
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Marco Onorati | + 1827 word(s) | 1827 | 2020-08-14 05:57:23 | | | |
| 2 | Conner Chen | + 1 word(s) | 1828 | 2020-10-30 09:35:48 | | |
Video Upload Options
Building and functioning of the human brain requires the precise orchestration and execution of myriad molecular and cellular processes, across a multitude of cell types and over an extended period of time. Neural Stem Cells (NSCs) represent the heart of these processes, since they increase the pool of neural progenitors and are the founders of all the neural progeny which will constitute the adult human brain.
1. Human Neural Stem Cells: When and Where?
The immense complexity of the human brain is reflected in its cellular organization and in the cognitive and behavioral repertoire that defines us as human. The human brain is the product of an evolutionary and developmental history that resulted in its progressive enlargement and specialization. Our central nervous system (CNS) develops through a dynamic and prolonged process in which myriad cell types are generated by neural stem cells (NSCs) and assembled into an intricate synaptic circuitry. Deviations from the normal course of development can lead to a variety of pathologies, including neurological and psychiatric disorders that affect some of the most distinctly human aspects of cognition and behavior [1][2][3][4].
During early CNS development, the neural tube is comprised of a pseudostratified layer of neuroepithelial cells lining the central cavity [3][5][6]. These cells constitute the ventricular zone (VZ) of the neural tube and are the founders from which all neurons and glial cells of the adult CNS will be generated (Figure 1).
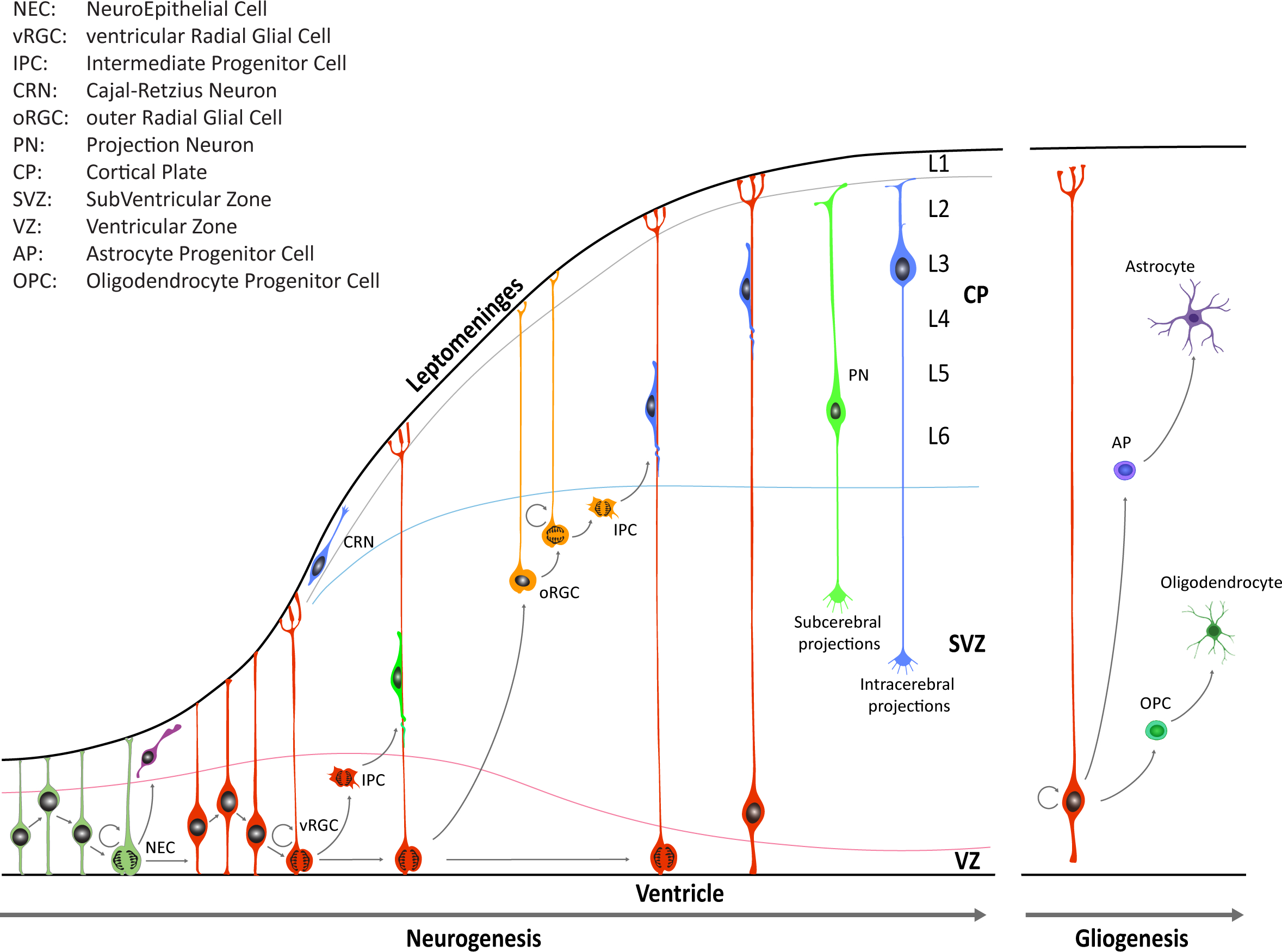
Figure 1. Schematic illustration of neocortical development. Neuroepithelial cells (NECs) undergo symmetric cell division to expand the initial pool and later transition into ventricular radial glia cells (vRGCs). vRGCs begin asymmetric cell division to generate another vRGC and a nascent projection neuron. Neurons then migrates radially from the ventricular zone (VZ) along the RGC basal processes into the cortical plate (CP). Early-born projection neurons (PNs) settle in the deep layers (Layers 5 and 6), and later-born neurons in upper layers. Additionally, some populations of RGC daughter cells convert themselves into intermediate progenitor cells (IPCs) or outer radial glial cells (oRGCs) in the subventricular zone (SVZ). After the neurogenic stages, gliogenesis occurs, generating astrocytes and oligodendrocytes.
Until the seventh post-conceptional week (pcw), depending on the region of the CNS, neuroepithelial cells undergo primarily symmetric divisions in order to expand the stem cell pool [3]. Dividing neuroepithelial cells are characterized by a radial movement of cell nuclei from the apical luminal side (apical surface) to the basal side of the neural tube (basal lamina at the pial surface), in concert with the progression of the cell cycle. Mitosis typically occurs when cell nuclei are close to the apical surface of the neuroepithelium, and this continuous relocation is commonly defined as interkinetic nuclear migration (IKNM). Later on, neuroepithelial cells transition into a distinct class of cells known as radial glial cells (RGCs) which reside in the VZ and in the inner and outer subventricular zone (iSVZ and oSVZ, respectively). RGCs contact, at least initially, both the ventricular and pial surface through their apical and basal process, respectively. These cell populations serve as progenitor cells to generate neurons and macroglia (i.e., astrocytes and oligodendrocytes) and to provide a scaffold for migrating nascent neurons [3][5][6]. RGCs divide, but unlike neuroepithelial cells, the divisions of RGCs are mostly asymmetric, giving rise to either a daughter RGC, an intermediate progenitor cell (IPC, also known as a transit amplifying cell), or a nascent neuron that subsequently migrates out of the VZ or the SVZ to its final location near the pial surface. In the human neocortex, neuroepithelial cells give rise first to ventricular RGCs (vRGCs), which later transit into outer RGCs (oRGCs) [7][8][9]. oRGCs have morphologically distinct features, retain the basal process, but lose apical contact, and their cell bodies translocate into the oSVZ. The oRGCs can be characterized by a distinct transcriptional signature compared with vRGCs and divide in a unique manner, called mitotic somal translocation (MST), a process where the cell soma moves rapidly up the basal fiber before cytokinesis [8]. A third population of RGCs, called truncated radial glia (tRGCs), develops later in neurogenesis [10]. Cell bodies of tRGCs reside near the apical surface and possess basal processes that do not reach the pial surface and appear truncated.
During early neurogenesis, at the beginning of the second trimester, the vRGCs and IPCs give rise to neurons present in the deep layers. On the other hand, oRGCs give rise to later born IPCs, and differentiate predominantly into upper layer neurons (Figure 1). The six cortical laminae are comprised of multiple molecularly defined excitatory neuron subtypes. These cells connect intra-cortically to regulate synaptic activity inside the cortex, as well as subcortically to provide executive regulation of sensory and motor activity [11].
Neural Stem Cells at a glance:
Neuroepithelial Cells: During mammalian embryogenesis, CNS development begins with the induction of the neuroectoderm, which forms the neural plate and then folds to give rise to the neural tube. Within these neural structures there exists a complex and heterogeneous population of neuroepithelial cells, the earliest neural progenitor type to arise. As CNS development proceeds, neuroepithelial cells give rise to temporally and spatially distinct neural stem/progenitor cell populations.
Radial Glia Cells (RGCs): Multipotent neural progenitors with glial-like properties. At the onset of neurogenesis, neuroepithelial cells in the ventricular zone transition into RGCs, bipolar cells with long radial processes extending from the apical surface of ventricular zone to the pial surface. RGCs also act as scaffolds along which newborn neurons can travel from their site of origin to their final destination in the adult CNS.
Intermediate Progenitor Cells (IPCs or Basal Progenitors): Neural progenitors generated from neuroepithelial cells and RGCs at the apical surface of the ventricular zone. IPCs migrate to the basal side of the ventricular zone forming the subventricular zone. Each IPC divides symmetrically to generate two or four neurons.
Adult Neural Stem Cells: Populations of multipotent neural stem cells mainly present in two specialized niches of the adult mammalian brain, the subventricular or subependymal zone of the lateral ventricle wall and the subgranular zone of the dentate gyrus. They maintain neurogenesis and gliogenesis throughout adult life in rodents and other mammals, but their presence and activity in humans is still debated. They derive from RGCs that in the postnatal brain convert into astrocytic-like NSCs.
2. In vitro systems of Neural Stem Cells
Our knowledge of NSCs has been revolutionized as optimized culture protocols have been put in place [12]. Since Reynolds and Weiss (1992) [13] made the landmark discovery that NSCs could be maintained in culture via propagation in free‐floating neurospheres, several works have progressively i mproved systems for NSC long‐term and homogeneous culture. In 2005, it has been shown that NSCs can be expanded as monolayer cultures, named NS cells, with full preservation of their neurogenic potential [14]. NS cells were derived from fetal and adult mouse CNS, but also from neural‐committed mouse embryonic stem cells (ESCs) and induced pluripotent stem cells (iPSCs) [14][15][16][17][18][19]. Additionally, NS cells were also derived from post‐mortem human fetal tissue [20][21]. Although NS cells showed features of neurogenic RGCs, they emerged to be restricted to the generation of GABAergic neurons, independently of the different sources they were derived from [19][20][21].
On the other hand, embryonic neuroepithelial cells possess a great self‐renewing potential and wide multilineage differentiation. Thanks to these unique properties, neuroepithelial cells represent an ideal candidate for in vitro studies related to NSC biology, neuronal and glial differentiation, and various neurodevelopmental diseases. Notably, Austin Smith’s group has described a population of NSCs derived from 5–7 pcw human hindbrain [22]. These cells, named hindbrain (hb) neuroepithelial stem (NES) cells, are neurogenic and preserve their original regional identity, exhibiting for the first time a stable wide degree of plasticity. More recently, we described the derivation and characterization of neocortical (NCX) NES cells [23]. NCX‐NES cell lines were derived from primary neuroepithelium of human post‐mortem specimens ranging from 5 to 8 pcw. After derivation, NCX‐NES cells form neural rosettes, reminiscent of the radial arrangement in the native neural tube. NES cells exhibit stem/progenitor cell characteristics as they express the neuroepithelial marker SOX1 and the pan‐neural stem cell markers Nestin, SOX2, and Vimentin. NES cells retain regional identity after long‐term expansion, as demonstrated by the expression of FOXG1 and OTX2, key transcription factors demarcating proliferative zones of the early human forebrain. NES cells show great neurogenic potentials, giving rise to mature neurons with extended complex neurites. Furthermore, they generate GFAP‐positive astroglial cells, thus demonstrating their multipotential stem cell capacity. We also reported that NCX‐NES cells could be expanded for more than 1 year and 38 passages with no evidence of chromosomal instability [23]. Single‐cell RNA‐sequencing (RNA‐seq) on expanded NCX‐NES cells and cells from donor‐matched brains demonstrated that the majority of cells from the donor‐matched brain tissue samples express canonical marker genes of neuroepithelial cells and RGCs of the dorsal forebrain [23]. Remarkably, NCX‐NES cells exhibit a close transcriptional signature of early NSCs as their donor‐matched genetically identical NCX cells. Together, these data establish NES cell lines as a consistent model of early human brain development. A similar NES population derived from developing human spinal cord has been described, able to maintain regional identity and neuronal commitment of the caudal CNS [24]. Of note, spinal‐cord NES cells were successfully tested in cell grafting approaches after spinal cord injury in mice [24].
Human pluripotent stem cells (hPSCs), which include both ESCs and iPSCs, represent an extraordinary ex vivo source of neural progenitors. The development of neural induction protocols provides the possibility to generate in vitro‐derived expandable NSC systems as a platform for studying basic human neurodevelopment, disease mechanisms, and potential therapeutics [12]. Seminal studies have identified rosette‐type NSCs that resemble neural tube‐stage progenitors, capable to respond to patterning instructions, but not long‐term expandable [25]. On the other hand, a long‐term population of NES cells (named lt‐hESNSCs or lt‐NES) was described by Koch et al. (2009) [26]. The study used the embryoid body (EB)‐based differentiation protocol and required manual isolation of the rosettes from plated EBs. However, in vitro culture conditions bias lt‐NES regional identity from rostral (first five passages) to more caudal midbrain‐hindbrain identity (later passages) [26]. In this direction, Li et al. (2011) [27] described a small molecule‐based neural induction method to derive primitive neural progenitors from hESCs. However, also in these conditions, the NSC population is biased towards a midbrain/hindbrain neural fate [27]. A more recent elegant work improved the isolation and propagation paradigm of neuroepithelial and RG‐like cells [28], but the true identity and physiological relevance of hPSC‐derived NSCs are open to interrogations because cells could acquire transcriptional and epigenetic programs that diverge from the cell state in vivo.
Acknowledgments
References
- Alejandro L. Diaz; Joseph G. Gleeson; The Molecular and Genetic Mechanisms of Neocortex Development. Clinics in Perinatology 2009, 36, 503-512, 10.1016/j.clp.2009.06.008.
- Jan H. Lui; David V. Hansen; Arnold R. Kriegstein; Development and Evolution of the Human Neocortex. Cell 2011, 146, 18-36, 10.1016/j.cell.2011.06.030.
- John C. Silbereis; Sirisha Pochareddy; Ying Zhu; Mingfeng Li; Nenad Sestan; The Cellular and Molecular Landscapes of the Developing Human Central Nervous System.. Neuron 2016, 89, 248-68, 10.1016/j.neuron.2015.12.008.
- André M.M. Sousa; Kyle A. Meyer; Gabriel Santpere; Forrest O. Gulden; Nenad Sestan; Evolution of the Human Nervous System Function, Structure, and Development. Cell 2017, 170, 226-247, 10.1016/j.cell.2017.06.036.
- Arnold R. Kriegstein; Arturo Alvarez-Buylla; The glial nature of embryonic and adult neural stem cells.. Annual Review of Neuroscience 2009, 32, 149-84, 10.1146/annurev.neuro.051508.135600.
- Joshua J. Breunig; Tarik F Haydar; Pasko Rakic; Neural Stem Cells: Historical Perspective and Future Prospects. Neuron 2011, 70, 614-625, 10.1016/j.neuron.2011.05.005.
- Simone A Fietz; Iva Kelava; Johannes Vogt; Michaela Wilsch-Bräuninger; Denise Stenzel; Jennifer L. Fish; Denis Corbeil; Axel Riehn; Wolfgang Distler; Robert Nitsch; et al.Wieland B. Huttner OSVZ progenitors of human and ferret neocortex are epithelial-like and expand by integrin signaling. Nature Neuroscience 2010, 13, 690-699, 10.1038/nn.2553.
- David V. Hansen; Jan H. Lui; Philip R. L. Parker; Arnold R. Kriegstein; Neurogenic radial glia in the outer subventricular zone of human neocortex. Nature 2010, 464, 554-561, 10.1038/nature08845.
- Isabel Reillo; Víctor Borrell; Germinal Zones in the Developing Cerebral Cortex of Ferret: Ontogeny, Cell Cycle Kinetics, and Diversity of Progenitors. Cerebral Cortex 2011, 22, 2039-2054, 10.1093/cercor/bhr284.
- Tomasz J. Nowakowski; Alex A. Pollen; Carmen Sandoval-Espinosa; Arnold R. Kriegstein; Transformation of the Radial Glia Scaffold Demarcates Two Stages of Human Cerebral Cortex Development.. Neuron 2016, 91, 1219-1227, 10.1016/j.neuron.2016.09.005.
- Bradley J. Molyneaux; Paola Arlotta; João R. L. Menezes; Jeffrey D. Macklis; Neuronal subtype specification in the cerebral cortex. Nature Reviews Neuroscience 2007, 8, 427-437, 10.1038/nrn2151.
- Luciano Conti; Elena Cattaneo; Neural stem cell systems: physiological players or in vitro entities?. Nature Reviews Neuroscience 2010, 11, 176-187, 10.1038/nrn2761.
- B. Reynolds; S Weiss; Generation of neurons and astrocytes from isolated cells of the adult mammalian central nervous system. Science 1992, 255, 1707-1710, 10.1126/science.1553558.
- Luciano Conti; Steven M. Pollard; Thorsten Gorba; Erika Reitano; Mauro Toselli; Gerardo Biella; Yirui Sun; Sveva Sanzone; Qi-Long Ying; Elena Cattaneo; et al.Austin Smith Niche-Independent Symmetrical Self-Renewal of a Mammalian Tissue Stem Cell. PLoS Biology 2005, 3, e283, 10.1371/journal.pbio.0030283.
- Steven M. Pollard; Luciano Conti; Yirui Sun; Donato Goffredo; Austin Smith; Adherent Neural Stem (NS) Cells from Fetal and Adult Forebrain. Cerebral Cortex 2006, 16, i112-i120, 10.1093/cercor/bhj167.
- D Goffredo; Luciano Conti; F Di Febo; G Biella; A Tosoni; G Vago; I Biunno; A Moiana; D Bolognini; M Toselli; et al.Elena Cattaneo Setting the conditions for efficient, robust and reproducible generation of functionally active neurons from adult subventricular zone-derived neural stem cells. Cell Death & Differentiation 2008, 15, 1847-1856, 10.1038/cdd.2008.118.
- Ilaria Albieri; Marco Onorati; Giovanna Calabrese; Alessia Moiana; Daniele Biasci; Aurora Badaloni; Stefano Camnasio; Dimitrios Spiliotopoulos; Zoltan Ivics; Elena Cattaneo; et al.Giangiacomo G. Consalez A DNA transposon-based approach to functional screening in neural stem cells. Journal of Biotechnology 2010, 150, 11-21, 10.1016/j.jbiotec.2010.07.027.
- Marco Onorati; Stefano Camnasio; Maurizio Binetti; Christian B. Jung; Alessandra Moretti; Elena Cattaneo; Neuropotent self-renewing neural stem (NS) cells derived from mouse induced pluripotent stem (iPS) cells. Molecular and Cellular Neuroscience 2010, 43, 287-295, 10.1016/j.mcn.2009.12.002.
- Marco Onorati; Maurizio Binetti; Luciano Conti; Stefano Camnasio; Giovanna Calabrese; Ilaria Albieri; Francesca Di Febo; Mauro Toselli; Gerardo Biella; Ben Martynoga; et al.François GuillemotGiangiacomo G. ConsalezElena Cattaneo Preservation of positional identity in fetus-derived neural stem (NS) cells from different mouse central nervous system compartments. Cellular and Molecular Life Sciences 2010, 68, 1769-1783, 10.1007/s00018-010-0548-7.
- Yirui Sun; Steven M. Pollard; Luciano Conti; Mauro Toselli; Gerardo Biella; Georgina Parkin; Lionel Willatt; Anna Falk; Elena Cattaneo; Austin Smith; et al. Long-term tripotent differentiation capacity of human neural stem (NS) cells in adherent culture. Molecular and Cellular Neuroscience 2008, 38, 245-258, 10.1016/j.mcn.2008.02.014.
- Lilian Hook; Joaquim Vives; Norma Fulton; Mathew Leveridge; Sarah Lingard; Martin D. Bootman; Anna Falk; Steven M. Pollard; Timothy E. Allsopp; Dennise Dalma-Weiszhausz; et al.Ann TsukamotoNobuko UchidaThorsten Gorba Non-immortalized human neural stem (NS) cells as a scalable platform for cellular assays. Neurochemistry International 2011, 59, 432-444, 10.1016/j.neuint.2011.06.024.
- Jignesh Tailor; Raja Kittappa; Ketty Leto; Monte Gates; Melodie Borel; Ole Paulsen; Sonia Spitzer; Ragnhildur Thora Karadottir; Ferdinando Rossi; Anna Falk; et al.Austin Smith Stem cells expanded from the human embryonic hindbrain stably retain regional specification and high neurogenic potency.. The Journal of Neuroscience 2013, 33, 12407-22, 10.1523/JNEUROSCI.0130-13.2013.
- Marco Onorati; Zhen Li; Fuchen Liu; André M.M. Sousa; Naoki Nakagawa; Mingfeng Li; Maria Teresa Dell’Anno; Forrest O. Gulden; Sirisha Pochareddy; Andrew T.N. Tebbenkamp; et al.Wenqi HanMihovil PletikosTianliuyun GaoYing ZhuCandace BichselLuis VarelaKlara Szigeti-BuckSteven LisgoYalan ZhangAnze TestenXiao-Bing GaoJernej MlakarMara PopovicMarie FlamandStephen M. StrittmatterLeonard K. KaczmarekE. S. AntonTamas HorvathBrett D. LindenbachNenad Sestan Zika Virus Disrupts Phospho-TBK1 Localization and Mitosis in Human Neuroepithelial Stem Cells and Radial Glia.. Cell Reports 2016, 16, 2576-2592, 10.1016/j.celrep.2016.08.038.
- Maria Teresa Dell’Anno; Xingxing Wang; Marco Onorati; Mingfeng Li; Francesca Talpo; Yuichi Sekine; Shaojie Ma; Fuchen Liu; William B. J. Cafferty; Nenad Sestan; et al.Stephen M. Strittmatter Human neuroepithelial stem cell regional specificity enables spinal cord repair through a relay circuit.. Nature Communications 2018, 9, 3419, 10.1038/s41467-018-05844-8.
- Yechiel Elkabetz; Georgia Panagiotakos; George Al Shamy; Nicholas D. Socci; Viviane Tabar; Lorenz Studer; Human ES cell-derived neural rosettes reveal a functionally distinct early neural stem cell stage. Genes & Development 2008, 22, 152-165, 10.1101/gad.1616208.
- Philipp Koch; Thoralf Opitz; Julius A. Steinbeck; Julia Ladewig; Oliver Brüstle; A rosette-type, self-renewing human ES cell-derived neural stem cell with potential for in vitro instruction and synaptic integration. Proceedings of the National Academy of Sciences 2009, 106, 3225-3230, 10.1073/pnas.0808387106.
- Wenlin Li; Woong Sun; Y. Zhang; Wanguo Wei; Rajesh Ambasudhan; Peng Xia; Maria Talantova; Tongxiang Lin; Janghwan Kim; Xiaolei Wang; et al.Woon Ryoung KimStuart A. LiptonKang ZhangSheng Ding Rapid induction and long-term self-renewal of primitive neural precursors from human embryonic stem cells by small molecule inhibitors. Proceedings of the National Academy of Sciences 2011, 108, 8299-8304, 10.1073/pnas.1014041108.
- Reuven Edri; Yakey Yaffe; Michael J. Ziller; Naresh Mutukula; Rotem Volkman; Eyal David; Jasmine Jacob-Hirsch; Hagar Malcov; Carmit Levy; Gideon Rechavi; et al.Irit Gat-ViksAlexander MeissnerYechiel Elkabetz Analysing human neural stem cell ontogeny by consecutive isolation of Notch active neural progenitors. Nature Communications 2015, 6, 6500, 10.1038/ncomms7500.





