
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Dennis Kyereh | + 3225 word(s) | 3225 | 2021-12-21 08:54:55 | | | |
| 2 | Dennis Kyereh | Meta information modification | 3225 | 2022-01-06 12:47:48 | | | | |
| 3 | Prasad S Hendre | Meta information modification | 3225 | 2022-01-06 13:00:00 | | | | |
| 4 | Peter Tang | -66 word(s) | 3159 | 2022-01-08 07:42:05 | | |
Video Upload Options
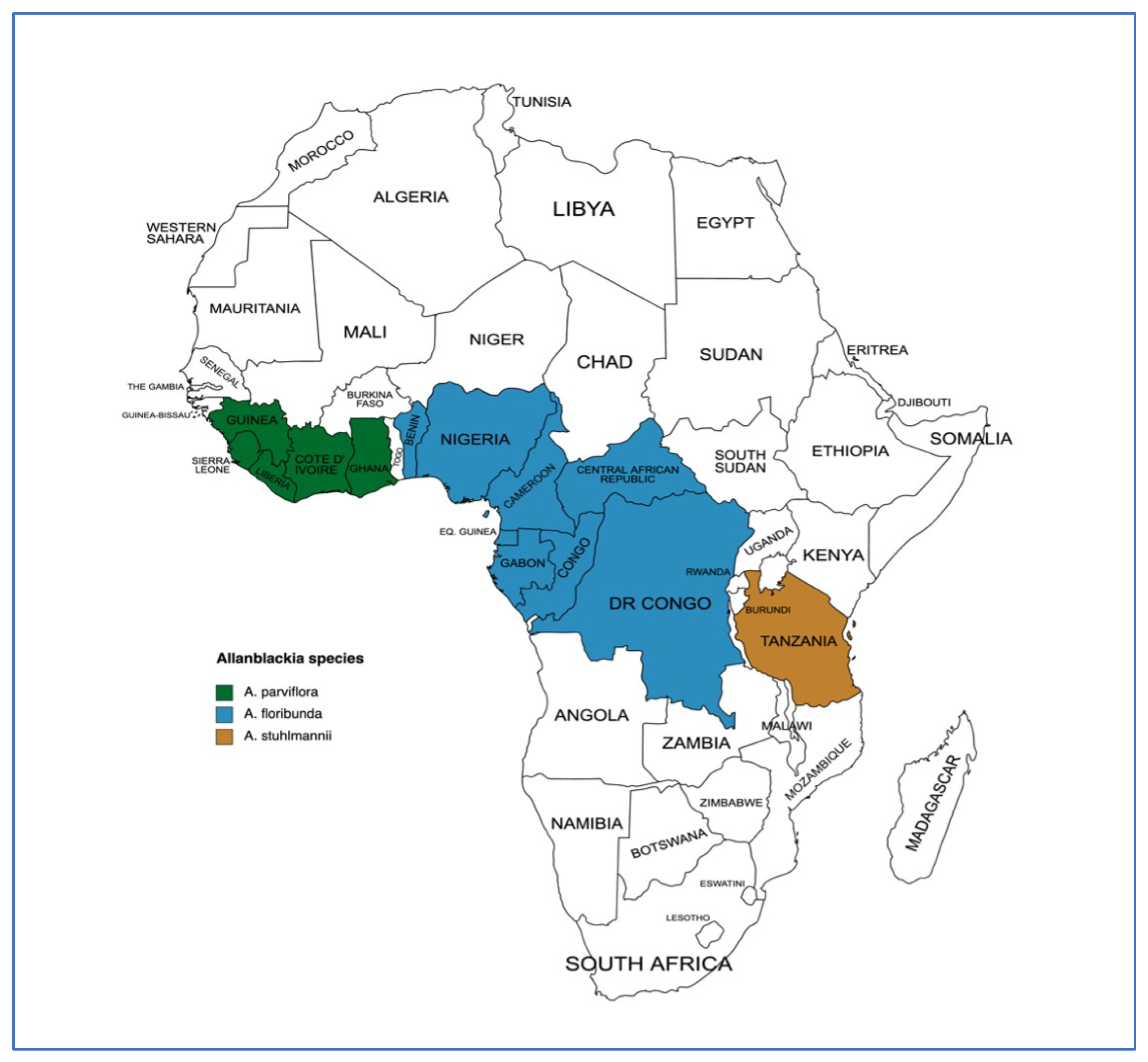
Allanblackia parviflora A. Chev. is an indigenous tree species that are found in West African rainforest zones. It is an underutilized fruit tree species that have been targeted for improvement as part of efforts to domesticate high-value indigenous multi-purpose trees for fruit and seed production in Africa. Allanblackia has several benefits, such as providing shade, timber, and medicine; however, the production of edible oil from its seeds is the economically most important use. There is evidence that the Allanblackia seed oil, which is used for cooking, the production of margarine and the manufacturing of ointments and soap, is being developed as a new agri-business in Ghana, Nigeria, Cameroon, and Tanzania. Despite the nutritional and socio-economic importance of A. parviflora, it is still at the early stages of its domestication process.
1. Introduction
2. Genus Allanblackia
3. Botanical Description



4. Propagation
5. Uses
6. Chemical Composition of A. parviflora Seeds
7. Morphological and Genetic Diversity
8. Conclusions
A. parviflora is one of the priority fruit tree species identified for improvement and domestication in Sub-Saharan Africa. It has been targeted for improvement for the production of oil from the seeds, for benefits not only to the local populations but also the economies of producing countries. Despite the great potential of this species, there are still significant knowledge gaps that require urgent research attention.
This entry shows that the most important economic use of A. parviflora is the production of edible oil from the seeds. This might justify why ICRAF and its partners already started with the first domestication steps by selecting superior individuals/populations in Ghana, Cameroon, Nigeria, and Tanzania, while Unilever remains a major buyer of the Allanblackia seed oil. However, scientific information on the seeds’ phytochemical composition and variability is scarce. Additionally, only a little or no information is available on the diversity and management of the species in the regions where it is naturally found. More specifically, basic information on genetic diversity, silvicultural management, productivity, methods of propagation and cultivation are not well documented. The development of morphological descriptors, the use of modern molecular markers for genetic diversity assessment, and studies on the phytochemistry of seeds are therefore highly recommended. Moreover, qualitative research on the preferences of local populations who produce A. parviflora seeds should be conducted to reveal their preferred traits of interest for development. The selection of superior mother tree populations may begin once these attributes are determined. The next step after identifying trees with superior desirable traits will be the multiplication of best individuals. Extensive studies on vegetative propagation and propagule regeneration of the species are urgently needed, as the currently available information is not ample. Seedlings from generative propagation are currently the easiest solution for farmers, but there is a limitation to such a method due to the dioecious nature of the species which makes it difficult to identify the sex of the tree at the juvenile stage. While research efforts should be focused on determining the sex of trees at a young age, studies on multiplication by vegetative propagation is crucial, since such a method of propagation assures the conservation of the traits of interest. The researchers conclude that A. parviflora has great potential to ensure food and nutritional security and alleviate poverty among rural populations in West Africa, and therefore could be domesticated to promote widespread planting in agroforestry systems and for the conservation of the genetic resource if the knowledge gaps identified here are fulfilled.
References
- Hetemäki, L.; Hurmekoski, E. Forest products markets under change: Review and research implications. Curr. For. Rep. 2016, 2, 177–188.
- Epanda, M.A.; Donkeng, R.T.; Nonga, F.N.; Frynta, D.; Adi, N.N.; Willie, J.; Speelman, S. Contribution of Non-Timber Forest Product valorisation to the livelihood assets of local people in the Northern periphery of the Dja Faunal Reserve, East Cameroon. Forests 2020, 11, 1019.
- Norris, K.; Asase, A.; Collen, B.; Gockowksi, J.; Mason, J.; Phalan, B.; Wade, A. Biodiversity in a Forest-Agriculture Mosaic—The changing face of West African rainforests. Biol. Conserv. 2010, 143, 2341–2350.
- Peprah, T.; Ofori, D.A.; Siaw, D.E.K.A.; Addo-Danso, S.D.; Cobbinah, J.R.; Simons, A.J.; Jamnadass, R. Reproductive biology and characterization of Allanblackia parviflora A. Chev. in Ghana. Genet. Resour. Crop Evol. 2009, 56, 1037–1044.
- AOCC. African Orphan Crops Consortium—Healthy Africa through Nutritious, Diverse and Local Food Crops. Available online: http://africanorphancrops.org/ (accessed on 15 November 2021).
- ICRAF. World Agroforestry. Transforming Lives and Landscapes with Trees. Available online: https://worldagroforestry.org/ (accessed on 15 November 2021).
- Hendre, P.S.; Muthemba, S.; Kariba, R.; Muchugi, A.; Fu, Y.; Chang, Y.; Song, B.; Liu, H.; Liu, M.; Liao, X.; et al. African Orphan Crops Consortium (AOCC): Status of developing genomic resources for African Orphan Crops. Planta 2019, 250, 989–1003.
- Tsobeng, A.; Akem, M.; Avana, M.L.; Muchugi, A.; Degrande, A.; Tchoundjeu, Z.; Jamnadass, R.; Na’a, F. Tree-to-Tree variation in fruits of three populations of Trichoscypha acuminata (Engl.) in Cameroon. Sci. Afr. 2020, 7, e00235.
- Mabhaudhi, T.; Chimonyo, V.G.P.; Hlahla, S.; Massawe, F.; Mayes, S.; Nhamo, L.; Modi, A.T. Prospects of Orphan Crops in climate change. Planta 2019, 250, 695–708.
- Leakey, R.R.B.; Simons, A.J. The domestication and commercialization of indigenous trees in Agroforestry for the alleviation of poverty. In Directions in Tropical Agroforestry Research. Forestry Sciences; Springer: Singapore, 1998; Volume 53, pp. 165–176.
- Leakey, R.R.B.; Tchoundjeu, Z.; Schreckenberg, K.; Shackleton, S.E.; Shackleton, C.M. Agroforestry Tree Products (AFTPs): Targeting poverty reduction and enhanced livelihoods. Int. J. Agric. Sustain. 2005, 3, 1–23.
- Tchoundjeu, Z.; Asaah, E.K.; Anegbeh, P.; Degrande, A.; Mbile, P.; Facheux, C.; Tsobeng, A.; Atangana, A.R.; Ngo-Mpeck, M.L.; Simons, A.J. Putting participatory domestication into practice in West and Central Africa. For. Trees Livelihoods 2006, 16, 53–69.
- Ofori, D.A.; Gyau, A.; Dawson, I.K.; Asaah, E.; Tchoundjeu, Z.; Jamnadass, R. Developing more productive African Agroforestry systems and improving food and nutritional security through tree domestication. Curr. Opin. Environ. Sustain. 2014, 6, 123–127.
- Siaw, D.; Cobbinah, J.R.; Kankam, B.O.; Derkyi, S.A.; Oduro, K.A.; Agyili, J.; Peprah, T. Final report on Allanblackia floribunda. Novella Project (69p). Submitted to Unilever Ghana Ltd. by Forestry Research Institute of Ghana. 2003. Available online: https://scirp.org/reference/ReferencesPapers.aspx?ReferenceID=1577077 (accessed on 12 November 2021).
- Sefah, W.; Adubofour, J.; Oldham, J.H. Extraction and Characterization of Vegetable Fat from Allanblackia floribunda. Master’s Thesis, Kwame Nkrumah University of Science and Technology, Kumasi, Ghana, 2006.
- Ofori, D.; Siaw, D.E.K.A.; Peprah, T.; Cobbinah, J.R. Domestication of Allanblackia in Ghana; Progress Report (January–June 2006); ICRAF: Nairobi, Kenya, 2006.
- Takase, M.; Essandoh, P.K.; Kipkoech, R. New non-edible Allanblackia parviflora seed oil as an alternative feedstock for biodiesel production and characterization of the fuel. Discov. Sustain. 2021, 2, 8.
- Shrestha, R.; Akangaamkum, A. Novella Partnership: A partnership for poverty reduction through sustainable enterprise development based on Allanblackia, SNV Ghana. SNV Ghana 2008, 20, 1–19.
- van Rompaey, R. Distribution and Ecology of Allanblackia Spp. (Clusiaceae) in African Rain Forests with Special Attention to the Development of a Wild Picking System of the Fruits. 2003. Available online: https://www.doc-developpement-durable.org/file/Culture/Arbres-Bois-de-Rapport-Reforestation/FICHES_ARBRES/Allanblackia/Ecology%20and%20distrib%20of%20Allanblackia%20in%20African%20rainforest%20Liberia.pdf (accessed on 6 November 2021).
- Bamps, P.; Robson, N.; Verdcourt, B. Flora of Tropical East Africa. Guttiferae; Royal Botanic Gardens, Kew: Richmond, UK, 1978.
- Crockett, S.L. Allanblackia oil: Phytochemistry and use as a functional food. Int. J. Mol. Sci. 2015, 16, 22333–22349.
- Bamps, P. Guttiferae. In Flore du Congo, du Rwanda et du Burundi, Spermatophytes; Boutique, R., Ed.; Jardin Botanique National de Belgique: Bruxelles, Belgium, 1970.
- Hawthorne, W.H.; Gyakari, N. Photo Guide to the Larger Trees of Ghana’s Forests; Oxford Forestry Institute: Oxford, UK, 2006.
- Orwa, C.; Oyen, L. Allanblackia parviflora A. Chev. In Vegetable Oils; Plant Resources of Tropical Africa; van Der Vossen, H.A.M., Mkamilo, G.S., Eds.; PROTA: Wageningen, The Netherlands, 2007; Volume 14, pp. 28–30.
- Hawthorne, W. FROGGIE: Forest Reserves of Ghana: Graphical Information Exhibitor. Programme, Database and Manual, Distributed with H & A Part 1 (Manual of the Program); ODA/FIMP/Ghana Forestry Department: Kumasi, Ghana, 1995; p. 117.
- Ofori, D.A.; Asomaning, J.M.; Peprah, T.; Agyeman, V.K.; Anjarwalla, P.; Tchoundjeu, Z.; Mowo, J.G.; Jamnadass, R. Addressing constraints in propagation of Allanblackia spp. through seed sectioning and air layering. J. Exp. Biol. Agric. Sci. 2015, 3, 89–96.
- Hall, J.; Swaine, M. Distribution and Ecology of Vascular Plants in a Tropical Rain Forest: Forest Vegetation in Ghana; Springer Science & Business Media: Berlin, Germany, 2013.
- Hawthorne, W. Field Guide to the Forest Trees of Ghana; Overseas Development Administration: London, UK, 1990.
- Peprah, T.; Oduro, K.A.; Siaw, D.E.K.A.; Cobbinah, J.R.; Tchoundjeu, Z.; Simons, A.J.; Jamnadass, R.; Ofori, D.A. Domestication and sustainable use of genetic resources of a native tree with high economic potential in Ghana. Open J. For. 2015, 5, 678–685.
- Ofori, D.A.; Kehlenbeck, K.; Munjuga, M.; Jamnadass, R.; Asaah, E.K.; Kattah, C.; Rutatina, F. Allanblackia species: A model for domestication of high potential tree crops in Africa. Acta Hortic. 2013, 979, 311–318.
- Ofori, D.A.; Peprah, T.; Cobbinah, J.R.; Atchwerebour, H.A.; Osabutey, F.; Tchoundjeu, Z.; Simons, A.J.; Jamnadass, R. Germination requirements of Allanblackia parviflora seeds and early growth of seedlings. New For. 2011, 41, 337–348.
- Anegbeh, P.O.; Iruka, C.; Nkirika, C. Enhancing germination of bitter cola (Garcinia kola), Heckel: Prospects for Agroforestry farmers in the Niger Delta. Sci. Afr. 2006, 5, 38–44.
- Atangana, A.R.; Tchoundjeu, Z.; Asaah, E.K.; Simons, A.J.; Khasa, D.P. Domestication of Allanblackia floribunda: Amenability to vegetative propagation. For. Ecol. Manag. 2006, 237, 246–251.
- Ofori, D.; Peprah, T.; Henneh, S.; von Berg, J.; Tchoundjeu, Z.; Jamnadass, R.; Simons, A. Utility of grafting in tree domestication programme with special reference to Allanblackia parviflora A. Chev. Gh. J. For. 2008, 23, 42–48.
- Asaah, E.; Tchoundjeu, Z.; Ngahane, W.; Tsobeng, A.; Kouodiekong, L.; Jamnadass, R.; Simons, A.; Asaah, E. Allanblackia floribunda: A new oil tree crop for Africa: Amenability to grafting. New For. 2011, 41, 389–398.
- Longman, K. Conservation and Utilization of Gene Resources by Vegetative Multiplication of Tropical Trees; AGRIS & FAO: Rome, Italy, 1976.
- Longman, K. Tropical Forest Tress. In CRC Handbook of Flowering; Halevy, A.H., Ed.; CRC Press Inc.: Roca Raton, FL, USA, 1985; Volume 1, pp. 23–39.
- Bawa, K. Breeding of Tropical Hardwoods: An Evaluation of underlying bases, current status and future prospects. In Tropical Trees; Variation, Breeding and Conservation; Burley, J., Styles, B.T., Eds.; Academic Press: London, UK, 1976; pp. 43–59.
- Ennos, R.; Cowie, N.; Legg, C.; Sydes, C. Which Measures of genetic variation are relevant in plant conservation? A case study of Primula Scotica. In The Role of Genetics in Conserving Small Populations; Tew, T.E., Crawford, T.J., Spencer, J.W., Stevens, D.P., Usher, M.B., Marren, J., Eds.; JNCC: Peterborough, UK, 1997; pp. 73–79.
- Abbiw, D. Useful Plants of Ghana: West African Uses of Wild and Cultivated Plants; Intermediate Technology Publications and The Royal Botanic Gardens Kew UK: London, UK, 1990; p. 337.
- Ofori, D.A.; Peprah, T.; Koech, G.; Anjarwalla, P.; Munjuga, M.; Jamnadass, R. Survival, growth performance and reproductive biology of Allanblackia Parviflora A. Chev., a high valued indigenous fruit tree species in Ghana. Genet. Resour. Crop. Evol. 2016, 63, 791–800.
- Orwa, C.; Munjuga, M. Allanblackia floribunda O Liv. In Vegetable Oils/Oléagineux; Van Der Vossen, H.A.M., Mkamilo, G.S., Eds.; Prota: Wageningen, The Netherlands, 2007.
- Orwa, C.; Mutua, A.; Kindt, R.; Jamnadass, R.; Simons, A. Agroforestree Database: A Tree Reference and Selection Guide. Version 4. 2009. Available online: http://www.worldagroforestry.org/sites/treedbs/treedatabases.asp (accessed on 18 October 2021).
- Attipoe, L.; van Andel, A.; Nyame, S.K. The Novella Project: Developing a sustainable supply chain for Allanblackia oil. Frontis 2006, 14, 179–189. Available online: https://library.wur.nl/ojs/index.php/frontis/article/view/982 (accessed on 10 October 2021).
- Sefah, W.; Adubofuor, J.; Oldham, J.H. Optimum conditions for expression of oil from Allanblackia floribunda seeds and assessing the quality and stability of pressed and solvent extracted oil. Afr. J. Food Sci. 2010, 4, 563–570.
- Irvine, F.R. Woody Plants of Ghana; Oxford University Press: Oxford, UK, 1961.
- Adubofuor, J.; Sefah, W.; Oldham, J.H. Nutrient composition of Allanblackia parviflora seed kernels and oil compared with some plant fats and oils and application of the oil in soap preparation. J. Cereals Oilseeds 2013, 4, 1–9.
- Oppong, K. Allanblackia seed oil supply chain management in Ghana. In Allanblackia Stakeholders’ Workshop; Sanaa Lodge: Cape Coast, Ghana, 2008.
- Kattah, C. Developing a sustainable Allanblackia business in Africa. In First Allanblackia Conference; Thinkers Village Hotel: Monrovia, Liberia, 2010; Volume 23.
- Hermann, M. The impact of the European novel food regulation on trade and food innovation based on traditional plant foods from developing countries. Food Policy 2009, 34, 499–507.
- Meara, M.; Zaky, Y. Fatty acids and glycerides of the seed fats of Allanblackia floribunda and Allanblackia parviflora. J. Soc. Chem. Ind. 1940, 59, 25–26.
- Atangana, A.R.; van der Vlis, E.; Khasa, D.P.; van Houten, D.; Beaulieu, J.; Hendrickx, H. Tree-to-Tree variation in stearic and oleic acid content in seed fat from Allanblackia floribunda from wild stands: Potential for tree breeding. Food Chem. 2011, 126, 1579–1585.
- Foma, M.; Abdala, T. Kernel oils of seven plant species of Zaire. J. Am. Oil Chem. Soc. 1985, 62, 910–911.
- Sefah, W.; Pierre, H.; Mary, C.B.; Lydia, S. Oil yields for Allanblackia parviflora (Tallow tree) in Ghana: The effects of oil extraction methods, tree morphology and environmental characteristics. Afr. J. Food Sci. 2019, 13, 163–181.
- Atangana, A.R.; Beaulieu, J.; Khasa, D.P. Wild genetic diversity preservation in a small-sized first generation breeding Population of Allanblackia floribunda (Clusiaceae). Tree Genet. Genomes 2010, 6, 127–136.
- Russell, J.R.; Kadu, C.A.C.; Jamnadass, R.; Booth, A.; Cordeiro, N.J.; Woodhead, M.; Dawson, I.K. AFLP and SSR diversity in the African fruit tree Allanblackia: Implications for management of a genus newly subject to domestication for the edible oil industry. Tree Genet. Genomes 2009, 5, 517–527.
- Leakey, R.R.B.; Greenwell, P.; Hall, M.N.; Atangana, A.R.; Usoro, C.; Anegbeh, P.O.; Fondoun, J.M.; Tchoundjeu, Z. Tree-to-Tree variation in food-thickening properties and in fat and protein contents of Dika Nut. Food Chem. 2005, 90, 365–378.
- Munjuga, M.; Ofori, D.; Sawe, C.; Asaah, E.; Anegbeh, P.; Peprah, T.; Mpanda, M.; Mwaura, L.; Mtui, E.; Sirito, C.; et al. Allanblackia Propagation Protocol; World Agroforestry Centre: Nairobi, Kenya, 2008.
- Mathew, M.M.; Munjuga, M.R.; Ndangalasi, H.J.; Cordeiro, N.J. Aspects of the floral and fruit biology of Allanblackia stuhlmannii (Clusiaceae), an Endemic Tanzanian tree. J. East Afr. Nat. Hist. 2009, 98, 79–93.
- Williams, J.G.K.; Kubelik, A.R.; Livak, K.J.; Rafalski, J.A.; Tingey, S.V. DNA polymorphisms amplified by arbitrary primers are useful as genetic markers. Nucleic Acids Res. 1990, 18, 6531–6535.
- Farooq, S.; Azam, F. Molecular markers in plant breeding- I: Concepts and characterization. Pak. J. Biol. Sci. 2002, 5, 1135–1140.
- Neale, D.; Ingvarsson, P. Population, quantitative and comparative genomics of adaptation in forest trees. Curr. Opin. Plant Biol. 2008, 11, 149–155.
- Kuhn, D.N.; Dillon, N.; Bally, I.; Groh, A.; Rahaman, J.; Warschefsky, E.; Freeman, B.; Innes, D.; Chambers, A.H. Estimation of genetic diversity and relatedness in a mango germplasm collection using SNP markers and a Simplified Visual Analysis method. Sci. Hortic. 2019, 252, 156–168.





