
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | eugenia quiros roldan | + 3414 word(s) | 3414 | 2021-12-14 04:14:23 | | | |
| 2 | Peter Tang | Meta information modification | 3414 | 2021-12-23 05:15:44 | | |
Video Upload Options
Antibodies are now a versatile tool for diagnostics and therapy of various conditions in humans and hyperimmune sera can be replaced by specific monoclonal antibodies (mAbs). mAbs have been known since the 1970s. Numerous mAbs have been developed against SARS-CoV 2 and have proven their effectiveness, especially in the management of the mild-to-moderate disease.
1. Introduction
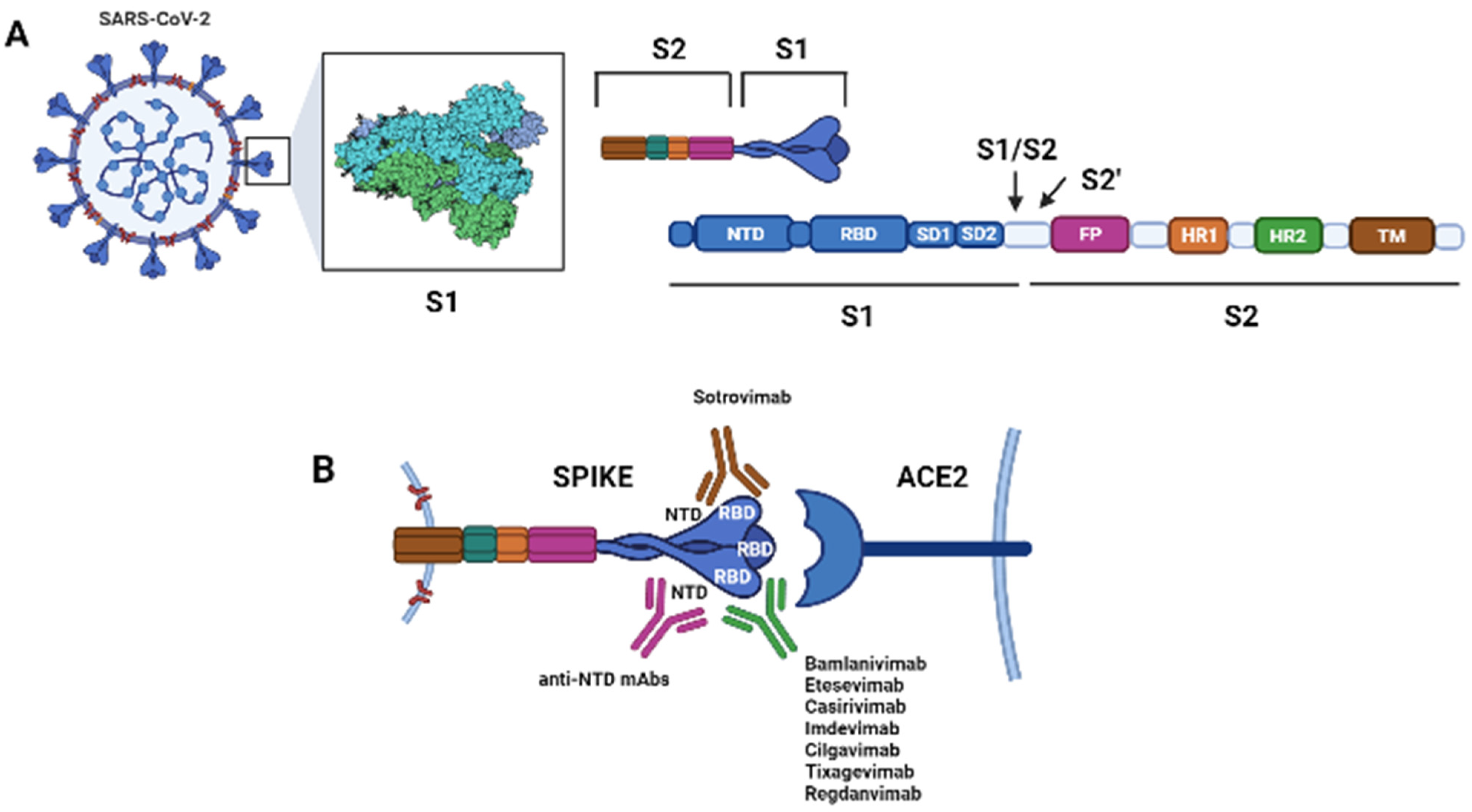
2. Antibodies Targeting SARS-CoV-2
3. Therapeutic and Prophylactic Indications
3.1. Prophylactic Use of mAb against SARS-CoV-2
3.2. Use of mAb in Patient Hospitalized for COVID-19
4. Challenges for Using mAbs against SARS-CoV-2
4.1. Activity against SARS-CoV-2 Variants
|
WHO Label |
Pango Lineage * |
GISAID Clade |
Nextstrain Clade |
Additional Aamino Acid Changes Monitored ° |
Earliest Documented Samples |
Date of Designation |
|---|---|---|---|---|---|---|
|
Alpha |
B.1.1.7 # |
GRY |
20I (V1) |
+S:484K +S:452R |
United Kingdom, Sep-2020 |
18 December 2020 |
|
Beta |
B.1.351 |
GH/501Y.V2 |
20H (V2) |
+S:L18F |
South Africa, May-2020 |
18 December 2020 |
|
Gamma |
P.1 |
GR/501Y.V3 |
20J (V3) |
+S:681H |
Brazil, Nov-2020 |
11 January 2021 |
|
Delta |
B.1.617.2 § |
G/478K.V1 |
21A, 21I, 21J |
+S:417N |
India, Oct-2020 |
VOI: 4 April 2021 VOC: 11 May 2021 |
|
WHO Label § |
Pango Lineage * |
GISAID Clade |
Nextstrain Clade |
Earliest Documented Samples |
Date of Designation |
|---|---|---|---|---|---|
|
Lambda |
C.37 |
GR/452Q.V1 |
21G |
Peru, December 2020 |
14 June 2021 |
|
Mu |
B.1.621 |
GH |
21H |
Colombia, January 2021 |
30 August 2021 |
-
Beta (B.1.351) variant: This VoC includes the E484K and K417N mutations, which results in a reduction in in vitro susceptibility to bamlanivimab and etesevimab [33][44]. In vitro studies also suggest that this variant has markedly reduced susceptibility to casirivimab, although the combination of casirivimab and imdevimab appears to retain activity; sotrovimab appears active as well against this VoC [34][45].
-
Gamma (P.1) variant: This VoC includes the E484K and K417T mutations, which results in a marked reduction in in vitro susceptibility to bamlanivimab and etesevimab [33][46]. Additionally, this variant shows reduced susceptibility to casirivimab, although the combination of casirivimab and imdevimab appears to retain activity; sotrovimab appears to retain activity as well [34][45].
-
Delta (B.1.617.2) variant: This is the prevalent VoC in the United States. It contains the L452R mutation, which results in a modest decrease in in vitro susceptibility to the combination of bamlanivimab and etesevimab, although the clinical implications of this finding are not fully known. Sotrovimab and casirivimab plus imdevimab appear to maintain activity [34][45][47].
|
Casirivimab Indevimab |
Bamlanivimab Etesevimab |
Sotrovimab |
Cilgavimab Tixagevimab |
Regdanvimab |
|
|---|---|---|---|---|---|
|
B.1.1.7 (UK) |
S S |
S S |
S |
S S |
S |
|
B.1.351 (South Africa) |
R S |
R R |
S |
S S |
I/R |
|
P.1 (Brazil) |
R S |
R R |
S |
S S |
Pot I/R |
|
B.1.429 (California) |
S S |
R S |
S |
S S |
I/R |
|
B.1.1.258 (Scotland) |
S R |
S U |
S |
U U |
Pot S |
|
B.1.525 (Nigeria) |
Pot I/R Pot S |
Pot I/R Pot S |
S |
Pot S Pot S |
U |
|
B.1.526 (New York) |
Pot I/R Pot S |
Pot I/R Pot S |
S |
Pot S Pot S |
U |
|
B.1.617.1 (India) |
S S |
R S |
S |
Pot S Pot S |
U |
S = neutralized (<10-fold loss of neutralization). I/R = poorly or not-neutralized (>10-fold loss of neutralization). Pot S = potential S. Pot I/R = potential I/R. U = Unknown.
4.2. Innovative Way of mAbs Administration
References
- Marston, H.D.; Paules, C.I.; Fauci, A.S. Monoclonal Antibodies for Emerging Infectious Diseases—Borrowing from History. N. Engl. J. Med. 2018, 378, 1469–1472.
- Guthmiller, J.J.; Dugan, H.L.; Neu, K.E.; Lan, L.Y.; Wilson, P.C. An Efficient Method to Generate Monoclonal Antibodies from Human B Cells. Methods Mol. Biol. 2019, 1904, 109–145.
- Roth, L.; Grzeschik, J.; Hinz, S.C.; Becker, S.; Toleikis, L.; Busch, M.; Kolmar, H.; Krah, S.; Zielonka, S. Facile generation of antibody heavy and light chain diversities for yeast surface display by Golden Gate Cloning. Biol. Chem. 2019, 400, 383–393.
- Burioni, R.; Lang, A.B.; Capra, J.D. Human monoclonal antibodies as a new class of antiinfective compounds. Clin. Dev. Immunol. 2013, 2013, 297120.
- Pelfrene, E.; Mura, M.; Cavaleiro Sanches, A.; Cavaleri, M. Monoclonal antibodies as anti-infective products: A promising future? Clin. Microbiol. Infect. 2019, 25, 60–64.
- U.S. Food and Drug Administration. FDA Approved Drug Products: Raxibacumab. Available online: https://www.accessdata.fda.gov/drugsatfda_docs/label/2012/125349s000lbl.pdf (accessed on 15 November 2021).
- U.S. Food and Drug Administration. FDA Approved Drug Products: Anthim. Available online: https://www.accessdata.fda.gov/drugsatfda_docs/label/2016/125509lbl.pdf (accessed on 15 November 2021).
- U.S. Food and Drug Administration. FDA Approved Drug Products: Trogarzo. Available online: https://www.accessdata.fda.gov/drugsatfda_docs/label/2018/761065lbl.pdf (accessed on 15 November 2021).
- Callaway, B.E.; Cyranoski, D.; Mallapaty, S.; Stoye, E.; Tollefson, J. Coronavirus by the numbers. Nature 2020, 579, 482–483.
- Kreuzberger, N.; Hirsch, C.; Chai, K.L.; Tomlinson, E.; Khosravi, Z.; Popp, M.; Neidhardt, M.; Piechotta, V.; Salomon, S.; Valk, S.J.; et al. SARS-CoV-2-neutralising monoclonal antibodies for treatment of COVID-19. Cochrane Database Syst. Rev. 2021, 9, CD013825.
- Kumar, D.; Gauthami, S.; Bayry, J.; Kaveri, S.V.; Hegde, N.R. Antibody Therapy: From Diphtheria to Cancer, COVID-19, and Beyond. Monoclon. Antibodies Immunodiagn. Immunother. 2021, 40, 36–49.
- Tian, X.; Li, C.; Huang, A.; Xia, S.; Lu, S.; Shi, Z.; Lu, L.; Jiang, S.; Yang, Z.; Wu, Y.; et al. Potent binding of 2019 novel coronavirus spike protein by a SARS coronavirus-specific human monoclonal antibody. Emerg. Microbes Infect. 2020, 9, 382–385.
- Wang, C.; Li, W.; Drabek, D.; Okba, N.M.A.; Van Haperen, R.; Osterhaus, A.; Van Kuppeveld, F.J.M.; Haagmans, B.L.; Grosveld, F.; Bosch, B.J. A human monoclonal antibody blocking SARS-CoV-2 infection. Nat. Commun. 2020, 11, 2251.
- Chen, X.; Li, R.; Pan, Z.; Qian, C.; Yang, Y.; You, R.; Zhao, J.; Liu, P.; Gao, L.; Li, Z.; et al. Human monoclonal antibodies block the binding of SARS-CoV-2 spike protein to angiotensin converting enzyme 2 receptor. Cell Mol. Immunol. 2020, 17, 647–649.
- Noy-Porat, T.; Makdasi, E.; Alcalay, R.; Mechaly, A.; Levy, Y.; Bercovich-Kinori, A.; Zauberman, A.; Tamir, H.; Yahalom-Ronen, Y.; Israeli, M.; et al. A panel of human neutralizing mAbs targeting SARSCoV-2 spike at multiple epitopes. Nat. Commun. 2020, 11, 4303.
- Yuan, M.; Wu, N.C.; Zhu, X.; Lee, C.D.; So, R.T.Y.; Lv, H.; Mok, C.K.P.; Wilson, I.A. A highly conserved cryptic epitope in the receptor binding domains of SARS-CoV-2 and SARS-CoV. Science 2020, 368, 630–633.
- Shi, R.; Shan, C.; Duan, X.; Chen, Z.; Liu, P.; Song, J.; Song, T.; Bi, X.; Han, C.; Wu, L.; et al. A human neutralizing antibody targets the receptor-binding site of SARS-CoV-2. Nature 2020, 584, 120–124.
- Ju, B.; Zhang, Q.; Ge, J.; Wang, R.; Sun, J.; Ge, X.; Yu, J.; Shan, S.; Zhou, B.; Song, S.; et al. Human neutralizing antibodies elicited by SARS-CoV-2 infection. Nature 2020, 584, 115–119.
- Lv, Z.; Deng, Y.-Q.; Ye, Q.; Cao, L.; Sun, C.-Y.; Fan, C.; Huang, W.; Sun, S.; Sun, Y.; Zhu, L.; et al. Structural basis for neutralization of SARSCoV-2 and SARS-CoV by a potent therapeutic antibody. Science 2020, 369, 1505–1509.
- Iyer, A.S.; Jones, F.K.; Nodoushani, A.; Kelly, M.; Becker, M.; Slater, D.; Mills, R.; Teng, E.; Kamruzzaman, M.; Garcia-Beltran, W.F.; et al. Persistence and decay of human antibody responses to the receptor binding domain of SARS-CoV-2 spike protein in COVID-19 patients. Sci. Immunol. 2020, 5, eabe0367.
- Chi, X.; Yan, R.; Zhang, J.; Zhang, G.; Zhang, Y.; Hao, M.; Zhang, Z.; Fan, P.; Dong, Y.; Yang, Y.; et al. A neutralizing human antibody binds to the N-terminal domain of the Spike protein of SARS-CoV-2. Science 2020, 369, 650–655.
- Liu, L.; Wang, P.; Nair, M.S.; Yu, J.; Rapp, M.; Wang, Q.; Luo, Y.; Chan, J.F.; Sahi, V.; Figueroa, A.; et al. Potent neutralizing antibodies against multiple epitopes on SARS-CoV-2 spike. Nature 2020, 584, 450–456.
- Huang, Y.; Yang, C.; Xu, X.F.; Xu, W.; Liu, S.W. Structural and functional properties of SARS-CoV-2 spike protein: Potential antivirus drug development for COVID-19. Acta Pharmacol. Sin. 2020, 41, 1141–1149.
- Abernathy, M.E.; Dam, K.A.; Esswein, S.R.; Jette, C.A.; Bjorkman, P.J. How Antibodies Recognize Pathogenic Viruses: Structural Correlates of Antibody Neutralization of HIV-1, SARS-CoV-2, and Zika. Viruses 2021, 13, 2106.
- Winkler, E.S.; Gilchuk, P.; Yu, J.; Bailey, A.L.; Chen, R.E.; Zost, S.J.; Jang, H.; Huang, Y.; Allen, J.D.; Case, J.B.; et al. Human neutralizing antibodies against SARS-CoV-2 require intact Fc effector functions for optimal therapeutic protection. Cell 2021, 184, 1804–1820.
- Lu, L.L.; Suscovich, T.J.; Fortune, S.M.; Alter, G. Beyond binding: Antibody effector functions in infectious diseases. Nat. Rev. Immunol. 2018, 18, 46–61.
- Bournazos, S.; Gupta, A.; Ravetch, J.V. The role of IgG Fc receptors in antibody-dependent enhancement. Nat. Rev. Immunol. 2020, 20, 633–643.
- Qi, T.; Cao, Y. In Translation: FcRn across the Therapeutic Spectrum. Int. J. Mol. Sci. 2021, 22, 3048.
- Vladislav, I.; Matlock, A.J.; Sawalha, A.H. COVID-19: A review of therapeutic strategies and vaccine candidates. Clin. Immunol. 2021, 222, 108634.
- Corey, L.; Mascola, J.R.; Fauci, A.S.; Collins, F.S. A strategic approach to COVID-19 vaccine R&D. Science 2020, 368, 948–950.
- Hurt, A.C.; Wheatley, A.K. Neutralizing Antibody Therapeutics for COVID-19. Viruses 2021, 13, 628.
- O’Brien, M.P.; Forleo-Neto, E.; Musser, B.J.; Isa, F.; Chan, K.C.; Sarkar, N.; Bar, K.J.; Barnabas, R.V.; Barouch, D.H.; Cohen, M.S.; et al. Subcutaneous REGEN-COV Antibody Combination to Prevent Covid-19. N. Engl. J. Med. 2021, 385, 1184–1195.
- U.S. Food and Drug Administration. Fact Sheet for Health Care Providers: Emergency Use Authorization (EUA) of Bamlanivimab and Etesevimab. Available online: https://www.fda.gov/media/145802/download (accessed on 15 November 2021).
- U.S. Food and Drug Administration. Fact Sheet for Health Care Providers: Emergency Use Authorization (EUA) of Casirivimab and Imdevimab. Available online: https://www.fda.gov/media/145611/download (accessed on 15 November 2021).
- CDC Quarantine and Isolation. Available online: https://www.cdc.gov/coronavirus/2019-ncov/your-health/quarantine-isolation.html?CDC_AA_refVal=https%3A%2F%2Fwww.cdc.gov%2Fcoronavirus%2F2019-ncov%2Fif-you-are-sick%2Fquarantine.html (accessed on 15 November 2021).
- Zost, S.J.; Gilchuk, P.; Case, J.B.; Binshtein, E.; Chen, R.E.; Nkolola, J.P.; Schäfer, A.; Reidy, J.X.; Trivette, A.; Nargi, R.S.; et al. Potently neutralizing and protective human antibodies against SARS-CoV-2. Nature 2020, 584, 443–449.
- Dong, J.; Zost, S.J.; Greaney, A.J.; Starr, T.N.; Dingens, A.S.; Chen, E.C.; Chen, R.E.; Case, J.B.; Sutton, R.E.; Gilchuk, P.; et al. Genetic and structural basis for SARS-CoV-2 variant neutralization by a two-antibody cocktail. Nat. Microbiol. 2021, 6, 1233–1244.
- Update on AZD7442 STORM CHASER Trial in Post-Exposure Prevention of Symptomatic COVID-19. Available online: https://www.astrazeneca.com/media-centre/press-releases/2021/update-on-azd7442-storm-chaser-trial.html (accessed on 15 November 2021).
- AZD7442 PROVENT Phase III Prophylaxis Trial Met Primary Endpoint in Preventing COVID-19. Available online: https://www.astrazeneca.com/media-centre/press-releases/2021/azd7442-prophylaxis-trial-met-primary-endpoint.html (accessed on 15 November 2021).
- RECOVERY Collaborative Group. Casirivimab and imdevimab in patients admitted to hospital with COVID-19 (RECOVERY): A randomised, controlled, open-label, platform trial. medRxiv. 2021. preprint. Available online: https://www.medrxiv.org/content/10.1101/2021.06.15.21258542v1.full (accessed on 15 November 2021).
- Focosi, D.; Maggi, F. Neutralising antibody escape of SARS-CoV-2 spike protein: Risk assessment for antibody-based COVID-19 therapeutics and vaccines. Rev. Med. Virol. 2021, 1–21.
- Tracking SARS-CoV-2 Variants. Available online: https://www.who.int/en/activities/tracking-SARS-CoV-2-variants (accessed on 15 November 2021).
- FDA (US Food and Drug Administration). Development of Monoclonal Antibody Products Targeting SARS-CoV-2, Including Addressing the Impact of Emerging Variants, During the COVID 19 Public Health Emergency. 2021. Available online: https://www.fda.gov/regulatory-information/search-fda-guidance-documents/development-monoclonal-antibody-products-targeting-sars-cov-2-includingaddressing-impact-emerging (accessed on 15 November 2021).
- Ho, D.; Wang, P.; Liu, L.; Iketani, S.; Luo, Y.; Guo, Y.; Wang, M.; Yu, J.; Zhang, B.; Kwong, P.; et al. Increased Resistance of SARS-CoV-2 Variants B.1.351 and B.1.1.7 to Antibody Neutralization. Res. Sq. 2021, 29, 747–751.
- U.S. Food and Drug Administration. Frequently Asked Questions on the Emergency Use Authorization of Sotrovimab. Available online: https://www.fda.gov/media/149534/download (accessed on 15 November 2021).
- Wang, P.; Wang, P.; Casner, R.G.; Nair, M.S.; Wang, M.; Yu, J.; Cerutti, G.; Liu, L.; Kwong, P.D.; Huang, Y.; et al. Increased resistance of SARS-CoV-2 variant P.1 to antibody neutralization. bioRxiv 2021.
- Liu, C.; Ginn, H.M.; Dejnirattisai, W.; Supasa, P.; Wang, B.; Tuekprakhon, A.; Nutalai, R.; Zhou, D.; Mentzer, A.J.; Zhao, Y.; et al. Reduced neutralization of SARS-CoV-2 B.1.617 by vaccine and convalescent serum. Cell 2021, 184, 4220–4236.e13, published online ahead of print. Available online: https://www.ncbi.nlm.nih.gov/pubmed/34242578 (accessed on 15 November 2021).
- Corti, D.; Purcell, L.A.; Snell, G.; Veesler, D. Tackling COVID-19 with neutralizing monoclonal antibodies. Cell 2021, 184, 3086–3108.
- Zhang, H.; Yang, Z.; Xiang, J.; Cui, Z.; Liu, J.; Liu, C. Intranasal administration of SARS-CoV-2 neutralizing human antibody prevents infection in mice. bioRxiv 2020.
- Nishat, F.; Vichitra, K.; Amjad, A. A Narrative Review of a Pulmonary Aerosolized Formulation or a Nasal Drop Using Sera Containing Neutralizing Antibodies Collected from COVID-19-Recovered Patients as a Probable Therapy for COVID-19. Iran. J. Med. Sci. 2021, 46, 151–168.
- Vivek, P.C.; Lalitkumar, K.V.; Anjali, K.P.; Vandana, B.P. Intranasal vaccines for SARS-CoV-2: From challenges to potential in COVID-19 management. Drug Discov. Today 2021, 26, 2619–2636.
- Tiboni, M.; Casettari, L.; Illum, L. Nasal vaccination against SARS-CoV-2: Synergistic or alternative to intramuscular vaccines? Int. J. Pharm. 2021, 603, 120686.
- Du, Y.; Xu, Y.; Feng, J.; Hu, L.; Zhang, Y.; Zhang, B.; Guo, W.; Mai, R.; Chen, L.; Fang, J.; et al. Intranasal administration of a recombinant RBD vaccine induced protective immunity against SARS-CoV-2 in mouse. Vaccine 2021, 39, 2280–2287.
- De Vries, R.D.; Schmitz, K.S.; Bovier, F.T.; Predella, C.; Khao, J.; Noack, D.; Haagmans, B.L.; Herfst, S.; Stearns, K.N.; Drew-Bear, J.; et al. Intranasal fusion inhibitory lipopeptide prevents direct contact SARSCoV-2 transmission in ferrets. Science 2021, 371, 1379–1382.
- Wan, Y.; Shang, J.; Sun, S.; Tai, W.; Chen, J.; Geng, Q.; He, L.; Chen, Y.; Wu, J.; Shi, Z.; et al. Molecular mechanism for antibody-dependent enhancement of coronavirus entry. J. Virol. 2020, 94, e02015-19.
- Parray, H.A.; Shukla, S.; Perween, R.; Khatri, R.; Shrivastava, T.; Singh, V.; Murugavelu, P.; Ahmed, S.; Samal, S.; Sharma, C.; et al. Inhalation monoclonal antibody therapy: A new way to treat and manage respiratory infections. Appl. Microbiol. Biotechnol. 2021, 105, 6315–6332.
- Piepenbrink, M.S.; Park, J.G.; Oladunni, F.S.; Deshpande, A.; Basu, M.; Sarkar, S.; Loos, A.; Woo, J.; Lovalenti, P.; Sloan, D.; et al. Therapeutic activity of an inhaled potent SARS-CoV-2 neutralizing human monoclonal antibody in hamsters. Cell Rep. Med. 2021, 2, 100218.
- In-patient COVID-19 Study of Intranasal Foralumab. Available online: https://clinicaltrials.gov/ct2/show/record/NCT04983446?term=foralumab&cond=COVID-19&draw=2&rank=1 (accessed on 15 November 2021).





