
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Surinder Singh | + 4391 word(s) | 4391 | 2021-12-23 08:37:17 | | | |
| 2 | Conner Chen | -2406 word(s) | 1985 | 2022-01-06 02:08:15 | | | | |
| 3 | Conner Chen | -2406 word(s) | 1985 | 2022-01-06 02:08:46 | | | | |
| 4 | Surinder Singh | -7 word(s) | 1978 | 2022-01-11 01:54:21 | | |
Video Upload Options
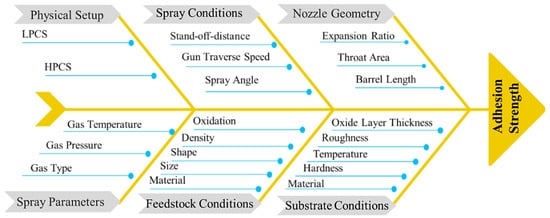
The cold spray process is governed by the impact of high velocity feedstock particles onto a substrate without melting. Hence, the bulk material properties are retained. However, it is challenging to achieve good adhesion strength. The adhesion strength depends on factors such as the cold spray process parameters, substrate conditions, coating/substrate interactions at the interface and feedstock material properties. This entry examines fundamental studies concerning the adhesion mechanisms of cold spray technology and considers the effect of cold spray input parameters such as temperature, stand-off-distance, pressure, process gas, spray angle, and traverse speed of the cold spray torch on the bonding mechanism and adhesion strength. Furthermore, the effects of substrate conditions such as temperature, hardness, roughness and material on the adhesion mechanism are highlighted. The effect of feedstock properties, such as feed rate, shape and size are summarized. Understanding the effect of these parameters is necessary to obtain the optimal input parameters that enable the best interfacial properties for a range of coating/substrate material combinations. It is expected that feedstock of spherical morphology and small particle size (<15 μm) provides optimal interfacial properties when deposited onto a mirror-finished substrate surface using high pressure cold spray. Deep insights into each parameter exposes the uncovered potential of cold spray as an additive manufacturing method.
1. Introduction
2. Adhesion Mechanism in Cold Spray
3. Concluding Remarks
References
- Ang, A.S.M.; Berndt, C.C.; Sesso, M.L.; Anupam, A.; Praveen, S.; Kottada, R.S.; Murty, B.S. Plasma-sprayed high entropy alloys: Microstructure and properties of AlCoCrFeNi and MnCoCrFeNi. Metall. Mater. Trans. 2015, 46, 791–800.
- Lima, R.S.; Kucuk, A.; Berndt, C.C.; Karthikeyan, J.; Kay, C.M.; Lindemann, J. Deposition efficiency, mechanical properties and coating roughness in cold-sprayed titanium. J. Mater. Sci. Lett. 2002, 21, 1687–1689.
- Niu, X.; Singh, S.; Garg, A.; Singh, H.; Panda, B.; Peng, X.; Zhang, Q. Review of materials used in laser-aided additive manufacturing processes to produce metallic products. Front. Mech. Eng. 2019, 14, 282–298.
- Espallargas, N. Future development of thermal spray coatings: Types, Designs, Manufacture and Applications. In Woodhead Publishing Series in Metals and Surface Engineering: Number 65; Elsevier, Woodhead Publishing: Cambridge, UK, 2015; pp. 27–28. ISBN 978-0-85709-774-3.
- Abadias, G.; Chason, E.; Keckes, J.; Sebastiani, M.; Thompson, G.B.; Barthel, E.; Doll, G.L.; Murray, C.E.; Stoessel, C.H.; Martinu, L. Review article: Stress in thin films and coatings: Current status, challenges, and prospects. J. Vac. Sci. Technol. A Vac. Surf. Film. 2018, 36, 020801.
- Meghwal, A.; Anupam, A.; Murty, B.S.; Berndt, C.C.; Kottada, R.S.; Ang, A.S.M. Thermal spray high-entropy alloy coatings: A review. J. Therm. Spray Technol. 2020, 29, 857–893.
- Singh, S.; Singh, P.; Singh, H.; Buddu, R.K. Characterization and comparison of copper coatings developed by low pressure cold spraying and laser cladding techniques. Mater. Today Proc. 2019, 18, 830–840.
- Frey, H.; Khan, H.R. Handbook of Thin-Film Technology; Springer: Berlin/Heidelberg, Germany, 2015; ISBN 978-3-642-05429-7.
- Singh, S.; Kumar, M.; Sodhi, G.P.S.; Buddu, R.K.; Singh, H. Development of thick copper claddings on SS316L steel for in-vessel components of fusion reactors and copper-cast iron canisters. Fusion Eng. Des. 2018, 128, 126–137.
- Papyrin, A.; Kosarev, V.; Klinkov, S.; Alkhimov, A.; Fomin, V.M. Cold Spray Technology; Elsevier Science: Amsterdam, The Netherlands, 2006; ISBN 978-0-08-046548-7. Available online: https://books.google.com.au/books?id=XjZMWNVvgLAC (accessed on 12 December 2021).
- Segall, A.E.; Papyrin, A.N.; Conway, J.C.; Shapiro, D. A cold-gas spray coating process for enhancing titanium. J. Miner. Met. Mater. Soc. 1998, 50, 52–54.
- Assadi, H.; Kreye, H.; Gärtner, F.; Klassen, T. Cold spraying—A materials perspective. Acta Mater. 2016, 116, 382–407.
- Assadi, H.; Gärtner, F.; Stoltenhoff, T.; Kreye, H. Bonding mechanism in cold gas spraying. Acta Mater. 2003, 51, 4379–4394.
- Hussain, T.; McCartney, D.G.; Shipway, P.H.; Zhang, D. Bonding mechanisms in cold spraying: The contributions of metallurgical and mechanical components. J. Therm. Spray Technol. 2009, 18, 364–379.
- Grujicic, M.; Zhao, C.; DeRosset, W.; Helfritch, D. Adiabatic shear instability based mechanism for particles/substrate bonding in the cold-gas dynamic-spray process. Mater. Eng. 2004, 25, 681–688.
- Hussain, T. Cold spraying of titanium: A review of bonding mechanisms, microstructure and properties. KEM 2012, 533, 53–90.
- Schmidt, T.; Assadi, H.; Gärtner, F.; Richter, H.; Stoltenhoff, T.; Kreye, H.; Klassen, T. From particle acceleration to impact and bonding in cold spraying. J. Therm. Spray Technol. 2009, 18, 794–808.
- Hassani-Gangaraj, M.; Veysset, D.; Nelson, K.A.; Schuh, C.A. Impact-bonding with aluminum, silver, and gold microparticles: Toward understanding the role of native oxide layer. Appl. Surf. Sci. 2019, 476, 528–532.
- Drehmann, R.; Grund, T.; Lampke, T.; Wielage, B.; Manygoats, K.; Schucknecht, T.; Rafaja, D. Splat formation and adhesion mechanisms of cold gas-sprayed Al coatings on Al2O3 substrates. J. Therm. Spray Technol. 2014, 23, 68–75.
- Borchers, C.; Stoltenhoff, T.; Gäartner, F.; Kreye, H.; Assadi, H. Deformation microstructure of cold gas sprayed coatings. MRS Proc. 2001, 673, P7.10.1–P7.10.6.
- Gilmore, D.L.; Dykhuizen, R.C.; Neiser, R.A.; Roemer, T.J.; Smith, M.F. Particle velocity and deposition efficiency in the cold spray process. J. Therm. Spray Technol. 1999, 8, 576–582.
- Raletz, F.; Vardelle, M.; Ezo’o, G. Critical particle velocity under cold spray conditions. Surf. Coat. Technol. 2006, 201, 1942–1947.
- Guetta, S.; Berger, M.H.; Borit, F.; Guipont, V.; Jeandin, M.; Boustie, M.; Ichikawa, Y.; Sakaguchi, K.; Ogawa, K. Influence of particle velocity on adhesion of cold-sprayed splats. J. Therm. Spray Technol. 2009, 18, 331–342.
- Wang, F.F.; Li, W.Y.; Yu, M.; Liao, H.L. Prediction of critical velocity during cold spraying based on a coupled thermomechanical eulerian model. J. Therm. Spray Technol. 2014, 23, 60–67.
- Yu, M.; Li, W.-Y.; Wang, F.F.; Suo, X.K.; Liao, H.L. Effect of particle and substrate preheating on particle deformation behavior in cold spraying. Surf. Coat. Technol. 2013, 220, 174–178.
- Ganesan, A.; Affi, J.; Yamada, M.; Fukumoto, M. Bonding behavior studies of cold sprayed copper coating on the PVC polymer substrate. Surf. Coat. Technol. 2012, 207, 262–269.
- Razavipour, M.; Rahmati, S.; Zúñiga, A.; Criado, D.; Jodoin, B. Bonding mechanisms in cold spray: Influence of surface oxidation during powder storage. J. Therm. Spray Technol. 2021, 30, 304–323.
- Nikbakht, R.; Seyedein, S.H.; Kheirandish, S.; Assadi, H.; Jodoin, B. Asymmetrical bonding in cold spraying of dissimilar materials. Appl. Surf. Sci. 2018, 444, 621–632.
- Neshastehriz, M.; Smid, I.; Segall, A.E.; Eden, T.J. On the bonding mechanism in cold spray of deformable Hex-BN-Ni clusters. J. Therm. Spray Technol. 2016, 25, 982–991.
- Loke, K.; Zhang, Z.Q.; Narayanaswamy, S.; Koh, P.K.; Luzin, P.; Herold, T.G.; Ang, A.S.M. Residual stress analysis of cold spray coatings sprayed at angles using through-thickness neutron diffraction measurement. J. Therm. Spray Technol. 2021, 30, 1810–1826.
- Xie, Y.; Chen, C.; Planche, M.-P.; Deng, S.; Huang, R.; Ren, Z.; Liao, H. Strengthened peening effect on metallurgical bonding formation in cold spray additive manufacturing. J. Therm. Spray Technol. 2019, 28, 769–779.
- Schmidt, T.; Gärtner, F.; Assadi, H.; Kreye, H. Development of a generalized parameter window for cold spray deposition. Acta Mater. 2006, 54, 729–742.
- Kromer, R.; Danlos, Y.; Costil, S. Cold gas-sprayed deposition of metallic coatings onto ceramic substrates using laser surface texturing pre-treatment. J. Therm. Spray Technol. 2018, 27, 809–817.
- Ichikawa, Y.; Tokoro, R.; Ogawa, K. Investigation of cold spray bonding mechanism focusing on the thin oxide film present in the deposition interface. In Proceedings of the International Thermal Spray Conference, Orlando, FL, USA, 7–10 May 2018; pp. 238–241, ISBN 9781627081603.
- Ichikawa, Y.; Tokoro, R.; Tanno, M.; Ogawa, K. Elucidation of cold-spray deposition mechanism by auger electron spectroscopic evaluation of bonding interface oxide film. Acta Mater. 2019, 164, 39–49.
- Chang, Y.; Mohanty, P.; Karmarkar, N.; Khan, M.T.; Wang, Y.; Wang, J. Microstructure and properties of Cu–Cr coatings deposited by cold spraying. Vacuum 2020, 171, 109032.
- Fukumoto, M.; Tanabe, K.; Yamada, M.; Yamaguchi, E. Clarification of deposition mechanism of copper particle in cold spray process. J. Jpn. Weld. Soc. 2007, 25, 537–541.
- Barradas, S.; Molins, R.; Jeandin, M.; Arrigoni, M.; Boustie, M.; Bolis, C.; Berthe, L.; Ducos, M. Application of laser shock adhesion testing to the study of the interlamellar strength and coating–substrate adhesion in cold-sprayed copper coating of aluminum. Surf. Coat. Technol. 2005, 197, 18–27.
- Fukumoto, M.; Mashiko, M.; Yamada, M.; Yamaguchi, E. Deposition behavior of copper fine particles onto flat substrate surface in cold spraying. J. Therm. Spray Technol. 2010, 19, 89–94.
- Flanagan, J.; Schütze, P.; Dunne, C.; Twomey, B.; Stanton, K.T. Use of a blast coating process to promote adhesion between aluminium surfaces for the automotive industry. J. Adhes. 2020, 96, 580–601.
- Wüstefeld, C.; Rafaja, D.; Motylenko, M.; Ullrich, C.; Drehmann, R.; Grund, T.; Lampke, T.; Wielage, B. Local heteroepitaxy as an adhesion mechanism in aluminium coatings cold gas sprayed on AlN substrates. Acta Mater. 2017, 128, 418–427.
- Ang, A.S.M.; Berndt, C.C.; Cheang, P. Deposition effects of WC particle size on cold sprayed WC–Co coatings. Surf. Coat. Technol. 2011, 205, 3260–3267.
- Lima, R.S.; Karthikeyan, J.; Kay, C.M.; Lindemann, J.; Berndt, C.C. Microstructural characteristics of cold-sprayed nanostructured WC–Co coatings. Thin Solid Film. 2002, 416, 129–135.
- Nunthavarawong, P.; Sacks, N.; Botef, I. Effect of powder feed rate on the mechanical properties of WC-5 wt% Ni coatings deposited using low pressure cold spray. Int. J. Refract. Met. Hard Mater. 2016, 61, 230–237.
- Christoulis, D.K.; Guetta, S.; Guipont, V.; Jeandin, M. The influence of the substrate on the deposition of cold-sprayed titanium: An experimental and numerical study. J. Therm. Spray Technol. 2011, 20, 523–533.
- Jami, H.; Jabbarzadeh, A. Effect of particle shape on mechanics of impact in the deposition of titanium nanoparticles on a titanium substrate. Surf. Coat. Technol. 2020, 394, 125880.
- Irinah Omar, N.; Selvami, S.; Kaisho, M.; Yamada, M.; Yasui, T.; Fukumoto, M. Deposition of titanium dioxide coating by the cold-spray process on annealed stainless steel substrate. Coatings 2020, 10, 991.
- Cao, C.; Li, W.; Yang, K.; Li, C.; Ji, G. Influence of substrate hardness and thermal characteristics on microstructure and mechanical properties of cold sprayed TC4 titanium alloy coatings. Cailiao Daobao/Mater. Rev. 2019, 33, 277–282.
- Dietrich, D.; Wielage, B.; Lampke, T.; Grund, T.; Kümme, S. Evolution of microstructure of cold-spray aluminum coatings on Al2O3 substrates. Adv. Eng. Mater. 2011, 14, 275–278.
- Ziemian, C.W.; Wright, W.J.; Cipoletti, D.E. Influence of impact conditions on feedstock deposition behavior of cold-sprayed Fe-based metallic glass. J. Therm. Spray Technol. 2018, 27, 843–856.
- Vidaller, M.V.; List, A.; Gaertner, F.; Klassen, T.; Dosta, S.; Guilemany, J.M. Single impact bonding of cold sprayed Ti-6Al-4V powders on different substrates. J. Therm. Spray Technol. 2015, 24, 644–658.
- Welk, B.A.; Williams, R.E.A.; Viswanathan, G.B.; Gibson, M.A.; Liaw, P.K.; Fraser, H.L. Nature of the interfaces between the constituent phases in the high entropy alloy CoCrCuFeNiAl. Ultramicroscopy 2013, 134, 193–199.
- Kumar, S.; Bae, G.; Lee, C. Influence of substrate roughness on bonding mechanism in cold spray. Surf. Coat. Technol. 2016, 304, 592–605.
- Singh, S.; Singh, H.; Chaudhary, S.; Buddu, R.K. Effect of substrate surface roughness on properties of cold-sprayed copper coatings on SS316L steel. Surf. Coat. Technol. 2020, 389, 125619.
- Lock Sulen, W.; Ravi, K.; Bernard, C.; Ichikawa, Y.; Ogawa, K. Deposition mechanism analysis of cold-sprayed fluoropolymer coatings and its wettability evaluation. J. Therm. Spray Technol. 2020, 29, 1643–1659.
- Hassani-Gangaraj, M.; Veysset, D.; Champagne, V.K.; Nelson, K.A.; Schuh, C.A. Adiabatic shear instability is not necessary for adhesion in cold spray. Acta Mater. 2018, 158, 430–439.
- Assadi, H.; Gärtner, F.; Klassen, T.; Kreye, H. Comment on “Adiabatic shear instability is not necessary for adhesion in cold spray”. Scr. Mater. 2019, 162, 512–514.
- Hassani-Gangaraj, M.; Veysset, D.; Champagne, V.K.; Nelson, K.A.; Schuh, C.A. Response to comment on “Adiabatic shear instability is not necessary for adhesion in cold spray”. Scr. Mater. 2019, 162, 515–519.
- Kim, G.H.; Park, C.K.; Ahn, H.J.; Kim, H.S.; Hong, K.H.; Jin, S.W.; Lee, H.G.; Fukanuma, H.; Huang, R.; Roh, B.R.; et al. Fabrication feasibility study on copper cold spray in tokamak system. Fusion Eng. Des. 2015, 98–99, 1576–1579.
- Ioki, K.; Barabash, V.; Bachmann, C.; Chappuis, P.; Choi, C.H.; Cordier, J.-J.; Giraud, B.; Gribov, Y.; Heitzenroeder, P.; Her, N.; et al. Design finalization and material qualification towards procurement of the ITER vacuum vessel. J. Nucl. Mater. 2011, 417, 860–865.
- Singh, S.; Chaudhary, S.; Singh, H. Effect of electroplated interlayers on properties of cold-sprayed copper coatings on SS316L steel. Surf. Coat. Technol. 2019, 375, 54–65.
- Singh, S.; Singh, H.; Buddu, R.K. Microstructural investigations on bonding mechanisms of cold-sprayed copper with SS316L steel. Surf. Eng. 2020, 36, 1067–1080.
- Arabgol, Z.; Vidaller, M.V.; Assadi, H.; Gärtner, F.; Klassen, T. Influence of thermal properties and temperature of substrate on the quality of cold-sprayed deposits. Acta Mater. 2017, 127, 287–301.
- Sun, W.; Tan, A.W.Y.; Bhowmik, A.; Marinescu, I.; Song, X.; Zhai, W.; Li, F.; Liu, E. Deposition characteristics of cold sprayed inconel 718 particles on inconel 718 substrates with different surface conditions. Mater. Sci. Eng. 2018, 720, 75–84.
- Ichikawa, Y.; Ogawa, K. Effect of substrate surface oxide film thickness on deposition behavior and deposition efficiency in the cold spray process. J. Therm. Spray Technol. 2015, 24, 1269–1276.
- Jodoin, B. Cold spray nozzle mach number limitation. J. Therm. Spray Technol. 2002, 11, 496–507.
- Dowding, I.; Hassani, M.; Sun, Y.; Veysset, D.; Nelson, K.A.; Schuh, C.A. Particle size effects in metallic microparticle impact-bonding. Acta Mater. 2020, 194, 40–48.
- Chun, D.-M.; Choi, J.-O.; Lee, C.S.; Ahn, S.-H. Effect of stand-off distance for cold gas spraying of fine ceramic particles (<5 μm) under low vacuum and room temperature using nano-particle deposition system (NPDS). Surf. Coat. Technol. 2012, 206, 2125–2132.
- Wang, Z.; Cai, S.; Jin, K.; Wang, X.; Chen, W. In-flight aggregation and deposition behaviour of particles in low pressure cold spray. Surf. Coat. Technol. 2021, 409, 126875.





