
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Wei Xu | + 1355 word(s) | 1355 | 2022-01-05 08:21:52 | | | |
| 2 | Conner Chen | Meta information modification | 1355 | 2022-01-19 03:18:25 | | |
Video Upload Options
Over the centuries, carbon allotropes have been playing a significant role in material science and engineering due to their outstanding electric, magnetic, optical, and mechanical properties. Compared to traditional carbon materials such as diamond and graphite, carbon nanomaterials consist of carbons with different hybridization types, namely linear (sp), planar (sp2), or tetrahedral (sp3) bond configurations, or even a combination of several types. Owning to the development of STM (scanning tunneling microscope) and nc-AFM (noncontact atomic force microscope), a wide variety of low-dimensional carbon nanostructures have been synthesized and characterized at the atomic scale on surfaces, such as linear polymers comprising of hydrocarbons, graphene nanoribbons, porous graphene, and polycyclic aromatic hydrocarbons. In particular, nanostructures containing sp-hybridized carbons are of great advantage for their structural linearity and small steric demands as well as intriguing electronic and mechanical properties.
1. 0D sp-Carbon Nanostructures
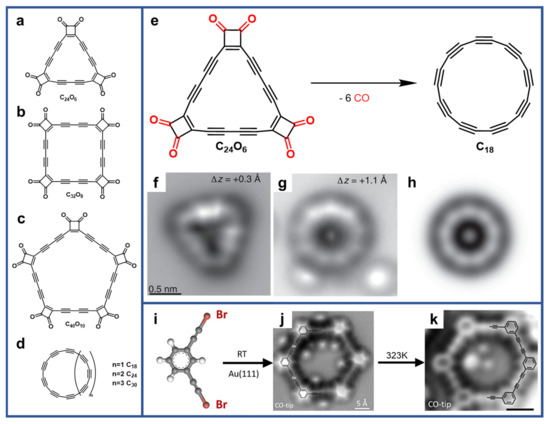
2. 2D sp-Carbon Nanostructures
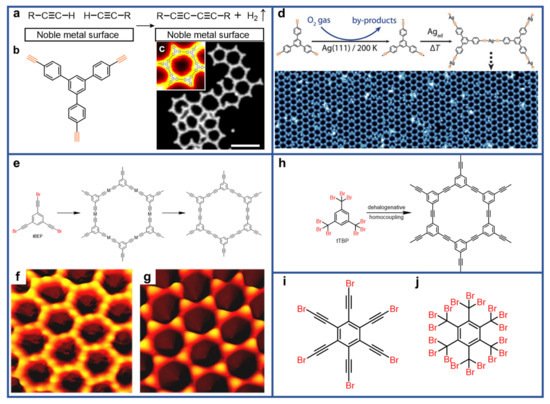
References
- Torelli, T.; Mitas, L. Electron Correlation in C4N+2 Carbon Rings: Aromatic Versus Dimerized Structures. Phys. Rev. Lett. 2000, 85, 1702–1705.
- Parasuk, V.; Almlof, J.; Feyereisen, M.W. The All-Carbon Molecule: Cumulene or Polyacetylene? J. Am. Chem. Soc. 1991, 113, 1049–1050.
- Neiss, C.; Trushin, E.; Gorling, A. The Nature of One-Dimensional Carbon: Polyynic Versus Cumulenic. ChemPhysChem 2014, 15, 2497–2502.
- McElvany, S.W.; Ross, M.M.; Goroff, N.S.; Diederich, F. Cyclocarbon Coalescence: Mechanisms for Tailor-Made Fullerene Formation. Science 1993, 259, 1594–1596.
- Kaiser, K.; Scriven, L.M.; Schulz, F.; Gawel, P.; Gross, L.; Anderson, H.L. An sp-Hybridized Molecular Carbon Allotrope, Cyclocarbon. Science 2019, 365, 1299–1301.
- Liu, M.; Li, S.; Zhou, J.; Zha, Z.; Pan, J.; Li, X.; Zhang, J.; Liu, Z.; Li, Y.; Qiu, X. High-Yield Formation of Graphdiyne Macrocycles through On-Surface Assembling and Coupling Reaction. ACS Nano 2018, 12, 12612–12618.
- Sun, Q.; Zhang, C.; Wang, L.; Li, Z.; Hu, A.; Tan, Q.; Xu, W. Surface-Assisted cis-trans Isomerization of an Alkene Molecule on Cu(110). Chem. Commun. 2014, 50, 1728–1730.
- Sun, Q.; Cai, L.; Ma, H.; Yuan, C.; Xu, W. The Stereoselective Synthesis of Dienes through Dehalogenative Homocoupling of Terminal Alkenyl Bromides on Cu(110). Chem. Commun. 2016, 52, 6009–6012.
- Baughman, R.H.; Eckhardt, H.; Kertesz, M. Structure-Property Predictions for New Planar Forms of Carbon: Layered Phases Containing sp2 and sp Atoms. J. Chem. Phys. 1987, 87, 6687–6699.
- Zhang, Y.Q.; Kepcija, N.; Kleinschrodt, M.; Diller, K.; Fischer, S.; Papageorgiou, A.C.; Allegretti, F.; Bjork, J.; Klyatskaya, S.; Klappenberger, F.; et al. Homo-Coupling of Terminal Alkynes on a Noble Metal Surface. Nat. Commun. 2012, 3, 1286.
- Zhang, Y.Q.; Paintner, T.; Hellwig, R.; Haag, F.; Allegretti, F.; Feulner, P.; Klyatskaya, S.; Ruben, M.; Seitsonen, A.P.; Barth, J.V.; et al. Synthesizing Highly Regular Single-Layer Alkynyl-Silver Networks at the Micrometer Scale via Gas-Mediated Surface Reaction. J. Am. Chem. Soc. 2019, 141, 5087–5091.
- Sun, Q.; Cai, L.; Ma, H.; Yuan, C.; Xu, W. Dehalogenative Homocoupling of Terminal Alkynyl Bromides on Au(111): Incorporation of Acetylenic Scaffolding into Surface Nanostructures. ACS Nano 2016, 10, 7023–7030.
- Sun, Q.; Yu, X.; Bao, M.; Liu, M.; Pan, J.; Zha, Z.; Cai, L.; Ma, H.; Yuan, C.; Qiu, X.; et al. Direct Formation of C–C Triple-Bonded Structural Motifs by On-Surface Dehalogenative Homocouplings of Tribromomethyl-Substituted Arenes. Angew. Chem. Int. Ed. 2018, 57, 4035–4038.





