin on Peroxisome Proliferator-Activated Receptors (PPARs)
2.2.3. Effect of Isorhamnetin on Hepatic Enzymes
Non-Alcoholic Steatohepatitis
Hepatic Fibrosis
Hepatocellular Carcinoma
2.2.4. Effect of Isorhamnetin on Pancreatic β-Cell Dysfunction
2.2.5. Effect of Isorhamnetin on NF-κB
References
- Liguori, I.; Russo, G.; Curcio, F.; Bulli, G.; Aran, L.; Della-Morte, D.; Gargiulo, G.; Testa, G.; Cacciatore, F.; Bonaduce, D. Oxidative stress, aging, and diseases. Clin. Interv. Aging 2018, 13, 757.
- Hayes, J.D.; Dinkova-Kostova, A.T.; Tew, K.D. Oxidative stress in cancer. Cancer Cell 2020, 38, 167–197.
- Wei, W.; Liu, Q.; Tan, Y.; Liu, L.; Li, X.; Cai, L. Oxidative stress, diabetes, and diabetic complications. Hemoglobin 2009, 33, 370–377.
- Madamanchi, N.R.; Vendrov, A.; Runge, M.S. Oxidative stress and vascular disease. Arterioscler. Thromb. Vasc. Biol. 2005, 25, 29–38.
- Barnham, K.J.; Masters, C.L.; Bush, A.I. Neurodegenerative diseases and oxidative stress. Nat. Rev. Drug Discov. 2004, 3, 205–214.
- De Souza Farias, S.A.N.; da Costa, K.S.; Martins, J.o.B. Analysis of Conformational, Structural, Magnetic, and Electronic Properties Related to Antioxidant Activity: Revisiting Flavan, Anthocyanidin, Flavanone, Flavonol, Isoflavone, Flavone, and Flavan-3-ol. ACS Omega 2021, 6, 8908–8918.
- Ronsisvalle, S.; Panarello, F.; Longhitano, G.; Siciliano, E.A.; Montenegro, L.; Panico, A. Natural flavones and flavonols: Relationships among antioxidant activity, glycation, and metalloproteinase inhibition. Cosmetics 2020, 7, 71.
- Seo, S.; Seo, K.; Ki, S.H.; Shin, S.M. Isorhamnetin inhibits reactive oxygen species-dependent hypoxia inducible factor (HIF)-1α accumulation. Biol. Pharm. Bull. 2016, 39, 1830–1838.
- Pengfei, L.; Tiansheng, D.; Xianglin, H.; Jianguo, W. Antioxidant properties of isolated isorhamnetin from the sea buckthorn marc. Plant Foods Hum. Nutr. 2009, 64, 141–145.
- Zuo, A.; Yanying, Y.; Li, J.; Binbin, X.; Xiongying, Y.; Yan, Q.; Shuwen, C. Study on the relation of structure and antioxidant activity of isorhamnetin, quercetin, phloretin, silybin and phloretin isonicotinyl hydrazone. Free Radic. Antioxid. 2011, 1, 39–47.
- Wu, Q.; Kroon, P.A.; Shao, H.; Needs, P.W.; Yang, X. Differential effects of quercetin and two of its derivatives, isorhamnetin and isorhamnetin-3-glucuronide, in inhibiting the proliferation of human breast-cancer MCF-7 cells. J. Agric. Food Chem. 2018, 66, 7181–7189.
- Wei, J.; Su, H.; Bi, Y.; Li, J.; Feng, L.; Sheng, W. Anti-proliferative effect of isorhamnetin on HeLa cells through inducing G2/M cell cycle arrest. Exp. Ther. Med. 2018, 15, 3917–3923.
- Li, C.; Yang, X.; Hu, J.-B.; Liao, J.-Z. Isorhamnetin suppresses the growth of gefitinib resistant human lung cancer PC9 cells. Her. Med. 2012, 31, 831–834.
- Aonuma, K.; Ferdousi, F.; Xu, D.; Tominaga, K.; Isoda, H. Effects of isorhamnetin in human amniotic epithelial stem cells in vitro and its cardioprotective effects in vivo. Front. Cell Dev. Biol. 2020, 8, 578197.
- Zhao, J.-J.; Song, J.-Q.; Pan, S.-Y.; Wang, K. Treatment with isorhamnetin protects the brain against ischemic injury in mice. Neurochem. Res. 2016, 41, 1939–1948.
- Ishola, I.O.; Osele, M.O.; Chijioke, M.C.; Adeyemi, O.O. Isorhamnetin enhanced cortico-hippocampal learning and memory capability in mice with scopolamine-induced amnesia: Role of antioxidant defense, cholinergic and BDNF signaling. Brain Res. 2019, 1712, 188–196.
- Seo, K.; Yang, J.H.; Kim, S.C.; Ku, S.K.; Ki, S.H.; Shin, S.M. The antioxidant effects of isorhamnetin contribute to inhibit COX-2 expression in response to inflammation: A potential role of HO-1. Inflammation 2014, 37, 712–722.
- Yang, B.; Li, X.-P.; Ni, Y.-F.; Du, H.-Y.; Wang, R.; Li, M.-J.; Wang, W.-C.; Li, M.-M.; Wang, X.-H.; Li, L. Protective effect of isorhamnetin on lipopolysaccharide-induced acute lung injury in mice. Inflammation 2016, 39, 129–137.
- Yang, J.H.; Kim, S.C.; Kim, K.M.; Jang, C.H.; Cho, S.S.; Kim, S.J.; Ku, S.K.; Cho, I.J.; Ki, S.H. Isorhamnetin attenuates liver fibrosis by inhibiting TGF-β/Smad signaling and relieving oxidative stress. Eur. J. Pharmacol. 2016, 783, 92–102.
- Dou, W.; Zhang, J.; Li, H.; Kortagere, S.; Sun, K.; Ding, L.; Ren, G.; Wang, Z.; Mani, S. Plant flavonol isorhamnetin attenuates chemically induced inflammatory bowel disease via a PXR-dependent pathway. J. Nutr. Biochem. 2014, 25, 923–933.
- Jnawali, H.N.; Jeon, D.; Jeong, M.-C.; Lee, E.; Jin, B.; Ryoo, S.; Yoo, J.; Jung, I.D.; Lee, S.J.; Park, Y.-M. Antituberculosis activity of a naturally occurring flavonoid, isorhamnetin. J. Nat. Prod. 2016, 79, 961–969.
- Qiu, S.; Sun, G.; Zhang, Y.; Li, X.; Wang, R. Involvement of the NF-κB signaling pathway in the renoprotective effects of isorhamnetin in a type 2 diabetic rat model. Biomed. Rep. 2016, 4, 628–634.
- Ibarra, M.; Pérez-Vizcaíno, F.; Cogolludo, A.; Duarte, J.; Zaragozá-Arnáez, F.; López-López, J.G.; Tamargo, J. Cardiovascular effects of isorhamnetin and quercetin in isolated rat and porcine vascular smooth muscle and isolated rat atria. Planta Med. 2002, 68, 307–310.
- Chang, Z.; Wang, J.l.; Jing, Z.c.; Ma, P.; Xu, Q.b.; Na, J.r.; Tian, J.; Ma, X.; Zhou, W.; Zhou, R. Protective effects of isorhamnetin on pulmonary arterial hypertension: In vivo and in vitro studies. Phytother. Res. 2020, 34, 2730–2744.
- Antunes-Ricardo, M.; Gutiérrez-Uribe, J.A.; Martínez-Vitela, C.; Serna-Saldívar, S.O. Topical anti-inflammatory effects of isorhamnetin glycosides isolated from Opuntia ficus-indica. BioMed Res. Int. 2015, 2015, 847320.
- Yang, L.; Chen, Q.; Wang, F.; Zhang, G. Antiosteoporotic compounds from seeds of Cuscuta chinensis. J. Ethnopharmacol. 2011, 135, 553–560.
- Rodríguez, L.; Badimon, L.; Méndez, D.; Padró, T.; Vilahur, G.; Peña, E.; Carrasco, B.; Vogel, H.; Palomo, I.; Fuentes, E. Antiplatelet Activity of Isorhamnetin via Mitochondrial Regulation. Antioxidants 2021, 10, 666.
- Dong, G.-Z.; Lee, J.-H.; Ki, S.H.; Yang, J.H.; Cho, I.J.; Kang, S.H.; Zhao, R.J.; Kim, S.C.; Kim, Y.W. AMPK activation by isorhamnetin protects hepatocytes against oxidative stress and mitochondrial dysfunction. Eur. J. Pharmacol. 2014, 740, 634–640.
- Zhang, D.; Lei, J.; Hong, E.-K.; Lu, D.; Yuan, W.; Yang, Z.; Ming, C. Anti-hypoxia effects of the ethanol extract of Oxytropis ochrocephala. Legume Res.-Int. J. 2016, 39, 914–920.
- Wang, H.; Zhang, Q.; Cheng, M.L.; Ma, L.; Meng, Q.Z.; Duan, L.; Chen, Y.; Tan, J.W.; Chen, M.; Liang, T.T. Effect of the Miaoyao Fanggan sachet-derived isorhamnetin on TLR2/4 and NKp46 expression in mice. J. Ethnopharmacol. 2012, 144, 138–144.
- Rigano, D.; Formisano, C.; Basile, A.; Lavitola, A.; Senatore, F.; Rosselli, S.; Bruno, M. Antibacterial activity of flavonoids and phenylpropanoids from Marrubium globosum ssp. libanoticum. Phytother. Res. 2007, 21, 395–397.
- Ren, X.; Bao, Y.; Zhu, Y.; Liu, S.; Peng, Z.; Zhang, Y.; Zhou, G. Isorhamnetin, hispidulin, and cirsimaritin identified in Tamarix ramosissima barks from southern Xinjiang and their antioxidant and antimicrobial activities. Molecules 2019, 24, 390.
- Bhattacharya, D.; Ghosh, D.; Bhattacharya, S.; Sarkar, S.; Karmakar, P.; Koley, H.; Gachhui, R. Antibacterial activity of polyphenolic fraction of Kombucha against Vibrio cholerae: Targeting cell membrane. Lett. Appl. Microbiol. 2018, 66, 145–152.
- Dayem, A.A.; Choi, H.Y.; Kim, Y.B.; Cho, S.-G. Antiviral effect of methylated flavonol isorhamnetin against influenza. PLoS ONE 2015, 10, e0121610.
- Zhan, Y.; Ta, W.; Tang, W.; Hua, R.; Wang, J.; Wang, C.; Lu, W. Potential antiviral activity of isorhamnetin against SARS-CoV-2 spike pseudotyped virus in vitro. Drug Dev. Res. 2021, 82, 1124–1130.
- Kim, H.-K.; Kang, B.-J.; Park, K.-J.; Ko, B.-S.; Whang, W.-K. Anti-Herpes Simplex Virus type I (HSV-1) Effect of Isorhamnetin 3-0-beta-D-Glucopyranoside Isolated from Brassica rapa. Yakhak Hoeji 1998, 42, 607–612.
- Matboli, M.; Saad, M.; Hasanin, A.H.; Saleh, L.A.; Baher, W.; Bekhet, M.M.; Eissa, S. New insight into the role of isorhamnetin as a regulator of insulin signaling pathway in type 2 diabetes mellitus rat model: Molecular and computational approach. Biomed. Pharmacother. 2021, 135, 111176.
- Kylin, E. Studien ueber das Hypertonie-Hyperglyka “mie-Hyperurika” miesyndrom. Zent. Inn. Med. 1923, 44, 105–127.
- Reaven, G.M. Role of insulin resistance in human disease. Diabetes 1988, 37, 1595–1607.
- Tan, S.Y.; Wong, J.L.M.; Sim, Y.J.; Wong, S.S.; Elhassan, S.A.M.; Tan, S.H.; Lim, G.P.L.; Tay, N.W.R.; Annan, N.C.; Bhattamisra, S.K. Type 1 and 2 diabetes mellitus: A review on current treatment approach and gene therapy as potential intervention. Diabetes Metab. Syndr. Clin. Res. Rev. 2019, 13, 364–372.
- Bruce, K.D.; Hanson, M.A. The developmental origins, mechanisms, and implications of metabolic syndrome. J. Nutr. Biochem. 2010, 140, 648–652.
- Oboh, G.; Ademiluyi, A.O.; Akinyemi, A.J.; Henle, T.; Saliu, J.A.; Schwarzenbolz, U. Inhibitory effect of polyphenol-rich extracts of jute leaf (Corchorus olitorius) on key enzyme linked to type 2 diabetes (α-amylase and α-glucosidase) and hypertension (angiotensin I converting) in vitro. J. Funct. Foods 2012, 4, 450–458.
- Ganbold, M.; Owada, Y.; Ozawa, Y.; Shimamoto, Y.; Ferdousi, F.; Tominaga, K.; Zheng, Y.-W.; Ohkohchi, N.; Isoda, H. Isorhamnetin alleviates steatosis and fibrosis in mice with nonalcoholic steatohepatitis. Sci. Rep. 2019, 9, 1–11.
- Jiang, H.; Yamashita, Y.; Nakamura, A.; Croft, K.; Ashida, H. Quercetin and its metabolite isorhamnetin promote glucose uptake through different signalling pathways in myotubes. Sci. Rep. 2019, 9, 2690.
- Lee, D.; Park, J.Y.; Lee, S.; Kang, K.S. In Vitro Studies to Assess the α-Glucosidase Inhibitory Activity and Insulin Secretion Effect of Isorhamnetin 3-O-Glucoside and Quercetin 3-O-Glucoside Isolated from Salicornia herbacea. Processes 2021, 9, 483.
- Lee, J.; Jung, E.; Lee, J.; Kim, S.; Huh, S.; Kim, Y.; Kim, Y.; Byun, S.Y.; Kim, Y.S.; Park, D. Isorhamnetin represses adipogenesis in 3T3-L1 cells. Obesity 2009, 17, 226–232.
- Manach, C.; Texier, O.; Régérat, F.; Agullo, G.; Demigné, C.; Rémésy, C. Dietary quercetin is recovered in rat plasma as conjugated derivatives of isorhamnetin and quercetin. J. Nutr. Biochem. 1996, 7, 375–380.
- Eid, H.M.; Martineau, L.C.; Saleem, A.; Muhammad, A.; Vallerand, D.; Benhaddou-Andaloussi, A.; Nistor, L.; Afshar, A.; Arnason, J.T.; Haddad, P.S. Stimulation of AMP-activated protein kinase and enhancement of basal glucose uptake in muscle cells by quercetin and quercetin glycosides, active principles of the antidiabetic medicinal plant Vaccinium vitis-idaea. Mol. Nutr. Food Res. 2010, 54, 991–1003.
- Kalai, F.Z.; Han, J.; Ksouri, R.; Abdelly, C.; Isoda, H. Oral administration of Nitraria retusa ethanolic extract enhances hepatic lipid metabolism in db/db mice model ‘BKS. Cg-Dock7m+/+ Leprdb/J’through the modulation of lipogenesis–lipolysis balance. Food Chem. Toxicol. 2014, 72, 247–256.
- Zar Kalai, F.; Han, J.; Ksouri, R.; El Omri, A.; Abdelly, C.; Isoda, H. Antiobesity effects of an edible halophyte Nitraria retusa Forssk in 3T3-L1 preadipocyte differentiation and in C57B6J/L mice fed a high fat diet-induced obesity. Evid.-Based Complement. Altern. Med. 2013, 2013, 368658.
- Kim, B.; Choi, Y.E.; Kim, H.S. Eruca sativa and its flavonoid components, quercetin and isorhamnetin, improve skin barrier function by activation of peroxisome proliferator-activated receptor (PPAR)-α and suppression of inflammatory cytokines. Phytother. Res. 2014, 28, 1359–1366.
- Lee, M.-K.; Yang, H.-K.; Ha, N.-R.; Sung, S.-H.; Kim, Y.-C. Isorhamnetin from Oenanthe javanica attenuates fibrosis in rat hepatic stellate cells via inhibition of ERK signaling pathway. Nat. Prod. Sci. 2008, 14, 81–85.
- Heim, M. The Jak–STAT pathway: Specific signal transduction from the cell membrane to the nucleus. Eur. J. Clin. Investig. 1996, 26, 1–12.
- Selvaraj, J. Identification of new antidiabetic agents targeting GLUT4 protein using in silico analysis. Int. J. Green Pharm. 2019, 12, S876.
- Eid, H.M.; Nachar, A.; Thong, F.; Sweeney, G.; Haddad, P.S. The molecular basis of the antidiabetic action of quercetin in cultured skeletal muscle cells and hepatocytes. Pharmacogn. Mag. 2015, 11, 74.
- Xu, M.; Hu, J.; Zhao, W.; Gao, X.; Jiang, C.; Liu, K.; Liu, B.; Huang, F. Quercetin differently regulates insulin-mediated glucose transporter 4 translocation under basal and inflammatory conditions in adipocytes. Mol. Nutr. Food Res. 2014, 58, 931–941.
- Calkin, A.C.; Thomas, M.C. PPAR agonists and cardiovascular disease in diabetes. PPAR Res. 2008, 2008, 245410.
- Holm, L.J.; Mønsted, M.Ø.; Haupt-Jorgensen, M.; Buschard, K. PPARs and the development of type 1 diabetes. PPAR Res. 2020, 2020, 6198628.
- Lamichane, S.; Dahal Lamichane, B.; Kwon, S.-M. Pivotal roles of peroxisome proliferator-activated receptors (PPARs) and their signal cascade for cellular and whole-body energy homeostasis. Int. J. Mol. Sci. 2018, 19, 949.
- Varga, T.; Czimmerer, Z.; Nagy, L. PPARs are a unique set of fatty acid regulated transcription factors controlling both lipid metabolism and inflammation. Biochim. Biophys. Acta -Mol. Basis Dis. 2011, 1812, 1007–1022.
- Zhang, Y.; Gu, M.; Cai, W.; Yu, L.; Feng, L.; Zhang, L.; Zang, Q.; Wang, Y.; Wang, D.; Chen, H. Dietary component isorhamnetin is a PPARγ antagonist and ameliorates metabolic disorders induced by diet or leptin deficiency. Sci. Rep. 2016, 6, 1–12.
- Krishnapuram, R.; Dhurandhar, E.J.; Dubuisson, O.; Hegde, V.; Dhurandhar, N.V. Doxycycline-regulated 3T3-L1 preadipocyte cell line with inducible, stable expression of adenoviral E4orf1 gene: A cell model to study insulin-independent glucose disposal. PLoS ONE 2013, 8, e60651.
- Ruiz-Ojeda, F.J.; Rupérez, A.I.; Gomez-Llorente, C.; Gil, A.; Aguilera, C.M. Cell models and their application for studying adipogenic differentiation in relation to obesity: A review. Int. J. Mol. Sci. 2016, 17, 1040.
- Lee, J.; Lee, J.; Jung, E.; Hwang, W.; Kim, Y.-S.; Park, D. Isorhamnetin-induced anti-adipogenesis is mediated by stabilization of β-catenin protein. Life Sci. 2010, 86, 416–423.
- Farias-Pereira, R.; Savarese, J.; Yue, Y.; Lee, S.-H.; Park, Y. Fat-lowering effects of isorhamnetin are via NHR-49-dependent pathway in Caenorhabditis elegans. Curr. Res. Food Sci. 2020, 2, 70–76.
- Sherwin, R.S. Role of liver in glucose homeostasis. Diabetes Care 1980, 3, 261–265.
- Roden, M. Mechanisms of disease: Hepatic steatosis in type 2 diabetes—Pathogenesis and clinical relevance. Nat. Clin. Pract. Endocrinol. Metab. 2006, 2, 335–348.
- Koyama, Y.; Brenner, D.A. Liver inflammation and fibrosis. J. Clin. Investig. 2017, 127, 55–64.
- Marks, J.B.; Raskin, P. Cardiovascular risk in diabetes: A brief review. J. Diabetes Its Complicat. 2000, 14, 108–115.
- Marra, F.; Gastaldelli, A.; Baroni, G.S.; Tell, G.; Tiribelli, C. Molecular basis and mechanisms of progression of non-alcoholic steatohepatitis. Trends Mol. Med. 2008, 14, 72–81.
- Ahmad, A.; Ahmad, R. Understanding the mechanism of hepatic fibrosis and potential therapeutic approaches. Saudi J. Gastroenterol. 2012, 18, 155.
- Sánchez-Valle, V.; Chavez-Tapia, N.C.; Uribe, M.; Méndez-Sánchez, N. Role of oxidative stress and molecular changes in liver fibrosis: A review. Curr. Med. Chem. 2012, 19, 4850–4860.
- Shek, F.W.; Benyon, R.C. How can transforming growth factor beta be targeted usefully to combat liver fibrosis? Eur. J. Gastroenterol. Hepatol. 2004, 16, 123–126.
- Liu, X.; Hu, H.; Yin, J.Q. Therapeutic strategies against TGF-β signaling pathway in hepatic fibrosis. Liver Int. 2006, 26, 8–22.
- Liu, N.; Feng, J.; Lu, X.; Yao, Z.; Liu, Q.; Lv, Y.; Han, Y.; Deng, J.; Zhou, Y. Isorhamnetin inhibits liver fibrosis by reducing autophagy and inhibiting extracellular matrix formation via the TGF-β1/Smad3 and TGF-β1/p38 MAPK pathways. Mediat. Inflamm. 2019, 2019, 6175091.
- Katyal, S.; Oliver, J.H., III; Peterson, M.S.; Ferris, J.V.; Carr, B.S.; Baron, R.L. Extrahepatic metastases of hepatocellular carcinoma. Radiology 2000, 216, 698–703.
- Singh, M.K.; Das, B.K.; Choudhary, S.; Gupta, D.; Patil, U.K. Diabetes and hepatocellular carcinoma: A pathophysiological link and pharmacological management. Biomed. Pharmacother. 2018, 106, 991–1002.
- Yang, J.D.; Ahmed, F.; Mara, K.C.; Addissie, B.D.; Allen, A.M.; Gores, G.J.; Roberts, L.R. Diabetes is associated with increased risk of hepatocellular carcinoma in patients with cirrhosis from nonalcoholic fatty liver disease. Hepatology 2020, 71, 907–916.
- Teng, B.-s.; Lu, Y.-H.; Wang, Z.-T.; Tao, X.-Y.; Wei, D.-Z. In vitro anti-tumor activity of isorhamnetin isolated from Hippophae rhamnoides L. against BEL-7402 cells. Pharmacol. Res. 2006, 54, 186–194.
- Youness, R.; Assal, R.; Gad, M.; Motaal, A.A. Hijacking hepatocellular carcinoma (HCC) tumour progression through restoring TP53/miR-15a/miR-16 tumour suppressor axis by a novel quercetin glycoside. ESMO Open 2018, 3, A160.
- Burganova, G.; Bridges, C.; Thorn, P.; Landsman, L. The Role of Vascular Cells in Pancreatic Beta-Cell Function. Front. Endocrinol. 2021, 12, 442.
- Marchetti, P.; Ferrannini, E. International Textbook of Diabetes Mellitus; Wiley-Blackwell: Hoboken, NJ, USA, 2015; Volume 2, p. 1240.
- Marchetti, P.; Bugliani, M.; De Tata, V.; Suleiman, M.; Marselli, L. Pancreatic beta cell identity in humans and the role of type 2 diabetes. Front. Cell Dev. Biol. 2017, 5, 55.
- Cerf, M.E. Beta cell dysfunction and insulin resistance. Front. Endocrinol. 2013, 4, 37.
- Wang, J.-L.; Quan, Q.; Ji, R.; Guo, X.-Y.; Zhang, J.-M.; Li, X.; Liu, Y.-G. Isorhamnetin suppresses PANC-1 pancreatic cancer cell proliferation through S phase arrest. Biomed. Pharmacother. 2018, 108, 925–933.
- Grdović, N.; Dinić, S.; Arambašić, J.; Mihailović, M.; Uskoković, A.; Marković, J.; Poznanović, G.; Vidović, S.; Zeković, Z.; Mujić, A. The protective effect of a mix of Lactarius deterrimus and Castanea sativa extracts on streptozotocin-induced oxidative stress and pancreatic β-cell death. Br. J. Nutr. 2012, 108, 1163–1176.
- Vessal, M.; Hemmati, M.; Vasei, M. Antidiabetic effects of quercetin in streptozocin-induced diabetic rats. Comp. Biochem. Physiol. Part C Toxicol. Pharmacol. 2003, 135, 357–364.
- Coskun, O.; Kanter, M.; Korkmaz, A.; Oter, S. Quercetin, a flavonoid antioxidant, prevents and protects streptozotocin-induced oxidative stress and β-cell damage in rat pancreas. Pharmacol. Res. 2005, 51, 117–123.
- Hii, C.; Howell, S. Effects of flavonoids on insulin secretion and 45Ca2+ handling in rat islets of Langerhans. J. Endocrinol. 1985, 107, 1–8.
- He, Z.; King, G.L. Microvascular complications of diabetes. Endocrinol. Metab. Clin. 2004, 33, 215–238.
- Patel, S.; Santani, D. Role of NF-κB in the pathogenesis of diabetes and its associated complications. Pharmacol. Rep. 2009, 61, 595–603.
- Suryavanshi, S.; Kulkarni, Y. NF-κβ: A potential target in the management of vascular complications of diabetes. Front. Pharmacol. 2017, 8, 798.
- Singh, V.P.; Bali, A.; Singh, N.; Jaggi, A.S. Advanced glycation end products and diabetic complications. Korean J. Physiol. Pharmacol. 2014, 18, 1–14.
- Leo, C.-H.; Woodman, O.L. Flavonols in the prevention of diabetes-induced vascular dysfunction. J. Cardiovasc. Pharmacol. 2015, 65, 532.
- Hämäläinen, M.; Nieminen, R.; Vuorela, P.; Heinonen, M.; Moilanen, E. Anti-inflammatory effects of flavonoids: Genistein, kaempferol, quercetin, and daidzein inhibit STAT-1 and NF-κB activations, whereas flavone, isorhamnetin, naringenin, and pelargonidin inhibit only NF-κB activation along with their inhibitory effect on iNOS expression and NO production in activated macrophages. Mediat. Inflamm. 2007, 2007, 045673.





